ARTICLES
Reaction of plant species to Meloidogyne enterolobii and the efficiency of their aqueous extracts in controlling the pathogen
Reação de espécies vegetais a Meloidogyne enterolobii e eficiência de seus extratos aquosos no controle do patógeno
Reaction of plant species to Meloidogyne enterolobii and the efficiency of their aqueous extracts in controlling the pathogen
Semina: Ciências Agrárias, vol. 39, no. 6, pp. 2385-2398, 2018
Universidade Estadual de Londrina
Received: 01 November 2017
Accepted: 18 May 2018
Abstract: The genus Meloidogyne includes species of the most common nematodes to affect crops around the world. The species M. enterolobii is notable for affecting and causing serious losses in the production of guava trees and various other economically important crops in Brazil. The aim of this study was to evaluate the susceptibility of 10 plant species to parasitism by M. enterolobii, and the in vitro and in vivo effects of their leaf extracts on the pathogen. Initially seedlings of Solenostemon scutellarioides, Dieffenbachia amoena, Spigelia anthelmia, Plumbago scandens, Ricinus communis, Chenopodium ambrosioides, Azadirachta indica, Morinda citrifolia, Jatropha curcas and Datura stramonium were inoculated with 5,000 eggs of M. enterolobii to evaluate their susceptibility to nematode infection. For the test in vitro, a 5% concentration of the leaf extracts were added to Petri dishes, and 50 J2 of M. enterolobii were placed in each dish. After 48 hours incubation, the juveniles were evaluated for motility and mortality in the extracts. For the test in vivo, leaf extracts were used at the same concentration, however with only the seven most-promising in vitro species. This assay included the following sequence: inoculation of 5,000 eggs in autoclaved and moist soil contained in 1L pots; application of 30 ml of extract to the soil after 24 hours; transplanting of ‘Santa Clara’ tomato seedlings the following day; and reapplying the extract after 7 and 14 days. The results were evaluated 45 days after nematode inoculation. It was seen that the species D. amoena, R. communis, A. indica, M. citrifolia, J. curcas and D. stramonium displayed highly resistant behaviour; S. anthelmia, P. scandens and C. ambrosioides were very resistant, whereas S. scutelarioides was susceptible to the nematode. With the in vitro test, extracts from seven of the 10 species caused 70.4% to 97.4% J2 mortality. Applying the best leaf extracts to the soil was efficient in reducing M. enterolobii infestation in roots of the tomato.
Key words: Alternative control, Plant extracts, Root-knot nematode, Susceptibility.
Resumo: O gênero Meloidogyne contempla as espécies de nematoides que mais comumente afetam as culturas em todo o mundo. A espécie M. enterolobii tem se destacado por afetar e provocar sérias perdas na produção de goiabeiras e diversas outras culturas de importância econômica no Brasil. O objetivo deste trabalho foi avaliar a suscetibilidade de 10 espécies vegetais quanto ao parasitismo pelo M. enterolobii e o efeito in vitro e in vivo de seus extratos foliares sobre o patógeno. Inicialmente mudas das plantas Solenostemon scutellarioides, Dieffenbachia amoena, Spigelia anthelmia, Plumbago scandens, Ricinus communis, Chenopodium ambrosioides, Azadirachta indica, Morinda citrifolia, Jatropha curcas e Datura stramonium foram inoculadas com 5.000 ovos de M. enterolobii para avaliação da sua suscetibilidade quanto à infecção pelo nematoide. No ensaio in vitro, os extratos foliares na concentração de 5% foram adicionados a placas de Petri, pondo-se em cada placa 50 J2 de M. enterolobii. Após 48 horas de incubação, os juvenis foram avaliados quanto à motilidade e mortalidade nos extratos. Para o ensaio in vivo, foram também utilizados extratos na concentração final de 5%, contudo, de apenas sete das espécies, as mais promissoras in vitro. O ensaio in vivo envolveu a seguinte sequência: inoculação de 5.000 ovos em solo autoclavado e umedecido, aplicação de 30 ml de extrato ao solo 24 horas depois; transplantio das mudas de tomateiro ‘Santa Clara’ no dia seguinte; repetição da aplicação do extrato/vaso após 7 e 14 dias. Os resultados foram avaliados 45 dias contados a partir da inoculação do nematoide. Observaram-se que as espécies D. amoena, R. communis, A. indica, M. citrifolia, J. curcas e D. stramonium comportaram-se como altamente resistentes. Spigelia anthelmia, P. scandens, e C. ambrosioides foram muito resistentes, enquanto que S. scutelarioides foi suscetível ao nematoide. No teste in vitro, extratos de sete das 10 espécies provocaram a mortalidade dos J2 variando de 70,4 a 97,4%. A aplicação desses extratos foliares ao solo, foi eficiente para reduzir a infestação de M. enterolobii em raízes de tomateiro.
Palavras-chave: Controle alternativo, Extratos vegetais, Nematoide das galhas, Suscetibilidade.
Introduction
Currently more than 100 nematode species of the genus Meloidogyne Goeldi 1887 have been reported worldwide, and numerous host plant species are known, most of which are commercially exploited. Root-knot nematodes, in addition to being widely distributed, are extremely harmful to agriculture, causing damage to world production of around 10% and losses of around USD 120 billion (NICOL et al., 2011; OKENDI et al., 2014).
Despite the large number of species included in the genus, those that have caused the greatest losses to world agriculture are M. incognita, M. javanica, M. arenaria and M. hapla. In Brazil, in addition to these four species, M. enterolobii has recently become prominent due to the damage it causes in guava (Psidium guajava L.), often meaning that four years after planting an orchard, it is often no longer viable to grow fruit (PEREIRA et al., 2009). The importance of M. enterolobii to the country may increase as infestation of the pathogen increases, not only in areas of guava production, but also in plantations of other crops (CARNEIRO, ALMEIDA, 2001). In the State of Ceará, the nematode was found associated with the roots of the guava tree in every orchard in the regions they were collected (SILVA; SANTOS, 2017).
Controlling phytonematodes with the use of conventional nematocides is expensive and damages the environment. For this reason, researchers have been looking for alternative measures, such as crop rotation, the use of antagonistic plants, biological control, and more recently, the use of natural substances in infested soils, such as plant extracts and essential oils (CARBONI; MAZZONETTO, 2013; MOREIRA et al., 2015; MARTINS; SANTOS, 2016).
The use of plant-based products may be a viable alternative in the control of phytonematodes, since many plant species contain chemical substances and secondary metabolites, such as alkaloids, saponins, tannins, heterosides, flavonoids, steroids and phenols, which have nematocidal action (FERRAZ et al., 2012).
Considering the importance and distribution of M. enterolobii in the guava orchards of Ceará, and its occurrence in various crops including vegetables, the aim of this study was to evaluate the susceptibility of 10 plant species to parasitism by M. enterolobii, and the in vitro and in vivo effect of their aqueous extracts in controlling the pathogen.
Material and Methods
The experiments were carried out in the laboratory and under greenhouse conditions at the Department of Plant Science of the Federal University of Ceará (UFC) in Fortaleza in the State of Ceará, Brazil. The plants and aqueous extracts of 10 species were evaluated: coelus (Solenostemon scutellarioides L.), dumbcane (Dieffenbachia amoena Bull), Spigelia (Spigelia anthelmia L.), plumbago (Plumbago scandens L.), castor bean (Ricinus communis L.), mastruz (Chenopodium ambrosioides L.), neem (Azadirachta indica A. Juss.), noni (Morinda citrifolia L.), jatropha (Jatropha curcas L.) and jimsonweed (Datura stramonium L.).
The plant species used were selected based on their therapeutic and vermifugal action in animals and humans, and the presence of secondary compounds that may have nematocidal action on M. enterolobii.
The plants came from commercially grown seeds or from cuttings obtained from nurseries at UFC and rooted in the greenhouse.
To extract the eggs for the inoculum used in the assays, and for later analysis in which the number of eggs was considered, the Coolen and D'Herde technique (1972) was used.
Evaluation of the susceptibility of the plant species to M. enterolobii
All 10 species were evaluated for susceptibility to M. enterolobii. Seeds or plant cuttings were distributed in pots containing previously autoclaved soil and goat manure (2:1 v/v). When the seedlings displayed two pairs of true leaves, they were inoculated with approximately 5,000 eggs of M. enterolobii per plant. The inoculum was derived from infected roots of tomato plants. The obtained suspension was calibrated in a Peters chamber under microscopic stereoscopy to determine egg concentration (eggs/ml) for inoculation into 'Santa Clara' tomatoes (positive control).
Sixty days after inoculation, the plants were removed from the pots to evaluate the number of galls (NG), egg mass index (EMI) and reproduction factor (RF), all used to determine species susceptibility. In analysing the NG, a numerical scale from 1 to 5 was applied, as defined by Taylor and Sasser (1978). This scale of numerical values, modified by Hadisoeganda and Sasser (1982), makes it possible to analyse the EMI and its respective classification using assigned concepts. The RF is the result of the calculation, final population/initial population of the pathogen (5,000 eggs), where RF = 0 for immune species, RF <1 for resistant species and RF >1 for susceptible species (OOSTENBRINK, 1966).
The experimental design was completely randomised, comprising 10 treatments and seven replications. Each experimental unit was represented by one pot containing one plant of each species. The results of the data were transformed into √x+1, and submitted to analysis of variance, the mean values being compared by Tukey’s test at 5% probability using the Assistat v 7.6 beta statistical software (SILVA; AZEVEDO, 2009).
Evaluation of the in vitro effect of the aqueous extracts of the plant species on the mobility and mortality of juveniles of M. enterolobii
The in vitro test was performed with leaf extracts of the 10 plant species. The leaves of each species were collected at 60 days and dried in a greenhouse (30 ± 4 ºC) for up to 15 days.
The leaf extracts were obtained following a procedure adopted by Martins and Santos (2016), in which 10 ml of pre-boiled cold water were added to each gram of dry leaf. After standing for 24 hours, the leaves were ground using a pestle and mortar, and the extract filtered through gauze and centrifuged (2000 rpm for 20 minutes) to obtain a 10% (w/v) or 0.1 g mL-1 extract. The extracts of each species were then diluted with an equal proportion of water to obtain the 5% or 0.05 g mL-1 extract for immediate use in the tests.
The 0.05g mL-1 extracts were distributed in Petri dishes, 3.5 cm in diameter, at a volume of 3 ml per dish. Fifty freshly hatched juveniles (J2) of M. enterolobii were added to each dish, which was numbered and closed for observation. After 48 hours, a count was made of active and inactive juveniles. The juveniles were transferred to a Petri dish containing water to observe any possible recovery of mobility. After 24 hours, a new count of active and inactive juveniles was made. Any that still appeared straight in the water were considered dead. In the assay, the control consisted of Petri dishes containing water and 50 juveniles.
The experimental design was completely randomised, consisting of 11 treatments and one control (water), with six replications per treatment. The data were analysed using the Assistat v 7.6 beta statistical software, with the variances compared by F-test at 5% probability and the mean values compared by Tukey’s test at 5% probability.
Evaluation of the efficiency of aqueous extracts applied to the infested soil before and after planting the tomato seedlings
For this assay, leaf extracts at 0.05 g mL-1 were obtained as described in assay 2. To set up the experiment, plastic pots with a capacity of 1 kg were used, filled with an autoclaved mixture of soil and manure (2:1 v/v).
The extracts from seven of the 10 plant species were used, which were the most promising in the in vitro test. First, the soil in each pot was moistened with running water (100 ml) prior to inoculation. The soil was then infested with a suspension containing approximately 5,000 eggs/J2 of M. enterolobii, distributed in two holes. On the following day, 30 ml pot-1 of each plant extract was distributed to irrigate the still-moist soil surface. On the third day, 30-day-old 'Santa Clara' tomato seedlings were individually transplanted to the pots. At 7 and 14 days after transplanting the seedlings, another 30 ml of the extracts was applied. Tomato plants, both inoculated and not inoculated with the nematode, were watered and kept as the positive and negative control for the assay, respectively.
Forty-five days after transplanting, the tomato plants were removed from the pots, and the roots washed to determine the number of galls (NG), number of egg masses (NEM) and number of eggs (NE) under microscopic stereoscopy. The percentage population reduction (PR) was also calculated using the formula PR = 100.(1-T/C), adapted from Vizard and Wallace (1987), where PR = population reduction, T = Mean values for NE of the treatment and C = Mean value for the NE of the positive control.
In evaluating the morphological aspects of the tomato plants, the following were considered: leaf colouration, plant height, size and weight of the roots, and fresh and dry biomass. Leaf colouration was observed for cases of phytotoxicity. Plant height and root length (RL) were measured. The aboveground part of each plant was weighed individually, the same being done for the roots. The dried biomass was determined after drying the aboveground part in an oven at 65 °C for 72 hours.
The design was completely randomised, with nine treatments (seven plant extracts and two controls) and seven replications, the experimental unit consisting of one plant in each pot.
To analyse the data for the number of galls, egg masses and eggs, the values were transformed into √x+1 for the analysis of variance and the Scott-Knot test of the mean values. Tukey’s test at 5% probability was used to compare the mean values of length and weight, using the Assistat v 7.6 beta statistical software (SILVA; AZEVEDO, 2009).
Results and Discussion
Evaluation of the susceptibility of the plant species to M. enterolobii
Due to the large number of zero mean values, adjustment by statistical methods was not possible, the data from this assay was therefore analysed based on the use of scales and classification systems.
The roots of the species dumbcane, plumbago, S. anthelmia, castor bean and mastruz were small (2 to 3 mm) and had from 3.2 to 24.7 galls with few or even no egg masses. No females were found when scarifying the roots of the dumbcane or castor bean, suggesting that the pathogen would not be able to complete its development cycle in these two species. There was no gall formation in the neem, noni or jimsonweed; whereas in the coleus and the control, a large number of galls (>200) and females were found in the roots (Table 1).
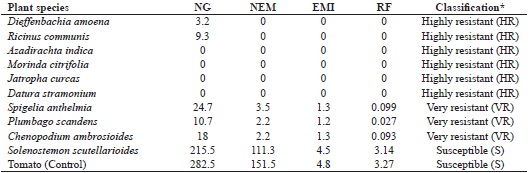
* Classification as per Taylor and Sasser (1978), modified by Hadisoeganda and Sasser (1982).
According to the Taylor and Sasser scale (1978) and the Hadisoeganda and Sasser classification (1982), the dumbcane, castor bean, neem, noni, jatropha and jimsonweed plants, which had an EMI of zero, were classified as highly resistant. In the plumbago, S. anthelmia and mastruz the EMI varied from 1.2 to 1.3, the plants being considered very resistant. Coleus, with the greatest EMI (4.5), was defined as susceptible, together with the control (Table 1).
With the classification proposed by Oostebrink (1966), the dumbcane, castor bean, neem, noni, jatropha and jimsenweed plants were classified as immune due to their RF of zero, and the plumbago, Spigelia and mastruz plants as resistant, since the RF was less than 1.0. An RF greater than 1.0, which classifies the plant as susceptible, was found only in coleus (Table 1) and in the control. Despite the low value for RF in coleus and the tomato, attributed to losses during extraction, the number of galls (215.5 and 282.5) and of external egg masses (111.3 and 151.5) was high, confirming strong multiplication of the pathogen in these plants under the conditions of the assay (Table 1).
There is controversy regarding the systems and variables that should be used in evaluating plant species for reactions of resistance/susceptibility to the root-knot nematode. Carneiro et al. (2012) evaluated 38 species of the Brassicaceae, Euphorbiaceae, Fabaceae and Poaceae families for susceptibility to M. enterolobii using the RF, and found that nine of them were considered immune to the pathogen, 26 were resistant and only three were susceptible. Silva and Silva (2009), evaluating the reaction of 11 species of the Poaceae and Fabaceae families to M. enterolobii, using the EMI as classification criterion, found that six were resistant and that two, crotalaria (Crotalaria paulina) and mucuna (Mucuna pruriens (L.) DC) reduced the number of galls and egg masses in tomatoes planted in pots in succession to these two species. In this test, comparing the EMI system with the RF system, a similar classification was seen for the 10 species under test.
Several plant species have been reported as antagonistic to Meloidogyne spp, as they produce bioactive compounds, such as proteins, alkaloids, tannins and terpenoids that may have a harmful effect on the nematodes. These compounds may be released by the roots or strengthen the plants, hampering penetration by the nematode or interfering in the life cycle and the interaction with the host plant (SOUZA et al., 2011; FERRAZ et al., 2012).
According to Campos et al. (2006), exudates of plants which are antagonistic or bad hosts to the root-knot nematode, as well as affecting the motility of these pathogens in the soil, may interfere in cell division in the nematode while still inside the egg, delaying formation of the second-stage juvenile and of the cuticle being broken by the J2 (hatching), such effects being a result of nematostatic action.
Compounds such as lipids, proteins and alkaloids are reported in dumbcane (FERREIRA et al., 2006). The plumbago plant contains naphthoquinones, flavonoids and terpenes (PAIVA et al., 2003). The active principles, actinidin and isoquinoline, are present in S. anthelmia (LORENZI; MATTOS, 2002). There is ricin in the castor bean, while neem plants have the presence of triterpenes, more specifically limonoids or tetranortriterpenoids and azadirachtin. Jimsonweed presents high rates of hyoscine in the tissue (FERRAZ et al., 2012). Various monoterpenes, alkaloids, saponins, glycosides and flavonoids have been reported in mastruz plants (ALMEIDA et al., 2012). Phenolic components, flavonoids and anthelmintic properties for humans have been reported in the noni, (DUSSOSSOY et al., 2011). Different species of Jatropha sp. present tannins, flavonoids, coumarins, saponins and others (DEVAPPA et al., 2010; SABANDAR et al., 2013). The supposition is that the above compounds might be involved in the resistance reaction to M. enterolobii seen in these species; on the other hand, the shikimic acid and flavonoids found in coleus do not show this antagonistic action (VOLP et al., 2008). The high infestation of the coleus roots and its easy multiplication through cuttings, together with its little-affected vegetative development, demonstrate that this species can be used for multiplication of the pathogen.
Despite the high degree of polyphagism and aggressiveness seen in M. enterolobii, nine non-host or nematode-resistant plant species were found in this research, some of which may contribute to a reduction of the pathogen in the soil, minimising the losses caused in infested areas.
Evaluation of the in vitro effect of the aqueous extracts of the 10 plant species on the motility and mortality of juveniles of M. enterolobii
Leaf extracts from the dumbcane, jimsonweed, plumbago, castor bean, mastruz and neem had a pronounced effect on the juveniles (J2) of M. enterolobii, with a marked reduction in their motility, which varied from 78.4 to 97.4%. However, extracts of the noni and jatropha had little effect on J2 motility. In turn, the coleus extract was inefficient, affecting only one in 50 individuals, the same as the control (water) (Table 2). The nematostatic effect was more expressive in the jatropha extract, with 48% of inactive juveniles recovering motility in the water.
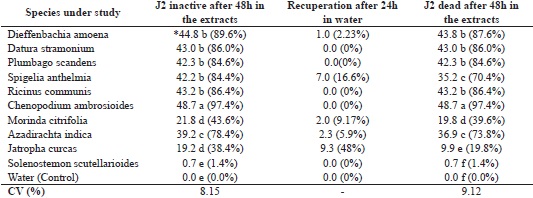
* Average of six replications, 50 juveniles dish-1. Mean values followed by the same letter in a column do not differ by Tukey’s test at 5% probability.
Placing juveniles in water after a time spent in plant extracts is essential, since the immobility induced by substances with a nematostatic effect can be reversed, i.e. a lack of motility does not imply nematocidal action (NEVES et al., 2008).
As for juvenile mortality, the extracts of dumbcane, jimsonweed, plumbago, S. anthelmia, castor bean, mastruz and neem had a high nematocidal effect, causing death in over 70% of the M. enterolobii J2, verified after placing them in water. The most efficient was the extract of mastruz, which caused 97.4% mortality. However, in the assays with jatropha, noni and coleus, the number of dead juveniles in the extracts was less than 40% (Table 2).
Santos (2015), studying the in vitro nematocidal potential of jimsonweed leaf extract at a concentration of 0.1 g mL-1, higher than the concentration used in this assay, found 100% mortality in second-stage juveniles of M. enterolobii. This result confirms the toxic action of jimsonweed on the nematoid.
The nematocidal effect of neem leaf extract has been demonstrated in several species of phytonematodes, among them Pratylenchus sp., Rotylenchulus reniformis, and M. incognita (FERRAZ et al,. 2012). In vitro tests of neem extract at a concentration of 0.25 g mL-1 caused death in 100% of M. incognita J2 (ADEGBITE; ADESIYAN, 2005). In this assay, neem leaf extract prepared at a lower concentration (0.05g mL-1) resulted in the death of 73.8% of M. enterolobii juveniles (Table 2).
Similar data to those obtained in this study were presented by Martins and Santos (2016) for leaf extracts of S. anthelmia and mastruz, in which strong nematocidal action on the J2 of M. incognita was found for aqueous extracts prepared at 0.1 g mL-1 and at 0.05g mL-1.
Aqueous extracts from the aboveground part of the castor bean were effective for in vitro mortality and the reduction of tumours in the roots of pepper plants infested with the false root-knot nematode, Nacobbus aberrans (MAREGGIANI et al., 2005).
A study carried out by Brito and Fernandes (2013), which evaluated the anthelmintic action of aqueous and ethanolic noni leaf extracts on the bird nematode, Heterakis gallinarum, found a nematode mortality of over 90%, demonstrating the high effectiveness of these extracts. However, in this study, the harmful effect of noni leaf extracts on the phytonematode, M. enterolobii, was low, with a mortality rate of only 39.6% (Table 3).
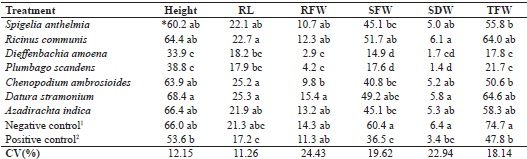
* Mean values followed by the same letter in a column do not differ by Tukey’s test at 5% probability.1 Uninoculated plants with no extract;2 Inoculated plants with no extract.
There are several studies in the literature that report the use of natural products, such as plant extracts and essential oils from various plant species (condiment, medicinal and toxic), as potential sources of nematocide and nematostatic compounds, especially from the leaves, roots and seeds (NEVES et al., 2008; MOREIRA et al., 2009; FERRAZ et al., 2012).
Studies of plant extracts in the control of phytonematodes are generally carried out in vitro or in the greenhouse, and confirmation of the results in the field is necessary.
The information obtained in this assay allowed the selection of promising plants for working with extracts in the greenhouse. The results of this research may encourage testing of the more-effective extracts under field conditions, with the aim of later adopting this practice as an alternative measure of nematode control.
Evaluation of the efficiency of aqueous extracts applied to the infested soil before and after planting the tomato seedlings
In evaluating the variables used in this assay, different action by the seven extracts was seen on plant growth and infection.
The tomato plants grown in soil inoculated with M. enterolobii and treated with the leaf extracts of S. anthelmia, castor bean, mastruz, neem and jimsonweed, showed no difference in height when compared to the negative control (66 cm) (healthy plant with no extract) (Table 3). On the other hand, where the extracts of dumbcane and plumbago were used, plant height was reduced (33.9 and 38.8 cm), and was even less than in the tomato plants of the positive control (53.6 cm) (Table 3).
It was found that for the tomato plants treated with extracts of S. anthelmia, castor bean, mastruz, jimsonweed and neem, there was no difference in RL (21.9 to 25.3 cm); this included the negative control. However, in plants treated with extracts of dumbcane and plumbago, root length was smaller (17.9 and 18.2 cm), differing from earlier treatments, but similar to the result obtained with the positive control (Table 3).
It was found that for plants in soil treated with extracts of Spigelia, castor bean, mastruz, jimsonweed and neem, the values for root fresh weight (RFW) were generally similar and did not differ from the results obtained with the controls. However, in the treatments where extracts of dumbcare and plumbago were applied to the soil, the plants presented values for RFW well below those of the other treatments (Table 3).
For shoot fresh weight (SFW), the best treatments were found to be those where extracts of castor bean and jimsonweed were used, and were no different from the negative control, where the average was 60.4 g. In the other treatments, with extracts of S. anthelmia, mastruz and neem, the values for SFW were no different from the positive control. The plants from soil treated with extracts of Spigelia and dumbcane again had the lowest values related to development (Table 3).
Ferreira et al. (2013), evaluating the aqueous extracts of six species applied to soil infested with M. incognita, found that the extracts increased aboveground weight in tomato plants, but found no increase in the fresh weight of the root system. Similarly, in the present study no increase in root development was seen in the tomato plants for any of the treatments.
Evaluation of shoot dry weight (SDW) for each treatment showed that the plants with an application of extracts of Spigelia, castor bean, mastruz, jimsonweed and neem did not differ in mean value from the negative control, but did differ from the positive control. This suggests that application of the extracts of these five plant species to the soil may have favoured the biomass of the tomato plants, since in the presence of nematode parasitism, development of the plants is usually affected (Table 3).
Tomato plants from the treatments with castor bean, jimsonweed and neem extract showed no statistical differences in total fresh weight (TFW) when compared to the negative control (70.7 g). Values for TFW in plants treated with extracts of Spigelia and mastruz were close to that of the positive control. Only the tomato plants treated with dumbcane and plumbago had the lowest values for TFW, both less than 22 g (Table 4).
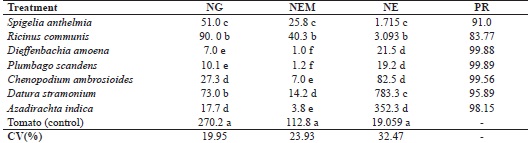
* Mean values followed by the same letter in a column do not differ by Skott-Knot test at 5% probability. The data for NG, NEM and NE were transformed into √x+1 for the statistical analysis. The original values are shown in the table.
The smaller growth of the plants in soil receiving extracts of dumbcare and plumbago may be related to a possible phytotoxic effect, since marked chlorosis was seen in the leaves seven days after the first application of the extracts. This toxic reaction may also have contributed to the lesser development of the treated plants.
The results of evaluating the effect of the seven plant extracts on M. enterolobii infestation in tomato plants showed that all treatments differed from the positive control for NG, NEM and NE, with a significant reduction in parasitism, indicating that the extracts were quite effective in controlling the pathogen (Table 4).
Considering the good development of the plant root system, the lowest mean values for NG were seen in treatments involving the extracts of neem (17.7) and mastruz (27.3), followed by S. anthelmia, jimsonweed and castor bean, all of which were significantly less than the result of the positive control (270.2) (Table 4).
The NEM in the roots of the treated plants varied from 3.8 in the treatment with neem extract to 40.3 where castor bean extract was applied to the soil. The positive control had a mean value of 112.8 eggs masses.root-1, differing from the other treatments (Table 4).
Soil treated with the extracts of mastruz and neem had the lowest mean values for NE, of 82.5 and 352.3 respectively. The jimsonweed, Spigelia, and castor bean extracts were also effective in reducing nematode infestation in the tomato plants, with even lower values for the number of eggs (783.3; 1,715 and 3,093) than the control with 19,059 eggs (Table 4).
The percentage population reduction (PR) of M. enterolobii in the root system of the tomato plants, calculated based on the number of eggs found per root, was greater than 90% in the treatments with mastruz, neem, jimsonweed or Spigelia extract. For the castor bean, the PR was still high, with a reduction of 83.77 in egg production per plant (Table 4).
The lower number of galls and reduced parasitism in the tomato plants from the treatments with dumbcare (7.0) and plumbago (10.1), as well as the NEM (1.0 and 1.2) and NE (21.5 and 19.2), may be associated with the lesser root development caused by the phytotoxic effect. New studies should therefore be carried out with these extracts, which were efficient in vitro, with the aim of evaluating the best concentration for their use in infested soil when it is intended to work with tomato plants.
The marked reduction in the number of eggs in the roots of the tomato plants is associated with the smaller number of females, and consequently of egg masses, in the root system, due to the death of infesting juveniles through the nematocidal action of the leaf extracts. Application of the extracts carried out the day after inoculation, and 7 and 14 days later, were effective in eliminating juveniles of M. enterolobii present in the inoculum and in the soil. Repeat applications of the extracts to the soil at seven-day intervals would have affected any hatched J2 through nematocidal action, as seen in the in vitro assays. Tests to investigate the in vitro effect of the extracts on the hatching of J2 were not carried out in this assay.
Results similar to those of this study were obtained by Almeida et al. (2012) using extracts of neem, where the authors verified the efficiency of the aqueous extract applied to the soil on M. javanica, with a reduction of 92% in the number of galls.
The nematocidal action of mastruz was reported by Rodrigues et al. (2008), who found that the aqueous extract of this species was able to reduce the number of galls of the M. exigua coffee nematode by 45.7%.
Ademola et al. (2007) reported from in vitro and in vivo tests that Spigelia has an anthelmintic action on sheep nematodes. Martins (2009) demonstrated the efficiency of the aqueous extract of this species on M. incognita when applied to the soil, reducing the number of galls in tomato plants by 59.8%.
Santos (2015), cultivating jimsonweed and D. metel before planting crispy lettuce (Lactuca sativa L.), showed a marked reduction (98-100%) in M. enterolobii parasitism when compared to the control.
Observations by Gardiano et al. (2009) showed that the aqueous extract of the castor bean when applied to the soil, gave a 54.4% and 56.6% reduction respectively in the number of galls and eggs of M. javanica in tomato plants.
According to Ferraz et al. (2012), research involving a greater number of botanical families is necessary to select plants with nematocidal potential, and to characterise the active compounds present in those plants, their mode of action, appropriate concentration and best method of application under field conditions.
In this study, the results obtained with leaf extracts were promising for further investigation of nematocidal compounds, aiming at their better use in the effective control of M. enterolobii in infested areas.
Conclusions
The plant species, dumbcane, Spigelia, plumbago, castor bean, mastruz, neem, noni, jatropha and jimsonweed can be recommended for crop rotation in areas infested with M. enterolobii.
Coleus plants can be employed in the multiplication of M. enterolobii.
The leaf extracts of dumbcane, Spigelia, plumbago, castor bean, mastruz, neem and jimsonweed displayed an in vitro nematocidal effect on the juveniles of M. enterolobii, with the extract of mastruz being the most efficient.
The extracts of coleus, neem and jatropha had little effect on M. enterolobii juveniles in vitro.
The leaf extracts of each species effective in vitro, were also effective in reducing M. enterolobii infestation in the roots of tomato plants.
Acknowledgements
The authors wish to thank CAPES for the scholarship grant. Thanks also go to the Federal University of Ceará where the research was carried out.
References
ADEGBITE, A. A.; ADESIYAN, S. O. Root extracts of plants to control root-knot nematode on edible soybean. World Journal of Agricultural Sciences, Ibadan, v. 1, n. 1, p. 18-21, jan. 2005.
ADEMOLA, I. O.; FAGBEMI, B. O.; IDOWU, S. O. Anthelmintic activity of Spigelia anthelmia extract against gastrointestinal nematodes of sheep. Parasitology Research, Ibadan, v. 1, n. 101, p. 63-69, jun. 2007.
ALMEIDA, F. A.; PETTER, F. A.; SIQUEIRA, V. C.; ALCÂNTARA NETO, F.; ALVES, A. U.; LEITE, M. L. T. Modos de preparo de extratos vegetais sobre Meloidogyne javanica no tomateiro. Nematropica, v. 42, n. 1, p. 9-15, 2012.
BRITO, D. R. B.; FERNANDES, R. M. Ação anti-helmíntica da Morinda citrifolia (noni) sobre Heterakis gallinarum. Semina : Ciências Agrárias, Londrina, v. 34, n. 4, p. 1775-1782, jul./ago. 2013.
CAMPOS, H. D. ; CAMPOS V. P.; COIMBRA , J. L. Efeito de exsudato radicular de Brachiaria decumbens e de sorgoleone de Sorghum bicolor no desenvolvimento de Meloidogyne javanica. Nematologia Brasileira, Rio Verde, v. 30, n. 1, p. 59-65, abr. 2006.
CARBONI, R. Z.; MAZZONETTO, F. Efeito do extrato aquoso de diferentes espécies vegetais no manejo de Meloidogyne incognita em tomateiro em ambiente protegido. Revista Agrogeoambiental, Pouso Alegre, v. 5, n. 2, p. 61-66, ago. 2013.
CARNEIRO, R. D. G.; ALMEIDA, M. R. A. Técnica de eletroforese usada nos estudos de enzimas dos nematoides de galhas para identificação de espécies. Nematologia Brasileira, Brasília, v. 25, n. 1, p. 35-44, jul. 2001.
CARNEIRO, R. M. D. G.; FREITAS, V. M.; MATTOS, J. K.; CASTRO, J. M.; GOMES, C. B.; CARNEIRO, R. M. Major guava nematodes and control prospects using resistance on Psidium spp. and non-host crops. Acta Horticulturae, Leuven, n. 959, p. 41-49, set. 2012.
COOLEN, W. A.; D´HERDE, C. J. A method for the quantitative extration of nematodes from plant tissue. Merelbeke: State Agriculture Research Center - GHENT, 1972. 77 p.
DEVAPPA, R. K.; MAKKAR, H. P. S.; BECKER, K. Jatropha toxicity- a review. Jornal Toxicol Environ Health B, Filadélfia, v. 13, n. 6, p. 476-507, ago. 2010.
DUSSOSSOY, E.; BRAT, P.; BONY, E.; BOUDARD, F.; POUCHERET, P.; MERTZ, C.; GIAIMIS, J.; MICHEL, A. Characterization, anti-oxidative and anti-inflammatory effects of Costa Rican noni juice (Morinda citrifolia L.). Journal of Ethnopharmacology, Bethesda, v. 133, n. 1, p. 108-115, jan. 2011.
FERRAZ, S.; FREITAS, L. G.; LOPES, E. A.; DIAS-ARIEIRA, C. R. Manejo sustentável de fitonematoiodes. Viçosa, MG: UFV, 2012. 304 p.
FERREIRA, I. C. M.; SILVA, G. S.; NASCIMENTO, F. S. Effect of aqueous extracts of Asteraceae species on Meloidogyne incognita. Summa Phytopathologica, Botucatu, v. 39, n. 1, p. 40-44, jan./mar. 2013.
FERREIRA, L. S.; MARSOLA, F. J.; TEIXEIRA, S. P. Anatomia dos órgãos vegetativos de Dieffenbachia picta Schott (Araceae) com ênfase na distribuição de cristais, laticíferos e grãos de amido. Revista Brasileira de Farmacognosia, Ribeirão Preto, v. 16, p. 664-670, dez. 2006. Suplemento.
GARDIANO, C. G.; FERRAZ, S.; LOPES, E. A.; FERREIRA, P. A.; AMORA, D. X.; FREITAS, L. G. Avaliação de extratos aquosos de várias espécies vegetais, aplicados ao solo, sobre Meloidogyne javanica (Treub, 1885) Chitwood, 1949. Semina : Ciências Agrárias, Londrina, v. 30, n. 3, p. 551-556, jul./set. 2009.
HADISOEGANDA, W. W.; SASSER, J. N. Resistance of tomato, bean, southern pea, and garden pea cultivars to root-knot nematodes based on host suitability. Plant Disease, Raleing, v. 66, n. 2, p. 145-150, fev. 1982.
LORENZI, H.; MATOS, F. J. A. Plantas medicinais no Brasil: nativas e exóticas. São Paulo: Instituto Plantarum, Nova Odessa, 2002. 512 p.
MAREGGIANI, G.; ZAMUNER, N.; MICHETTI, M.; FRANZETTI, D.; COLLAVINO, C. Impact of natural extracts on target and non target soil organisms. Boletín de Sanidad Vegetal, Plagas, Buenos Aires, v. 31, n. 3, p. 443-448, jun. 2005.
MARTINS, M. C. B. Reação de plantas medicinais e efeito de seus extratos sobre Meloidogyne incógnita. 2009. Dissertação (Mestrado em Fitotecnia) - Centro de Ciências Agrárias, Universidade Federal do Ceará, Fortaleza.
MARTINS, M. C. B.; SANTOS, C. D. G. Ação de extratos de plantas medicinais sobre juvenis de Meloidogyne incognita raça 2. Revista Ciências Agronômica, Fortaleza, v. 47, n. 1, p. 135-142, jan./mar. 2016.
MOREIRA, F. J. C.; SANTOS, C. D. G.; INNECCO, R. Eclosão e mortalidade de juvenis J2 de Meloidogyne incognita raça 2 em óleos essenciais. Revista Ciências Agronômica, Fortaleza, v. 40, n. 3, p. 441-448, jul./set. 2009.
MOREIRA, F. J. C.; SANTOS, C. D. G.; INNECCO, R.; SILVA, G. S. Controle alternativo de nematoide das galhas (Meloidogyne incognita) raça 2, com óleos essenciais em solo. Summa Phytopathologica, Botucatu, v. 41, n. 3, p. 207-213, jun. 2015.
NEVES, W. S.; FREITAS, L. G.; CLEIA, F. S.; DALLEMOLE-GIARETTA, F. R.; FERREIRA, P. A.; FERRAZ, L. O.; ONKAR, D. D.; FERRAZ, S. Ação nematicida de óleo, extratos vegetais e de dois produtos à base de Capsaicina, Capsainóides e Alil Isotiocianato sobre juvenis de Meloidogyne javanica (Treub) Chitwood. Nematologia Brasileira, Piracicaba, v. 32, n. 2, p. 93-100, mar. 2008.
NICOL, J. M.; TURNER, S. J.; COYNE, D. L.; DEN NIJS, L.; HOCKLAND, S.; TAHNA MAAFI, Z. Current nematode threats to world agriculture. In: JONES, J.; GHEYSEN, G.; FENOLL, C. (Ed.). Genomics and molecular genetics of plant-nematode interactions. Heidelberg: Springer, 2011. p. 21-43.
OKENDI, E. M.; KARIUKI, G. M.; MARAIS, M.; MOLELEKI, L. N. The threat of root-knot nematodes (Meloidogyne spp.). Plant Pathology, v. 63, n. 4, p. 727-737, fev. 2014.
OOSTENBRINK, M. Major characteristic of relation between nematodes and plants. Mededelingen Landbouwhogeschool, Wageningen, v. 66, n. 4, p. 1-46, 1966.
PAIVA, S. R.; FIGUEIREDO, M. R.; ARAGO, T. V.; KAPLAN, M. A. C. Antimicrobial activity in vitro of plumbagin isolated from Plumbago species. Memórias do Instituto Oswaldo Cruz, Rio de Janeiro, v. 98, n. 7, p. 959-961, jan. 2003.
PEREIRA, F. O. M.; SOUZA, R. M.; SOUZA, P. M.; DOLINSKI, C.; SANTOS, G. K. Estimativa do impacto econômico e social direto de Meloidogyne mayaguensis na cultura da goiaba no Brasil. Nematologia Brasileira, Piracicaba, v. 33, n. 2, p. 176-181, out. 2009.
RODRIGUES, A. A.; RABELLO, L. K. C.; ALVES, F. R.; JESUS JUNIOR, W. C. de; MORAES, W. B. de;GUERRA, P. A. Efeito dos extratos aquosos de guiné e erva de santa maria na redução populacional de Meloidogyne exigua em cafeeiro. In: ENCONTRO LATINO AMERICANO DE INICIAÇÃO CIENTÍFICA E ENCONTRO LATINO AMERICANO DE PÓS-GRADUAÇÃO, 12., 8., 2008, São José dos campos. Anais... São José dos campos: Universidade do Vale do Paraíba. 2008.
SABANDAR, C. W.; AHMAT, N.; JAAFAR, F. M.; SAHIDIN, I. Medicinal property, phytochemistry and pharmacology of several Jatropha species (Euphorbiaceae): a review. Phytochemistry, Amsterdã, v. 85, p. 7-29, jan. 2013.
SANTOS, M. L. L. Emprego de plantas com princípios tóxicos no controle de Meloidogyne enterolobii. 2015. Dissertação (Mestrado em Agronomia/Fitotecnia) - Centro de Ciências Agrárias, Universidade Federal do Ceará, Fortaleza.
SILVA, F. de A. S.; AZEVEDO, C. A. V. Principal components analysis in the software assistat-statistical attendance. In: WORLD CONGRESS ON COMPUTERS IN AGRICULTURE, 2009, Orlando. Anais… Reno: American Society of Agricultural and Biological Engineers, 2009. CD-ROM.
SILVA, K. C.; SILVA, G. S. Reaction of grasses and legumes to Meloidogyne mayaguensis. Nematologia Brasileira, Piracicaba, v. 33, n. 2, p. 198-200, mar. 2009.
SILVA, M. C. L.; SANTOS, C. D. G. Distribution of Meloidogyne enterolobii in guava orchards in the state of Ceará, Brazil. Revista Caatinga, Mossoró, v. 30, n. 2, p. 335- 342, abr./jun. 2017.
SOUZA, J. D.; SILVA, M. B. R. ; ARGOLO, A .C. C.; NAPOLEÃO, T. H.; SÁ, R. A.; CORREIA, M. T. S.; PAIVA, P. M. G.; SILVA, M. D. C.; COELHO, L. C. B. B. A new Bauhinia monandra galactose-specific lectin purified in milligram quantities from secondary roots with antifungal and termiticidal activities. International Biodeterioration & Biodegradation, v. 65, n. 5, p. 696-702, ago. 2011.
TAYLOR, A. L.; SASSER, J. N. Biology, identification and control of root-knot nematodes (Meloidogyne sp.). Raleigh: International Meloidogyne Project, NCSU & USAID Coop. Publ., 1978. 111 p.
VIZARD, A. L.; WALLACE, R. J. A simplified egg count reduction test. Australian Veterinary Journal, v. 64, n. 4, p. 109-111, abr. 1987.
VOLP, A. C. P.; RENHE, I. R. T.; BARRA, K.; STRINGUETA, P. C. Flavonóides antocianinas: características e propriedades na nutrição e saúde. Revista Brasileira Nutrição Clinica, Viçosa, MG, v. 23, n. 2, p. 141-149, jan. 2008.
Author notes
* Author for correspondence