ARTICLES
In vitro evaluation of the safety and probiotic and technological potential of Pediococcus pentosaceus isolated from sheep milk
Avaliação in vitro do potencial de segurança, probiótico e tecnológico de Pediococcus pentosaceus isolado de leite de ovelhas
In vitro evaluation of the safety and probiotic and technological potential of Pediococcus pentosaceus isolated from sheep milk
Semina: Ciências Agrárias, vol. 39, no. 1, pp. 113-131, 2018
Universidade Estadual de Londrina
Received: 31 March 2017
Accepted: 19 October 2017
Abstract: Six isolates (Ac1Pd, Ac3Pd, Ac4Pd, Ac5Pd, Ac7Pd, and Ac22Pd) of Pediococcus pentosaceus from sheep milk were tested for safety and for probiotic and technological potential. The results showed that none of the isolates were able to produce biogenic amines or virulence factors. The isolates tested showed low hydrophobicities, high auto-aggregation capacities and co-aggregation with L. monocytogenes ATCC 7644, L. sakei ATCC 15521 and E. faecalis ATCC 19444, but none produced β-galactosidase and bacteriocins. The isolates did not show growth at pH values 3 and 12, while in a pH range from 4 to 10 the growth was variable. In the absence of bile, all the isolates showed growth, with suppression at bile concentrations of 0.1%, 0.3%, 0.6% and 1.0%. In the disc-diffusion test, the isolates tested were resistant to oxacillin, sulfatrimethoprim and vancomycin but were sensitive to chloramphenicol and tetracycline. The isolates showed variable responses to penicillin G and were resistant to most of the drugs tested, except for amoxicillin trihydrate and ibuprofen. All cultures showed a high milk-acidification capacity after 24 hours and none produced exopolysaccharides. The isolates of P. pentosaceus were able to produce diacetyl; however, no culture showed extracellular proteolytic activity and the autolysis varied from 21.3% to 30.5% after 24 h. The isolates grew at NaCl concentrations of 4.0 and 6.0%, but the growth was lower at 10.0%. Finally, all the isolates were found to be safe but had limited application as probiotics and in some technological uses.
Key words: Dairy technology, Lactic acid bactéria, Starter.
Resumo: Testes de avaliação de potencial de segurança, probiótico e tecnológicos foram realizados em seis isolados de Pediococcus pentosaceus (Ac1Pd, Ac3Pd, Ac4Pd, Ac5Pd, Ac7Pd, Ac22Pd) provenientes de leite de ovelha. Os resultados obtidos demonstraram que nenhum dos isolados foi capaz de produzir aminas biogênicas ou fatores de virulência. Os isolados testados apresentaram baixa hidrofobicidade, alta capacidade de autoagregação e coagregação com L. monocytogenes ATCC 7644, L. sakei ATCC 15521 e E. faecalis ATCC 19444, porém nenhum produziu β-galactosidase e bacteriocinas. Não foi observado desenvolvimento dos isolados em pH 3 e 12, sendo que na faixa de pH de 4 a 10 o desenvolvimento foi variável. Na ausência de bile todos os isolados apresentaram desenvolvimento, observando-se supressão quando em concentrações de 0,1%, 0,3%, 0,6% e 1%. No teste de disco-difusão os isolados testados foram resistentes à oxaciclina, sulfatrimetropim e vancomicina, mas foram sensíveis ao cloranfenicol e à tetraciclina e com resultados variáveis para a penicilina G e foram resistentes à maioria dos medicamentos testados, exceto à amoxicilina tri-hidratada e ibuprofeno. Foi observada acidificação do leite somente após 24 h e nenhum produziu exopolissacarídeos. Os isolados de P. pentosaceus foram capazes de produzir diacetil, no entanto, nenhuma cultura apresentou atividade proteolítica extracelular e a produção de autólise foi variada de 21,3% a 30,5%, após 24 h. Os isolados se desenvolveram em concentrações de NaCl a 4% e 6%, porém o desenvolvimento foi menor na concentração de 10%. Por fim, todos os isolados apresentaram boa capacidade de segurança, mas aplicação limitada como probióticos e alguns aspectos de potencial tecnológico.
Palavras-chave: Bactérias ácido-láticas, Culturas iniciadoras, Tecnologia do leite.
Introduction
According to FAO/WHO (2002) probiotics are defined as living microorganisms that are capable of conferring health benefits to their host when administered in adequate quantities. Most probiotic microorganisms are lactic acid bacteria (LAB). LAB can have several beneficial properties in food, such as the conservation of its nutrition and an increase in its flavor as well as an increase in food safety (MEIRA, 2011). For a microorganism to be considered a probiotic it is necessary that it present characteristics desirable for safety, functionality and technological use.
With regard to safety, probiotic microorganisms must come from the normal gut inhabitants of healthy animals, must not be toxic or pathogenic, and it is preferable that the isolates used be host-specific, in order to obtain maximum effectiveness. The functionality of the lineage is linked with its ability to withstand the adverse conditions of its host’s gastrointestinal tract (GIT) and its antagonistic ability against resident pathogens (ALVIM, 2011). In addition, with regard to technological properties, probiotics should contain isolates that grow fast in vitro, are easy to handle, have good industrial production conditions and survive in the final product with their function intact (PANCHENIAK, 2005).
Pediococcus is a genus of LAB that has great economic importance in the fermented-food industry because Pediococcus bacteria are used as starter cultures in the fermentation processes of milk, meats and sausages, sauerkraut, cucumber, green beans, soybeans, bread and silage. P. pentosaceus, in particular, is relevant due to its biotechnological potential (IVANOVA et al., 2013). Although there are many well-characterized probiotic lineages available for commercial use, the isolation and characterization of new strains is desirable for the formulation of modern probiotic foods because the health benefits are specific to each lineage. Thus, the present work seeks to evaluate in vitro the probiotic technological and safety potential of P. pentosaceus isolated from sheep milk.
Materials and Methods
Strains and culture conditions
Isolates of P. pentosaceus were obtained from sheep milk and are part of the LAB collection of the Laboratory of Milk Analysis and Derivatives, Faculty of Agronomy and Veterinary Medicine, UnB; they were previously identified by Koch (2014). In total, six isolates were evaluated and identified: Ac1Pd, Ac3Pd, Ac4Pd, Ac5Pd, Ac7Pd and Ac22Pd. The isolates are stored frozen (-80 ºC) in de Man, Rogosa and Sharpe (MRS) broth (Oxoid Ltd., Basingstoke, England) containing 20% (w/v) glycerol. For this study, the isolates were recovered in MRS broth and stored in MRS agar until the analyses were performed at the Animal Products Inspection Laboratory, Federal University of Viçosa-MG.
Virulence factors
The isolates were subjected to different tests to identify virulence factors, as described by Barbosa et al. (2010). Hemolytic activity was assessed by streaking the cultures on Trypticase Soy Agar (TSA) (Oxoid) supplemented with 5% (w/v) defibrinated horse blood. Subsequently, the plates were incubated at 37 °C for 48 h. In this test, halo formation indicates hemolysis. The hemolysis by each isolate was classified as total or β-hemolysis (clear halos around the colonies), partial or α-hemolysis (halos around the greenish colonies), and absent or γ-hemolysis. Gelatinase production was determined by placing 1 µL aliquots of the cultures on the surface of Luria-Bertani agar (1.0% tryptone, 0.5% yeast extract, 1.0% NaCl) supplemented with 3.0% (w/v) gelatin (BD) and incubating at 37 °C for 48 h. After incubation, the plates were maintained at 4 °C for 4 h and gelatin hydrolysis was identified by the formation of opaque halos around the colonies. Lipase production was assessed by spotting 1 µL of the cultures on Luria-Bertani agar plates supplemented with CaCl2 (Sigma-Aldrich, 0.2%, w/v) and Tween 80 (Sigma-Aldrich, 1.0%, w/v) and incubating at 37 °C for 48 h. The formation of clear halos around the colonies was recorded as lipase production. Deoxyribonuclease production was identified by spotting 1 µL aliquots of the cultures on the surface of DNAse agar (BD) and incubating at 37 °C for 48 h. Positive results were identified by the formation of translucid halos around the colonies.
Production of biogenic amines
The production of biogenic amines by the selected isolates was evaluated as described by Bover-Cid and Holzapfel (1999). The decarboxylase production was induced by five successive transfers of 0.5 mL aliquots of the cultures in MRS broth (Oxoid) supplemented with 0.005% pyridoxal 5-phosphate (w/v, Sigma-Aldrich) and with 0.1% (w/v) of each of the precursors of biogenic amines: free tyrosine base (for tyramine), histidine monohydrochloride (for histamine), ornithine monohydrochloride (for putrescine) and lysine monohydrochloride (for cadaverine), all from Sigma Aldrich. Each culture was incubated at 37 °C for 24 h, and after 4 days the final cultures were seeded on decarboxylase agar - a modified MRS agar as described by Joosten and Northolt (1989) - supplemented with one of each biogenic amine precursor at 0.1% (w/v). Plates were incubated at 37 °C for 24 h. Positive results were identified by a change in color from yellow to purple.
Cell surface hydrophobicity
The abilities of the isolates to adhere to the hydrophobic substrates were determined according to the methodology of Santos et al. (2014). Cells were centrifuged (7000xg for 5 min at 4 °C), washed twice with 0.1 mol L-1 phosphate buffer, and resuspended in the same solution, and then the optical density (OD) of the resuspension was measured in a spectrophotometer, at a wavelength of 560 nm, before incubation (OD0). N-hexadecane was added to the cell suspension in the ratio 1:5 (N-hexadecane (Sigma-Aldrich)/cell suspension), and the mixture was homogenized for 2 min. After 1 h of incubation at 37 °C, the A560 (A) value of the aqueous layer was measured. One milliliter of the aqueous phase was removed to determine the OD. The hydrophobicity percentage was calculated from the following equation: , where A0 and A are the absorbance values before and after extraction with the organic solvent, respectively.
Auto-aggregation and co-aggregation
As described by Todorov et al. (2011) and Santos et al. (2014), the isolates were reactivated in MRS broth for 24 h at 37 °C, and the solutions were centrifuged (7000xg for 10 min at 20 °C), washed, resuspended and diluted in sterile saline solution (0.85% NaCl). Then, 1 mL of the cell suspension was transferred to the cuvette to obtain an OD-reading of approximately 0.3. Then, the samples were incubated in Eppendorf-type microcentrifuge tubes at 37 °C for 1 h. The OD was measured in a spectrophotometer at a wavelength of 660 nm before (OD0) and after a 60 min (OD60) incubation. To determine OD60, the cultures were centrifuged at 3000xg for 2 min at 20 °C. Auto-aggregation was determined as described by Todorov et al. (2008), using the equation: , where OD0 refers to the initial OD and OD60 refers to the OD obtained after 60 min of incubation.
To evaluate co-aggregation, cultures of L. sakei ATCC 15521 were grown in MRS broth, and L. monocytogenes ATCC 7644 and E. faecalis ATCC 19444 in BHI (Oxoid) for 24 h and 48 h, respectively, at 37 °C. The cells were prepared in a similar manner so the cell suspensions reached an OD660nm of 0.3. Then, 750 µL of each isolate was mixed for 30 seconds with 750 µL of the indicator microorganism: L. monocytogenes ATCC 7644 (bacteriocin-sensitive), L. sakei ATCC 15521 and E. faecalis ATCC 19443 (bacteriocin-resistant); the OD660nm was recorded at the start of incubation (time 0) and after 60 min of incubation at 37 °C. To determine the OD60, the cells were centrifuged at 3000xg for 2 min at 20 °C. For the calculations, the same formula was used as described above. The tests were performed in triplicate.
Growth at different pH and bile concentrations
The isolates were inoculated in MRS broth their pH adjusted to 3, 4, 5, 6, 7, 8, 10 and 12 by adding 1.0 mol L-1 HCl or 1.0 mol L-1 NaOH before sterilization in an autoclave. If necessary, the pH was readjusted with the addition of 1.0 mol L-1 HCl or 1.0 mol L-1 NaOH. In addition, the isolates were inoculated in MRS broth containing ox bile (Sigma) adjusted to bile 0, bile 0.1%, bile 0.3%, bile 0.6% and bile 1.0% (w/v). All tests were subjected to a background microtitration in 96-well sterile plates (TPP; testplatte Zellkultur, Trasadingen, Switzerland). Each well was filled with 150 µL of MRS and 10 µL of culture grown in MRS broth (OD 650 nm) at 37 °C. The optical density was read hourly for 12 h at 650 nm using the plate microtiter reading (TPP). Cultures grown in MRS broth without bile served as controls. The experiments were carried out in triplicate (TODOROV et al., 2011, adapted).
β-Galactosidase activity
The β-galactosidase activities of the isolates were determined using paper discs impregnated with o-nitrophenyl-β-D-galactopyranoside (ONPG Discs, Fluka) according to the supplier’s instructions. The isolates were reactivated for 24 h at 37 °C, streaked on Petri dishes containing MRS agar and incubated at 37 °C for 48 h in aerobic conditions. One colony of each culture was transferred to and mixed in a tube containing 0.1 mL of sterile saline, and the ONPG disc was then added. The tubes were incubated at 37 °C and were observed for 6 h, at 1 h intervals. The appearance of a yellow color due to the release of the chromogenic compound o-nitrophenyl indicates the presence of β-galactosidase (SANTOS et al., 2014).
Resistance to drugs and antibiotics
The disc-diffusion test (Oxoid) was used to evaluate the susceptibility and resistance of the selected P. pentosaceus cultures to antibiotics with different modes of action: penicillin G (10 µg/disc), vancomycin (30 µg/disc), tetracycline (30 µg/disc), chloramphenicol (30 µg/disc), sulfa-trimethoprim (23.75 µg + 1.25 µg) and oxacillin (1 µg/disc). MRS agar plates containing 105-106 CFU mL-1 of the P. pentosaceus cultures were prepared after culturing in MRS broth at 37 °C for 48 h. The discs were applied to the plates and subsequently incubated at 37 °C for 24 h. The diameters of the inhibition zones around the discs were measured (mm) with the help of a ruler, to evaluate the sensitivities (CHARTERIS et al., 1998). Resistance and sensitivity levels were measured according to the Clinical and Laboratory Standards Institute standard (CLSI, 2011).
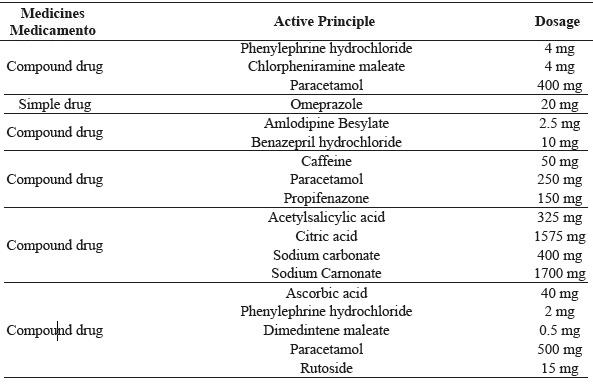
Isolates were tested for resistance to 34 drugs (Table 1), which were purchased in a pharmaceutical establishment and dissolved in sterile water to achieve the desired concentration. The cultures were inoculated separately into 10 mL of MRS broth, incubated at 37 °C for 24 h and mixed in MRS agar (2.0%, w/v; Oxoid) in order to reach a cell density of 106 CFU mL-1. After solidifying the agar, one drop of each drug was added to the surfaces of the plates and incubated at 37 °C for 24 h. The plates were examined for the presence of zones of inhibition around the drop of the medicament.
| MedicinesMedicamento | Active Principle | Dosage |
|---|---|---|
| Simple drug | Dipyrone | 500 mg |
| Compound drug | Scopolamine butylbromide | 10 mg |
| Dipyrone | 250 mg | |
| Simple drug | Allergosan | 25 mg |
| Simple drug | Vitamin and Mineral Supplement A to Z | - |
| Simple drug | Polyvitamin | - |
| Compound drug | Caffeine | 50 mg |
| Orphenadrine citrate | 35 mg | |
| Dipyrone | 300 mg | |
| Compound drug | Pyridoxine hydrochloride | 10 mg |
| Dimenhydrate | 50 mg | |
| Simple drug | Bisacodyl | 5 mg |
| Simple drug | Acetylcysteine | 600 mg |
| Generic | Aceclofenac | 100 mg |
| Generic | Acetylcysteine | 100 mg |
| Generic | Mefenamic acid | 500 mg |
| Generic | Aminophylline | 100 mg |
| Generic | Amoxicillin trihydrate | 500 mg |
| Generic | Bromopride | 10 mg |
| Generic | Cinarizine | 75 mg |
| Generic | Propanolol hydrochloride | 40 mg |
| Generic | Diclofenac potassium | 50 mg |
| Generic | Ketotifen fumarate (Syrup) | 0.2 mg mL-1 |
| Generic | Lisinopril | 20 mg |
| Generic | Loratadina | 10 mg |
| Generic | Paracetamol | 500 mg |
| Generic | Paracetamol | 750 mg |
| Simple drug | Peumus boldus (Boldo) | 67 mg |
| Simple drug | Ibuprofen | 600 mg |
| Simple drug | Prenoxdiazine hydrochloride | 100 mg |
| Simple drug | Loperamide | 2 mg |
| Simple drug | Nimesulide | 100 mg |
| Compound drug | Phenylephrine hydrochloride | 4 mg |
| Chlorpheniramine maleate | 4 mg | |
| Paracetamol | 400 mg | |
| Simple drug | Omeprazole | 20 mg |
| Compound drug | Amlodipine Besylate | 2.5 mg |
| Benazepril hydrochloride | 10 mg | |
| Compound drug | Caffeine | 50 mg |
| Paracetamol | 250 mg | |
| Propifenazone | 150 mg | |
| Compound drug | Acetylsalicylic acid | 325 mg |
| Citric acid | 1575 mg | |
| Sodium carbonate | 400 mg | |
| Sodium Carnonate | 1700 mg | |
| Compound drug | Ascorbic acid | 40 mg |
| Phenylephrine hydrochloride | 2 mg | |
| Dimedintene maleate | 0.5 mg | |
| Paracetamol | 500 mg | |
| Rutoside | 15 mg |
Acidifying activity
The isolates were revived in MRS broth with overnight incubation at 37 °C. For the acidifying-activity test, tubes containing 10 mL of sterile reconstituted skim milk (RSM) (10% w/v, Oxoid) were inoculated (1.0% w/v) with revived isolates and incubated at 37 °C. The pH was measured after 6 and 24 h with a pH meter (Microprocessor pHMeter 213, Hanna Instruments, Ireland). The data were expressed as the mean of the duplicate of the analysis (DAL BELLO et al., 2012).
Extracellular proteolytic activity
Extracellular proteolytic activity was determined as described by Franciosi et al. (2009) and Dal Bello et al. (2012). One microliter of each revived culture was inoculated onto the agar surface consisting of 10% (w/v) RSM (Oxoid) and 2.0% (w/v) agar (Oxoid) and incubated at 37 °C for 4 days. The proteolytic activity was indicated by the presence of a translucid zone around the colonies.
Exopolysaccharide formation (EPS)
Aliquots (1.0% w/v) of each culture were inoculated into 10 mL of sterile RSM (10% w/v) and incubated at 37 °C for 24 h. The production of EPS from lactose was determined qualitatively by measuring the degree of yarn formation, as described by Cogan (1996) and Dal Bello et al. (2012).
Growth ability at different salt concentration
Aliquots (100 µL) of the strains were inoculated into 350 µL of MRS broth previously distributed in microtiter plates with different concentrations of NaCl: 0.0, 4.0, 6.0 and 10.0% (w/v). The ability of the cultures to grow at different concentrations was evaluated after 24 h at 37 °C by measuring the optical density (OD650nm) using a spectrophotometer (Spectrophotometer DU® 640 UV/Vis - Beckman Coulter, CA, USA). The multiplication-potential of the cultures in the different concentrations of NaCl was evaluated from the difference in the ODs recorded in the two readings (DAL BELLO et al., 2012).
Autolytic activity
Cell autolysis was measured as described by Mora et al. (2003) and Dal Bello et al. (2012), with modifications. Cultures were grown in MRS broth (Oxoid) for 24 h at 37 °C to achieve an OD650nm of 0.8-1.0. Cells were washed in tripotassium phosphate buffer solution (50 mmol-1, pH 6.5) (Sigma-Aldrich, Ireland), resuspended in the same buffer and incubated at 37 °C. The degree of autolysis was expressed as the percentage of the decrease in OD650 nm after 24 h.
Diacetyl production
The production of diacetyl was determined as described by King (1948) and Dal Bello et al. (2012). Revived cultures (1.0% w/v) were inoculated into 10 mL of sterile RSM and incubated at 37 °C for 24 h. One milliliter of each cell suspension was combined with 0.5 mL α-naphthol (1.0% w/v) and KOH (16.0% w/v) (Sigma-Aldrich) and incubated at 37 °C for 10 min. The production of diacetyl was indicated by the formation of a red ring at the top of the tube.
Experimental design
A completely randomized design was used, with three replicates. Initially, the analysis of variance was performed at 5% probability, and the Scott-Knott mean test was used for the variables hydrophobicity, auto-aggregation and co-aggregation. Regarding the development of the isolates, as a function of pH and bile concentration over time, descriptive statistics were used. Assistat Version 7.7 beta software was used for the analysis of variance and Scott-Knott’s mean test, and SigmaPlot 10.0 software was used for plotting the graphs.
Results and Discussion
Safety potential
All P. pentosaceus isolates (Ac1Pd, Ac3Pd, Ac4Pd, Ac5Pd, Ac7Pd, and Ac22Pd) showed negative results for the expression of the virulence factors tested (gelatinase, hemolysis, lipase, and DNAse) by phenotypic methods. This result is interesting given the use of LAB as starter cultures; the use of isolates requires caution due to their possible virulence since some types of LAB can harbor virulence genes and, therefore, express them in food products, which would present a risk to the consumers (SANTOS et al., 2014). LAB may also be resistant to different antibiotics due to the presence of genes conferring antibiotic resistance, increasing their virulence potential (PERIN et al., 2014). It should be emphasized that there is a possibility of the horizontal transfer of these genes between LAB and other bacteria, which further increases the concerns for the food industry. These genes are usually located in transferable plasmids (SANTOS et al., 2014).
All the isolates presented negative results for the production of biogenic amines (BA). As cheeses represent a good environment for the production and accumulation of BA produced by starter and non-starter strains, high concentrations of BA in dairy products indicate a low microbiological quality of milk and a lack of hygienic manufacturing practices. In addition, there are several toxicological problems resulting from the ingestion of foods containing relatively high levels of specific histamine and tyramine substrate enzymes of the microorganisms (PERIN et al., 2017). The results obtained, which proved the absence of virulence factors and biogenic amines, demonstrate the safety of all P. pentosaceus isolates evaluated.
In the study by Perin et al. (2017), no culture was able to produce putrescine, histamine, or cadaverine, but 12 cultures (five Lactococcus spp. and seven Enterococcus spp.) were able to form tyramine. In the study by Bover-Cid and Holzapfel (1999), tyramine was the main amine formed by the investigated LAB cultures, but the isolates of P. pentosaceus and P. acidilactici had no decarboxylase activity.
Probiotic potential
There were significant differences in hydrophobicities between P. pentosaceus isolates. The isolates of P. pentosaceus that presented higher values of hydrophobicity were Ac4Pd and Ac22Pd (Figure 1). It should be noted that all isolates presented hydrophobicity values below 10%. The determination of microbial adhesion to N-hexadecane is considered a valid qualitative approach to estimate the ability to adhere to epithelial cells (KIELY; OLSON, 2000). Adherence in the gastrointestinal tract is a determinant factor for colonization of and residency in the host and is considered to be one of the main selection criteria for probiotic potential. The hydrophobicity of the cells is related to the ability of the microorganisms to adhere to intestinal epithelial cells. In general, bacterial cells with high hydrophobicities have strong interactions with mucosal cells that can make them more competitive, providing a strong anchorage of the bacterial cells to the gastrointestinal tract (TODOROV et al., 2011; SANTOS et al., 2014).
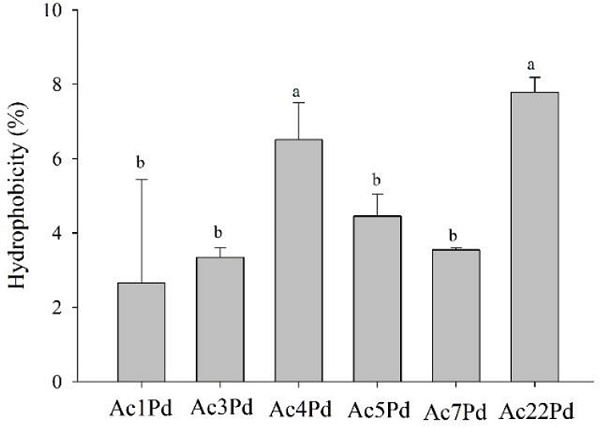
Figure 1
Hydrophobicity of P. pentosaceus isolates. Mean values followed by the same letter did not differ statistically according to the Scott-Knott test at 5% probability.
However, this feature is not a prerequisite for strong adherence since this process involves several mechanisms. Even cells that exhibit low hydrophobicity can adhere effectively to the cell surface (TODOROV et al., 2007). According to Bautista-Gallego et al. (2013) of a total of 109 isolates from spontaneously fermented green olive pickles, most of their Lactobacillus isolates showed hydrophobicities between 0.0% and 5.0%. In contrast, Vidhyasagar and Jeevaratnam (2013) obtained six isolates of P. pentosaceus from Idli (typical food from India) that showed high hydrophobicity with n-hexadecane, especially the VJ49 and VJ13 isolates, which presented 79% and 77%, respectively. The hydrophobicity of the strain should always be observed because it varies genetically among species as well as between strains of the same species (TODOROV et al., 2011).
With regards to auto-aggregation (Figure 2), a significant difference (p<0.05) was observed between P. pentosaceus isolates and the indicator microorganisms. It should be noted that P. pentosaceus isolates presented values above 82%. Higher auto-aggregation was observed in the Ac7Pd, Ac3Pd and Ac4Pd isolates, with mean values equal to 92.2, 90.9 and 89.9%, respectively. On the other hand, the indicator microorganism, L. monocytogenes ATCC 7644, presented the auto-aggregation at a value of 41.5%.
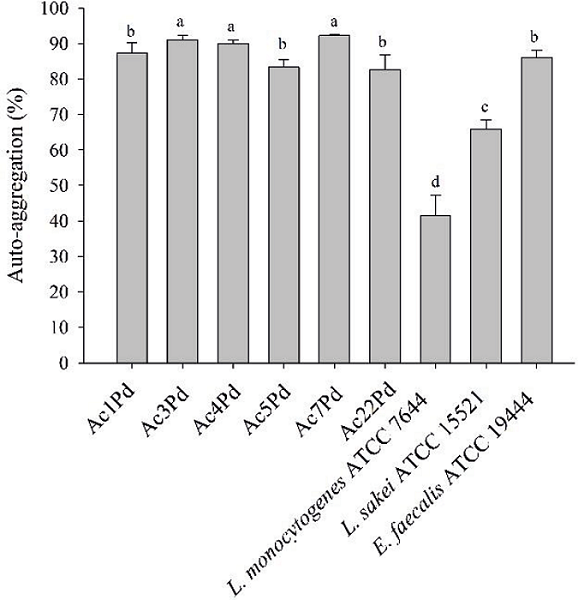
Figure 2
Auto-aggregation of isolates of P. pentosaceus, L. monocytogenes ATCC 7644, L. sakei ATCC 15521 and E. faecalis ATCC 19444. Mean values followed by the same letter did not differ statistically according to the Scott-Knott test at 5% of probability.
Aggregation between bacterial cells is considered an important criterion in adhesion and biofilm formation on several surfaces, such as on the mucosa of the GIT. Cellular aggregation, in addition to facilitating the transient colonization of GIT, contributes to the persistence of beneficial microorganisms in that GIT and its effects on health and may also prevent pathogen adhesion (SANTOS et al., 2014). In the study by Vidhyasagar and Jeevaratnam (2013), P. pentosaceus presented a similar result, in which its isolate VJ41 showed a maximum aggregation of 89%, which corroborates the present study. According to Todorov et al. (2011), auto-aggregation is strain-specific and may vary within the same taxonomic group, as observed in a study conducted by the authors, in which the percentages found were 7.2% for L. fermentum and 12.1% for E. faecium.
Figure 3 shows the average values of co-aggregation between P. pentosaceus isolates and different indicator microorganisms. There was no significant difference (p>0.05) when co-aggregation was analyzed between P. pentosaceus isolates with L. monocytogenes ATCC 7644 (Figure 3A) and L. sakei ATCC 15521 (Figure 3B). It is noteworthy that the co-aggregation percentages of P. pentosaceus isolates with L. monocytogenes ATCC 7644 and L. sakei ATCC 15521 were higher than 60% and 83%, respectively. With regard to co-aggregation with E. faecalis ATCC 19444 (Figure 3C), the Ac7Pd isolate allowed for greater co-aggregation with an equivalent value of 93.3%, according to the Scott-Knott’s test at 5% probability.
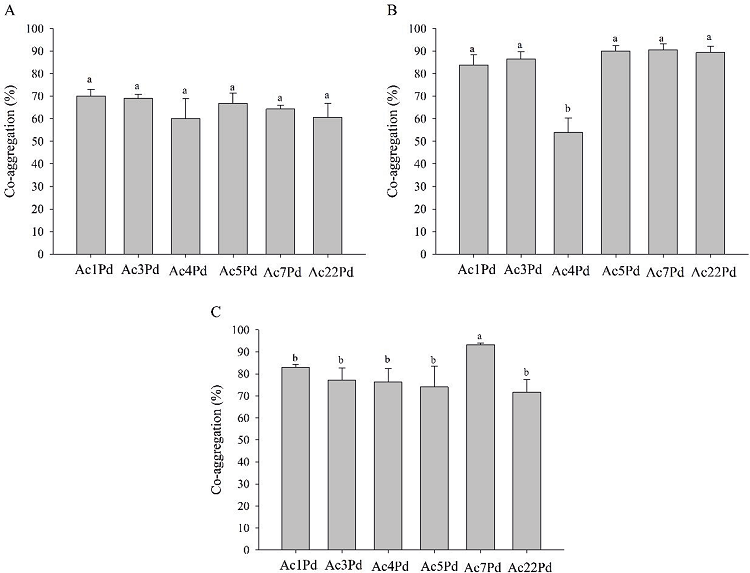
Figure 3
Co-aggregation of P. pentosaceus isolates with L. monocytogenes ATCC 7644 (A), L. sakei ATCC 15521 (B) and E. faecalis ATCC 19444 (C). Mean values followed by the same letter did not differ statistically according to the Scott-Knott test at 5% probability.
Aggregation between cells (auto-aggregation) and with genetically different organisms (co-aggregation) are probiotic criteria that are essential for maintaining a bacterial population in the intestine. The co-aggregation potential of LAB can be considered a positive trait since it can exert antagonistic effects against pathogens, such as L. monocytogenes, through mechanisms involving the production of antimicrobial compounds, such as organics, hydrogen peroxide and bacteriocins (SANTOS et al., 2014). Low levels of co-aggregation with pathogens can play an important role in preventing the formation of biofilms and thus eliminate pathogens from the GIT. Co-aggregation between LAB and other cells, especially L. monocytogenes, can be considered a positive feature since it is one of the steps necessary for the elimination of undesirable GIT cultures. In contrast, higher levels of co-aggregation with L. sakei ATCC 15521, a non-pathogenic microorganism, may facilitate the presence of probiotics in the human GIT (TODOROV et al., 2011).
According to a study by Todorov et al. (2011), L. curvatus, L. fermentum, L. delbrueckii, P. acidilactici and E. faecium strains were found to have low levels of co-aggregation with L. monocytogenes and E. faecalis ATCC 19443, but higher levels of co-aggregation with L. Sakei ATCC 15521. Vidhyasagar and Jeevaratnam (2013) reported that their P. pentosaceus isolates (VJ13 culture) aggregated with L. monocytogenes and E. coli in 90% and 81%, respectively.
All P. pentosaceus isolates presented intense growth in a pH range of 6 to 10, especially after 8 h (Figure 4). However, the growth of P. pentosaceus isolates in a pH range between 3 and 5, and at pH 12, was unimpressive. The ability to survive through the GIT and to persist temporarily in the intestinal environment of the host are the main functional characteristics of a probiotic culture (SANTOS et al., 2014). Thus, due to the conditions in the stomach, where the pH is approximately 2.0, it is essential to select probiotics that have a high tolerance to acidic conditions (BAUTISTA-GALLEGO et al., 2013; PATEL et al., 2014). Similarly, bile salts secreted into the small intestine also present a challenge to bacterial survival in the GIT, so it is important to select probiotic isolates with resistance to high concentrations of bile so that they may develop better in the small intestine (TODOROV et al., 2011; BAUTISTA-GALLEGO et al., 2013).
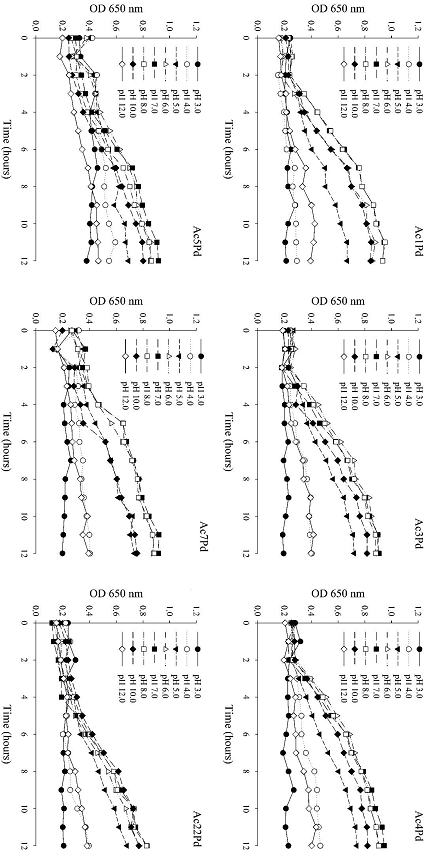
Figure 4
Growth of six P. pentosaceus isolates in MRS broth, as a function of pH over 12 h.
In the study by Brink et al. (2006) the development of L. plantarum, L. casei, L. salivarius, L. curvatus and P. pentosaceus was suppressed in the first 10 h of incubation at pH 3.0 but was more vigorous at pH values between 5.0 and 6.5. An in vitro assay conducted by Argyri et al. (2013), demonstrated that five L. plantarum isolates showed high resistance to low pH, and a similar result was obtained for L. casei Shirota and L. ramnosus GG reference cultures, which are known to maintain viability at pH values 2.5 to 4.0.
P. pentosaceus isolates lost their capacity to grow when bile concentrations of 0.1%, 0.3%, 0.6% and 1% were tested (Figure 5). Thus, such conditions are inadequate for the development of these microorganisms. However, all isolates developed satisfactorily in the absence of bile over 12 h. In the study by Patel et al. (2014), none of the Lactobacillus spp., Weissella spp. and Pediococcus spp. cultures showed growth ability in the presence of 0.3% bile. However, in another study, all L. rhamnosus and L. plantarum strains resisted the simulated enteric fluid, containing 0.5% of bile salts, well (SANTOS et al., 2014).
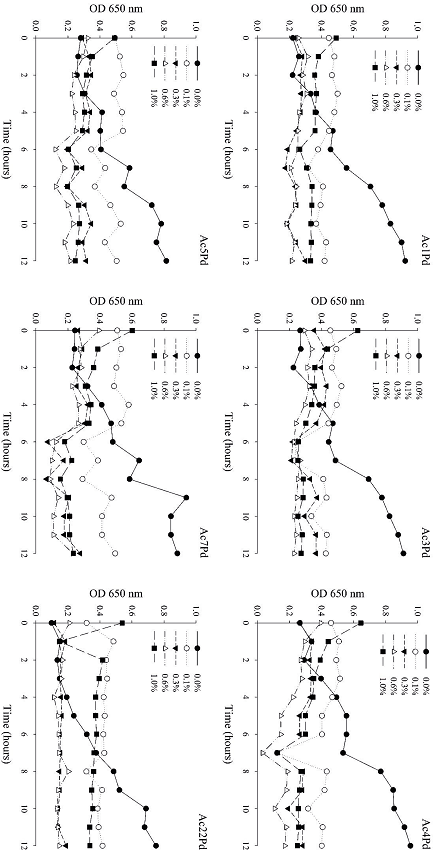
Figure 5
Growth of six P. pentosaceus isolates in MRS broth, as a function of bile concentration over 12 h.
It should be noted that the pH used for the testing of probiotic potentials is selected very judiciously and is not a commonly found pH in the human stomach, which can be altered by diet. Therefore, this value only ensures the isolation of acid-tolerant probiotics. It should be noted that probiotic cultures can be buffered by food or other molecular-matrix supports after consumption and, therefore, will not be exposed to the pH of the stomach. Similarly, some food components can protect and promote bacterial resistance to bile salts (ARGYRI et al., 2013; BAUTISTA-GALLEGO et al., 2013).
The isolates tested did not present β-galactosidase production. This activity is considered a positive trait in probiotic and starter isolates, conferring an advantage for growth and proliferation in milk. This activity is essential for the removal of sugars from milk, allowing lactose-intolerant individuals to consume the product (VIDHYASAGAR; JEEVARATNAM, 2013). In a study by Santos et al. (2014), a L. rhamnosus isolate also presented negative results in the β-galactosidase-production test. In the Vidhyasagar and Jeevaratnam (2013) analysis, among P. pentosaceus isolates, the VJ49 strain produced 5990 Miller units of β-galactosidase, which according to the authors, was considered the maximum value.
In the present study, all P. pentosaceus isolates were resistant to oxacillin, sulfatrimethoprim and vancomycin. However, they were sensitive to chloramphenicol and tetracycline. With regards to penicillin G, the Ac1Pd isolate was sensitive, Ac5Pd was resistant, and the others had intermediate sensitivity (Table 2). Antimicrobial drugs can reduce the effects of LAB and still make them resistant. Another concern related to antibiotic resistance is the horizontal transfer of resistance genes from LAB to the other bacteria present in the human GIT (DICKS et al., 2009; TODOROV et al., 2011). Resistance may be intrinsic to a bacterial genus or species but may also be obtained through the exchange of genetic material, mutations, and the incorporation of new genes. According to Ivanova et al. (2013), since the pediococci are resistant to vancomycin they can be confused with enterococci in routine analyses, and this could lead to the under-reporting of Pediococcus spp. in clinical settings.
| Isolated | Ac1Pd | Ac3Pd | Ac4Pd | Ac5Pd | Ac7Pd | Ac22Pd |
|---|---|---|---|---|---|---|
| Tetracycline | S | S | S | S | S | S |
| Oxaciclin | R | R | R | R | R | R |
| Sulfatrimetoprim | R | R | R | R | R | R |
| Vancomycin | R | R | R | R | R | R |
| Chloramphenicol | S | S | S | S | S | S |
| Penicillin G | S | I | I | R | I | I |
In the study by Hummel et al. (2007), two Pediococcus cultures (P. acidilactici and P. pentosaceus) showed low resistance to tetracycline, but P. pentosaceus was resistant to ampicillin. Santos et al. (2014) reported that all the cultures tested showed susceptibility to penicillin G, chloramphenicol and tetracycline; however, they showed resistance to cotrimoxazole (sulfatrimethoprim). This demonstrates the variability of resistance among members of the same genus and species. It is known that the commercial introduction of probiotics-containing antibiotic-resistant cultures can have negative consequences, for example, when resistance is transferred to intestinal pathogens. However, sometimes resistance to antibiotics can be considered intrinsic or natural because it is chromosomally encoded and thus non-transmissible (ARGYRI et al., 2013).
It is also important to determine the effects of various drugs and the survival of probiotic cultures. All evaluated cultures were resistant to most of the drugs tested except for amoxicillin trihydrate (500 mg) and ibuprofen (600 mg). Because the CLSI protocol is basically for antimicrobial agents, only the formation of the inhibition halo was observed for other types of drugs. All isolates showed sensitivity to amoxicillin trihydrate. There are studies, such as that by de Botes et al. (2008), that have reported that L. casei Shirota is sensitive to several commercial antibiotics, including amoxicillin. Ibuprofen, a nonsteroidal anti-inflammatory drug (NSAID) that, as reported in another study, inhibits the growth of L. lactis subsp. lactis (TODOROV et al., 2007). Interference from other NSAIDs, such as diclofenac, has also been reported in other studies. Thus, the action of these substances in the GIT can affect the probiotic potential of LAB strains.
Technological potential
The results obtained for Technological Potential characterization (acidification capacity, exopolysaccharide and diacetyl production, and extracellular proteolytic activity) are shown in Table 3. In this study, no isolate was able to significantly reduce the pH after 6 h, but acidification was observed after 24 h. Several studies corroborate these results, as they have also shown that most laboratory isolates are initially slow in acid production (FRANCIOSI et al., 2009; MORANDI et al., 2011; DAL BELLO et al., 2012; PICON et al., 2015). According to Ribeiro et al. (2013), microorganisms with low acidification capacities can be used as coadjuvant organisms based on their other characteristics. A rapid decrease in pH is very important during cheese production and contributes to its texture and flavor, preventing the growth of undesirable and pathogenic microorganisms (PERIN et al., 2017). In addition, it may contribute not only to the preservation of organoleptic properties but also to the extension of the shelf-lives of fermented foods (BERESFORD et al., 2001; SALVUCCI et al., 2016). According to Beresford et al. (2001), starter cultures should be capable of producing sufficient acid to reduce the pH of the milk to 5.3 or less after 6 h at 30 to 37 °C.
| Isolates | Capacity of Acidification | EPS Production* | Production of Diacetyl* | Extracellular Proteolytic Activity Autolysis | Autolysis** | Growth at different concentrations NaCl*** | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| pH in RSM | |||||||||||
| 0h | 6h | 24h | 24 h | 0% | 4% | 6% | 10% | ||||
| Ac1Pd | 6.45 | 6.19 | 4.68 | - | + | - | 22.55 | +++ | +++ | +++ | + |
| Ac3Pd | 6.45 | 6.26 | 5.26 | - | + | - | 29.27 | +++ | +++ | +++ | + |
| Ac4Pd | 6.45 | 6.23 | 5.10 | - | + | - | 26.70 | +++ | ++ | +++ | + |
| Ac5Pd | 6.45 | 6.21 | 4.64 | - | + | - | 23.48 | +++ | ++ | +++ | + |
| Ac7Pd | 6.45 | 6.22 | 4.73 | - | + | - | 25.90 | +++ | ++ | +++ | + |
| Ac22Pd | 6.45 | 6.17 | 4.95 | - | + | - | 23.23 | +++ | +++ | +++ | + |
None of the isolates studied showed production of extracellular polysaccharides _ EPS; the same results were obtained by Franciosi et al. (2009), Dal Bello et al. (2012) and Perin et al. (2017). The generation of EPS is considered an important resource for cultures used in dairy environments, mainly in yogurt production. During this process, the EPS aid the rheological characteristics of the product, for example, by increasing the viscosity and improving water binding. Thus, they create a smooth and creamy texture and can also confer beneficial effects on health (FRANCIOSI et al., 2009; PERIN et al., 2017).
All P. pentosaceus isolates were able to produce diacetyl. In the study by Perin et al. (2017), L. lactis subsp. lactis and Enterococcus spp. isolates were also able to produce diacetyl. A similar result was observed in a study by Dal Bello et al. (2012), in which 7 of the 20 L. lactis isolates tested produced high diacetyl concentrations. Diacetyl is a volatile and flavored compound generated as a final product of the conversion of citrate to pyruvate, which contributes to flavor in, for instance, butter, buttermilk and other dairy products (LEROY; DE VUYST, 2004; PERIN et al., 2017), and can still influence the texture and aroma of fermented products (RIBEIRO et al., 2013).
Diacetyl production is considered to be a strain-dependent trait because not all LAB have the ability to metabolize citrate. Therefore, this behavior may differ between species and cultures (PERIN et al., 2017). In addition, it is known that homofermentative LAB transform sugar into lactic acid, resulting in rapid acidification of the raw material and production of other metabolites, such as ethanol, diacetyl, acetate, acetaldehyde, etc., which improve the taste, texture, storage and safety of the final products. Therefore, strategies aimed at direct modification of the equilibrium have led to the overproduction of the desired metabolites mentioned above. Examples include increased diacetyl production by L. lactis subsp. lactis biovar. diacetylactis in buttermilk by redirection of the pyruvate catabolism (LEROY; DE VUYST, 2004).
None of the isolates showed extracellular proteolytic activity. Proteolytic activity is an essential property for starter cultures since it contributes to the taste and texture of the product (RIBEIRO et al., 2013). During cheese maturation, various aromatic compounds are generated due to the action of milk enzymes such as proteolytic enzymes which are derived from the LAB present in the cheese. The proteolytic activity of LAB consists of the activities of proteinases and peptidases, which hydrolyze casein into small peptides and free amino acids, which generate most of the aromatic precursors that influence the flavors in yogurt and cheeses. Thus, proteolytic enzymes play an important role in the fermentation of dairy products (PERIN et al., 2017).
However, it is preferable to not use strains with high proteolytic activity for the production of some types of cheese because excessive proteolysis can produce bitter-tasting peptides and other undesirable compounds or excessive casein hydrolysis can result in an undesirable texture of the cheese (PERIN et al., 2017). In the study by Perin et al. (2017)Enterococcus and L. lactis subsp. lactis isolates did not present extracellular proteolytic activity. Similar results were found for some Enterococcus and L. lactis isolates in other studies (DAL BELLO et al., 2012; RIBEIRO et al., 2013). The results of proteolytic-activity studies by different authors should, however, be cautiously analyzed due to differences in the methods used (PICON et al., 2015).
The percentages of autolysis after 24 h presented by P. pentosaceus cultures are shown in Table 3. After this period, the autolytic abilities of the isolates ranged from 22% to 29%. Autolysis by LAB favors the access of intracellular peptidases to their substrates but also accelerates the maturation of cheeses. Intact bacterial cells are required for physiological activities, such as fermentation, oxygen withdrawal from lactose, and for a series of flavor reactions. However, the main consequence of cheese autolysis is the acceleration of the peptidolytic reactions (PERIN et al., 2017). Compared to other studies, such as that by Perin et al. (2017), the autolysis by L. lactic subsp. lactis was 16.45% after 24 h. In the study by Dal Bello et al. (2012), the autolytic capacity for all L. lactis cultures ranged from 20 to 40% after 24 h. Research by Mora et al. (2003) has demonstrated that the autolytic property is widely distributed among P. acidilactici and P. pentosaceus species. Although autolysis of starter cultures is beneficial, undesirable consequences, such as acid production and insufficient removal of residual lactose may occur if autolysis is too rapid. In practice, a balance in autolysis is required for the ideal maturation of certain cheeses and for flavor development. Furthermore, isolates with low autolytic activity may be desirable since lower production of bacteriocins or other antagonistic activities may lead to them surviving longer in the product and continuing to exhibit such activities (PERIN et al., 2017).
The tolerance of P. pentosaceus isolates for the different concentrations of sodium chloride (4.0%, 6.0% and 10.0%) is shown in Table 3. Growth in MRS medium without NaCl and with 4.0% NaCl was not significantly different (p <0.05). A similar result was found in several studies in which the majority of the Lactococcus spp. and Enterococcus spp. isolates tested were able to grow at a NaCl concentration of 4.0% (DAL BELLO et al., 2012; PERIN et al., 2017). Growth of all isolates in 6.0% NaCl was observed. In the study by Salvucci et al. (2016), approximately 88% of the evaluated Lactobacillus spp. and Enterococcus spp. strains were able to grow in the presence of 6.5% NaCl, but the authors did not observe the growth of P. pentosaceus at this concentration. In the study by Dal Bello et al. (2012), none of the isolates of Lactococcus spp. were able to grow at concentrations above 6%. In the study by Perin et al. (2017), the addition of 6.0 and 10.0% (w/v) NaCl resulted in lower growth of Lactococcus spp. and Enterococcus spp. when compared to the control group. At a 10.0% NaCl concentration, the growth of the isolates was significantly lower when compared to the others. Ribeiro et al. (2013), reported that only one isolate of E. faecalis showed low growth at the highest salt concentration tested (10.0%), while the other Enterococcus isolates analyzed were resistant. The ability of LAB to adapt and survive in different salt concentrations is important, especially in the production of certain types of cheeses and in other foods that contain high concentrations of salt. In addition, growth in media with high salt content is desirable for starter cultures since NaCl is one of the most important additives for food preservation (SALVUCCI et al., 2016).
The results showed that the isolates of P. pentosaceus from sheep milk are safe with regard to the analyzed factors; however, they have limited application as probiotics because they do not present essential characteristics such as survival in an acidic environment. In addition, when evaluated for their technological potential, the isolates did not present results that would be necessary for their use in the production of milk derivatives.
Regarding the drugs, it is important to point out that the indiscriminate use of antimicrobials can lead to resistance of multiple drugs, so it is often necessary to find alternative treatment to fight infections and thus also decrease the resistance of the LAB. The present research provides evidence that sheep milk can be considered as an appropriate source of LAB cultures. It should also be noted that the data generated were obtained from in vitro tests and if the tests were performed in vivo, the results could be different. As future prospects of research, it is intended to initiate studies of genetic improvement, in order to modify characteristics of the isolates so that they present conditions of use as probiotics.
Acknowledgments
To Capes for financial support.
References
ALVIM, L. B. Identificação molecular e seleção de bactérias láticas com potencial probiótico isoladas de diferentes mucosas de suínos. 2011. Dissertação (Mestrado em Biologia Geral) - Universidade Federal de Minas Gerais, Belo Horizonte.
ARGYRI, A. A.; ZOUMPOPOULOU, G.; KARATZAS, K. G.; TSAKALIDOU, E.; NYCHAS, G. E.; PANAGOU, E. Z.; TASSOU, C. C. Selection of potential probiotic lactic acid bacteria from fermented olives by in vitro tests. Food Microbiology, London, v. 33, n. 2, p. 282-291, 2013.
BARBOSA, J.; GIBBS, P. A.; TEIXEIRA, P. Virulence factors among enterococci isolated from traditional fermented meat products produced in the North of Portugal. Food Control, Oxford, v. 21, n. 5, p. 651-656, 2010.
BAUTISTA-GALLEGO, J.; ARROYO-LÓPEZ, F. N.; RANTSIOU, K.; JIMÉNEZ-DÍAZ, R.; GARRIDO-FERNÁNDEZ, A.; COCOLIN, L. Screening of lactic acid bacteria isolated from fermented table olives with probiotic potential. Food Research International, Amsterdam, v. 50, n. 1, p. 135-142, 2013.
BERESFORD, T. P.; FITZSIMONS, N. A.; BRENNAN, N. L.; COGAN, T. M. Recent advances in cheese microbiology. International Dairy Journal, Oxford, v. 11, n. 4, p. 259-274, 2001.
BOTES, M.; VAN REENEN, C. A.; DICKS, L. M. T. Evaluation of Enterococcus mundtii ST4SA and Lactobacillus plantarum 423 as probiotics by using a gastro-intestinal model with infant milk formulations as substrate. International Journal of Food Microbiology, London, v. 128, n. 2, p. 362-370, 2008.
BOVER-CID, S.; HOLZAPFEL, W. H. Improved screening procedure for biogenic amine production by lactic acid bacteria. International Journal of Food Microbiology, London, v. 53, n. 1, p. 33-41, 1999.
BRINK, M.; TODOROV, S. D.; MARTIN, J. H.; SENEKAL, M.; DICKS, L. M. T. The effect of prebiotics on production of antimicrobial compounds, resistance to growth at low pH and in the presence of bile, and adhesion of probiotic cells to intestinal mucus. International Journal of Food Microbiology, London, v. 100, n. 4, p. 813-820, 2006.
CHARTERIS, W. P.; KELLY, P. M.; MORELLI, L.; COLLINS, J. K. Antibiotic susceptibility of potentially probiotic Lactobacillus species. Journal of Food Protection, Des Moines, v. 61, n. 12, p. 1636-1643, 1998.
CLINICAL AND LABORATORY STANDARDS INSTITUTE - CLSI. Performance standards for antimicrobial susceptibility testing; twenty-first informational supplement. Pennsylvania: Clinical and Laboratory Standards Institute, 2011.
COGAN, T. M. History and taxonomy of starter cultures. In: COGAN, T. M.; ACCOLAS, J. P. (Ed.). Dairy starter cultures. New York: John Wiley and Sons Inc., 1996. p. 1-23.
DAL BELLO, B.; COCOLIN, L.; ZEPPA, G.; DES FIELD, D.; COTTER, P.; HILL, C. Technological characterization of bacteriocin producing Lactococcus lactis strains employed to control Listeria monocytogenes in Cottage cheese. International Journal of Food Microbiology, London, v. 153, n. 1-2, p. 58-65, 2012.
DICKS, L. M. T.; TODOROV, S. D.; FRANCO, B. D. G. M. Current status of antibiotic resistance in lactic acid bacteria. In: BONILLA, A. R.; MUNIZ, K. P. (Ed.). Antibiotic resistance: causes and risk factors, mechanisms and alternatives. Pharmacology - research, safety testing and regulation. New York: Nova Publisher, 2009. p. 379-425.
FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS. WORLD HEALTH ORGANIZATION - FAO/WHO. Organization Guidelines for Evaluation of Probiotics in Food. London, Ontario, Canada, April 30 and May 1, 2002. 11 p.
FRANCIOSI, E.; SETTANNI, L.; CAVAZZA, A.; POZNANSKI, E. Biodiversity and technological potential of wild lactic acid bacteria from raw cows’ milk. International Dairy Journal, Oxford, v. 19, n. 1, p. 3-11, 2009.
HUMMEL, A. S.; HERTEL, C.; HOLZAPFEL, W. H.; FRANZ, C. M. A. P. Antibiotic resistances of starter and probiotic strains of lactic acid bacteria. Applied and Environmental Microbiology, Washington, v. 73, n. 3, p. 730-739, 2007.
IVANOVA, I.; ILIEV, I.; HAERTLÉ, T.; CHOBERT, J. Food or medicine? Future of lactic acid bacteria. In: ______. Lactic acid bacteria-Most important genuses. Sofia University: Diagalprint Printing House, 2013. p. 26-30.
KIELY, L. J.; OLSON, N. F. The physicochemical surface characteristics of Lactobacillus casei. Food Microbiology, London, v. 17, n. 3, p. 277-291, 2000.
KING, N. Modifications of the Voges-Proskauer test for rapid colorimetric determination of acetylmethylcarbinol plus diacetyl in butter cultures. Dairy Industries, Gravesend, v. 13, p. 860-866, 1948.
KOCH, A. C. C. Características físico-químicas e microbiológicas do leite de ovelha e atividade antagonista de sua microbiota lática. 2014. Tese (Doutorado em Ciências Animais) - Universidade de Brasília, Brasília.
LEROY, F.; DE VUYST, L. Lactic acid bacteria as functional starter cultures for the food fermentation industry. Trends in Food Science & Technology, London, v. 15, n. 2, p. 67-78, 2004.
JOOSTEN, H. M. L. J.; NORTHOLD, M. D. Detection, growth and amine-producing capacity of lactobacilli in cheese. Applied and Environmental Microbiology, Washington, v. 55, n. 9, p. 2356-2359, 1989.
MEIRA, S. M. M. Potencial probiótico de bactérias lácticas e atividades biológicas de leite e queijos de ovelha. 2011. Dissertação (Mestrado em Ciência e Tecnologia de Alimentos) - Universidade Federal Rio Grande do Sul, Porto Alegre.
MORA, D.; MUSACCHIO, F.; FORTINA, M. G.; SENINI, L.; MANACHINI, P. L. Autolytic activity and pediocin-induced lysis in Pediococcus acidilactici and Pediococcus pentosaceus strains. Journal of Applied Microbiology, London, v. 94, n. 4, p. 561-570, 2003.
MORANDI, S.; BRASCA, M.; LODI, R. Technological, phenotypic and genotypic characterization of wild lactic acid bacteria involved in the production of Bitto PDO Italian cheese. Dairy Science & Technology, Paris, v. 9, n. 3, p. 341-359, 2011.
PANCHENIAK, E. F. R. Isolamento, seleção, caracterização bioquímica e molecular para produção e avaliação do potencial probiótico de Lactobacillus reuteri LPB P01-001 em suínos. 2005. Tese (Doutorado em Tecnologia de Alimentos) - Universidade Federal do Paraná, Curitiba.
PATEL, A.; PRAJAPATI, J. B.; HOLST, O.; LJUNGH, A. Determining probiotic potential of exopolysaccharide producing lactic acid bacteria isolated from vegetables and traditional Indian fermented food products. Food Bioscience, Amsterdam, v. 5, p. 27-33, 2014.
PERIN, L. M.; BELVISO, S.; DAL BELLO, B.; NERO, L. A.; COCOLIN, L. Technological properties and biogenic amines production by bacteriocinogenic Lactococci and Enterococci strains isolated from raw goat milk. Journal of Food Protection, Des Moines, v. 80, n. 1, p. 151-157, 2017.
PERIN, L. M.; MIRANDA, R. O.; TODOROV, S. D.; FRANCO, B. D. G. M.; NERO, L. A. Virulence, antibiotic resistance and biogenic amines of bacteriocinogenic lactococci and enterococci isolated from goat milk. International Journal of Food Microbiology, London, v. 185, p. 121-126, 2014.
PICON, A.; GARDE, S.; ÁVILA, M.; NUÑEZ, M. Microbiota dynamics and lactic acid bacteria biodiversity in raw goat milk cheeses. International Dairy Journal, Oxford, v. 58, p. 14-22, 2015.
RIBEIRO, S. C.; COELHO, M. C.; TODOROV, S. D.; FRANCO, B. D. G. M.; DAPKEVICIUS, M. L. E.; SILVA, C. C. G. Technological properties of bacteriocin-producing lactic acid bacteria isolated from Pico cheese an artisanal cow’s milk cheese. Journal of Applied Mcrobiology, London, v. 116. n. 3, p. 573-585, 2013.
SALVUCCI, E.; LEBLANC, J.; PEREZ, G. Technological properties of lactic acid bacteria isolated from raw cereal material. LWT - Food Science and Technology, Amsterdam, v. 70, p. 185-191, 2016.
SANTOS, K. M. O.; VIEIRA, A. D. S.; BURITI, F. C. A.; FRANCO, B. D. G. M.; BRUNO, L. M.; BORGES, M. F.; TODOROV, S. D.; NASCIMENTO, J. C. F.; ROCHA, C. R. C.; MELO, M. E. S.; SOUZA LOPES, A. C. Artisanal Coalho cheeses as source of beneficial Lactobacillus plantarum and Lactobacillus rhamnosus strains. Dairy Science & Technology, Paris, v. 95, n. 2, p. 209-230, 2014.
TODOROV, S. D.; BOTES, M.; DANOVA, S. T.; DICKS, L. M. T. Probiotic properties of Lactococcus lactis ssp. lactis HV219, isolated from human vaginal secretions. Journal of Applied Microbiology, London, v. 103, n. 3, p. 629-639, 2007.
TODOROV, S. D.; BOTES, M.; GUIGAS, C.; SCHILLINGER, U.; WIID, I.; WACHSMAN, M. B.; HOLZAPFEL, W. H.; DICKS, L. M. T. Boza, a natural source of probiotic lactic acid bacteria. Journal of Applied Microbiology, London, v. 104, n. 2, p. 465-477, 2008.
TODOROV, S. D.; FURTADO, D. N.; SAAD, S. M. I.; TOME, E.; FRANCO, B. D. G. M. Potential beneficial properties of bacteriocin-producing lactic acid bacteria isolated from smoked salmon. Journal of Applied Microbiology, London, v. 110, n. 4, p. 971-986, 2011.
VIDHYASAGAR, V.; JEEVARATNAM, K. Evaluation of Pediococcus pentosaceus strains isolated from Idly batter for probiotic properties in vitro. Journal of Functional Foods, St. John’s, v. 5, n. 1, p. 235-243, 2013.
Author notes
*Author for correspondence