ARTICLES
Sodium bicarbonate and HEPES as buffer components for cooling the semen of pony stallions
Bicarbonato de sódio e HEPES como tampões para o resfrigeração de sêmen de pôneis
Sodium bicarbonate and HEPES as buffer components for cooling the semen of pony stallions
Semina: Ciências Agrárias, vol. 39, no. 2, pp. 631-642, 2018
Universidade Estadual de Londrina
Received: 20 July 2017
Accepted: 08 January 2017
Abstract: The composition of semen diluents can modify its viability during cooling. The buffering effects of HEPES and sodium bicarbonate were evaluated considering the pH and sperm viability. The semen of seven adult Brazilian ponies was evaluated before and after cooling at 5ºC for 24 h and 48 h. A non-buffered skim milk powder extender (C) and the same extender buffered with sodium bicarbonate (SB) and HEPES (H) were used. After dilution, semen (three ejaculates/pony) was centrifuged and the seminal plasma discarded. Sperm was then diluted with SB, H or C and its concentration adjusted to 50 x 106 sptz/mL. Progressive motility evaluated after dilution showed similar results with all extenders (71.42% (SB), 74.28% (H), and 74.52% (C)). Sperm motility was evaluated 24 h and 48 h after cooling for SB (44.76% and 25.23%), H (51.42% and 38.09%) and C (54.05% and 41.66%, respectively). Plasma membrane integrity was similar after exposure to the three extenders (62.71% (SB), 68.76% (H), and 69.23% (C)). Mitochondrial activity was higher in SB immediately after dilution (SB= 1.05nm, H= 0.81nm, C= 0.79nm), and after 24 h (0.83nm (SB), 0.73nm (H) and 0.64nm (C)). After 48 h, the mitochondrial activity decreased to 0.72nm (SB), 0.69nm (H), and 0.63nm (C) (P > 0.05). The pH, osmolarity and pH after 48 h of cooling of the diluted semen were higher in SB (8; 382; 7.9), intermediate in H (7.5; 362; 7.32) and lower in C (7.16; 350; 7.07). Lipid peroxidation and its induction were similar in all groups. Data were analyzed by analysis of variance (ANOVA), and Duncan's test was used to evaluate the significant differences (P < 0.05). Sodium bicarbonate reduced the progressive motility and increased the semen pH. The extender C was considered more appropriate for immediate use in artificial insemination. The non-buffered and HEPES-buffered extenders were considered appropriate for the cooling of equine semen for 48 h at 5°C.
Key words: Dilution, Equine, Extender, Motility, pH, Spermatozoa.
Resumo: A composição dos diluentes de sêmen pode modificar sua viabilidade durante o processo de resfriamento. O efeito de tamponamento do HEPES e Bicarbonato de Sódio foi avaliado considerando o pH e a viabilidade espermática. Sete pôneis brasileiros adultos tiveram seu sêmen avaliado antes e após a refrigeração a 5°C durante 24 h e 48 h. Um diluente de leite em pó desnatado não tamponado (C) e um diluente tamponado com bicarbonato de sódio (SB) ou HEPES (H) foram utilizados. Após a diluição, o sêmen (três ejaculados/ pônei) foi centrifugado e o sobrenadante foi descartado. O sêmen foi então diluído com SB, H ou C e a concentração ajustada para 50 x 106 espermatozoides/mL. A motilidade progressiva avaliada após a diluição apresentou resultados similares para todos os diluentes (71,42% (SB), 74,28% (H), 74,52% (C)). A motilidade espermática foi avaliada 24 h e 48 h após o resfriamento, respectivamente, para SB (44,76%, 25,23%), H (51,42%, 38,09%) e C (54,05%, 41,66%). A integridade da membrana plasmática foi semelhante após a exposição aos três diluentes (62,71% (SB), 68,76% (H), 69,23% (C)). A atividade mitocondrial após a diluição foi maior em SB (SB = 1.05nm, H = 0.81nm, C = 0.79nm) e após 24 h foi 0.83nm (SB), 0.73nm (H) e 0.64nm (C). A atividade mitocondrial após 48 h diminuiu para 0.72nm (SB), 0.69nm (H) e 0.63nm (C) (P > 0.05). O pH, a osmolaridade e o pH do sêmen diluído após as 48 h de refrigeração foram maiores em SB (8; 382; 7,9), intermediário em H (7,5; 362; 7,32) e menor em C (7,16; 350; 7,07). A peroxidação lipídica e sua indução foram semelhantes em todos os grupos. As médias foram avaliadas através de análise de variância (ANOVA) e o Teste Duncan foi utilizado para analisar as diferenças significativas (P < 0.05). O bicarbonato de sódio reduziu a motilidade progressiva e aumentou o pH do sêmen. O diluente C foi considerado mais adequado para uso imediato na inseminação artificial. O diluente não tamponado e tampado com HEPES foram considerados apropriados para o resfriamento de sêmen de equino durante 48 h a 5°C.
Palavras-chave: Diluente, Diluição, Espermatozoides, Equino, Motilidade, pH.
Introduction
One of the extenders used for cooling equine semen is skim milk, glucose and antibiotic (KENNEY et al., 1975; BERGERON; MANJUNATH, 2006; FLOREZ-RODRIGUEZ et al., 2014). Extenders are known for their practicality and for the protection afforded to the sperm during cooling (AURICH, 2008), but the longevity of spermatozoa seems to vary according to the extender composition (PAPA et al., 2014; MARTINS et al., 2016). Most diluents are based on skim milk or egg yolk. The protection mechanisms involved are not well elucidated. In milk, the protective effect seems to be related to casein micelles (BATELLIER et al., 2001; LUSIGNAN et al., 2011), while in egg yolk, low density lipoproteins are important (BERGERON; MANJUNATH, 2006; PUGLIESI et al., 2012).
The storage temperature, the rate of cooling, exposure to oxygen, the presence of bacteria, the type of extender, the presence and type of antibiotic added to the extender, and the concentration of seminal plasma (LOVE et al., 2005; KARESKOSKI, KATILA, 2008) during cooling can all influence sperm viability (ORTEGA-FERRUSOLA et al., 2011; LOVE et al., 2012; VIDAMENT et al., 2012; RODRIGUEZ et al., 2014). Besides the preservation of motility, the extender should also guarantee and preserve the integrity of spermatozoa membranes (CRESPILHO et al., 2012), their mitochondria (POMMER et al., 2002), and sperm chromatin.
Substances used in semen extenders must, at the very least, meet the following requirements: a similar or slightly higher osmolarity than the semen (300-350 mOsm/L), and slightly acidic (6.8) to neutral (7.0) pH. Essential components include substances that stabilize the pH and control microorganism growth, a readily available source of energy, and ingredients that stabilize membranes and maintain the metabolic function of sperm, as well as neutralizing metabolic substances (MARTINS et al., 2016). Ideally, extender components should not interfere with the results of semen evaluation (AURICH, 2011); however, some components in extenders can interfere in computer-assisted semen analysis (FLOREZ-RODRIGUEZ et al., 2014), even reducing the efficiency of sperm sexing by flow cytometry (MORRIS, 2005).
During storage, sperm and contaminating bacteria generally produce metabolites that may reduce the pH of the extender, reducing both sperm metabolism and motility (YÁNIZ et al., 2011). These alterations can cause sperm death due to drastic changes in pH, justifying the addition of buffers to the extender (GRAHAM et al., 1972; HOLT, 2000).
Kenney et al. (1975) previously described sodium bicarbonate as a buffer. Later, HEPES (N-2-hydroxyethyl piperazine-N'-2-ethanesulfonic acid), an organic buffer, was the subject of studies on maturation media for embryo production (GOOD et al., 1996; DOWNS; MASTROPOLO, 1997), and its use is currently still indicated (SWAIN, 2010; PARRISH, 2014).
Ponies and miniature horses are used both as working animals and as riding animals for children. In addition, data generated by research work using ponies can also be considered relevant to horses. Preliminary studies in our laboratory have demonstrated that sodium bicarbonate can have a detrimental effect on cooled semen. The aim of this study was to evaluate an alternative to sodium bicarbonate as an extender for cooled semen.
Multiple extenders have been developed in the last few years to preserve equine semen in storage (GIBB, AITKEN, 2016). While buffers have been compared, there is need for further investigation in this area. The objective of this study was to evaluate the effect of an equine semen extender containing skim milk (powder), without buffer and buffered with sodium bicarbonate or HEPES, on sperm viability and pH in fresh semen, and in semen cooled for 24 h or 48 h at 5°C.
Materials and Methods
Locale of the experiment and animal management
The study was performed from October through December (springtime in the southern hemisphere) during the 2014 breeding season at the Laboratory of Animal Embryology of the Federal University of Santa Maria (UFSM), Santa Maria/RS, Brazil (29°41'-03° South, 53°48'-25° West). Seven pony stallions of the Brazilian breed, aged 9 to 13 years, were fed once daily with oat grains and alfalfa, with water freely available. They were located in a paddock and kept under the same management conditions. The Committee of Ethics in Animal Experimentation from the Federal University of Santa Maria approved all of the procedures performed in these experiments, according to protocol number 065/2013.
Semen collection and analyses
Semen of all ponies was routinely collected with an artificial vagina (Hannover Model; Minitube, Tiefenbach, Germany) twice a week, before and during the experiment. Three ejaculates per pony were used in the study. Ejaculates were filtered with sterile gauze and analyzed macroscopically to assess their volume, color and appearance. Evaluation of sperm concentration was performed in a Neubauer hemocytometer counting chamber. The total volume of the ejaculate was split into three fractions and diluted 1:1 (semen:extender) in three different extenders (Table 1), followed by centrifugation at 450 x g for 10 min to remove seminal plasma. The pH and osmolarity of the extenders was not fixed, in order to evaluate the effects of the buffers in the diluent, which already contains other chemical components. The concentrations of the buffers were defined after preliminary testing in order to generate a pH between 7 and 8. The supernatant was removed, the pellet was resuspended in the extender, and the concentration was adjusted to 50 x 106 sptz/mL. Unless otherwise mentioned, the chemical components were purchased from Sigma Aldrich (St. Louis, MO, USA).
| Extender/Component | Sodium bicarbonate | HEPES | Control |
|---|---|---|---|
| Skim milk powder | 2.4 g | 2.4 g | 2.4 g |
| Glucose | 4.9 g | 4.9 g | 4.9 g |
| Sodium bicarbonate | 0.150 g | - | - |
| HEPES | - | 0.100 g | - |
| Ultra-pure water | 95 mL | 95 mL | 95 mL |
One sample from each group of ponies was used for analysis of motility, pH, membrane function (hypoosmotic - HOST), mitochondrial activity (MTT), lipid peroxidation through thiobarbituric acid-reactive substances (TBARS), and peroxidation induced by iron sulfate. The remaining semen samples were refrigerated at 5°C. New evaluations were performed after refrigeration for 24 h and 48 h. Before assessment, the samples were warmed for 5 minutes in a dry bath adjusted to 37°C.
Semen evaluation
Evaluation of sperm motility was performed subjectively using an optical phase contrast microscope (CBRA, 2013). The pH of the fresh diluted semen, and of semen that had been stored refrigerated for 24 h and 48 h, was determined (pH Meter Tec-2, Tecnal).
Membrane functionality test (HOST)
The functional membrane integrity was evaluated using the hypoosmotic test, in a 2:1 dilution (distilled water:semen), adjusting the osmolarity to around 100 mOsm / kg H2O-1. The samples were incubated at 37°C for eight minutes according a protocol adjusted for horses (LAGARES et al., 2000). Subsequently, analysis was conducted using a phase contrast microscope (400x), on a slide and under a cover glass, counting 100 sperm per sample.
Mitochondrial activity assessed by reduction of tetrazolium (MTT assay)
The MTT reduction assay depends on the ability of metabolically active cells to reduce the tetrazolium salt (3 [4,5-dimethylthiazol-2-y1] -2,5-diphenyl tetrazolium bromide) to formazan (AZIZ et al., 2005). MTT is a yellow salt that is reduced to formazan by the action of dehydrogenases of metabolically active cells, forming insoluble purple crystals in water. These crystals can be solubilized with organic solvents. The intensity of the purple color can be measured spectrophotometrically and has a direct relationship with the metabolic activity of the cells in the samples.
To assess the mitochondrial activity, a semen sample in each extender (sodium bicarbonate, HEPES, or unbuffered extender) was centrifuged at 600 x g for 10 min, the supernatant discarded, and the pellet resuspended in the appropriate extender, adjusting the concentration to 100 x 106 sptz/mL. Two aliquots of 200 µL at 100 x 106 sptz/mL were placed in 2 mL microcentrifuge tubes. Tetrazolium solution, 20 µL (5 mg/mL thiazolyl blue tetrazolium bromide; M2128, Sigma-Aldrich) in saline PBS, was added to the tubes, which were then incubated for 30 min in a water bath at 37°C.
The microcentrifuge tubes were centrifuged at 12000 x g for 5 min after the addition of 200 µL of a 0.04N HCl-isopropanol solution. The mitochondrial metabolic activity was estimated by UV visible spectrophotometry of the supernatant at a wavelength of 540 nm. The reference sample used was the skim milk extender solution containing glucose, with the addition of tetrazolium and 0.04N HCl-isopropanol. The aliquots from each stallion were analyzed in triplicate.
Assay of lipid peroxidation by thiobarbituric acid (TBARS)
A sample of each tested extender (containing sodium bicarbonate, HEPES, or unbuffered) containing 10 x 106 sperm adjusted to 80 µL was used for the lipid peroxidation test by thiobarbituric acid (TBARS).
The TBARS concentration in the semen samples was determined in microcentrifuge tubes containing 0.2 mL of thiobarbituric acid (TBA), 0.2 mL of acetic acid solution, 0.04 mL of ultra-pure water, 0.08 mL of the sample, and 0.08 mL of sodium dodecyl sulfate (SDS). The tubes containing this mixture were incubated at 100 °C for 2 h, and then cooled in a container with ice. The samples were centrifuged at 800 x g for 5 min, and the absorbances of the supernatants were measured in a spectrophotometer. The assay was performed in triplicate and the absorbance of the samples was measured with a 532 nm filter. As reference samples, extenders without semen samples were also evaluated. All analyses were performed in a UV-VIS spectrophotometer (Perkin Elmer - Lambda 25) at 25 °C.
Induction of lipid peroxidation by iron sulfate
To induce lipid peroxidation, samples of each tested extender containing 10 x 106 sperm adjusted to 80 µL were added to 0.02 mL of ferrous sulfate and 0.02 mL of ascorbic acid. These samples were incubated in a water bath at 37 °C for one hour. Then, 0.2 mL of trichloroacetic acid was added to each sample. All samples were centrifuged at 3000 x g for 10 min. The supernatant was transferred to a microcentrifuge tube and 0.2 ml of thiobarbituric acid (TBA) was added. The specimens were incubated at 100°C for one hour and cooled. The samples were then centrifuged following the protocol used for TBARS. This procedure is known as induced or catalyzed lipid peroxidation by iron. It is designed to measure the full potential of the sample to generate the radical in question, and is related to lipid peroxidation.
Statistical analysis
All statistical analyses were performed using SAS 9.2 software (SAS Institute Inc., Cary, NC, USA). Data were investigated using analysis of variance (ANOVA) and comparisons among the extenders containing sodium bicarbonate, HEPES, or without buffer at 0 h, 24 h, and 48 h were performed using Duncan test. Graphs were prepared using Graph Prism software. Differences among groups were considered statistically significant when the probability (P-value) was less than or equal to 0.05.
Results
The progressive motility of the semen immediately after dilution was similar (P = 0.1367) in the extenders containing sodium bicarbonate (SB) and HEPES (H) and in the non-buffered skim milk powder extender (C = control). Semen in the extender containing SB showed a lower percentage of spermatozoa exhibiting progressive motility compared with the HEPES (H), and C extenders after 24 h (P = 0.0083) and 48 h (P < 0.0001) of refrigeration. The integrity of the sperm membrane (HOST) was similar among the SB, H, and C extenders immediately after dilution, and after 24 h and 48 h of cooling (Table 2; Figure1).
| DilutionPreservation | Sodium bicarbonate | HEPES | Control | P Value |
|---|---|---|---|---|
| Progressive motility | ||||
| Fresh | 71.42 ± 5.94 Aa | 74.28 ± 5.31Aa | 74.52 ± 5.22 Aa | > 0.05 |
| 24 h | 44.76 ± 12.59 Bb | 51.42 ± 6.91Ba | 54.04 ± 8.47 Ba | < 0.0083 |
| 48 h | 25.23 ± 12.59 Cb | 38.09 ± 9.67 Ca | 41.66 ± 6.58 Ca | < 0.0001 |
| Hypoosmotic | ||||
| Fresh | 62.71 ± 11Aa | 68.76 ± 9.75Aa | 69.23 ± 9.5Aa | > 0.05 |
| 24 h | 35.95 ± 13.08Ba | 42.47 ± 13.67 Ba | 39.47 ± 12.1Ba | > 0.05 |
| 48 h | 27.76 ± 11.96Ca | 33.09 ± 11.97 Ca | 33.09 ± 13.6Ba | > 0.05 |
| MTT(nm) | ||||
| Fresh | 1.05 ± 0.35Aa | 0.818 ± 0.23Ab | 0.79 ± 0.28Ab | 0.011 |
| 24 h | 0.83 ± 0.32Ba | 0.73 ± 0.26Aab | 0.64 ± 0.18Ab | 0.0685 |
| 48 h | 0.72 ± 0.3Ba | 0.69 ± 0.31Aa | 0.63 ± 0.27Aa | > 0.05 |
| pH | ||||
| Fresh | 7.82 ± 0.1Aa | 7.46 ± 0.14 Ab | 7.08 ± 0.23 Ac | < 0.0001 |
| 24 h | 7.85 ± 0.18Aa | 7.43 ± 0.14 Ab | 7.07 ± 0.12 Ac | < 0.0001 |
| 48 h | 7.9 ± 0.15Aa | 7.32 ± 0.21Bb | 7.07 ± 0.15Ac | < 0.0001 |
A, B, C values with different uppercase superscripts differ within a column (P < 0.05).
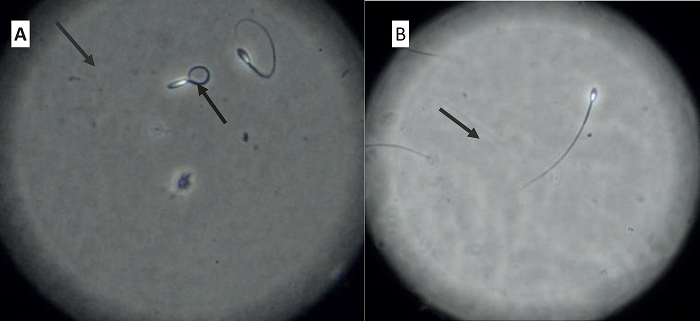
Figure 1
Spermatozoa reactive (A), and non-reactive (B), to the hypoosmotic test.
After dilution, the highest mitochondrial activity was observed in semen in extender SB, with lower activity in the extenders with H and without buffer (P = 0.01). After 24 h of cooling, the activity in extenders SB and H was similar, while semen in extender C showed lower activity. Mitochondrial activity of semen samples after 48 h of cooling was similar among the three extenders (P = 0.585). Comparisons with the absorbance of the reference sample solution (skim milk extender solution with glucose) showed that extender SB had a higher absorbance than the other extenders.
The average pH of the pony semen was 7.52 ± 0.04. Assessing the pH results, it is interesting to note that the extender containing sodium bicarbonate had the highest pH value before cooling, after 24 h refrigeration, and also after 48 h of cooling. Intermediate pH values were observed in diluent H. Lower pH values were observed in diluent C (Figure 2), which remained stable after up to 48 h of cooling (P < 0.0001). The pH of the extenders (Figure 3) immediately after preparation, and after 24 h and 48 h of cooling, behaved similarly to pH of the semen diluted in the same extenders (extender SB = highest pH value; extender H = intermediate pH, and solvent C = lower pH) (P < 0.0001).
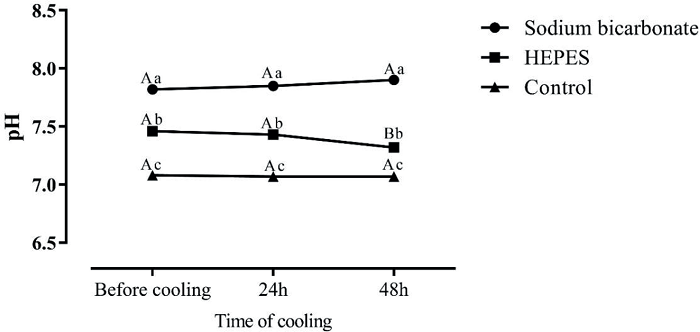
Figure 2
The mean pH values of diluted semen of Brazilian ponies in extender with sodium bicarbonate, HEPES or control (without buffer) before cooling or cooled at 5°C for 24 or 48 h. Capital letters indicate differences (P < 0.0288) between time points for each extender.
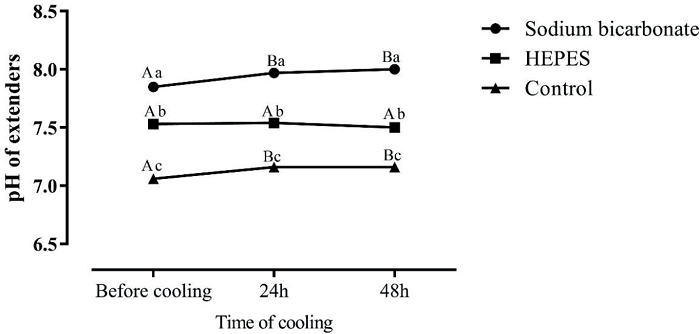
Figure 3
Average pH values of Brazilian pony semen in extenders containing sodium bicarbonate, HEPES or control (without buffer) after dilution, or after 24 h or 48 h of cooling at 5°C (P = 0.0278). Capital letters indicate differences (P < 0.05) between time points for each extender.
The results indicate a higher osmolarity for the sodium bicarbonate extender, intermediate osmolarity of the HEPES extender, and a lower osmolarity for the unbuffered extender (P < 0.0001) after 48 h of cooling (Figure 4).
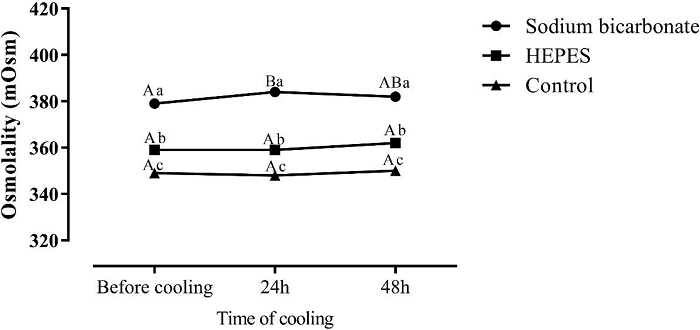
Figure 4
Mean osmolarity values semen in extenders containing sodium bicarbonate, HEPES or control (without buffer) immediately after dilution, or after refrigeration at 5°C for 24 or 48 h (P < 0.0001). Capital letters indicate differences (P < 0.05) between time points for each extender.
Evaluation of lipid peroxidation and induction of peroxidation immediately after sample preparation showed similar values (P > 0.05) among the extenders SB (0.16 ± 0.28 and 0.09 ± 0.12), H (0.19 ± 0.22 and 0.06 ± 0.08) and Control (0.24 ± 0.18 and 0.06 ± 0.11, respectively) in both tests. Lipid peroxidation and induction of peroxidation after 48 h of cooling was also similar among the extenders.
Discussion
The use of HEPES in unbuffered skim milk extender solution containing glucose showed better results than did sodium bicarbonate in all evaluations, except for mitochondrial activity before cooling. This observation can be explained because sperm hyperactivation occurs spontaneously in most spermatozoa incubated in media containing bicarbonate and Ca2+ (HINRICHS; LOUX, 2012; MACÍAS-GARCÍA et al., 2015).
Assessment of sperm motility allowed us to state that there was no difference effect between sperm diluted in HEPES buffered extender (H) and unbuffered extender (C). This suggests that the addition of the buffer did not affect the motility of the spermatozoa at 5°C; nonetheless, its addition was not necessary.
In the absence of sodium bicarbonate, sperm do not lose their membrane integrity and sperm capacitation is minimal, but when exposed to sodium bicarbonate, sperm becomes vulnerable to membrane rupture and cell death (RATHI et al., 2001). Furthermore, sodium bicarbonate leads to changes in the structure of the plasma membrane lipid in equine sperm (GADELLA; HARRISON, 2000). Induction of the acrosome reaction was observed when sperm were incubated in medium containing sodium bicarbonate; in the absence of bicarbonate, the acrosome reaction was not observed (RATHI et al., 2001). Interestingly, Foster et al. (2011) reported that observed damage to sperm membranes is significant only after 48 h of cooling.
The pH of equine semen ranges between 6.8 and 7.0 but is influenced by several factors. Extenders should have a buffering effect on the pH of semen diluted to balance the production of metabolic substances from sperm or bacteria (AURICH, 2011). Most milk-based extenders have a pH of about 6.6 (PICKETT; AMANN, 1987). Here, addition of buffers to the extender led to changes in the pH value. When assessing only the pH of the extender during 48 h of cooling, our evaluation highlights that the HEPES extender showed the least variation between 0 and 48 hours; nevertheless, in extender C values closer to physiological conditions were observed. It must be emphasized here that the necessity of adding a buffer to semen should be carefully examined, since the absence of the buffer in the diluent did not impair sperm membrane integrity or motility. Furthermore, the addition of buffers to the extenders caused an increase in the pH of the medium using either sodium bicarbonate or HEPES.
Osmotic stress reduces the viability and mitochondrial membrane potential of sperm, and also increases superoxide anion generation and alters protein tyrosine phosphorylation in equine spermatozoa (BALL; VO, 2001; POMMER et al., 2002; BURNAUGH et al., 2010). Equine spermatozoa are more prone to hypotonic- rather than hypertonic-induced damage (POMMER et al., 2002; BURNAUGH et al., 2010). Meanwhile, the incubation of spermatozoa in nonisosmotic media resulted in an increase in oxygen free radical generation (BURNAUGH et al., 2010). Stallion spermatozoa are more sensitive to osmotic shocks than to a given anisosmotic environment, and changes in osmolarity are more deleterious to mitochondria than to the plasma membrane (GARCÍA et al., 2012). The osmolarity of semen extenders ranges between 250 and 400 mOsm/L (KATILA, 1997). In our study, the osmolarity was higher (379 ± 4.18 before cooling, 384 ± 4.18 in 24 h after cooling, and 382 ± 2.67 mOsm/L in 48 h after cooling) in the extender containing sodium bicarbonate.
It has been proposed that sodium bicarbonate activates adenylate cyclase, either directly or indirectly, causing Ca2+ increases (CHRISTENSEN et al., 1996) and, thus, raising intracellular concentrations of cAMP. This, in turn, induces sperm hyperactivation (WHITE; AITKEN, 1989) that may be associated with increased degradation of the tetrazolium salt, which would explain why the extender with sodium bicarbonate showed the highest value for mitochondrial activity before cooling. After cooling and following the decline in motility, the mitochondrial activity in extender SB also declined, becoming similar to the other diluents after cooling for 48 h.
In milk-based extenders, native phosphocaseinate and β-lactoglobulin seem to be responsible for maintaining sperm motility (BATELLIER et al., 1997; MANJUNATH, 2012). The usefulness of the skim milk powder extender may be attributed to the antioxidant capacity of the diluted semen, which would explain the similarity of lipid peroxidation among the extenders, as the three diluents contain the same amount of skim milk. It has been reported (KANKOFER et al., 2005) that dilution of semen only with milk diluent results in a significant increase in the antioxidant capacity of the diluted semen. The addition of skim milk to equine semen extender for cooling at 5°C seems to play a crucial role in sperm preservation (KENNEY et al., 1975; FLOREZ-RODRIGUEZ et al., 2014), but to our knowledge there is little research about semen extenders in ponies (NEVES et al., 2006).
Conclusion
In ponies, skim milk powder extender with or without HEPES buffer provides superior sperm viability compared with the same extender buffered with sodium bicarbonate. Sodium bicarbonate in pony semen extender reduces sperm motility, and causes increased pH. There is no need to buffer the skim milk extender for routine use. HEPES extender and the diluent without buffer are both appropriate for cooling pony sperm for up to 48 h at 5°C. The addition of buffers results in increased extender pH, resulting in reduced viability. An extender without buffer can be used for cooling pony semen at 5°C for 24 h or 48 h.
References
AURICH, C. Recent advances in cooled-semen technology. Animal Reproduction Science, Amsterdam, v. 107, n. 3-4, p. 268-275, 2008.
AURICH, C. Semen extenders for cooled semen (Europe). In: MCKINNON, A. O.; SQUIRES, E. L.; VAALA, W. E.; VARNER, D. D.. Equine reproduction 2th ed. Chichester: Wiley-Blackwell, 2011. v. 1, chap. 131, p. 1336-1340.
AZIZ, D. M.; AHLSWEDE, L.; ENBERGS, H. Application of MTT reduction assay to evaluate equine sperm viability. Theriogenology, New York, v. 64, n. 6, p. 1350-1356, 2005.
BALL, B. A.; VO, A. Osmotic tolerance of equine spermatozoa and the effects of soluble cryoprotectants on equine sperm motility viability and mitochondrial membrane potential. Journal of Andrology, Philadelphia, v. 22, n. 6, p. 1061-1069, 2001.
BATELLIER, F.; MAGISTRINI, M.; FAUQUANTZ, J.; PALMER, E. Effect of milk fractions on survival of equine spermatozoa. Theriogenology, New York, v. 48, n. 3, p. 391-410, 1997.
BATELLIER, F.; VIDAMENT, M.; FAUQUANT, J.; DUCHAMP, G.; ARNAUD, G.; YVON, J. M.; MAGISTRINI, M. Advances in cooled semen technology. Animal Reproduction Science, Amsterdam, v. 68, n. 3-4, p. 181-190, 2001.
BERGERON, A.; MANJUNATH, P. New insights towards understanding the mechanisms of sperm protection by egg yolk and milk. Molecular Reproduction and Development, Rhode Island, v. 73, n. 10, p. 1338-1344, 2006.
BURNAUGH, L.; BALL, B. A.; SABEUR, K.; THOMAS, A. D.; MEYERS, S. A. Osmotic stress stimulates generation of superoxide anion by spermatozoa in horses. Animal Reproduction Science, Amsterdam, v. 117, n. 3-4, p. 249-260, 2010.
CHRISTENSEN, P.; WHITFIELD, C. H.; PARKINSON, T. J. In vitro induction of acrosome reaction in stallion spermatozoa by heparin and A23187. Theriogenology, New York, v. 45, n. 6, p. 1201-1210, 1996.
COLÉGIO BRASILEIRO DE REPRODUÇÃO ANIMAL - CBRA. Manual para exame andrológico e avaliação de sêmen animal. 3. ed. Belo Horizonte: CBRA, 2013. 104 p.
CRESPILHO, A. M.; SÁ FILHO, M. F.; DELL'AQUA JUNIOR, J. A.; NICHI, M.; MONTEIRO, G. A.; AVANZI, B. R.; MARTINS, A.; PAPA, F. O. Comparison of in vitro and in vivo fertilizing potential of bovine semen frozen in egg yolk or new lecithin based extenders. Livestock Science, Amsterdam, v. 149, n. 1-2, p. 1-6, 2012.
DOWNS, S. M.; MASTROPOLO, A. M. Culture conditions affect meiotic regulation in cumulus cell-enclosed mouse oocyte. Molecular Reproduction and Development, Rhode Island, v. 46, n. 4, p. 551-566, 1997.
FLOREZ-RODRIGUEZ, S. A.; PAES DE ARRUDA, R.; BIANCHI-ALVES, M. R.; AFFONSO, F. J.; CARVALHO, H. F.; LEMES, K. M.; LANÇONI, R.; CESAR DE ANDRADE, A. F.; CARVALHO CELEGHINI, E. C. Morphofunctional characterization of cooled sperm with different extenders to use in equine-assisted reproduction. Journal of Equine Veterinary Science, New York, v. 34, n. 7, p. 911-917, 2014.
FOSTER, M. L.; VARNER, D. D.; HINRICHS, K.; TEAGUE, S.; LACAZE, K.; BLANCHARD, T. L. Agreement between measures of total motility and membrane integrity in stallion sperm. Theriogenology, New York, v. 75, n. 8, p. 1499-1505, 2011.
GADELLA, B. M.; HARRISON, R. A. The capacitating agent bicarbonate induces protein kinase A-dependent changes in phospholipid transbilayer behavior in the plasma membrane. Development, Washington, v. 127, n. 11, p. 2407-2420, 2000.
GARCÍA, B. M.; MORAN, A. M.; FERNÁNDEZ, L. G.; FERRUSOLA, C. O.; RODRIGUEZ, A. M.; BOLAÑOS, J. M. G.; SILVA, C. M. da; MARTÍNEZ, H. R.; TAPIA, J. A.; PEÑA, F. J. The mitochondria of stallion spermatozoa are more sensitive than the plasmalemma to osmotic-induced stress: role of c-Jun N-terminal Kinase (JNK) pathway. Journal of Andrology, Philadelphia, v. 33, n. 1, p. 105-113, 2012.
GIBB, Z.; AITKEN, R. J. Recent developments in stallion semen preservation. Journal of Equine Veterinary Science, New York, v. 43, p. S29-S36, 2016.
GOOD, N. E.; WINGET, G. D.; WINTER, W.; CONNOLLY, T. N.; IZAWA, S.; SINGH, R. M. M. Hydrogen ion buffers for biological research. Biochemistry, Washington, v. 5, n. 2, p. 467-477, 1996.
GRAHAM, E. F.; CRABO, B.; BROWN, K. I. Effects of some zwitterion buffers on the freezing and storage of spermatozoa: 1. Bull. Journal of Dairy Science, Champaign, v. 55, n. 3, p. 372-378, 1972.
HINRICHS, K.; LOUX, S. C. Hyperactivated sperm motility: Are equine sperm different? Journal of Equine Veterinary Science, New York, v. 32, n. 8, p. 441-444, 2012.
HOLT, W. V. Basic aspects of frozen storage of semen. Animal Reproduction Science, Amsterdam, v. 62, n. 1-3, p. 3-22, 2000.
KANKOFER, M.; KOLM, G.; AURICH, J. E.; AURICH, C. Activity of glutathione peroxidase, superoxide dismutase and catalase and lipid peroxidation intensity in stallion semen during storage at 5°C. Theriogenology, New York, v. 63, n. 5, p. 1354-1365, 2005.
KARESKOSKI, M.; KATILA, T. Components of stallion seminal plasma and the effects of seminal plasma on sperm longevity. Animal Reproduction Science, Amsterdam, v. 107, n. 3-4, p. 249-256, 2008.
KATILA, T. Procedures for handling fresh stallion semen. Theriogenology, New York, v. 46, n. 7, p. 1217-1227, 1997.
KENNEY, R. M.; BERGMAN, R. V.; COOPER, W. L.; MORSE, G. W. Minimal contamination techniques for breeding mares: techniques and preliminary findings. In: 21ST ANNUAL CONVENTION OF THE AMERICAN ASSOCIATION OF EQUINE PRACTITIONERS, 21., 1975, San Antonio. Proceedings... Lexington: American of Equine Practitioners, 1975. v. 21, p. 327-336..
LAGARES, M. A.; PETZOLDT, R.; SIEME, H.; KLUG, E. Assessing equine sperm-membrane integrity. Andrologia, Berlin, v. 32, n. 3, p. 163-167, 2000.
LOVE, C. C.; BLANCHARD, T. L.; VARNER, D. D.; BRINSKO, S. P.; VOGE, J.; BLISS, S.; SUDDERTH, K.; TEAGUE, S.; LACAZE, K. Effect of daily semen centrifugation and resuspension on the longevity of equine sperm quality following cooled storage. Theriogenology, New York, v. 77, n. 9, p. 1911-1917, 2012.
LOVE, C. C.; BRINSKO, S. P.; RIGBY, S. L.; THOMPSON, J. A.; BLANCHARD, T. L.; VARNER, D. D. Relationship of seminal plasma level and extender type to sperm motility and DNA integrity. Theriogenology, New York, v. 63, n. 6, p. 1584-1591, 2005.
LUSIGNAN, M.; BERGERON, A.; LAFLEUR, M.; MANJUNATH, P. The major proteins of bovine seminal plasma interact with caseins and whey proteins of milk extender. Biology of Reproduction, Utrecht, v. 85, n. 3, p. 457-464, 2011.
MACÍAS-GARCÍA, B.; GONZÁLEZ-FERNÁNDEZ, L.; LOUX, S. C.; ROCHA, A. M.; GUIMARÃES, T.; PEÑA, F. J.; VARNER, D. D.; HINRICHS, K. Effect of calcium, bicarbonate, and albumin on capacitation-related events in equine sperm. Reproduction, Bristol, v. 149, n. 1, p. 87-99, 2015.
MANJUNATH, P. New insights into the understanding of the mechanism of sperm protection by extender components. Animal Reproduction, Belo Horizonte, v. 9, n. 4, p. 809-815, 2012.
MARTINS, H. S.; SOUZA, M. R.; PENNA, C. F. A. M.; SILVA, G. C. da; CÔRTES, S. F.; STAHLBERG, R.; LAGARES, M. A. Milk, caseinate and lactoferrin addition to equine semen cooling extenders. Andrologia, Berlin, v. 48, n. 9, p. 950-956, 2016.
MORRIS, L. H. A. Challenges facing sex preselection of stallion spermatozoa. Animal Reproduction Science, Amsterdam, v. 89, n. 1-4, p. 147-157, 2005.
NEVES, A. P.; BUSTAMANTE FILHO, I. C.; TREIN, C. R.; MALSCHITZKY, E.; JOBIM, M. I. M.; MATTOS, R. C. Reproductive parameters and sperm freezability of stallions of the Brazilian pony breed. In: INTERNATIONAL SYMPOSIUM ON EQUINE REPRODUCTION, 9., 2006, Kerkrade. Proceedings... Amsterdam: Elsevier, 2006. v. 94, p. 67-69.
ORTEGA-FERRUSOLA, C.; JOHANNISSON, A.; VEJA, F. J. P.; TAPIA, J. Á.; RODRIGUEZ-MARTINEZ, H.; DALIN, A. M.; MORRELL, J. M. Effect of different extenders and seminal plasma on the susceptibility of equine spermatozoa to lipid peroxidation after single-layer centrifugation, through Androcoll-E. Journal of Equine Veterinary Science, New York, v. 31, n. 7, p. 411-416, 2011.
PAPA, P. M.; RAMIRES-NETO, C.; MONTEIRO, G. A.; SANCLER-SILVA, Y. F. R.; RESENDE, H. L.; FREITAS-DELL'AQUA, C. P.; DELL'AQUA JUNIOR, J. A.; ALVARENGA, M. A.; PAPA, F. O. Sperm parameters and fertility of stallion semen cooled with different extenders. Journal of Equine Veterinary Science, New York, v. 34, n. 1, p. 79, 2014.
PARRISH, J. J. Bovine in vitro fertilization: in vitro oocyte maturation and sperm capacitation with heparin. Theriogenology, New York, v. 81, n. 1, p. 67-73, 2014.
PICKETT, B. W.; AMANN, R. P. Extension and storage of stallion spermatozoa: a review. Equine Veterinary Science, California, v. 7, n. 5, p. 289-302, 1987.
POMMER, A. C.; RUTLLANT, J.; MEYERS, S. A. The role of osmotic resistance on equine spermatozoal function. Theriogenology, New York, v. 58, n. 7, p. 1373-1384, 2002.
PUGLIESI, G.; CARVALHO, G. R.; RATES, D. M.; KER, P. G.; PEREIRA, M. O.; RENAN, R. O.; MONTEIRO, J. D. S. Viability and fertility of cooled equine semen diluted with skimmed milk or glycine egg yolk-based extenders. Revista Brasileira de Zootecnia, Viçosa, MG, v. 41, n. 12, p. 2411-2417, 2012.
RATHI, R.; COLENBRANDER, B.; BEVERS, M. M.; GADELLA, B. M. Evaluation of in vitro capacitation of stallion spermatozoa. Biology of Reproduction, Utrecht, v. 65, n. 2, p. 462-470, 2001.
RODRIGUEZ, S. A. F.; ARRUDA, R. P.; BIANCHI-ALVES, M. R.; AFFONSO, F. J.; LANÇONI, R.; CARVALHO, H. F.; LEMES, K. M.; ANDRADE, A. F. C.; CELEGHINI, E. C. C. Morphofunctional characterization of cooled sperm using different extenders in equine-assisted reproduction. In: ISER. Journal of Equine Veterinary Science, New York, v. 34, n. 1, p. 94, 2014.
SWAIN, J. E. Optimizing the culture environment in the IVF laboratory: impact of pH and buffer capacity on gamete and embryo quality. Reproductive BioMedicine Online, Oxford, v. 21, n. 1, p. 6-16, 2010.
VIDAMENT, M.; MAGISTRINIA, M.; LE FOLL, Y.; LAVILLAINA, N.; YVONA, J. -M.; DUCHAMPE, G.; BLESBOIS, E. Temperatures from 4 to 15°C are suitable for preserving the fertilizing capacity of stallion semen stored for 22 h or more in INRA96 extender. Theriogenology, New York, v. 78, n. 2, p. 297-307, 2012.
WHITE, D. R.; AITKEN, R. J. Relationship between calcium, cyclic AMP, ATP, and intracellular pH and the capacity of hamster spermatozoa to express hyperactivated motility. Molecular Reproduction and Development, Rhode Island, v. 22, n. 2, p. 163-177, 1989.
YÁNIZ, J. L.; MATEOS, J. A.; SANTOLARIA, P. Zwitterionic buffers preserve ram semen quality more efficiently than TRIS during storage at 15°C. Small Ruminant Research, Amsterdam, v. 95, n. 1, p. 54-60, 2011.
Author notes
*Author for correspondence