Geometridae Stephens, 1829 from different altitudes in Western Himalayan Protected Areas of Uttarakhand, India. (Lepidoptera: Geometridae)
Geometridae Stephens, 1829 de diferentes altitudes de las áreas protegidas del Himalaya occidental de Uttarakhand, India (Lepidoptera: Geometridae)
Geometridae Stephens, 1829 from different altitudes in Western Himalayan Protected Areas of Uttarakhand, India. (Lepidoptera: Geometridae)
SHILAP Revista de lepidopterología, vol. 45, no. 177, pp. 143-163, 2017
Sociedad Hispano-Luso-Americana de Lepidopterología

This work is licensed under Creative Commons Attribution 4.0 International.
Received: 24 May 2016
Accepted: 06 November 2016
Abstract: The Geometridae Stephens, 1829 are considered as an excellent model group to study insect diversity patterns across elevational gradients globally. This paper documents 168 species of Geometridae belonging to 99 genera and 5 subfamilies from different Protected Areas in a Western Himalayan state, Uttarakhand in India. The list includes 36 species reported for the first time from Uttarakhand, which hitherto was poorly explored and reveals significant altitudinal range expansion for at least 15 species. We sampled different vegetation zones across an elevation gradient stretching from 600 m up to 3600 m, in Dehradun-Rajaji landscape, Nanda Devi National Park, Valley of Flowers National Park, Govind Wildlife Sanctuary, Gangotri National Park and Askot Wildlife Sanctuary. The subfamily Ennominae represented the maximum number of species, and the species of subfamily Larentiinae were found to be more restricted to higher elevation areas. Western Mixed Coniferous forest held the greatest number of species, whereas the Subalpine forest was characterized by the highest number of indicator species identified through Indicator Species Analysis. While Indo-Malayan species dominated the assemblage composition, the maximum number of Himalayan endemics suggested that these species are long adapted to the Himalayan climatic gradient and ongoing climate-mediated perturbation may hamper their future survival.
Keywords: Lepidoptera, Geometridae, diversity, altitude, Western Himalaya, indicator species, Uttarakhand, India.
Resumen: Los Geometridae Stephens, 1829 se consideran como un grupo modelo excelente para estudiar a nivel global, los patrones de diversidad de los insectos a lo largo de gradientes de elevación. Este trabajo documenta a 168 especies de Geometridae pertenecientes a 99 géneros y 5 subfamilias de diferentes áreas protegidas en el estado del Himalaya occidental, Uttarakhand, en India. La lista incluye 36 especies registradas por primera vez de Uttarakhand, que ha sido mal explorado hasta ahora y revela la expansión del alcance altitudinal de al menos 15 especies. Estudiamos muestras de distintas zonas de vegetación de elevación entre los 600 m y los 3.600 m, en el área de Dehradun-Rajaji, Parque Nacional de Nanda Devi, Parque Nacional del Valle de las Flores, Reserva Natural de Govind, Parque Nacional de Gangotri y Reserva Natural de Askot. La subfamilia Ennominae supone el mayor número de especies, pero se encontró que las especies de la subfamilia Larentiinae eran más restringidas a las áreas más elevadas. El bosque mixto de coníferas occidental reúne el mayor número de especies, mientras que el bosque subalpino se caracteriza por incluir el mayor número de especies indicadoras, identificadas por medio del análisis de especies indicadoras. Mientras que las especies indo-malayas dominan la composición del conjunto, el máximo número de endemismos del Himalaya, sugiere que estas especies están más adaptadas al gradiente climático del Himalaya desde hace tiempo y las perturbaciones climáticas actualmente en curso, podrían dificultar su futura supervivencia.
Palabras clave: Lepidoptera, Geometridae, diversidad, altitud, Oeste del Himalaya, indicador de especies, Uttarakhand, India.
Introduction
The distribution of species and higher taxa like families is known to change along environmental gradients globally (BREHM & FIEDLER, 2003). One such gradient is the altitudinal gradient which serves as a natural system for various experiments (BREHM & FIEDLER, 2003) in ecology providing a diverse array of habitat and micro-climatic regimes and associated steep changes in the biotic and abiotic factors in a small geographic area (HODKINSON, 2005). Thus, mountain ecosystems have turned out to be an ideal system to study the factors governing the diversity and distribution of organisms and to predict responses due to subtle climatic variation (ASHTON et al., 2016). These ecosystems have now become critical areas for conservation need globally because of the high number of endemic and climate sensitive species (FOSTER, 2001) and can be used as tools to monitor climate change responses (BENISTON et al., 1997).
Different species show different patterns in altitudinal stratification, where some species occupy very small altitudinal ranges and have a high turnover across altitudes, but there are species spread across a wide range of environmental conditions (ASHTON et al., 2016). Many studies have found altitudinal stratification in insect assemblages like ants (BURWELL & NAKAMURA, 2011), moths (BREHM & FIEDLER, 2003; ASHTON et al., 2011), beetles (ESCOBAR et al., 2005), as well as birds (WILLIAMS et al., 2010) and mammals (WILLIAMS, 1997). It is known from these studies that different groups respond distinctively to altitude (STORK & BRENDALL, 1990), with particular species staying restricted to high altitudes showing endemism (KESSLER, 2002; SZUMIK et al., 2012) and high phylogenetic diversity (ZOU et al., 2016), thus demanding increased conservation efforts of their habitats.
Moth assemblages react sensitively to environmental gradients and are distinctly stratified altitudinally in tropical and subtropical forests (ASHTON et al., 2016). This kind of database certainly is lacking in the Indian Himalayan Region (IHR) which as part of the world’s largest mountain ecosystem, harbours a diverse and unique assemblage of faunal diversity due to its unique position at the junction of the Palaearctic and Oriental regions. The Himalayan system, recognized as a globally important biodiversity hotspot, is characterized by sharp environmental gradients due to rapid geo- climatic variations generating diverse vegetation and community types. A baseline data of the distribution of different families of nocturnal Lepidoptera along the altitude needs to be created, as little information is available so as to pile on future research addressing the ecological patterns governing the distribution and diversity as well as the effects of climate change.
The Geometridae Stephens, 1829, generally known as Looper moths, are the second most speciose family of moths worldwide. They occur in every biogeographical region (SCOBLE et al., 1995) and are a well-established model group for biodiversity studies in temperate and tropical regions. Their altitudinal distribution patterns have been studied extensively in tropical South America (BREHM et al., 2003, 2007; HILT et al., 2006), Africa (AXMACHER et al., 2004), Australia (KITCHING et al., 2000) and South-east Asia (HOLLOWAY, 1985; CHEY et al., 1997; INTACHAT et al., 1997; BECK et al., 2002). They have been proposed and experimentally demonstrated as a good biological indicator in habitat assessment and monitoring program, as well as in climate change studies (CHEN et al., 2009). Although the taxonomy of this family is well established for the temperate regions, the tropical areas need large-scale revisions. Around 23,000 species have been described to date worldwide (SCOBLE & HAUSMANN, 2007) with high synonymy at the species level which suggests much more revisionary work to be done. The study of patterns of species description reveals that much revision of the taxonomy is still required at the species level.
The present study aims to document Geometridae moths across different habitat types, along the elevation and vegetation gradient in the Indian state of Uttarakhand located in the Western Himalayan Biogeographic Province. Our primary objective was to prepare a species compilation from primary field data which can be compared with old records as well as be a baseline for future study. We also investigated how major species groups of this important family are distributed along elevational and vegetation gradients, how different biogeographic elements influence the overall faunal composition and which would be the target species to monitor in future.
Materials and methods
STUDY AREA
The Uttarakhand state of India provides an epitome of the geological architecture of the entire Himalaya. The 88% hilly state has 62% of its geographical area under forest cover (CHAWLA et al., 2008). As making an initial inventory of particular taxa is an important first step towards any conservation management program, we tried to cover as many as possible different forest and habitat types according to major biomes and selected five heterogeneous landscapes. We sampled in Dehradun-Rajaji Landscape (600 m to 800 m) harbouring Moist Sal forest habitat. Subtropical hill forest habitats were sampled in Askot Wildlife Sanctuary (600 m to 1000 m). This landscape, located along the India-Nepal border is also significant as a junction between the Western and Central Himalaya, as floral elements from both these biogeographic zones converge here. Himalayan Moist
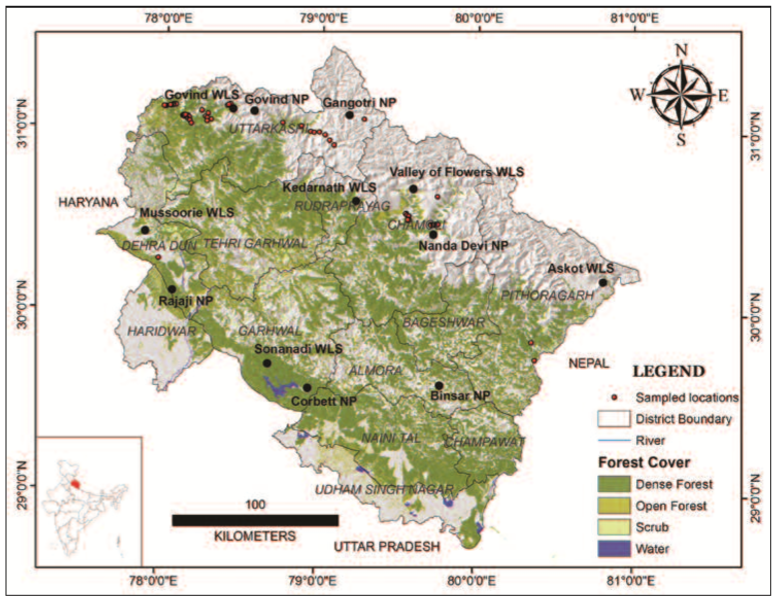
Fig. 1
The distribution of the light-trapping sites for Geometridae across 5 Protected Areas in the Indian State of Uttarakhand.
Temperate habitat was sampled in Govind Wildlife Sanctuary ranging from an elevation of 1400 m to 3600 m including major forest types like Subtropical Pine Broadleaved Mix forest, Moist Temperate Deciduous forest, Western Mix Coniferous forest, Western Himalayan Upper Oak forest, and Subalpine forest (CHAMPION & SETH, 1968). The sampling sites within Gangotri National Park, owing to its special location as great vertical orientation, included habitats similar to the Trans- Himalayan condition of the Tibetan Plateau. The Nanda Devi Biosphere Reserve, including Nanda Devi National Park and Valley of Flowers National Park, harbours varied habitats like Himalayan Dry Temperate forest and Alpine pastures. In total, 223 sampling nights (Table 1) were performed between 2008 and 2015 in 197 sites (all the sites sampled are shown in Figure 1) across 5 Protected Areas.
SAMPLING GEOMETRID MOTHS
Geometrid moths were sampled manually using a light trap running for 4 hours, in two sessions on consecutive nights, from 8 pm to 12 midnight and from 12 midnight to 4 am at a particular site, to ensure all the moths flying in a particular location or habitat in different quarters of the night could be recorded. Details of sampling sessions and sampling effort in various PAs are provided in Table 1. Light traps were set using a solar powered lantern and gas petromax equivalent to Mercury Vapour (MV) bulb. The combination of light sources was placed in front of a white 3 x 1.8 m cloth sheet hung between two vertical poles in such a way that it touched the surface and extended forward over the ground slightly. This was to ensure enough resting place for individual moths after they were attracted to light for live photography and collection. Some species were very active around a light trap and never settled on the white sheet. They were collected using an insect net. No sampling occurred within the period five days before to four days after the full moon, as light trapping is much less efficient during these periods (MCGEACHIE, 1989; YELA & HOLYOAK, 1997).
| ProtectedArea | Sampling session | Samplingnights | Seasons sampled | Altitudinal rangecovered (m) |
| Dehradun- Rajaji Landscape | April-June, July-September, October-November, 2009-2014 | 32 | Pre-Monsoon, Monsoon, Post-monsoon | 600-800 |
| Gangotri NP | October-November, 2008; October, 2012 | 18 | Post-monsoon | 2400-3600 |
| Govind WLS | April-June, July-September, October-November, 2009-2012 | 84 | Pre-Monsoon, Monsoon, Post-monsoon | 1400-3600 |
| Nanda Devi Biosphere Reserve | April-June, August-October, 2013-2015 | 65 | Pre-monsoon, Post- monsoon | 2000-3800 |
| Askot WLS | May-June, September, 2013- 2014 | 24 | Pre-monsoon, Post- monsoon | 600-1000 |
Individual moths were collected in a wide-mouth glass jar filled with Benzene vapour evaporating from a cotton swab soaked in liquid Benzene and placed at the bottom of the glass jar. The specimens were first sorted into morphospecies and later identified with the help of the available literature and by comparison with the reference collections available at the Zoological Survey of India, Jabalpur and Kolkata. The identification was done following HAMPSON (1892, 1894, 1895 and 1896), HOLLOWAY (1993, 1996, 1997), BARLOW (1982), and HARUTA (2002). The nomenclature has been followed after SCOBLE & HAUSMANN (2007). The voucher specimens were submitted to the national repository at the Zoological Survey of India.
Results
Altogether 168 species of Geometridae moths belonging to 99 genera of 5 subfamilies were recorded from different Protected Areas (PAs) of Uttarakhand. The detailed species account with their recorded altitudinal range, past altitudinal record and host plant information is provided in Appendix 1.
We recorded 20 species from Askot Landscape, 42 species from Dehradun-Rajaji Landscape, 112 species from Govind Wildlife Sanctuary, 15 Species from Gangotri National Park and 37 species from Nanda Devi Biosphere Reserve. Among major forest types sampled, maximum numbers of species were recorded from Western Mixed Coniferous forest (55 species) which was mainly the mid-elevation area stretching from 2200 m to 2800 m altitude zone. Among other species-rich areas were Pine (Pinus roxburghii) Mix forests (46 species) extending from 1400 m to 1800 m and Subalpine forest (43 species) between 3200 m to 3600 m. Riverine forest (9 species) and Moru Oak (Quercus dilatata) forest (12 species) were among species-poor regions. The alpine scrubland, the semi-arid altitudinal zone above 3600 m beyond tree-line yielded 20 species (Figure 2).
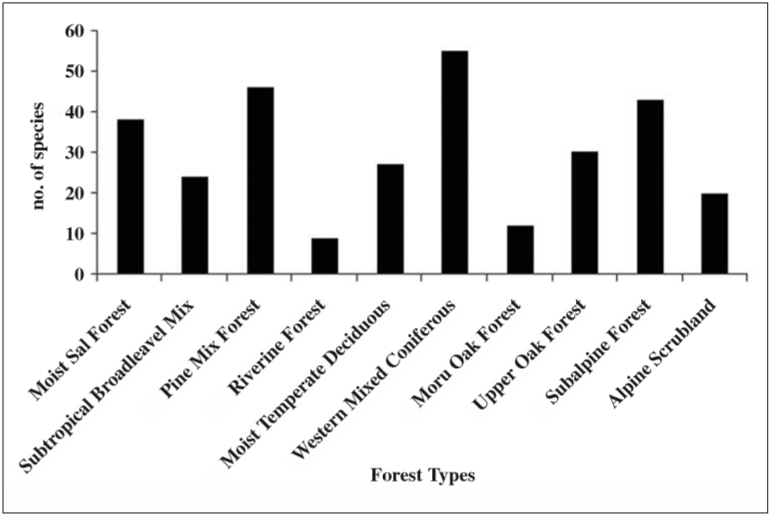
Fig. 2
The number of species recorded in the different types of forest sampled across all the sampling areas. Western Mixed Coniferous forest was the most species-rich habitat followed by Pine Mix forest, Subalpine forest and Moist Sal forest.
Among five subfamilies of Geometridae sampled across different elevation and forest types, Ennominae were dominant (92 species), followed by Larentiinae (37 species), Geometrinae (28 species), Sterrhinae (11 species) and Desmobathrinae (1 species). Altitudinal distribution of the four major subfamilies (Figure 3) showed that the subfamily Larentiinae was exceptionally distributed towards higher altitude while the other three were diverse in lower and middle elevation zones. Mean species distribution of the dominant subfamily Ennominae was recorded around 1400 m while most of the species were recorded between 600 m to 2300 m and the species range extended up to 3400 m. The mean species distribution of the subfamily Larentiinae was recorded around 2800 m while most of the species were recorded between 2500 m to 3300 m and the species range extended from 1800 m to 3600 m. The mean species distribution of Geometrinae was around 700 m while most of the species were recorded from 600 m to 1300 m, and the species range extended up to 2500 m. For Sterrhinae, the mean species distribution was around 1400 m, while most of the species were recorded from 700 m to 1700 m, and the species range extended up to 2900 m (Figure 3).
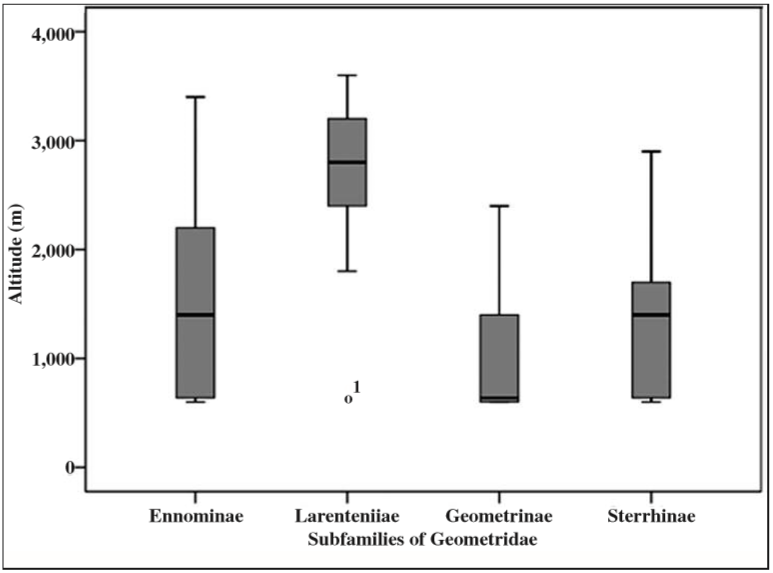
Fig. 3
The altitudinal distribution of four major subfamilies of the family Geometridae collected across all sampling sites. While the subfamily Ennominae was widely distributed, species of subfamily Larentiinae had clear preference for higher altitudes.
The subfamily composition of the Geometridae also changes according to various PAs covered, depending on their elevational position (Figure 4). While there was a dominance of subfamily Ennominae in all the PAs, except Gangotri NP, which being truly a high-altitude PA ranging above 3000 m, was dominated by Larentiinae. Notably, the lower altitude PAs like Askot and Dehradun (Rajaji Landscape) were almost devoid of Larentiinae species, with no record from Dehradun at all. Whereas, in other PAs, which had significant representation of high altitude forest types, like the Nanda Devi Biosphere Reserve and the Govind Wildlife Sanctuary, Larentiinae species were present in high numbers along with Ennominae species.
Among 12 tribes recorded of the subfamily Ennominae, Boarmiini was the dominant (37.5%) followed by Hypochrosini (12.5%). The other main tribes were Eutoeini, Abraxini, Gnophini, Ourapterygini and Macariini (6.25% each). Nine tribes were recorded of Larentiinae, among which, 30% of the species were from Cidariini, followed by Larentiini, Asthenini and Xanthorhoini (14.81% each). Specimens of Tribe Eupitheciini and Perizomini were mostly excluded from the analysis since their identification up to species level could not be confirmed except one species of Eupithecia and two species of Perizoma. Among Geometrinae, 43% species were recorded of tribe Geometrini, 29% species were of tribe Pseudoterpnini and 23% of Hemitheini. Among Sterrhinae, nearly 50% specimens were of Scopulini, whose identification up to species level was not very successful except one species, viz. Scopula pulchellata.
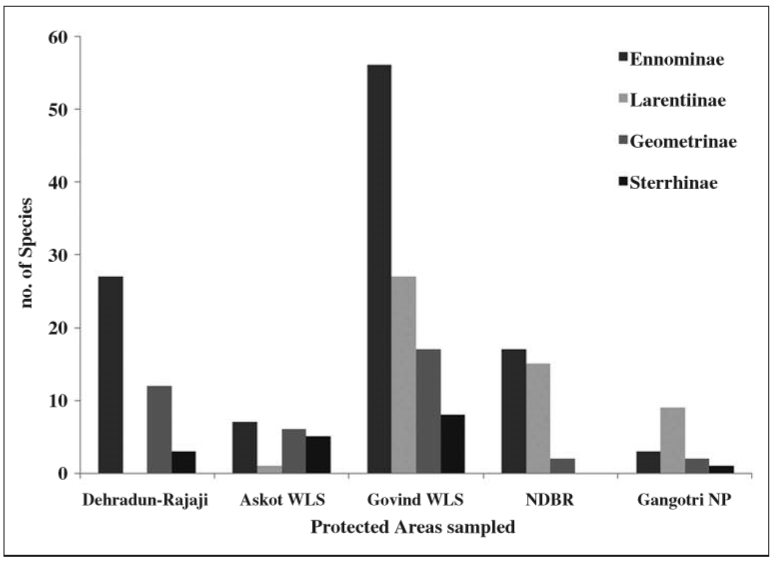
Fig. 4
No. of species in each subfamily of Geometridae sampled across different Protected Areas in the Indian state Uttarakhand. The subfamily Ennominae was most numerous all through except in Gangotri NP. The subfamily Larentiinae had significant representation in high altitude protected areas and almost absent from lower altitude areas like Dehradun and Askot WLS.
We categorized each species into four Biogeographic components based on their regional and global distribution from literature survey. Within Indian sub-region, 65% species were endemic to Himalayan region, while 16% species were also common in Gangetic plains. Around 19% species had common distribution throughout India (Figure 5a). Globally, 60% species were of Indo-Malayan origin, while significant portion (22%) was of Sino-Himalayan origin. A minor representation (9%) was also there of Eastern Palaearctic element while a similar proportion of species were also recorded which are globally distributed (Figure 5b).
We compared each species’ maximum altitude record from past literature with highest altitude recorded in the current study and were able to document possible range expansion for at least 15 species. Among these species we recorded altitudinal range expansion of more than 1000 m for 12 species: Abraxas irrorata (2000 m to 3400 m), Abraxas picaria (2000 m to 3400 m), Heterolocha phoenicotaeniata (2000 m to 3200 m), Odontopera heydena (1500 m to 3200 m), Odontopera lentiginosaria (600 m to 3200 m), Arichanna tenebraria (2000 m to 3400 m), Psyra debilis (2100 m to 3400 m), Eupithecia rajata (1500 m to 2800 m), Docirava aequilineata (Indian plains to 3400 m), Docirava pudicata (Central India to 3200 m); for 2 species, around 1000 m expanse were recorded: Laciniodes plurilinearia (2400 m to 3200 m) and Xanthorhoe hampsoni (2200 m to 3200 m).
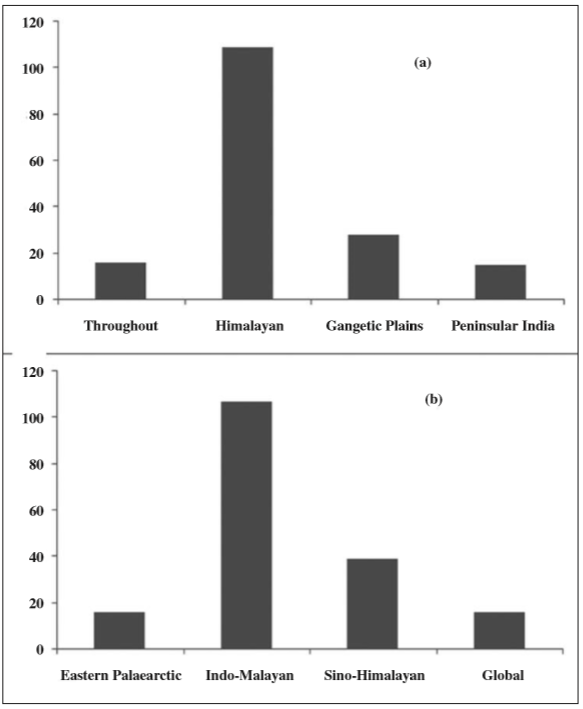
Fig. 5
Biogeographic composition of sampled Geometridae assemblage: (a) Within Indian Subcontinent, Himalayan species dominated, the rest commonly distributed throughout. (b) The global pattern was dominated by Indo-Malayan species distributed along entire Himalayan breadth. There was significant proportion of Sino- Himalayan species as well as Eastern Palaearctic species
Characteristic moth species restricted to specific altitude or forest types were identified for each vegetation type using the Indicator Species Analysis (DUFRÊNE & LEGENDRE, 1997) using program PC-ORD. This method combines measures of specificity and fidelity and provides an indicator value (IndVal) for each species, as a percentage with an associated test of significance, with high and significant percentages designating good indicator species. Three species were identified to be characteristic of low altitude Pine-broadleaved mix forest, Semiothisa sufflata, Menophra subplagiata, Scopula pulchellata; two species to Moist Temperate Deciduous forest: Sirinopteryx rufivinctata, Odontopera kametaria; single species each, were restricted to Western Mixed Coniferous forest and Kharsu Oak forest, Pseudopanthera himaleyica and Odontopera lentiginosaria respectively. The highest altitude forest, Subalpine forest was characterized by nine specialized species which were not recorded from any other forest types: Arichanna tenebraria, Photoscotosia amplicata, Opisthograptis tridentifera, Photoscotosia multilinea, Venusia crassisigna, Abraxas gunsana, Triphosa rubrodotata, Eustroma chalcoptera and Opisthograptis sulphurea (Table 2).
| Forest Types | Species | Subfamily | Indicator Value | Sig (P) |
| SPBM | Semiothisa sufflata | Ennominae | 81.6 | 0.001 |
| SPBM | Menophra subplagiata | Ennominae | 78.4 | 0.0008 |
| SPBM | Scopula pulchellata | Sterrhinae | 75.4 | 0.0013 |
| MTD | Sirinopteryx rufivinctata | Ennominae | 77.5 | 0.009 |
| MTD | Odontopera kametaria | Ennominae | 56.4 | 0.0048 |
| WMC | Pseudopanthera himaleyica | Ennominae | 56.9 | 0.0577 |
| WHUOF | Odontopera lentiginosaria | Ennominae | 81 | 0.0051 |
| SAF | Arichanna tenebraria | Ennominae | 87.1 | 0.0004 |
| SAF | Photoscotosia amplicata | Larentiinae | 74.6 | 0.0009 |
| SAF | Opisthograptis tridentifera | Ennominae | 64.7 | 0.0033 |
| SAF | Photoscotosia multilinea | Larentiinae | 62.9 | 0.0024 |
| SAF | Venusia classisigna | Larentiinae | 62.9 | 0.0024 |
| SAF | Abraxas gunsana | Ennominae | 62.4 | 0.0032 |
| SAF | Triphosa rubrodotata | Larentiinae | 60.1 | 0.0166 |
| SAF | Eustroma chalcoptera | Larentiinae | 58.4 | 0.0069 |
| SAF | Opisthograptis sulphurea | Ennominae | 57.8 | 0.0099 |
Discussion
This study was an initial step towards better understanding of a long-neglected but diverse and charismatic herbivorous insect assemblage in Himalayan temperate altitudinal gradient. The diversity of this crucial group of nocturnal Lepidoptera has not been systematically inventoried in the Indian Himalaya except WALIA (2005) and SMETACEK (2008). Thus, the study recorded several species which were either first-time record from India, or from the Western Himalayan state of Uttarakhand. After intensive literature survey, we documented 36 species which were previously unrecorded from Uttarakhand. Among them 19 species were of subfamily Ennominae: Anonychia violacea, Biston falcata, Psilalcis inceptaria, Medasina interruptaria, Medasina cervina, Erebomorpha fulguraria, Ourapteryx convergens, Arichanna tenebraria, Gnophos albidior, Hypomecis ratotaria, Loxaspilates hastigera, Odontopera heydena, Odontopera lentiginosaria, Plagodis inustaria, Psyra debilis, Opisthograptis sulphurea, Opisthograptis tridentifera, Sirinopteryx rufivinctata and Tanaoctenia haliaria; 3 species of subfamily Geometrinae: Chlorochaeta inductaria, Chlorochaeta pictipennis, Pingasa rubicunda; and 13 species were of subfamily Larentiinae: Photoscotosia multilinea, Photoscotosia metachryseis, Cidaria aurata, Electrophaes recta, Eustroma chalcoptera, Hydrelia bicolorata, Stamnodes pamphilata, Trichopterigia rufinotata, Triphosa rubrodotata, Perizoma albofasciata, Euphyia stellata, Xanthorhoe hampsoni and Heterothera dentifasciata. One species Rhodostrophia pelloniaria of subfamily Sterrhinae was also the first record from Western Himalaya.
Latitudinal species richness gradients are studied in mountain ecosystems in a much smaller scale but are more ecologically informative (SANDERS & RAHBEK, 2012). In high altitude areas, the geographical distance between different habitat or environments is very small, resulting in steep ecological gradients and the influence of various factors on biodiversity can easily be teased apart (AXMACHER et al., 2004). BREHM et al. (2003) studied elevational patterns of Geometrid moths in the Andean rainforest and found a maximum diversity between 1040 m and 2670 m, revealing a distinctive pattern, whereas SCHULZE (2000) showed that high levels of diversity in geometrid moth communities existed over a broad elevational range in a tropical mountain rainforest in Mt. Kinabalu, Borneo. There was a gap in studies from Himalayan temperate altitudinal gradient leading to no robust or generalized pattern of species diversity across these mountain ecosystems. The present study covering a wide altitudinal and geographical stretch tried to achieve equal sampling effort all through the gradient. Initial analysis suggested multi-modal peaks in diversity around 1400 m, 2600 m, and 3200 m.
Biotic interactions coupled with ecological and physiological characteristics of the species act as environmental filters (WEBB et al., 2002; GRAHAM et al., 2009) governing the species assemblages along the elevational and vegetational gradient. Not much is known about the climatic barriers influencing the moth assemblages, but the larval host plant availability must be substantial for the specialist species. But this constraint will not apply to specialists whose host plants are distributed across different elevations (BREHM et al., 2013). The host plant information compiled here for each species reflected that majority of the geometrid species are not even specialists as most belonging to the subfamily Ennominae are polyphagous. Polyphagy was more prominent for the species distributed in wider altitudinal range than restricted-range species.
The result from this study showed a similar pattern of distribution of subfamilies as in Ecuadorian Andes (BREHM & FIEDLER, 2003) with Ennominae being the most abundant family at the lower altitudes and higher altitude places showing more abundance of the subfamily Larentiinae. Species found at lower elevations are intolerant to environmental stochasticity according to Rapoport’s “rescue” hypothesis. Thus, species which occupy higher elevations have a larger range of tolerances and large elevational range (BREHM et al., 2007). Species that occupy high altitude areas must have the physiological characters to comply with the cooler temperatures and affiliation to the host plants that have colonised the upper areas (BREHM et al., 2013). The underlying factors are yet to be known, but it can be speculated that the Larentiinae moths are better suited to the cooler environments than the member of other subfamilies, especially Sterrhinae and Geometrinae (BREHM et al., 2013). The montane characteristics of Larentiinae was already explained by HOLLOWAY (1987), but the physiological properties that allow the moths of this subfamily to be unusually tolerant of unfavourable conditions remain unknown (BREHM & FIEDLER, 2003). The primary predators of moths (bats and birds) also show a decline in species richness and abundance as we go up the elevation (RAHBECK, 1997). Larentiinae moths have a much weaker body structure than the other sub-families making them weak flyers and thus might benefit in a predator- free environment (BREHM & FIEDLER, 2003). However, the Geometridae moths are found to be less affected by temperature limitations than the other nocturnal moths (BECK et al., 2011). Thus, moderate host plant specificity coupled with adaptability to cooler temperatures describes the patterns in species distribution across the elevation (BREHM et al., 2013).
This study has covered an elevational range from 600 m to 3800 m spread across different protected areas of Uttarakhand. Still there is a gap in moth samples between 1000 m to 1500 m, which is mainly due to the absence of suitable natural sites in this range which are free from human disturbance. The sampling of entire elevational gradient would generate a more discernible pattern with relevant ecological explanations. The proportion of one taxon, when compared to other can be used for determining the species numbers (COLWELL & CODDINGTON, 1994), but it requires ample representation throughout the sampling effort. Determining the subfamily composition along environmental gradients allowed us to explore a significant pattern which complements the measures of species diversity (BREHM & FIEDLER, 2003). It was found that preference of the subfamily Larentiinae for higher altitude sites holds true even in Himalayan context, and this pattern can be regarded as a universal phenomenon, irrespective of biogeographic positions. Concerning Lepidoptera, Himalaya represents a mixing ground of Palaearctic and Indo-Malayan communities which have caused a proliferation of species usually not found outside tropics. Biogeographically, the Himalayan range straddles a transition zone between the Palaearctic and Indo-Malayan realms. Species from both realms are found in the hotspot. High percentage of Himalayan endemics among sampled Geometridae species suggested that this assemblage is long adapted to Himalayan climatic gradient and human or climate-induced habitat alteration may threaten their future survival. For at least 15 species, a new altitudinal limit has been documented. In majority of the cases, the previous records being more than hundred years old and the shift recorded more than 1000 m, these species can be targeted for detailed life history and distribution study to confirm whether these range expansions are due to climate alteration or other stochastic factors. Climate induced shift in altitudinal range has already been recorded for moth assemblages in Finland (PARMESAN, 2006) and Borneo (CHEN et al., 2009).
The selection of suitable indicator species depends on several criteria. An effective indicator needs to be present in large numbers, be easily recognizable, as well as being sensitive to environmental variables (SCOBLE, 1995; HOLLOWAY, 1998). Moth groups that are sensitive to floristic change and which have low vagility (ASHTON et al., 2011) fulfil these criteria and have been demonstrated to be good indicators across a variety of ecological investigations (HOLLOWAY, 1985; SCOBLE, 1995; KITCHING et al., 2000; BECK et al., 2002). The analyses presented here suggested a set of 16 species of Indicators which may be useful as part of a multi-taxon predictor set for future monitoring of the impact of global warming on forest biodiversity. The existence of clear cut patterns of altitudinally delimited moth assemblages, with particular species having restricted altitudinal distributions, suggests that selected moth taxa will be useful in tracking any upward shifts in distribution and invasions of higher altitudes, a likely consequence of global warming. It also suggests that the highly distinctive upper elevation assemblage (the subalpine set of indicators) must be regarded as vulnerable and of conservation concern.
Although our data is still scattered and more intensive sampling can result in more addition to this species record of Geometridae, future research on this current database should benefit the conservation of entire moth assemblage and their habitats in Western Himalayan Biogeographic province.
BIBLIOGRAPHY
ASHTON, L. A., ODELL, E. H., BURWELL, C. J., MAUNSELL, S. C., NAKAMURA, A., MCDONALD, W. J. F. & KITCHING, R. L., 2016.– Altitudinal patterns of moth diversity in tropical and subtropical Australian rainforests.– Austral Ecology, 41(2): 197-208.
ASHTON, L. A., KITCHING, R. L., MAUNSELL, S., BITO, D. & PUTLAND, D., 2011.– Macrolepidopteran assemblages along an altitudinal gradient in subtropical rainforest exploring indicators of climate change.– Memoirs of the Queensland Museum, 55: 375-389.
AXMACHER, J., HOLTMANN, G., SCHEUERMANN, L., BREHM, G., MÜLLER-HOHENSTEIN, K. & FIEDLER, K., 2004.– Diversity of geometrid moths (Lepidoptera: Geometridae) along an Afrotropical elevational rainforest transect.– Diversity and Distributions, 10: 293-302.
BARLOW, H. S., 1982.– An Introduction to the Moths of South East Asia: 305 pp. Malayan Nature Society, Kuala Lumpur.
BECK, J., BREHM, G. & FIEDLER, K., 2011.– Links between the environment, abundance and diversity of Andean moths.– Biotropica, 43: 208-217.
BECK, J., SCHULZE, C. H., LINSENMAIR, K. E. & FIEDLER, K., 2002.– From forest to farmland: diversity of geometrid moths along two habitat gradients in Borneo.– Journal of Tropical Ecology, 17: 33-51.
BENISTON, M., DIAZ, H. F. & BRADLEY, R. S., 1997.– Climate change at high elevation sites: an overview.– Climate Change, 36: 233-251.
BREHM, G. & FIEDLER, K., 2003.– Faunal composition of geometrid moths changes with altitude in an Andean montane rain forest.– Journal of Biogeography, 30: 431-440.
BREHM, G., COLWELL, R. K. & KLUGE, J., 2007.– The role of environment and mid-domain effect on moth species richness along a tropical elevational gradient.– Global Ecology and Biogeography, 16: 205-217.
BREHM, G., STRUTZENBERGER, P. & FIEDLER, K., 2013.– Phylogenetic diversity of geometrid moths decreases with elevation in the tropical Andes.– Ecography, 36: 1247-1253.
BREHM, G., SÜSSENBACH, D. & FIEDLER, K., 2003.– Unique elevational patterns of geometrid moths in an Andean montane rainforest.– Ecography, 26: 456-466.
BURWELL, C. J. & NAKAMURA, A., 2011.– Distribution of ant species along an altitudinal transect in continuous rainforest in sub-tropical Queensland, Australia.– Memoirs of Queensland Museum, 55: 391- 411.
CHAMPION, H. G. & SETH, S. K., 1968.– A revised survey of forest types of India: 404 pp. Government of India Press, New Delhi.
CHAWLA, A., RAJKUMAR, S., SINGH, K. N., LAL B. & THUKRAL, A. K., 2008.– Plant species diversity along an altitudinal gradient of Bhabha Valley in Western Himalaya.– Journal of Mountain Science, 5: 157–177.
CHEN, I. C., SHIU, H. J., BENEDICK, S., HOLLOWAY, J. D., CHEY, V. K., BARLOW, H. S., HILL, J. K. & THOMAS, C. D., 2009.– Elevation increases in moth assemblages over 42 years on a tropical mountain.– Proceedings of the National Academy of Sciences, 106(5.: 1479-1483.
CHEY, V. K., HOLLOWAY, J. D. & SPEIGHT, M. R., 1997.– Diversity of moths in forest plantations and natural forests in Sabah.– Bulletin of Entomological Research, 87: 371-385.
COLWELL, R. K. & CODDINGTON, J. A., 1994.– Estimating terrestrial biodiversity through extrapolation.– Philosophical transactions of the Royal Society London, 345: 101-118.
DUFRENE, M. & LEGENDRE, P., 1997.– Species assemblages and indicator species: the need for a flexible asymmetrical approach.– Ecological Monographs, 67: 345-366.
FOSTER, P., 2001.– The potential negative impacts of global climate change on tropical montane cloud forests.– Earth-Science Reviews, 55: 73-106.
GRAHAM, C. H., PARRA, J. L., RAHBEK, C. & MCGUIRE, J. A., 2009.– Phylogenetic structure in tropical hummingbird communities.– Proceedings of the National Academy of Science, 106: 19673-19678.
HAMPSON, G. F., 1892.– Fauna of British India including Ceylon and Burma-Moths, .: 527 pp. Taylor & Francis, London.
HAMPSON, G. F., 1894.– Fauna of British India including Ceylon and Burma-Moths, 2: 609 pp. Taylor & Francis, London.
HAMPSON, G. F., 1895.– Fauna of British India including Ceylon and Burma-Moths, 3: 546 pp. Taylor & Francis, London.
HAMPSON, G. F., 1896.– Fauna of British India including Ceylon and Burma-Moths, 4: 594 pp. Taylor & Francis, London.
HILT, N., BREHM, G. & FIEDLER, K., 2006.– Diversity and ensemble composition of geometrid moths along a successional gradient in the Ecuadorian Andes.– Journal of Tropical Ecology, 22: 155-166.
HODKINSON, I. D., 2005.– Terrestrial insects along elevation gradients: species and community responses to altitude.– Biological Reviews, 80: 489-513.
HARUTA, T. (eds.), 1994.– Moths of Nepal, Part 3, Tinea. 14(Supplement 1): 171 pp. Japan Heterocerists’ Society, Tokyo.
HARUTA, T. (eds.), 1995.– Moths of Nepal, Part 4, Tinea. 14(Supplement 2): 206 pp. Japan Heterocerists’ Society, Tokyo.
HARUTA, T. (eds.), 1998.– Moths of Nepal, Part 5, Tinea. 15(Supplement 1): 330 pp. Japan Heterocerists’ Society, Tokyo.
HARUTA, T. (eds.), 2000.– Moths of Nepal, Part 6, Tinea. 16(Supplement 1): 163 pp. Japan Heterocerists’ Society, Tokyo.
HOLLOWAY, J. D., 1985.– Moths as indicator organisms for categorizing rain-forest and monitoring changes and regeneration process.– In A. C. CHADWICK & S. L. SUTTON (eds). Tropical rain-forest. the Leeds Symposium. Leeds Philosophical and Literary Society: 235-242 pp. Leeds.
HOLLOWAY, J. D., 1987.– Macrolepidoptera diversity in the Indo-Australian tropics, geographic, biotopic and taxonomic variations.– Biological Journal of the Linnean Society, 30: 325-341.
HOLLOWAY, J. D., 1993.– The moths of Borneo (part 11); Family Geometridae: Subfamilies Ennominae.– Malayan Nature Journal, 47: 1-309.
HOLLOWAY, J. D., 1996.– The moths of Borneo (part 9); Family Geometridae: Subfamilies Oenochrominae, Desmobathrinae, Geometrinae.– Malayan Nature Journal, 49: 147-326.
HOLLOWAY, J. D., 1997.– The moths of Borneo (part 10); Family Geometridae: Subfamilies Subfamilies Sterrhinae, Larentiinae, Addenda to other subfamilies.– Malayan Nature Journal, 51: 1-242.
HOLLOWAY, J. D., 1998.– The impact of traditional and modern cultivation practices, including forestry, on Lepidoptera diversity in Malaysia and Indonesia.– In D. M. NEWBERY. Dynamics of Tropical Communities: 567-597 pp. Cambridge University Press, London.
INTACHAT, J., HOLLOWAY, J. D. & SPEIGHT, M. R., 1997.– The effects of different forest management practices on geometrid moth populations and their diversity in Peninsular Malaysia.– Journal of Tropical Forest Science, .: 411-430.
KESSLER, M., 2002.– The elevational gradient of Andean plant endemism: varying influences of taxon-specific traits and topography at different taxonomic levels.– Journal of Biogeography, 29: 1159-1165.
KITCHING, R. L., ORR, A. G., THALIB, L., MITCHELL, H., HOPKINS, M. S. & GRAHAM, A. W., 2000.– Moth assemblages as indicators of environmental quality in remnants of upland Australian rain forest.– Journal of Applied Ecology, 37: 284-297.
MCGEACHIE, W. J., 1989.– The effects of moonlight illuminance, temperature and wind speed on light-trap catches of moths.– Bulletin of Entomological Research, 79: 185-192.
PARMESAN, C., 2006.– Ecological and evolutionary responses to recent climate change.– Annual Review of Ecology, Evolution, and Systematics, 37: 637-669.
RAHBECK, C., 1997.– The relationship among area, elevation, and regional species richness in Neotropical birds.– The American Naturalist, 149: 875-902.
SANDERS, N. J. & RAHBEK, C., 2012.– The patterns and causes of elevational diversity gradients.– Ecography, 35: 1-3.
SCHULZE, C. H., 2000.– Auswirkungen anthropogener Störungen auf die Diversität von Herbivoren-Analyse von Nachtfalterzönosen entlang von Habitatgradienten in Ost- Malaysia. 350 pp. Ph. D. Thesis, University of Bayreuth.
SCOBLE, M. J. & HAUSMANN, A., 2007.– Online list of valid and nomenclaturally available names of the Geometridae of the world. Available from http://www.lepbarcoding.org/cl_geometridae.php (accessed 4th February, 2016).
SCOBLE, M. J., 1995.– The Lepidoptera. Form, Function and Diversity: 420 pp. The Natural History Museum and Oxford University Press, Oxford.
SCOBLE, M. J., GASTON, K. J. & CROOK, A., 1995.– Using taxonomic data to estimate species richness in Geometridae.– Journal of the Lepidopterists’ Society, 49: 136-147.
SMETACEK, P., 2008.– Moths recorded from different elevations in Nainital District, Kumaon Himalaya, India.– Bionotes, 10: 5-15.
STORK, N. & BRENDALL, M., 1990.– Variation in the insect fauna of Sulawesi trees with season, altitude and forest type. Insects and the rain forests of South East Asia.– Wallacea, 7: 173-190.
SZUMIK, C., AAGESEN, L., CASAGRANDA, D., ARZAMENDIA, V., BALDO, D., CLAPS, L. E., CUEZZO, F., GOMEZ, J. M. D., DI GIACOMO, A., GIRAUDO, A., GOLOBOFF, P., GRAMAJO, C., KOPUCHIAN, C., KRETZSCHMAR, S., LIZARRALDE, M., MOLINA, A., MOLLERACH, M., NAVARRO, F., NOMDEDEU, S., PANIZZA, A., PEREYRA, V.V., SANDOVAL, M., SCROCCHI, G. & ZULOAGA, F. O., 2012.– Detecting areas of endemism with a taxonomically diverse data set: plants, mammals, reptiles, amphibians, birds and insects from Argentina.– Cladistics, 28: 317-329.
WALIA, V. K., 2005.– Insecta: Lepidoptera: Geometridae: 181-190.– In H. S. MEHTA (ed.). Fauna of Western Himalaya, 2: 358 pp. Zoological Survey of India, Kolkata.
WEBB, C. O., ACKERLY, D. D., MCPEEK, M. A. & DONOGHUE, M. J., 2002.– Phylogenies and community ecology.– Annual Review of Ecological Systematics, 33: 475-505.
WILLIAMS, S., 1997.– Patterns of mammalian species richness in the Australian tropical rainforests: are extinctions during historical contractions of the rainforest the primary determinants of current regional patterns in biodiversity?- Wildlife Research, 24: 513-530.
WILLIAMS, S. E., SHOO, L. P., HENROID, R. & PEARSON, R. G., 2010.– Elevational gradients in species abundance, assemblage structure and energy use of rainforest birds in the Australian Wet Tropics bioregion.– Austral Ecology, 35: 650-664.
YELA, J. L. & HOLYOAK, M., 1997.– Effects of moonlight and meteorological factors on light and bait trap catches of noctuid moths (Lepidoptera: Noctuidae).– Population Ecology, 26: 1283-1290.
ZOU, Y., SANG, W., HAUSMANN, A. & AXMACHER, J. C., 2016.– High phylogenetic diversity is preserved in species- poor high- elevation temperate moth assemblages.– Scientific Reports 6: 23045; doi:10.1038/srep23045(2016).
Appendix I
The complete species account of 168 Geometridae recorded and identified in this study. The current valid name of species is provided after consultation of Lepindex (http://www.nhm.ac.uk/our-science/data/lepindex/). Host plant information is compiled from Host (http://www.nhm.ac.uk/our-science/data/hostplants/) and other relevant species-specific publications. Current altitudinal range from where the species is recorded is provided along with old altitudinal record of the species compiled from SMETACEK (2008), WALIA (2005) and original description of the species published mainly in Proceedings of Zoological Society, London in the years 1835-1897.
| Subfamily | Species | Author, Year | Localities - PA | Altitudedistribution (m) | Old altitudinal distribution (m)(Year of publication: Indian State) | Host plant (Global record) |
| Ennominae | Abraxas irrorata | Moore, 1867 | Govind WLS | 3200-3400 | 2000 (1867: West Bengal) | No Record |
| Ennominae | Abraxas peregrina | Inoue, 1995 | Govind WLS | 1200-1400 | 1600 (1995: Nepal) | No Record |
| Ennominae | Abraxas picaria | Moore, 1867 | Govind WLS, NDBR | 2000-3000, 3000-3400 | 2000 (1868: Uttarakhand) | No Record |
| Ennominae | Abraxas sylvata | Scopoli, 1763 | Govind WLS, Dehradun, NDBR | 600-800, 2600-3400 | 450-2400 (2008: Uttarakhand) | Betulaceae (Betula sp., Corylus sp.), Ulmaceae (Ulmus sp.), Rosaceae (Prunus sp.), Fagaceae (Fagus sp.), Rhamnaceae (Frangula sp.) |
| Ennominae | Alcis variegata | Moore, 1888 | Dehradun | 600-800 | 2062 (1867: West Bengal) / 450-2200 (2008: Uttarakhand) | Fagaceae (Quercus sp.), Rosaceae (Rubus, Malus), Pinaceae (Pinus sp.) as Genus host plant |
| Ennominae | Alcis prosoica | Wehrli, 1943 | NDBR | 2500-2700 | No old altitude record | Fagaceae (Quercus sp.), Rosaceae (Rubus, Malus), Pinaceae (Pinus sp.) as Genus host plant |
| Ennominae | Amblychiaangeronaria | Guenée, 1858 | Dehradun | 600-800 | 450-1500 (2008: Uttarakhand) | Lauraceae |
| Ennominae | Anonychia lativitta | Moore, 1888 | Govind WLS, NDBR | 2600-3000 | 2000 (1888: West Bengal) | No Record |
| Ennominae | Anonychia violacea | Moore, 1888 | Gangotri NP, Govind WLS, NDBR | 1800-3200 | 2000 (1881: West Bengal) | No Record |
| Ennominae | Anonychia exilis | Yazaki, 1994 | NDBR | 2200-2400 | No old altitude record | No Record |
| Ennominae | Arichanna flavinigra | Hampson, 1907 | NDBR | 2200-2600, 3000-3200 | No old altitude record | Ericaceae (Rhododendron sp.) |
| Ennominae | Arichanna picaria | Wileman, 1910 | NDBR | 3000-3200 | No old altitude record | Ericaceae (Rhododendron sp.) |
| Ennominae | Arichanna tenebraria | Moore, 1867 | Govind WLS, NDBR | 2400-2600, 3000-3400 | 2000 (1888: West Bengal) | Ericaceae (Rhododendron sp.) |
| Ennominae | Biston (Buzura) suppressaria | Guenée, 1858 | Askot WLS, Govind WLS, Dehradun | 600-800, 2200-2400 | 450-1500 (2008: Uttarakhand) | Apocynaceae (Carissa carandas), Lauraceae (Cassia auriculata, Cassia fistula, Litsea monopetala), Lythraceae (Lagerstroemia indica), Fabaceae (Acacia catechu), Euphorbiaceae (Aleurites montana), Fabaceae (Bauhinia variegata), Bombacaceae (Bombax ceiba), Theaceae (Camellia sinensis), apindaceae (Dodonaea viscosa), Myrtaceae (Eugenia cumini) |
| Ennominae | Biston falcata | Warren, 1893 | Govind WLS | 2800-3200 | No old altitude record | Polyphagous |
| Ennominae | Buzura bengaliaria | Guenée, 1858 | Govind WLS | 2000-2200 | 1500 (2008: Uttarakhand) | Theaceae (Camellia sinensis) |
| Ennominae | Corymica arnearia | Walker, 1860 | Dehradun | 600-800 | 450-1500 (2008: Uttarakhand) | Lauraceae (Cinnamomum camphora Oriental region) |
| Ennominae | Corymica deducta | Walker, 1866 | Dehradun | 600-800 | 450-1500 (2008: Uttarakhand) | Lauraceae (Alseodaphne semecarpifolia) |
| Ennominae | Corymica specularia(oblongimacula) | Warren, 1896 | Dehradun | 600-800 | 450-1500 (2008: Uttarakhand) | Lauraceae (Lindera praecox recorded from Japan) |
| Ennominae | Dalima patularia | Walker, 1860 | Dehradun | 600-800 | 450-1500 (2008: Uttarakhand) | No Record |
| Ennominae | Dasyboarmiasubpilosa | Warren, 1894 | Dehradun | 600-800 | 450-1500 (2008: Uttarakhand) | Apocynaceae |
| Ennominae | Ectropiscrepuscularia | Duponchel, 1829 | Dehradun | 600-800 | No old altitude record | Pinaceae (Tsuga sp., Abies sp., Pseudotsuga sp., Larix sp., Picea sp.), Cupressaceae (Thuja sp.), Rosaceae (Rubus sp., Sorbus sp.), Betulaceae (Alnus sp., Betula sp.), Salicaceae (Salix sp.) |
| Ennominae | Elphos pardicelata | Walker, 1862 | Govind WLS | 1600-2400 | 2400 (2008: Uttarakhand) | Lauraceae |
| Ennominae | Erebomorphafulguraria | Walker, 1860 | Govind WLS | 2400-2800 | No old altitude record | Theaceae (Camellia sinensis) |
| Ennominae | Fascellinachromataria | Walker, 1860 | Dehradun | 600-800 | 450-1500 (2008: Uttarakhand) | Lauraceae (Alseodaphne semecarpifolia, Cinnamomum zeylanicum, Litsea monopetala, Persea gamblei,Phoebe lanceolata) |
| Ennominae | Fascellina plagiata | Walker, 1866 | Askot WLS, Govind WLS, Dehradun | 600-800, 1200-1400 | 450-2400 (2008: Uttarakhand) | Lauraceae (Alseodaphne sp., Beilschmiedia sp., Cinnamommum sp.) |
| Ennominae | Gnophos albidior | Hampson, 1895 | Govind WLS, NDBR | 1600-1900, 2000-2200 | 1700 (1895: Nagaland) | No Record |
| Ennominae | Heterocalliatemeraria | Swinhoe, 1891 | Govind WLS | 1200-1400, 1800-2000 | 1500 (2008: Uttarakhand) | No Record |
| Ennominae | Heterolocha patalata | Felder, 1874 | NDBR | 2000-2200 | 1500 (2008: Uttarakhand) | No Record |
| Ennominae | Heterolochaphoenicotaeniata | Kollar, 1844 | Govind WLS | 1800-3200 | 2000 (1844: Uttarakhand) | Plumbaginaceae (Plumbago auriculata) |
| Ennominae | Heterostegane sp. | Askot WLS | 600-800 | No old altitude record | Leguminosae | |
| Ennominae | Heterostegane subtessellata | Walker, 1863 | Govind WLS, Dehradun | 600-800, 1400-1600 | 450-1500 (2008: Uttarakhand) | Fabaceae (Acacia sp., Mimosa sp.) |
| Ennominae | Hirasa muscosaria | Walker, 1866 | Govind WLS | 1200-2800 | No old altitude record | Fabaceae (Quercus sp.) |
| Ennominae | Hyperythra lutea | Stoll, 1781 | Govind WLS, Dehradun | 2400-2600 | 600 (2008: Uttarakhand) | Rhamnaceae (Gouania leptostachya), (Ziziphus oenoplia) |
| Ennominae | Hypomecis cineracea | Moore, 1888 | Dehradun | 600-800 | 450-600 (2008: Uttarakhand) | No Record |
| Ennominae | Hypomecis ratotaria | Swinhoe, 1894 | Govind WLS | 1200-2400 | No old altitude record | Betulaceae, Rosaceae, Fagaceae |
| Ennominae | Hyposidra violescens | Hampson, 1895 | Dehradun | 600-800 | 450-1500 (2008: Uttarakhand) | Theaceae (Camellia sinensis) |
| Ennominae | Krananda sp. | Govind WLS, Askot WLS | 600-800, 1200-1400 | No old altitude record | No Record | |
| Ennominae | Leptomiza calcearia | Walker, 1860 | Dehradun | 600-800 | 450-2400 (2008: Uttarakhand) | Rosaceae (Rubus sp.) |
| Ennominae | Lomographa distans | Warren, 1894 | NDBR | 2000-2200 | 1200-2400 (2005: Himachal Pradesh) | Rosaceae (Malus sp.) |
| Ennominae | Lomographa sp.1 | Govind WLS | 1200-1400, 2200- 2400, 2800-3000, 3400-3600 | No old altitude record | Leguminosae, Rosaceae | |
| Ennominae | Lomographa sp.2 | NDBR | 2400-2600 | No old altitude record | Leguminosae, Rosaceae | |
| Ennominae | Loxaspilates hastigera | Butler, 1889 | Govind WLS, Dehradun, NDBR | 600-800, 1200-1400, 3400-3600 | 3142 (1889: Himachal Pradesh) | No Record |
| Ennominae | Loxaspilatesobliquaria | Moore, 1897 | NDBR | 3400-3600 | No old altitude record | No Record |
| Ennominae | Luxiaria phyllosaria | Walker, 1860 | Dehradun | 600-800 | 450-600 (2008: Uttarakhand) | Melastomataceae |
| Ennominae | Luxiaria sp. | Govind WLS | 1200-1400 | No old altitude record | Melastomataceae |
| Ennominae | Medasina albidaria | Walker, 1866 | Govind WLS, Gangotri NP, NDBR | 1400-3200 | 1500 (2008: Uttarakhand) | Pinaceae (Pinus wallichiana), Rosaceae (Prunus sp., Rosa sp.) |
| Ennominae | Medasina cervina | Warren, 1893 | Govind WLS, NDBR | 2000-3200 | No old altitude record | No Record |
| Ennominae | Medasinainterruptaria | Moore, 1867 | Govind WLS | 2400-3000 | No old altitude record | No Record |
| Ennominae | Menophra bicornuta | Inoue, 1990 | Govind WLS | 1400-1600 | 2000 (1990) | Polyphagous |
| Ennominae | Menophrasubplagiata | Walker, 1860 NDBR | Govind WLS, | 1200-1600, 2200-2400 | 1500 (2005: Himachal Pradesh) | Fagaceae (Castanea crenata, Quercus serrata) |
| Ennominae | Odontopera heydena | Swinhoe, 1894 | Govind WLS | 2000-2200, 3000-3200 | 1500 (1894: Meghlaya) | Theaceae (Camellia sinensis) |
| Ennominae | Odontoperakametaria | Felder, 1873 | Govind WLS, NDBR | 1800-2600 | No old altitude record | Fabaceae (Bauhinia variegata), Oleaceae (Jasminium sp.) |
| Ennominae | Odontoperalentiginosaria | Moore, 1867 | Govind WLS | 2200-3200 | 670 (2005: Himachal Pradesh) | No Record |
| Ennominae | Odontoperaobliquaria | Moore, 1867 | Govind WLS | 3200-3400 | No old altitude record | Theaceae (Camellia sinensis) |
| Ennominae | Ophthalmitis herbidaria | Guenée, 1858 | Govind WLS | 1200-1400 | 450-1500 (2008: Uttarakhand) | Flacourtiaceae (Caesaria elliptica) |
| Ennominae | Opthalmitissp. | Askot WLS | 600-800 | No old altitude record | No Record | |
| Ennominae | Opisthograptissulphurea | Butler, 1880 | Govind WLS | 2400-3600 | 2000 (1880: West Bengal) | Rosaceae, Betulaceae |
| Ennominae | Opisthograptistridentifera | Moore, 1888 | Govind WLS | 1800-2000, 2800-3400 | 2000 (1888: Uttarakhand) | Rosaceae, Betulaceae |
| Ennominae | Opisthograptisluteola | Linnaeus, 1758 | NDBR | 2000-2800 | No old altitude record | Betulaceae (Betula sp.), Rosaceae (Malus, Sorbus, Prunus), Salicaceae (Salix) |
| Ennominae | Ourapteryx clara | Butler, 1880 | Dehradun | 600-800 | 450-1500 (2008: Uttarakhand) | No Record |
| Ennominae | Ourapteryx convergens | Warren, 1897 | Govind WLS | 2400-2600 | 2200 (1897: Himachal Pradesh) | No Record |
| Ennominae | Ourapteryx ebuleata | Guenée, 1858 | Govind WLS, Gangotri NP | 1200-1400, 2400- 2600, 3200-3400 | 1500-2400 (2008: Uttarakhand) | Symplocaceae (Symplocos sp.) |
| Ennominae | Ourapteryxsciticaudaria | Walker, 1862 | Govind WLS | 2400-2600 | 1500 (2008: Uttarakhand) | No Record |
| Ennominae | Peratophygahyalinata | Kollar, 1844 | Govind WLS, Dehradun | 600-800, 1200- 1400, 1800-2000 | 2000, 1500 (2005: Himachal Pradesh) / 450-2400 (2008: Uttarakhand) | Hypericaceae (Hypericum sp.) |
| Ennominae | Percnia belluaria | Guenée, 1858 | Govind WLS | 1200-1400, 2000- 2200, 3000-3200 | No old altitude record | Lauraceae |
| Ennominae | Petelia distracta | Walker, 1860 | Dehradun | 600-800 | 450-1500 (2008: Uttarakhand) | Rhamnaceae (Gouania sp., Ziziphus sp., Hovenia sp.) |
| Ennominae | Phthonandriaatrilineata | Butler, 1881 | Govind WLS | 1800-2000 | 850 (1990: West Bengal) / 1500 (2008: Uttarakhand) | Moraceae (Morus sp.) |
| Ennominae | Plagodis inusitaria | Moore, 1867 | Govind WLS | 2800-3000 | No old altitude record | Sapindaceae (Acer sp.), Betulaceae (Betula sp.), Salicaceae (Salix sp.), Pinaceae (Picea sp.) |
| Ennominae | Plagodis reticulata | Warren, 1893 | Govind WLS | 2400-3000 | 1500 (2008: Uttarakhand) | Sapindaceae (Acer sp.), Betulaceae (Betula sp.), Salicaceae (Salix sp.), Pinaceae (Picea sp.) |
| Ennominae | Pseudomizacruentaria | Moore, 1867 | Govind WLS, NDBR | 1200-1400, 2000-2600 | 1500-2400 (2008: Uttarakhand) | No Record |
| Ennominae | Pseudopantherahimaleyica | Kollar, 1848 | Govind WLS | 1600-2600 | 2200 (2005: Uttarakhand), 2000 (1844: Uttarakhand) | Labiatae |
| Ennominae | Psilalcis inceptaria | Walker, 1866 | Govind WLS | 1400-3000 | No old altitude record | Polyphagous |
| Ennominae | Psyra angulifera | Walker, 1866 | Govind WLS, NDBR | 2000-3200 | 2400 (2008: Uttarakhand) | Polyphagous |
| Ennominae | Psyra debilis | Warren, 1888 | Govind WLS, NDBR | 1600-2800, 3200-3400 | 2100(1889: Himachal Pradesh) | Polyphagous |
| Ennominae | Psyra falcipennis | Yazaki, 1994 | Govind WLS | 2200-2600 | No old altitude record | Polyphagous, Rosaceae |
| Ennominae | Psyra similaria | Moore, 1888 | Govind WLS | 2200-3000 | 2000 (1868: Himachal Pradesh) | Polyphagous |
| Ennominae | Psyra crypta | Yazaki, 1994 | NDBR | 2400-2800 | No old altitude record | No Record |
| Ennominae | Semiothisa eleonora | Cramer, 1780 | Dehradun | 600-800 | 450-1500 (2008: Uttarakhand) | No Record |
| Ennominae | Semiothisa nora | Walker, 1861 | Askot WLS, Govind WLS | 2200-2400 | 2000 (1861: West Bengal) | Cupressaceae (Juniperus sp.) |
| Ennominae | Semiothisa sufflata | Guenée, 1858 | Govind WLS | 1200-1600 | No old altitude record | Betulaceae (Alnus sp.), Salicaceae (Salix sp.) |
| Ennominae | Sirinopteryxrufivinctata | Walker, 1862 | Govind WLS | 1600-2200 | 2000 (1863: West Bengal) | No Record |
| Ennominae | Stenorumia ablunata | Guenée, 1858 | NDBR | 2000-2200 | 1500-2400 (2008: Uttarakhand) | Solanaceae (Solanum tuberosum) |
| Ennominae | Stenorumia sp. | Govind WLS | 3000-3200 | No old altitude record | Solanaceae | |
| Ennominae | Tanaoctenia haliaria | Walker, 1861 NDBR | Govind WLS, | 2200-2800 | No old altitude record | Fagaceae |
| Ennominae | Thinopteryxcrocoptera | Kollar, 1844 | Govind WLS, Dehradun | 1200-1400 | 2000 (1844: Uttarakhand) / 450-1500 (2008: Uttarakhand) | Vitaceae (Parthenocissusquinquefolia), (Vitis sp.) |
| Ennominae | Thinopteryx nebulosa | Butler, 1883 | Dehradun | 600-800 | 450-1500 (2008: Uttarakhand) | Vitaceae (Vitis sp., Amelopsis sp.) |
| Ennominae | Xandrames latiferaria | Walker, 1860 | Govind WLS | 1400-1800 | No old altitude record | Lauraceae (Lindera praecox Recorded from Japan) |
| Ennominae | Zamarada symmetra | Fletcher, 1974 | Dehradun | 600-800 | No old altitude record | No Record |
| Ennominae | Zeheba aureatoides | Holloway, 1983 | Askot WLS | 600-800 | 2000 (1887: West Bengal) | No Record |
| Ennominae | Zeheba sp. | Govind WLS | 1200-1400 | No Record | ||
| Ennominae | Ctenognophos sp. | NDBR | 2000-3600 | No Record | ||
| Larentiinae | Chartographa sp. | Govind WLS | 2200-2400 | - | No Record | |
| Larentiinae | Chartographatrigoniplaga | Hampson, 1895 | NDBR | 2600-2800 | No old altitude record | No Record |
| Larentiinae | Cidaria aurata | Moore, 1867 | Govind WLS | 1400-1600, 2200- 2400, 3200-3400 | No old altitude record | Rosaceae |
| Larentiinae | Cidaria catenaria | Moore, 1971 | NDBR | 2400-2800 | No old altitude record | No Record |
| Larentiinae | Colostygiaalbigirata | Kollar, 1844 | Govind WLS, Gangotri NP | 1400-3600 | 2000 (1844: Uttarakhand) | Rubiaceae (Galium sp. recorded from Europe) |
| Larentiinae | Dociravaaequilineata | Walker, 1863 | Govind WLS, Gangotri NP | 2400-2600, 3200-3400 | No old altitude record | Rosaceae |
| Larentiinae | Docirava pudicata | Guenée, 1858 | Govind WLS, NDBR | 1800-2000, 2400- | No old altitude record 2600, 3000-3200 | Rosaceae, Labiatae |
| Larentiinae | Dysstroma sp. | Govind WLS, Gangotri NP | 1400-1600, 2000-2 | 600, 3000-3400 | Betulaceae (Alnus sp.), Salicaceae (Salix sp.), Rosaceae (Sorbus sp., Rubus sp.) | |
| Larentiinae | Ecliptoperapostpallida | Prout, 1940 | Govind WLS, Gangotri NP | 1400-1600, 2200- 2400, 2800-3600 | No old altitude record | Balsaminaceae (Impatiens sp.) |
| Larentiinae | Electrophaes aliena | Butler, 1880 | Askot WLS | 600-800 | 1300 (1940: Himachal Pradesh) / 1500 (2008: Uttarakhand) | No Record |
| Larentiinae | Electrophaes recta | Yazaki, 1994 | Govind WLS, NDBR | 1600-2800, 3200-3400 | No old altitude record | Betulaceae, Rosaceae, Fagaceae |
| Larentiinae | Electrophaesmarginata | Yazaki, 1994 | NDBR | 3000-3200 | No old altitude record | Betulaceae, Rosaceae, Fagaceae |
| Larentiinae | Euphyia coangulata | Prout, 1914 | Govind WLS, NDBR | 1600-3400 | No old altitude record | Betulaceae (Betula sp.), Salicaceae (Salix sp.), Ulmaceae (Ulmus sp.), Caryophyllaceae (Stellaria sp.), Rosaceae (Rubus sp.) |
| Larentiinae | Euphyia stellata | Warren, 1893 | Govind WLS | 2600-3600 | No old altitude record | Betulaceae (Betula sp.), Salicaceae (Salix sp.), Ulmaceae (Ulmus sp.) |
| Larentiinae | Euphyia subangulata | Kollar, 1844 | NDBR | 2400-2600 | 2000 (1844: Uttarakhand) | No Record |
| Larentiinae | Eupithecia rajata | Guenée, 1858 NDBR | Gangotri NP, 2400-2800 | 1400-1600, | 1500 (2008: Uttarakhand) | Pinaceae (Abies sp.), Betulaceae(Alnus sp.) |
| Larentiinae | Eustromachalcoptera | Hampson, 1895 | Govind WLS | 2000-2200, 3200-3600 | 3048 (1895: Sikkim) | Balsaminaceae (Impatiens sp.) |
| Larentiinae | Heterotheradentifasciata | Hampson, 1895 | Govind WLS, NDBR | 1400-2000 | 2100 (1895: Himachal Pradesh) | Pinaceae (Cedrus deodara) |
| Larentiinae | Hydrelia bicolorata | Moore, 1867 | Govind WLS, NDBR | 1800-2400 | No old altitude record | Betulaceae (Betula sp.), Ulmaceae (Ulmus sp.) |
| Larentiinae | Laciniodesplurilinearia | Moore, 1867 | Govind WLS | 2000-2400, 3000-3200 | 2000 (1868: West Bengal) / 2400 (2008: Uttarakhand) | Rubiaceae, Rosaceae, Oleaceae |
| Larentiinae | Larentia nigralbata | Warren, 1888 | NDBR | 2400-2800 | No old altitude record | No Record |
| Larentiinae | Perizoma albofasciata | Moore, 1888 | Govind WLS, Gangotri NP, NDBR | 1400-2600, 3000-3400 | 2000 (1888: Uttarakhand) | No Record |
| Larentiinae | Perizoma seriata | Moore, 1888 | Govind WLS, Gangotri NP, NDBR | 1400-1600, 2200-3600 | 2000 (1888: Uttarakhand) | No Record |
| Larentiinae | Photoscotosiaamplicata | Walker, 1862 | Govind WLS | 2200-3600 | No old altitude record | Rosaceae, Fagaceae |
| Larentiinae | Photoscotosiametachryseis | Hampson, 1896 | Govind WLS, NDBR | 2200-2400, 2800-3200 | No old altitude record | Rosaceae |
| Larentiinae | Photoscotosiaminiosata | Walker, 1862 | Govind WLS, Gangotri NP, NDBR | 1600-3600 | 1500 (2008: Uttarakhand) | Rosaceae (Rubus sp., Rubus ellipticus) |
| Larentiinae | Photoscotosiamultilinea | Warren, 1893 | Govind WLS | 3000-3600 | No old altitude record | Rosaceae |
| Larentiinae | Rheumapteramelanoplagia | Hampson, 1902 | NDBR | 3000-3200 | No old altitude record | Betulaceae (Betula sp., Alnus sp.), Salicaceae (Salix sp.), Berberidaceae (Berberis sp.) |
| Larentiinae | Rheumaptera sp. | Govind WLS | 3400-3600 | No old altitude record | Betulaceae (Betula sp., Alnus sp.), Salicaceae (Salix sp.), Berberidaceae (Berberis sp.) | |
| Larentiinae | Stamnodespamphilata | Felder, 1875 | Govind WLS | 2400-3400 | No old altitude record | Rosaceae |
| Larentiinae | Trichopterigiarufinotata | Butler, 1889 | Govind WLS | 1200-1400 | 2740 (1889: Himachal Pradesh) | Fagaceae (Quercus sp.) |
| Larentiinae | Triphosarubrodotata | Walker, 1862 | Govind WLS, Gangotri NP | 1400-1600, 2400-3400 | No old altitude record | Rosaceae (Pyrus sp., Prunus sp.), Rhamnaceae (Rhamnus sp.) |
| Larentiinae | Venusia crassisigna | Inoue, 1987 | Govind WLS, NDBR | 2400-2600, 3000-3600 | No old altitude record | Betulaceae (Alnus sp., Betula sp.), Salicaceae (Salix sp.), Fagaceae (Quercus sp.), Rosaceae (Malus sp., Sorbus sp.) |
| Larentiinae | Venusia roseicosta | Yazaki, 1994 | Govind WLS | 3000-3600 | No old altitude record | Betulaceae (Alnus sp., Betula sp.), Salicaceae (Salix sp.), Fagaceae (Quercus sp.), Rosaceae (Malus sp., Sorbus sp.) |
| Larentiinae | Xanthorhoe hampsoni | Prout, 1925 | Govind WLS | 3000-3200 | No old altitude record | Polyphagous |
| Larentiinae | Lobogonodes sp. | NDBR | 2300-2500 | No old altitude record | No Record | |
| Larentiinae | Aplocera uniformata | Urbahn, 1971 | NDBR | 2200-2400 | No old altitude record | Guttiferae (Hypericum) as Genus host plant |
| Sterrhinae | Chrysocraspedaolearia | Guenée, 1858 | Govind WLS, Dehradun | 600-800, 2200-2400 | No old altitude record | Myrtaceae (Syzygium cumini) |
| Sterrhinae | Organopodacarnearia | Walker, 1861 | Askot WLS | 600-800 | 1500 (2008: Uttarakhand) | No Record |
| Sterrhinae | Problepsis albidior | Warren, 1899 | Askot WLS, Govind WLS | 600-800, 1600-1800 | 1300 (1899: Himachal Pradesh) | Oleaceae |
| Sterrhinae | Problepsis vulgaris | Butler, 1889 | Askot WLS, Govind WLS, Dehradun | 600-800, 1400-1600 | 733 (1889: Himachal Pradesh) / 450-1500 (2008: Uttarakhand) | No Record |
| Sterrhinae | Rhodometra sacraria | Linnaeus, 1767 | Askot WLS, Govind WLS | 600-800, 2800-3000 | 1500 (2008) | Polygonaceae (Polygonum sp., Rumex sp., Oxygonum sp.), Rosaceae (Malus sp.), Anacardiaceae (Rhus sp.) |
| Sterrhinae | Rhodostrophiapelloniaria | Guenée, 1858 | Govind WLS, Gangotri NP | 1400-1600, 2400-2800 | 1300 (1935: Himachal Pradesh) | No Record |
| Sterrhinae | Rhodostrophiaolivacea | Warren, 1895 | NDBR | 2300-2500 | 2200 (1895: West Bengal) | No Record |
| Sterrhinae | Scopula pulchellata | Fabricius, 1794 | Askot WLS, Govind WLS | 600-800, 1200-1600 | No old altitude record | Plumbaginaceae (Plumbago sp. East Africa) |
| Sterrhinae | Timandra griseata | Petersen 1902 | Govind WLS | 1400-1600 | No old altitude record | Polygonaceae (Polygonum chinense) |
| Sterrhinae | Timandra ruptilinea | Warren, 1897 | Govind WLS | 1400-1600 | No old altitude record | No Record |
| Sterrhinae | Traminda mundissima | Walker, 1861 | Dehradun | 600-800 | 450-1500 (2008: Uttarakhand) | No Record |
| Geometrinae | Agathia carissima | Butler, 1878 | Dehradun | 600-800 | No old altitude record | Asclepiadaceae (Cynanchum wilfordii, Metaplexis japonica) (Recorded from Japan) |
| Geometrinae | Agathia hemithearia | Guenée, 1858 | Govind WLS | 1200-1600 | 1500 (2008: Uttarakhand) | Apocynaceae (Carissa sp., Holarrhena sp., Nerium sp., Tabernaemontana sp.) |
| Geometrinae | Agathia hilarata | Guenée, 1858 | Askot WLS | 600-800 | 1500 (2008: Uttarakhand) | Apocynaceae (Trachelospermum carissa,T. jasminoides) |
| Geometrinae | Agathia lycaenaria | Kollar, 1844 | Dehradun | 600-800 | 2000 (1848: Uttarakhand) / 450-1500 (2008: Uttarakhand) | Apocynaceae (Nerium sp., Nerium oleander, Tabernaemontana heyneana, T. divaricata) |
| Geometrinae | Anisozyga gavissima | Walker, 1861 | Dehradun, Govind WLS | 600-800, 1400-1600 | 1500 (2008: Uttarakhand) | No Record |
| Geometrinae | Aporandriaspecularia | Guenée, 1858 | Askot WLS, Govind WLS, | 600-800, 1200-1400 | 450-1500 (2008: Uttarakhand) | No Record |
| Geometrinae | Hemitheaaquamarina | Hampson, 1895 | Dehradun | 600-800 | 2100 (1895: Uttarakhand) / 450-2400 (2008: Uttarakhand) | No Record |
| Geometrinae | Chlorissadistinctaria | Walker, 1866 | Govind WLS, Gangotri NP | 1200-1600 | 1500 (2008: Uttarakhand) | Apocynaceae |
| Geometrinae | Chlorissa gelida | Butler, 1889 | Askot WLS, Govind WLS | 600-800, 1200- 1400, 1800-2000 | 2100 (1889: Uttarakhand) / 1500 (2008: Uttarakhand) | Apocynaceae (Carissa sp.), Fabaceae (Acacia sp.) |
| Geometrinae | Comibaenaquadrinotata | Butler, 1889 | Gangotri NP | 2600-2800 | 2100 (1889: Uttarakhand) | Fagaceae (Quercus sp.), Rosaceae (Malus sp.), Betulaceae (Betula sp.), Juglandaceae (Juglans sp.), Anacardiaceae (Rhus sp.) |
| Geometrinae | Argyrocosmainductaria | Guenée, 1858 | Govind WLS | 1400-1600 | No old altitude record | No Record |
| Geometrinae | Chloromachiadivapala | Walker, 1861 | Askot WLS, Dehradun | 600-800 | 450-1500 (2008: Uttarakhand) | No Record |
| Geometrinae | Chlororithra fea | Butler, 1890 | Askot WLS | 600-800 | 2100 (1889: Himachal Pradesh) | No Record |
| Geometrinae | Comibaenapictipennis | Butler, 1880 | Govind WLS | 2400-2600 | 2000 (1888: West Bengal) | Fagaceae (Quercus sp.), Myrtaceae (Syzigium sp.) |
| Geometrinae | Comostola subtiliaria | Bremer, 1864 | Govind WLS | 1200-2000 | 1500 (2008: Uttarakhand) | Theaceae (Eurya sp.), Rosaceae (Malus sp.), Adoxaceae (Viburnum sp.) |
| Geometrinae | Dysphaniamilitaris | Linnaeus, 1758 | Dehradun | 600-800 | 450-1500 (2008: Uttarakhand) Oriental region), Carallia brachiata, | Rhizophoraceae (Carallia sp. recorded from (Kandelia candel recorded from Hongkong) |
| Geometrinae | Hemithea tritonaria | Walker, 1863 | Govind WLS | 1400-1600 | 1500 (2008: Uttarakhand) | Fabaceae (Acacia sp.) |
| Geometrinae | Herochroma cristata | Warren, 1894 | Govind WLS, Dehradun | 600-800, 2000-2400 | 450-2400 (2008: Uttarakhand) | Araliaceae (Araliaceae Schefflera recorded from Hongkong) |
| Geometrinae | Herochromaorientalis | Holloway, 1982 | Dehradun | 600-800 | No old altitude record | Araliaceae (Araliaceae Schefflera recorded from Hongkong) |
| Geometrinae | Mixochlora vittata | Moore, 1867 | Govind WLS, NDBR | 2400-2600, 3200-3400 | 1500 (2008: Uttarakhand) | Fabaceae (Quercus sp. specially Quercus incana) |
| Geometrinae | Ornithospilaavicularia | Guenée, 1858 | Govind WLS, Dehradun | 600-800, 2200-2400 | 450-2400 (2008: Uttarakhand) | No Record |
| Geometrinae | Pingasa alba | Swinhoe, 1891 | Govind WLS | 1200-1400 | 1500 (2008: Uttarakhand) | Fabaceae (Dalbergia sp.), Lauraceae (Litsea sp.), Malvaceae (Sterculia sp.) |
| Geometrinae | Pingasa lariaria | Walker, 1860 | Dehradun | 600-800 | No old altitude record | No Record |
| Geometrinae | Pingasa rubicunda | Warren, 1894 | Govind WLS | 1200-1400 | No old altitude record | Dipterocarpaceae (Shorea recorded from Malaysia) |
| Geometrinae | Pingasa ruginaria | Guenée, 1858 | Govind WLS | 1200-1800 | No old altitude record | Fabaceae (Dalbergia monetaria,Xylia xylocarpa), Lauraceae (Litsea elongata), Malvaceae (Sterculia villosa), Rubiaceae (Wendlandia notoniana) |
| Geometrinae | Tanaorhinusreciprocata | Walker, 1861 | Govind WLS | 1200-1400 | 450-2400 (2008: Uttarakhand) | Fagaceae (Quercus recorded from Japan), Quercus cerris, Q. serrata) |
| Geometrinae | Thalassodes veraria | Guenée, 1858 | Askot WLS, Govind WLS, Dehradun, NDBR | 600-800, 2200-3200 | 450-2400 (2008: Uttarakhand) | Fabaceae (Xylia sp.) |
| Desmobathinae | Eumelea rosalia | Stoll, 1781 | Govind WLS, Dehradun | 600-800, 1400-1600 | 450-1500 (2008: Uttarakhand) | Euphorbiaceae (Mallotus sp., Macaranga sp.), Zingiberaceae (Elettaria sp.) |