Abstract: Heliothis nubigeraHerrich-Schäffer, 1851 and Trichoplusia ni (Hübner, [1803]) (Lepidoptera: Noctuidae) are serious polyphagous for economic crops searching for a host to complete their life cycle. The present study was conducted to estimate the relationship between Heliothis nubigera and Trichoplusia ni with Ferula communis L. in two locations, Hassat Al-dab and Almotadal, AlUla Government, Almadina Almunawarah, Saudi Arabia. Genomic DNA was isolated from Heliothis nubigera and Trichoplusia ni for molecular identification. The results indicate that Heliothis nubigera and Trichoplusia ni completes their larval stages during the growth of Ferula communis. Molecular identification by gene sequencing of the target fragment of COI showed 100% identical to Heliothis nubigera with Accession number HM875008.1 and 98.44% identical to Trichoplusia ni with Accession number KX862907.1 using BLAST/N. The study concludes that the target fragment of the COI mitochondrial gene is a useful method for the identification of Heliothis nubigera and Trichoplusia ni. The study recorded that Heliothis nubigera and Trichoplusia ni use Ferula communis as a new host plant located in Hassat Al-dab and Almotadal, AlUla Government, Saudi Arabia.
Keywords: Lepidoptera, Noctuidae, Heliothis nubigera, Trichoplusia ni, Ferula communis, COI mitochondrial gene, interaction insects with plants, Saudi Arabia.
Resumen: Heliothis nubigeraHerrich-Schäffer, 1851 y Trichoplusia ni (Hübner, [1803]) (Lepidoptera: Noctuidae) son graves polífagos de cultivos económicos que buscan un hospedador para completar su ciclo biológico. El presente estudio se llevó a cabo para estimar la relación entre Heliothis nubigera y Trichoplusia ni con Ferula communis L. en dos localidades, Hassat Al-dab y Almotadal, Gobierno de AlUla, Almadina Almunawarah, Arabia Saudí. Se aisló el ADN genómico de Heliothis nubigera y Trichoplusia ni para su identificación molecular. Los resultados indican que Heliothis nubigera y Trichoplusia ni completan sus estadios larvarios durante el crecimiento de Ferula communis. La identificación molecular mediante secuenciación génica del fragmento diana de la COI mostró una identidad del 100% con Heliothis nubigera, con el número de acceso HM875008.1, y del 98,44% con Trichoplusia ni, con el número de acceso KX862907.1. KX862907.1 mediante BLAST/N. El estudio concluye que el fragmento diana del gen mitocondrial COI es un método útil para la identificación de Heliothis nubigera y Trichoplusia ni. El estudio registró que Heliothis nubigera y Trichoplusia ni utilizan Ferula communis como nueva planta huésped localizada en Hassat Al-dab y Almotadal, Gobierno de AlUla, Arabia Saudí.
Palabras clave: Lepidoptera, Noctuidae, Heliothis nubigera, Trichoplusia ni, Ferula communis, gen mitocondrial COI, interacción insectos con plantas, Arabia Saudí.
Artículos
Bioecological and Molecular Studies of Heliothis nubigera Herrich-Schäffer, 1851 and Trichoplusia ni (Hübner, [1803]) associated with Ferula communis L. as a new host in AlUla, Saudi Arabia (Lepidoptera: Noctuidae)
Estudios bioecológicos y moleculares de Heliothis nubigera Herrich-Schäffer, 1851 y Trichoplusia ni (Hübner, [1803]) asociados a Ferula communis L. como nuevo hospedante en AlUla, Arabia Saudita (Lepidoptera: Noctuidae)
Received: 28 October 2023
Accepted: 10 January 2024
Published: 30 June 2024
Interactions between insects and plants have been regulated with many chemical signals leading to multiple behavioral responses (Peyrovi et al. 2011). Many insects use visual cues and olfactory perception stimuli to find their target such as food resources, choice of oviposition site, and recognition of conspecifics for mating depending on the chemical senses, which may be volatile oils or aromatic compounds (Thacker, 2002). Insects search for the plant host by attractant chemicals to complete their life cycle by preferences of female oviposition and acceptance of food for larvae. Acceptance and aversive behavior of insects for the potential hosts depends on the gustatory chemoreceptors in insects that work when an insect contacts a plant with its legs that respond to various chemicals (Ozaki et al. 2011; Zhang et al. 2013).
Ferula communis L. is considered the plant host for some Lepidoptera by female oviposition preferences, larval food acceptance, and taste sensitivity (Sollai et al. 2018). Ferula communis has an unpleasant-smelling, aromatic smell (volatile oils) that attracts insects; it belongs to the family Apiaceae (Umbelliferae), where Ferula L. from Latin means “rod” (Fernch, 1971). It is commonly used as spices, in folklore medicine, in preparation of local drugs, good source of gum-resin, rich source of biologically active compounds, and local drugs (Sahebkar & Iranshahi, 2010; Mohammadhosseini et al. 2019). Ferula communis L. is known for its toxicity, where it causes intoxication of goats, hemorrhagic diathesis in grazing sheep, and neurological toxicity (Shlosberg & Egyed, 1983; Lahmar et al. 2018; Kharchoufa et al. 2021). Saudi Arabia contains four species of Ferula i.e: Ferula sinaica Boiss., Ferula rutbaensis C. C. Towns., Ferula ovina (Boiss) Boiss., and Ferula communis. Ferula species is native to arid regions and detected in desert areas, mountainous regions, central and south-west Asia Mediterranean area (Yaqoob & Nawchoo, 2016).
Heliothinae species are serious polyphagous vegetable pests and have high reproduction rates and contain about 400 species worldwide (Kravchenko, 1984; Mitter et al. 1993; Matov et al. 2008). A variety of common names for Heliothinae are bollworm, flower caterpillar, budworm, pod borer, and earworm (Zalucki et al. 1986; Cunningham & Zalucki, 2014). Since the 1950’s Heliothis Ochsenheimer, 1816 species have been considered the major pests of cotton. Heliothis species is considered the world’s most damaging pest Lepidoptera, where they infest and damage several important economic crops such as zygophyllum, cotton, tomatoes, chickpeas, sunflowers, tobacco Echinops, watermelons, and lonicera (Matthews, 1991; 1999). The common species of Heliothinae are Helicoverpa armigera (Hübner, [1805]), Heliothis nubigeraHerrich-Schäffer, 1851, Heliothis peltigera and the rare species are Heliothis albida (Hampson, 1905) and Schinia scutosa ([Denis & Schiffermüller], 1775) (Amer & El Torkey, 2019). Heliothis nubigera is a polyphagous pest of significant economic importance in agriculture, it attacks many fields and vegetable crops throughout the world (Amer & El Torkey, 2019). Kravchenko et al. (2005) reported that Heliothis nubigera has multivoltine and probably bivoltine with summer aestivation. Heliothis nubigera is known as a common migratory species flying from February to November in North Africa, and Middle East. Larvae of Heliothis nubigera are polyphagous and prefer Solanaceae and Fabaceae families in feeding (Matov et al. 2008). The probability of the successful spread of Heliothis nubigera is because there are many host choices that change through space and time and depend on availability and attractiveness. Heliothis nubigera occupies semi-desert, desert, arid areas, tropical, and subtropical areas. Geographical records of Heliothis nubigera are Algeria, Afghanistan, Cape Verde, Armenia, Cyprus, Israel, Italy, Czechoslovakia, Iran, Saudi Arabia, Jordan, Kazakhstan, Lebanon, Tunisia, Morocco, Turkey, Libya, Malta, Central Asia, India, Yemen, Mauritania, Niger, Sicily, Somalia, Sudan, Oman, Syria, Tadzhikistan, Turkmenistan, and United Arab Emirates (Amer & El Torkey, 2019).
Trichoplusia ni (Hübner, [1803]), is a highly migratory pest, fly long distances, spreads in the spring and early summer, late summer, and fall (Mitchell & Chalfant, 1984; Cervantes et al. 2011; Franklin et al. 2011). This species comes from the crawling behavior of larvae and the preference for cruciferous vegetables (broccoli, cabbages, bok choy) as host plants (Capinera, 2001). It is a polyphagous pest on cruciferous plants and attacks a diverse range of crops, over 160 plants can serve as hosts of cabbage looper larvae such as cotton, weedy species, peas, ornamental plants, beets, celery, tomatoes, and lettuce (Shikano & Isman, 2009; Mpumi et al. 2020). Trichoplusia ni adult is a migratory moth and successfully spread in a wide range of host plants, which change through space and time and depend on availability and attractiveness (Chomchalow, 2003). Trichoplusia ni causes significant economic crop losses either in vegetable production greenhouses or in traditional agriculture, where it has strong resistance to bioinsecticides based on Bacillus thuringiensis toxins (Caron & Myers, 2008). The cabbage looper larvae are marked with irregular holes with variable shapes during feeding on the host leaves (Mpumi et al. 2020). Trichoplusia ni becomes difficult to manage and control due to resistance to many insecticides and broad distribution (Capinera, 2001; Fening et al. 2013). Trichoplusia ni could overwinter in the pupal stage or not (Cervantes et al. 2011).
There are no bioecological studies on the relation of Heliothis nubigera and Trichoplusia ni with Ferula communis as a new host. Thus, the present work aimed to investigate the geographical location of Ferula communis, Heliothis nubigera, and Trichoplusia ni. insert Ferula communis as a new host of Heliothis nubigera and Trichoplusia ni. In addition, use the molecular tool to identify the Heliothis nubigera and Trichoplusia ni. The current work is the first paper discuss the relation of these insects with Ferula.
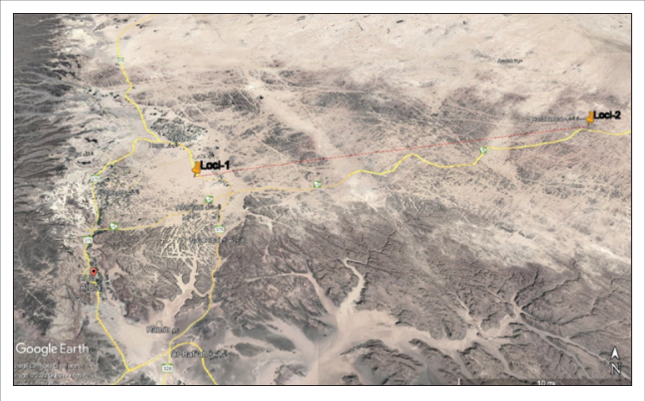
The present work was conducted to evaluate the occurrence of Heliothis nubigera and Trichoplusia ni on Ferula communis from two locations, Almotadal region (26º44’18.7”N 37º59’55.6”E) (17 km away from AlUla Government) and Hasat Al-dab (26º50’29.7”N 38º31’58.9”E) (70 km away from AlUla Government) (Figure 1), Almadina Al-Munawarah, Saudi Arabia. The larvae of Heliothis nubigera and Trichoplusia ni were collected from Ferula communis from January to May during 2019, 2020, and 2021. Some larvae were left in the field for next trip observations. The morphological identification of Heliothis nubigera and Trichoplusia ni was performed (Matov et al. 2008; Mpumi et al. 2020). The Ferula communis was identified by Prof. Dr. Kadry Abdelkhalik (Biology Department, Faculty of Science, Umm Al-Qura University, Saudi Arabia) and by utilizing available literature (Anzalone et al. 1991). The collected larvae were reared in the laboratory until the emergence of the adult stage to confirm the kind of studied insect. The larvae feed upon fresh leaves of Ferula communis and complete development until the pupa stage. The rearing beaker was provided with sand and sawdust to be suitable for the pupa stage. The laboratory specimens were developed into adult moths then, killed, pinned, and dried. Some samples were frozen at -20 for DNA extraction. The insect traps have been used in the field to catch the adult stage.
Genomic DNA was isolated from the legs of Heliothis nubigera and Trichoplusia ni according to the manufacturer’s protocol of the Geneaid kit (gSYNCTM DNA extraction kit quick protocol; catalog number GS100). Genomic DNA was loaded in 1% agarose gel electrophoresis in Tris-borate-EDTA to check the quality of extraction. The polymerase chain reaction (PCR) machine used in this study is a Veriti 96 well thermal cycler, s. n. 2990238536, and Applied biosystems company. Polymerase chain reaction (PCR) was performed with primers LCO(F) (5 - GGT CAA CAA ATC ATA AAG ATA TTG G - 3) and HCO(R) (5 - TAA ACT TCA GGG TGA CCA AAA AAT CA - 3) for target of a fragment of the COI mitochondrial gene of Heliothis nubigera (Folmer et al. 1994; Leite et al. 2014). The primers for the target of a fragment of the COI mitochondrial gene Trichoplusia ni are forward primer (5- TTGTGACAGCCCATGCATTT 3-) and reverse primer (5- GGGGCCCCTAATATAAGAGGTACTA 3-) based on Primer-BLAST as a tool for design target-specific primers (Ye et al., 2012). Amplification reactions were done in the final volume of 25 µL according to procedures (Leite et al. 2014). All reagents used in PCR are GOTag®Green Master Mix, 2x (Ref number: M712)(Promega). The aliquots after amplification were loaded in agarose gel electrophoresis (1% w/v). Purification of amplicons was done by ethanol precipitation. DNA sequencing was done by using the same primer used in amplification by DNA sequencing equipment (3500 Genetic Analyzer, model: 622-0010, serial number: 291-23-030, company: Applied biosystems) at the Center of Excellence in Genomic Medicine Research in King Abdulaziz University, Kingdom of Saudi Arabia. The reagents kit used in the cycle sequence step is the Big Dye Terminator v3.1 cycle sequencing kit (ref number: 4336917) and 5X Sequencing buffer v1.1/3.1 (ref number: 4336697). BlastN tool was used to determine phylogenetic tree construction, sequence alignment, and available homology online at NCBI (Madden, 2002).
Heliothis nubigera larvae move freely on the parts of Ferula communis searching for food during the period of development. Small and large larvae (1st, 2nd, 3rd instar) were observed on the leaves, flowers, follicles, and stems (Figures 2A-F). The breeding season of Heliothis nubigera was accompanied with the growth of Ferula communis, which grows in Hassat Al-dab and Almotadal in January and finishes in May depending on the rain season in this region. Small larvae were fed preferentially on leaves, while large larvae were detected in the leaves and flowers of Ferula communis. Larvae used plant follicles during rest, bad weather, and escaping from enemies (Figure 2D). Larvae used follicles or leaves of Ferula communis as housing for the dormant stage (pupae) in cocoons (field observation). Larvae feed and damage some parts of Ferula communis such as the stem (Figures 3A-E). The larvae of Heliothis nubigera are characterized by small gray caterpillars that undergo a change in mosaic colors during instars to brown, green, and orange with distinct white or yellow longitudinal lines (Figures 2-3). Excretory products of larvae were left in the follicles of Ferula communis (Figures 3B, D, E). The study records the time penetration of Heliothis nubigera larvae to the sand between the roots of Ferula communis or far away from plants (nearly 50 cm) to form pupa at 15 cm depth, where they drop to the ground leaving the feeding site and burrow into the soil and pupate (Figures 4 A-C). Also, the pupa was found enclosed in a network of white threads on the leaves or follicles of Ferula communis in the field (field observations). The adult stage was collected from the bottom of the Ferula communis during daytime hidden between Ferula communis leaves.

In the laboratory, the larvae of Heliothis nubigera were reared in the lab and fed with fresh leaves of Ferula communis. The pupa appears in green color and then develops into a reddish-brown color, where the length is approximately 20 mm. The mature larvae use available sand mixed with sawdust to form a cocoon to complete the pupa stage or sometimes use a covered net for the pupation stage (lab observation). The duration time of pupation is about 18 days to develop into an adult in the lab. The pupa contains a cremaster spine shape. The distance between genital and anal opening slots in the pupa uses as a marker to distinguish between male and female. It is small in male pupa and large in female pupa (Figures 4 C-D). The adult stage of Heliothis nubigera has forewings mosaic greyish ochreous flushed with pale brown, pale reniform stigma dark grey, and a narrow margin containing separated dark spots. The hind wings are creamy white in the anterior part and blackish in the posterior part. The female contains two white rings on the hind wing that were not detected in the adult male (Figures 5A, B).

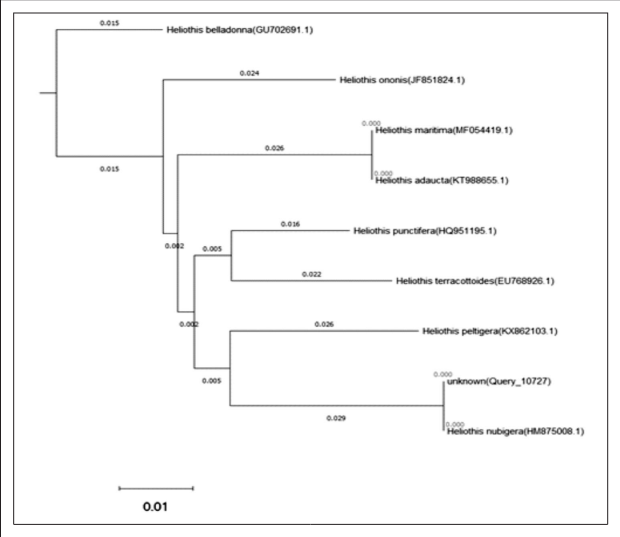
The obtained sequenced regions (one direction for forward primer and one direction for reverse primer) of the target fragment of the COI mitochondrial gene were deposited in BLAST/N programs. The results of query 10727 using BLAST/N program for cytochrome oxidase (COI) gene revealed 100% of percent identical with Heliothis nubigera (Accession no.HM875008.1), 94.71% of percent identical with Heliothis peltigera (Accession no. KX862103.1), 94.52% of percent identical with Heliothis punctifera (Accession no. HQ951195.1), 94.32 % of percent identical with Heliothis terracottoides Rothschild, 1921 (Accession no. EU768926.1), 94.13% of percent identical with Heliothis ononis ([Denis & Schiffermüller], 1775) (Accession no. JF851824.1), 93.93% of percent identical with Heliothis belladonna (Edwards, 1881) (Accession no. GU702691.1), 93.74% of percent identical with Heliothis maritima Graslin, 1855 (Accession no. MF054419.1), and 93.74% of percent identical with Heliothis adaucta Butler, 1878 (Accession no. KT988655.1). The current results confirm the molecular identification of Heliothis nubigera for the current studied samples (Figure 6).





Trichoplusia ni larvae, pupa, and adult on Ferula communis: The breeding season of Trichoplusia ni was associated with the growth of Ferula communis, which depends on the rainy season in the studied locations (Hassat Al-dab and Almotadal) from January to May 2019, 2020, and 2021. Trichoplusia ni larvae occur on the different parts of Ferula communis; on leaves; in follicles, and on flowers (Figures 7A-D). Trichoplusia ni larva has 3º prolegs, therefore it is a looper and it arches its back into a loop when it crawls. Trichoplusia ni larva has four distinct white longitudinal lines on the body. The young larvae were fed on the leaves of small Ferula communis, whereas growing and elder larvae chew larger holes in the stems. The pupa stage was found enclosed in a network of white threads on the leaves or in the follicle of Ferula communis (Figures 8A, B). Trichoplusia ni larvae used Ferula communis follicles during rest, bad weather, escaping from enemies, and as a housing for the dormant stage (pupae) in cocoons (Figures 8A, B). The damagefeeding effect of Trichoplusia ni larvae on the Ferula communis before pupation was noted (Figures 8A, B). Excretory products of Trichoplusia ni larvae were left in the follicles and used for pupation in follicles (Figures 8A, B). The larvae sometimes form a network of white threads (a cocoon) on a covered net or drop into a mixture of sand and sawdust and burrow for the pupation stage (lab observation). Trichoplusia ni larvae (3rd instar) take around three weeks in the laboratory to develop into a pupa (20 mm), which appears initially green in color and then turns dark brown or reddish brown. The pupa end has two large, curved spines and small spines at the end (Figures 8C, D). Trichoplusia ni adult moths are mottled in the lab after pupation time (around 23 days) either in the sand or in the covered net. The forewing of the adult moth is unique and distinguishes Trichoplusia ni from most other Noctuidae. The forewing has silvery white spots, which take U or Y and are often connected with a circle or dot (Figures 9 A, B). The hind wings are blackish in the posterior part and light brown in the anterior portions. The forewing of males is shaded with gray and brown, and the forewings of females are uniformly grayish brown (Figures 9A, B). Long dense hairs occur on the abdomen of the male, while small little hairs on the female abdomen (Figures 9 C, D).
The obtained sequenced regions of the target fragment of the COI mitochondrial gene after DNA sequencing by one direction for reverse primer were deposited in BLAST/N programs. The results of the cytochrome oxidase subunit I (COI) gene in BLAST/N of query 35127 showed similarly 98.44% with Trichoplusia ni (Accession no. KX862907.1). Caenides lota (Evans, 1937) (Accession no. KY019786.1) showed similarly 96.88% with query 35127. Hyperaeschrella nigribasis (Hampson, [1893]) (Accession no. MN131551.1) showed similarly 96.88% with query 35127. The current results confirm the molecular identification of Trichoplusia ni for the current studied samples (Figure 10).

There is no information on the relation between Heliothis nubigera and Trichoplusia ni associated with Ferula communis, which are important for understanding and aiding in predicting the ability of these species to successfully live in different locations, different host plants, and understanding the biology of the pest complex in the target crop. The present study reported the occurrence of small larvae on the small plant leaves that means the chemicals compounds in leaves attract the female for oviposition. Many signals such as chemical, visual cues, and olfactory perception stimuli are involved in the interactions between insects and plants, which lead to various behavioral responses to find their target such as food resources, attractant, antifeedent, recognition for mating, repellent, and deterrent (Nordlund, 1981; Peyrovi et al. 2011). Insects select a specific oviposition site on plant might be due to the type of surface texture, plant morphology, prior experience from larval feeding memory, chemoreceptors on the ovipositor, nectar production, nutrients of plant quality, plant chemical attractiveness, and suitability for offspring development (Cullen, 1969; Adjei-Maafo & Wilson, 1983; Hassan, 1985; Mabbett & Nachapong 1983; Zalucki et al. 1986; Shikano & Isman, 2009). Behavioral adaptations are the first barrier in the insect-plant relationship, where the insect discriminates and senses before nutritional and toxic factors become operative (Dethier, 1973). Some authors support the relationship between oviposition preferences and larval performance (Gripenberg et al. 2010; Sollai et al. 2018).
Insects have a gustatory neuron system that is sensory organs and has an important role in host plant acceptance, host selection, food recognition, and final oviposition choice, where it is located in the lateral and medial styloconic sensilla of larvae and in foretarsi and mouthparts of adults (Sollai et al. 2017a, b, c; Sollai et al. 2018). Abundant evidence suggests that insects searching on the plant host depend on the detection of unique combinations in the host chemicals and the presence of shared attractant chemicals; the oviposition and larval preadaptation occur on plants that are chemically similar to hosts (Berenbaum, 1990). Attractant chemicals in plants (volatile oils or aromatic compounds) and gustatory chemoreceptors in insects are the most considered signals for the insects to female oviposition preferences, food acceptance for larvae, and complete their life cycle (Dethier, 1973; Ozaki et al. 2011; Zhang et al. 2013). Electrophysiological studies established that larvae recognize and respond to complex mixtures of chemicals rather than to a specific host-recognition signal (Dethier, 1970; Berenbaum, 1990). Field bioassay and electroantennogram tests showed the role of green leaf volatile compounds through long-distance olfaction in contact chemoreception (Guerin & Visser, 1980; Guerin & Stadler, 1984). Host recognition received an amount of attention because the oviposition stimulant mechanism for adult females was identified (Stadler & Buser, 1984; Feeny et al. 1988).
The interactions between umbelliferous plants and insects have received considerable attention in insect-plant associations depending on the umbellifer chemistry. Previous reports documented that some orders of insects are represented in association exclusively or almost exclusively with the Umbelliferae species; i.e., lepidopterous larvae are the most conspicuous of umbel-lifer specialists (Berenbaum, 1990). All members of the Umbelliferae family contain essential oils that are primarily terpenoids and phenylpropanoids, flavonoids, and resins in schizogenous canals in all parts of the plant that attract the adult stage for oviposition (Hegnauer, 1971; Berenbaum, 1990). The presence of shared attractant chemicals in Ferula species and a progression of chemosensory responses are responsible for the attraction of insects to specific plants (Berenbaum, 1990). In contrast, the umbellifer chemicals are toxic to nonspecialist insect species. Generalist insects that feed regularly on umbellifers plants might depend on avoidance mechanisms that probability serve as preadaptations to deal with toxins and metabolic adaptation, which means metabolizing toxic material in the plant to non-toxic substances (Camm et al. 1976; Berenbaum, 1990). The principal toxicants in the umbellifer family are the furanocoumarins, and p coumaric acid derivatives, which are activated by ultraviolet light (Berenbaum, 1983). Umbellifer compounds include phenylpropanoids, terpenes, furanocoumarins, coumarins, and flavonoids that have been shown toxic to generalist herbivores (Berenbaum, 1990). Some umbellifer insects have metabolic adaptation, which means the ability to tolerate or metabolize some toxic material in the plant without suffering ill effects such as furanocoumarins (Camm et al. 1976). The furan ring double bond is oxidized through an epoxide intermediate to produce two dicarboxylic acid derivatives that are nonphototoxic metabolites (Ivie et al. 1983; Bull et al. 1986). Other insects metabolize the furanocoumarin xanthotoxin by cytochrome P450 mechanism (Ivie et al. 1983; Bull et al. 1986). Trichoplusia ni shows tolerance of aflatoxin B1, which increases throughout the development (Zeng et al. 2013; Berenbaum et al. 2021). Recent work records the death of some insects on the flowers of Ferula communis L. (Alansari, et al., 2024).
Heliothis nubigera and Trichoplusia ni search for a suitable plant host far away from attack or pesticide spray, where Hasat Al-dab and Almotadal regions are considered open fields or wild-type fields. Small larvae feed preferentially on leaves, while large larvae feed preferentially on leaves and flowers of Ferula communis, which may suggest developmental differences in feeding site preferences. Heliothis species oviposition on its host plants was noted before peak flowering and nectar production, on pre-flowering soybeans, cotton, tobacco, the leaf-whorls of seedling sorghum, leaves of sunflowers at any stage, tomatoes and lucerne at any stage, and pre-tassled maize (Zalucki et al. 1986; Cunningham & Zalucki, 2014). Oviposition by Heliothis sp. includes many cues such as nectar production, plant morphology, chemical attractiveness, nutrients of plant quality, and individual females which vary in their responses to these cues (Adjei-Maafo, 1980; Adjei-Maafo & Wilson, 1983; Mabbett & Nachapong, 1983). Heliothis spp. choose only the plants that will support the development of their larvae to pupation or adulthood (Manjunath et al. 1989). Heliothines may use similar cues to identify their hosts, where they prefer Asteraceae, Fabaceae, Malvaceae, Solanaceae, Poaceae, and Rosaceae families as host plants (Cunningham & Zalucki, 2014). The study concludes that gene sequencing of the target fragment of the COI mitochondrial gene is a useful method for Heliothis nubigera and Trichoplusia ni identification. Also, the study recorded the occurrence of Ferula communis in two locations, Hassat al-dab and Almotadal, AlUla Government, KSA. In addition, insert Ferula communis as a new host for Heliothis nubigera and Trichoplusia ni.
* Autor para la correspondencia / Corresponding author: E-mail: amin_seleem@science.sohag.edu.eg, E-mail: amin_seleem@yahoo.com









