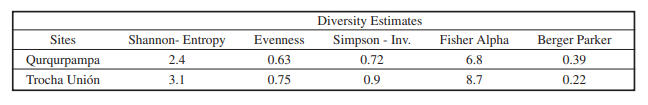Abstract: The subtribe Pronophilina Reuter, 1896 is a group of Papilionoidea that is particularly useful for assessing biodiversity and human-made disturbance, especially in cloud forests. This is because of their high diversity, abundance, distribution patterns, ecological fidelity, and usefulness in formulating conservation policies. A rapid assessment (RAP) was conducted on the Pronophilina in two locations in the upper montane area of Manu National Park, southeastern Peru. The assessment included an evaluation of species richness, faunal composition, similarity, and community structure of Lepidoptera in the area. The two locations were only 30 km apart but in different elevations, ranging from 2800 to 3200 m above sea level. The study recorded a total of 1941 specimens of 45 species, with 18 species (40%) belonging to the genus Pedaliodes Butler, 1867, and as many as 1356 individuals (70%) were found in this genus. Some local species were found to be restricted to very narrow elevation ranges at the forest-puna grassland ecotone. The community structure showed good adjustment with the RAD log-normal model in Trocha Unión and with the Zipf-Mandelbrot model in Qurqurpampa, indicating that the sample obtained is representative of well-preserved uppermost cloud forests. The Sørensen similarity index was relatively low, despite the short distance between the two sites and no apparent geographic isolation. Sampling by “Van Someren-Rydon” traps was confirmed as a very effective method for studying cloud forest species, Satyrinae in particular, to evaluate community structure, diversity, and altitudinal distribution.
Keywords: Lepidoptera, Nymphalidae, Satyrinae, conservation policies, forest disturbance, RBA, RAD, Peru.
Resumen: La subtribu Pronophilina Reuter, 1896 es un grupo de Papilionoidea particularmente útil para evaluar la biodiversidad y las perturbaciones causadas por el hombre, especialmente en los bosques nubosos. Esto se debe a su gran diversidad, abundancia, patrones de distribución, fidelidad ecológica y utilidad para formular políticas de conservación. Se llevó a cabo una evaluación rápida (RAP) de la Pronophilina en dos localidades de la zona montañosa superior del Parque Nacional del Manu, al sureste de Perú. La evaluación incluyó una valoración de la riqueza de especies, la composición faunística, la similitud y la estructura comunitaria de los Lepidoptera de la zona. Las dos localidades estaban separadas por sólo 30 km pero en elevaciones diferentes, entre 2.800 y 3.200 m sobre el nivel del mar. El estudio registró un total de 1941 ejemplares de 45 especies, de las que 18 especies (40%) pertenecían al género Pedaliodes Butler, 1867, y se encontraron hasta 1356 individuos (70%) de este género. Algunas especies locales se encontraron restringidas a rangos de elevación muy estrechos en el ecotono bosquepradera de puna. La estructura de la comunidad mostró un buen ajuste con el modelo log-normal RAD en Trocha Unión y con el modelo Zipf-Mandelbrot en Qurqurpampa, lo que indica que la muestra obtenida es representativa de bosques nubosos superiores bien conservados. El índice de similitud de van Sørensen fue relativamente bajo, a pesar de la corta distancia entre los dos lugares y de no existir un aislamiento geográfico aparente. El muestreo mediante trampas “Van Someren-Rydon” se confirmó como un método muy eficaz para estudiar las especies de los bosques nubosos, Satyrinae en particular, para evaluar la estructura de la comunidad, la diversidad y la distribución altitudinal.
Palabras clave: Lepidoptera, Nymphalidae, Satyrinae, políticas de conservación, perturbación forestal, RBA, RAD, Perú.
Artículos
A Rapid Assessment of Pronophilina Reuter, 1896 community structure and diversity in the uppermost cloud forests near timberline in Manu National Park, Peru (Lepidoptera: Nymphalidae, Satyrinae)
Una evaluación rápida de la estructura y diversidad de la comunidad de Pronophilina Reuter, 1896 en los bosques nublados más altos cerca del límite maderero en el Parque Nacional del Manu, Perú (Lepidoptera: Nymphalidae, Satyrinae)
Received: 10 June 2022
Accepted: 29 January 2024
Published: 30 June 2024
Tropical regions around the world are known for their high biodiversity and endemism of flora and fauna (Hamer & Hill 2004; Lodh & Agarwala, 2016; Meseguer et al. 2022). However, they are under threat of species extinction due to deforestation and habitat loss caused by increasing human demands (Lodh & Agarwala, 2016; Moreira et al. 2023). Among these regions, the Andean tropical montane forest (ATMF) is considered the principal hotspot of global biodiversity, and it has extremely high species richness in most taxonomic groups of plants and animals and contains sites with high speciation rates (Pennigton & Lavin, 2010; Toledo-Aceves et al. 2011; Ledo et al. 2012; Horwath et al. 2019; Bax et al. 2021; Meseguer et al. 2022), and it is also home to many endemic species with restricted geographic ranges (Luebert & Weigend, 2014; Hutter et al. 2017; Pérez-Escobar et al. 2017; Meseguer et al. 2022). However, the ATMF is among the most threatened ecosystems on the planet, with a speedy rate of loss of its natural vegetation cover due to human land-use change, such as agricultural expansion, urbanization, and deforestation (Armenteras et al. 2011; Toledo-Aceves et al. 2011; Quitián et al. 2018; Aide et al. 2019; Horwath et al. 2019; Moreira et al. 2023). This has put many endemic species at risk of extinction due to the bottleneck process in their populations (ToledoAceves et al. 2011; Mahecha-Jiménez et al. 2011; Soh et al. 2019; Bax et al. 2021). Moreover, due to their restricted elevation range, many ATMF plant and animal species have naturally limited distribution in the cloud belt of tropical mountains, ATMF distribution is often considered analogous to an archipelago or islands, with deforestation enhancing the isolation of remaining ATMF fragments (Toledo-Aceves et al. 2011; Mahecha et al. 2019).
Over the last two decades, there has been a gradual loss of tropical forests in Latin America, specifically the ATMF, that have been cleared and replaced with pastures and crops, leading to a permanent loss of forest cover, and due to their high population density, the ATMF has been subjected to intense agricultural activity, which has led to their reduction (Castellanos-Mora & Agudelo-Hz, 2020; Moreira et al. 2023). Extensive areas of cloud forests are being cut down for timber, cleared for agriculture at mid-elevations, and burnt for grazing at high elevations and the act of burning is likely the primary reason behind the widespread and dramatic disappearance of vast portions of lowland and montane tropical forests in South America for centuries (Arroyo-Kalin, 2012; Aide et al. 2019). It is increasingly clear that the impact of human activity in the tropical Andes has been intense and longlasting, with evidence dating back to the pre-Columbian era, and this activity has greatly contributed to the deforestation of the region, particularly at high elevations near timberline, and on a larger scale than what we see today (Loughlin et al. 2018). For the above reasons, ATMF is widely regarded as a conservation priority worldwide because provide important ecosystem services to millions of people (Ivory & Misrachi, 2014; Moreira et al. 2023) due to its critical role in the maintenance of hydrological and nutrient cycles, carbon sequestration and storage, and prevention of erosion and its high biodiversity (Toledo-Aceves et al. 2011; Soh et al. 2019; Bax et al. 2021). For that reason, there is an immediate need for conservation in highly diverse tropical ecosystems, especially in remote areas like mountain habitats. However, the lack of data has been a barrier to effective planning and implementation of conservation efforts (Lodh & Agarwala, 2016).
Conservation International introduced the Rapid Assessment Program (RAP) in 1990 for speciesrich areas, to create baseline data which includes species checklists, records of habitat structure, and of disturbance and anthropogenic pressures in a short time, and vegetation type, etc. (Alonso et al. 2011; Lodh & Agarwala, 2016). To address this, Rapid Biodiversity Assessment (RBA) is a cost- and timeeffective method for exploring the biodiversity of tropical habitats and it is considered a good method for predicting species richness using indicator taxa (Pearson, 1994; Lodh & Agarwala, 2016; Oliveira et al., 2020; Tettey et al. 2020). In addition, the RBAs only use a taxonomic group to indicate the site’s status (Tettey et al. 2020). Butterflies are commonly used to assess the health of both pristine and anthropogenic ecosystems. They have also been utilized to study various aspects of tropical forest ecology in natural, managed, and degraded ecosystems (Tettey et al. 2020). Moreover, butterflies are a promising group of insects for rapid assessment because they are widespread, conspicuous, and easily recognizable, taxonomically well-known, and effective indicators of forest health (Lodh & Agarwala, 2016; Henao-Bañol & Gantiva-Q., 2020; Tettey et al. 2020; Sharma & Sharma, 2021). Also, Lepidoptera diversity is an indicator of biodiversity because they depend on plant diversity and different habitats according to ecosystem ecology (Ferrer-Paris et al. 2013; Lodh & Agarwala, 2016; Tettey et al. 2020; Sharma & Sharma, 2021).
The subtribe Pronophilina Reuter, 1896 is one of the groups of Lepidoptera almost entirely restricted to ATMF region (Casner & Pyrcz, 2010; Mahecha et al. 2019; Pyrcz et al. 2019). Thanks to numerous contributions, over the last two decades, in all aspects of their taxonomy, phylogeny and natural history it turned from one of the least known into one of the better known, if not the best-known group of Andean Lepidoptera, and they present a number of characteristics which make them a group particularly suitable for researching biodiversity (Pyrcz et al. 2009; Casner & Pyrcz, 2010; Pyrcz et al. 2019; Díaz-Suárez et al. 2022). In particular, Pronophilina are one of the most abundant and arguably the most species-rich group among AMTF with well over 600 known species (Lamas et al. 2004; Pyrcz, 2010). They are highly habitat-specific, restricted to well-defined ranges of altitude, an issue which has been studied with considerable focus, and are extremely sensitive to habitat degradation which affects their elevational ranges (Mahecha-Jiménez et al. 2011; Pyrcz & Garlacz, 2012; Díaz-Suárez et al. 2022). Species are often geographically limited, confined to single ranges or valleys, with only a few having wider distributions (Pyrcz et al. 2020). Even though their highly complicated alphataxonomy still require some research, it can be considered as fair, and fast species identification only occasionally presents any serious obstacles (Marín et al. 2017; Padrón et al. 2021). In addition, the spatial distribution patterns of Pronophilina are well-known and fine-tuned both geographically and altitudinally, and this is fundamental from the perspectives of biogeography and population ecology. (Padrón et al. 2021). The presence of Pronophilina is generally high in bamboo thickets, where the adults remain close to their host plants, which are mainly Chusquea Kunth bamboos (Poaceae) (Pyrcz et al. 2009; Mahecha et al. 2019; Pyrcz et al. 2020; Padrón et al. 2021). Finally, and importantly, the use of van Someren-Rydon traps baited with decomposing animal matter guarantees the obtaining of a numerically large, statistically viable sample, frequently representative of local Pronophilina communities within a limited time frame (Pyrcz et al. 2009; Díaz-Suárez et al. 2022).
On a global scale, the number of species in most animal groups, including Lepidoptera, either decreases steadily with increasing elevation or has a peak at mid-elevation (Brehm et al. 2007; Grytnes & Maccain, 2007; Despland et al. 2018). However, recent studies conducted throughout the Andes yield a somewhat different pattern, with several groups of Lepidoptera whose diversity increases with altitude, sometimes reaching the highest values close to timberline at some 2600-3000 m, for example some taxa of the Larentiinae subfamily of Geometridae (Brehm et al. 2003), or to that matter the Pronophilina (Pyrcz, 2010). In this study, we did an RBA of Pronophilina butterfly community structure and diversity along two transects in the uppermost cloud forests near timberline in Manu National Park, Cusco department in SE Peru with the aim to confirm that Pronophilina are good habitat preservation indicators in the Andes.
Study area is situated in the upper Madre de Dios watershed, in the upper montane area of the Manu National Park, located in the department of Cusco in southern Peru (Figure 1). The Manu NP covers 17163 km2 of Amazonian rainforest, montane forests and Andean grasslands between 150 m and 4200 m. The Park was created in 1973 in order to preserve natural habitats of the upper River Manu in the basin of the Madre de Dios. It was recognized as Biosphere Reserve by the UNESCO in 1977, and it is the second largest national park of Peru and corresponds to the one of the top diversity terrestrial areas of the World (Myers et al. 2000).
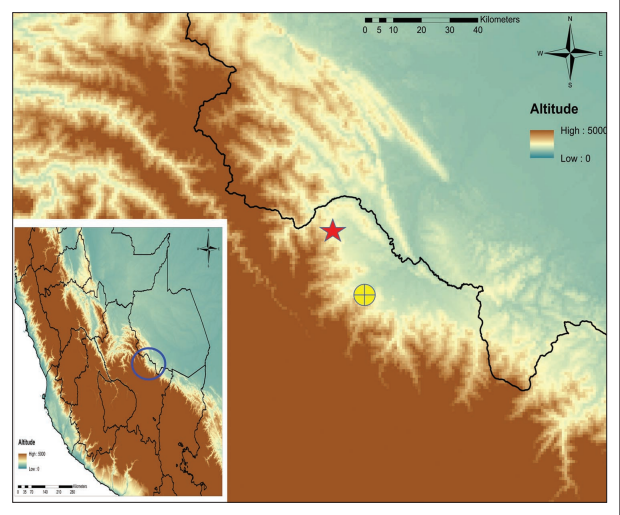
Two altitudinal transects were set at 2800-3500 m.a.s.l. One of them situated 4 km north of the park rangers’ station Qurqurpampa (12º54’ S 71º47’W), along the trails with links it with the village of Callanga. The second transect was set along the so-called trail Trocha Union (13º06’ S 71º36’ W), which links the sector of Tres Cruces with the road going down the Valley of Kosñipata. Both transects covered three kinds of habitats: puna grassland, transitional shrubby forest (tree-line ecotone), and montane forest. Papilionoidea were sampled using entomological nets and 20 Van Someren-Rydon traps, baited with decomposing fish. Traps were separated by 50 m each, starting at grassland forest ecotone at 3200 m and going down to 2800 m. Transect walks and traps were surveyed for five consecutive days each sampling period. Traps were revised daily. Only entomological nets were used in the habitat of puna grassland for four consecutive days, traps were not installed due to poor sampling efficiency demonstrated in previous studies (Pyrcz et al. 2013). Field work was carried out by two people. Field work/sampling took place three times in October (2012), April (2013) and August (2013).
All the material was set and examined in the laboratory of entomology at the Museo de Historia Natural de la Universidad Nacional de San Agustín (MUSA), Arequipa, Perú, and crossed-checked, also against the type specimens kept at the Nature Education Centre of the Jagiellonian University, Kraków, Poland (CEPUJ) and the Museo de Historia Natural de la Universidad Nacional Mayor San Marcos (MUSM), Lima, Perú. Additionally, the types illustrated at Butterflies of America website were examined. Most of the material is deposited at MUSA and voucher specimens in the collections of CEPUJ and MUSM. In the case of specimens requiring more sophisticated methods of taxonomic analysis, male and female genitalia were dissected and compared to type specimens, using standard procedures (Pyrcz et al. 2019).
As a quantitative measure both the abundance, the number of species, doubletons and singletons recorded at each station were given. For the evaluation of alpha diversity three diversity measures: Fisher alpha, Shannon-Entropy and Simpson-Inv. estimative were calculated. As a measure of dominance, the Berger-Parker, and of evenness the Pielou estimative were used (Mahecha-Jiménez et al. 2011; Pyrcz & Garlacz, 2012; Urbano et al. 2018). Moreover, the Hill numbers (q= 0, 1, 2) were estimated for each site as a complementary measure of alpha diversity (Hill, 1973; Moreno et al. 2011; Chao et al. 2014; Marín et al. 2014; Hsieh et al. 2016). The sample coverage and species diversitybased rarefaction/extrapolation (RE) sampling curves were performed by site sampling, in which computes the Hills numbers for rarefied and extrapolated samples with sample completeness up to the coverage value of double the reference sample size (Hsieh et al. 2016). Moreover, the sample-size and coverage-based RE of Hill numbers as a unified framework for estimating species diversity. Likewise, a sample coverage RE curve can compare sites that have different sizes in their samples (Hsieh et al. 2016). Sørensen similarity index was computed to assess the similarity among both sites. Sørensen’s coefficient of similarity was calculated based on presence/absence data for each species. It varies from 0 to 1, and a value close to 1 indicates greater similarity between sites, hence a low diversity ß (Magurran, 2013; Ganamé et al. 2020).
The Relative Abundance Distribution (RAD) was calculated to describe the sampled community (Chao et al. 2015; Cusack et al. 2015). The Akaike’s Information Criteria (AIC) was carried out to select the best RAD model for each site, with subsequent inferences based on the model with the lowest AIC value (Burnham & Anderson, 2002; Cusack et al. 2015). However, the fitting of models to RADs is an intuitive way of representing and comparing community structures, it does not in itself provide information on the relative rank each species occupies within the observed communities (Cusack et al. 2015). All analyses were carried out in R version 3.6.3 (R DEVELOPMENT CORE TEAM, 2020). Species accumulation curves were plotted using the package iNEXT (HSIEH et al. 2013), and analyses of community composition and structure were carried out in package diverse (Guevara et al. 2016) and BiodiversityR (Kindt & Coe, 2005).
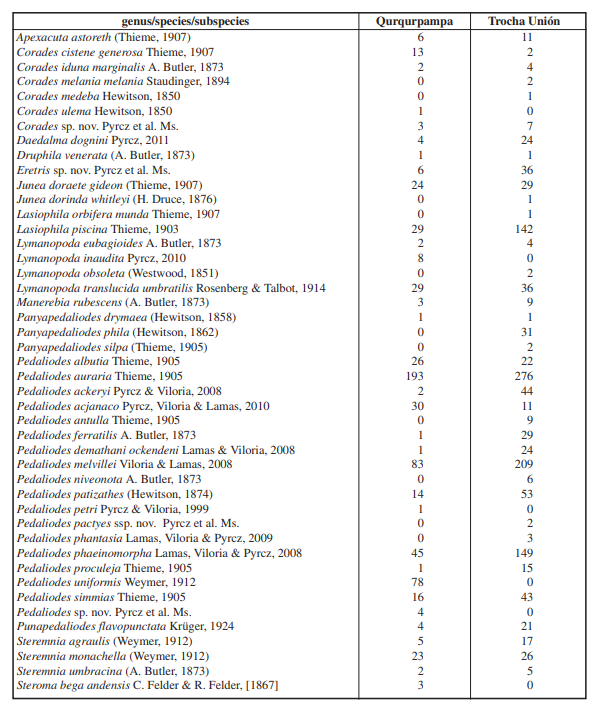
A total of 1941 specimens of the subtribe Pronophilina were collected, 664 along the Qurqurpampa, and 1310 along the Trocha Unión transects respectively. They belong to 15 genera and 45 species. Overall, 18 species (40%) belong to the genus Pedaliodes Butler, 1867. All the species of Pedaliodes account for a total of 1358 individuals which constitutes as much as 70% of the sample. The most abundant species in both transects is Pedaliodes auraria with 469 individuals overall, representing 24% of the sample. The second and third most abundant species also belong to the genus Pedaliodes (P. melvillei - 293, P. pheinomorpha - 194 specimens). Four species singletons and six doubletons were reported (Table I).
The sample of Trocha Unión presents slightly higher values of all principal diversity estimates (in particular Shannon, Simpson, and Fisher alpha) (Table II), and significant difference between the two samples was evidenced (U- Mann-Whitney test: p-value = 0.039). These results are supported by Hill numbers of values for both sites (Table III). On the other hand, there is a major value of dominance in the Qurqurpampa sample as shown by Berger-Parker index.
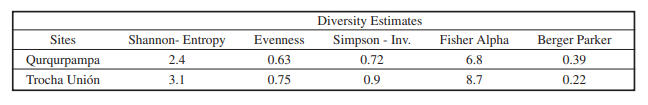
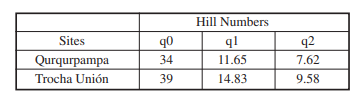
The analysis of sample coverage (Figure 2) and species diversity based on the RE curve (Figure 3) indicate to the sampling effort was appropriate for both sites. Additionally, the species diversity-sample coverage curves allowed to corroborate the good sampling effort (Figure 4). Although it is noteworthy that no curve reached an asymptote, this suggests that a more extensive sampling could potentially increase the number of species in the study area.
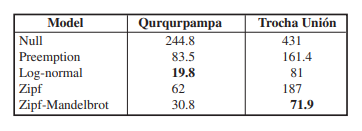
Five models of RAD were tested (null, preemption, lognormal, Zipf and Zipf-Mandelbrot models) (Table IV). According to the AIC criterion, the best adjustment for the sample of Trocha Unión turned out to be the Zipf-Mandelbrot model, whereas for Qurqurpampa the log-normal model (Table IV) (Figure 5). Our results are evidence of a similar pattern from the abundance distribution between dominant and rare species of butterflies in the study area.
Overall, 13 species were found in one of the localities only. Significantly, however, as many as nine of the species reported in only one of the localities were either singletons or doubletons. Among five species which were reported in more individuals, four apparently indicate some faunal dissimilarities between the two sites. In fact, two species were reported in Trocha Unión only, the most southerly site of the two are known to occur most southwards as well, in the well-sampled Acjanaco area, in particular Pedaliodes antulla Thieme, 1905 and Pedaliodes niveonota Butler, 1873. On the other hand, Lymanopoda inaudita Pyrcz, 2004 reported in Qurqupampa only has never been collected in Acjanaco but, on the other hands, it was known from the Abra Malaga-San Luis area situated just northwards of the study area. The most significant absentee in Trocha Unión is Pedaliodes uniformis Weymer, 1912 one of the co-dominant species in Qurqurpampa. The absence of Panyapedaliodes phila (Hewitson, 1862) in Qurqupampa is hardly understandable since it is a very widely distributed, nearly Panandean species found frequently above 2800 m.
Sørensen similarity index is 0.66, which is a rather low value considered that the distance between the two collecting sites is only 30 km, and both are situated on the eastern slopes of the Cordillera de Vilcanota.
An overwhelming majority of the species recorded in both transects are those known to be associated with the highest strata of cloud forests, occurring elsewhere at 2800-3200 m, and found in well-preserved cloud forest. In particular, some species reported here are known to present extremely narrow elevational ranges and are basically restricted to the forest-paramo ecotone. Among such species we should list Corades sp. nov., Apexacuta astoreth (Thieme, 1907), Daedalma dognini Pyrcz, 2011, Pedaliodes acjanacoLamas, Viloria & Pyrcz, 2010, Pedaliodes proculeja Thieme, 1905 and Pedaliodes phantasia Viloria, Lamas & Pyrcz, 2009. On the other hand, some species reported here are generally found at lower elevations than those covered by the transects, in particular Lasiophila orbifera Butler, 1868, Corades medeba Hewitson, 1850, Lymanopoda obsolete (Westwood, [1851]), Pedaliodes petri Pyrcz & Viloria, 1999 and Steroma bega Westwood, [1850] occurring elsewhere at lower elevations, generally at 1800-2600 m. They were recorded either as singletons, doubletons or in three individuals in the latter species, which shows the elevational band covered in this study was above their usual altitudinal range. All five are, however, widespread, and common species and occasionally can be found elsewhere above 2800 m. One species collected on both transects as singletons, Panyapedaliodes drymaea (Hewitson, 1858), is an indicator of secondary areas. Punapedaliodes flavopunctata (Staudinger, 1894) is the only species in the sample which is associated with puna grasslands and is found usually at considerably higher elevations than 3200 m. Its presence in both transects indicates the sampling occasionally took place slightly above the upper forest limit.
This is the first RBA study for Pronophilina butterflies diversity in a sanctuary protected by law in Manu National Park, Cusco department in SE Peru, and our study proves once again that sampling with van Someren-Rydon baited traps is an extremely efficient method in carrying out quick surveys of Lepidoptera diversity and community structure in both lowland and montane rainforest habitats for local or regional studies (De Vries & Walla, 2001; Maicher et al. 2020), as well as in ecological assessments of habitat preservation versus anthropogenic activity within a limited time frame (Aduse-Poku et al. 2012; Whitworth et al. 2016). Such a method is efficient not only by attracting large numbers of individuals which enables to produce viable samples for statistical analysis, but it is also relatively specifically unbiased being equally attractive for all the species of a butterfly community, at least true for cloud forest Satyrinae (Pyrcz et al. 2009; Pyrcz & Garlacz, 2012). Biased sampling method is frequently a problem in similar studies, thus the use a wide array of baits is necessary (Maicher et al. 2020).
This study is, in our opinion, also valuable because it is one of the few to offer a comprehensive comparison of two ecologically similar and geographically marginally set apart localities, allowing a fine-tuned insight into intricate zoogeography of one of topographically most complex areas of the Andes. In fact, the two sampling transects are only 30 km away, both situated on the eastern slopes of the Vilcanota range and are not separated by any noticeable topographical event. And yet, their Sørensen similarity index is surprisingly low, and several interesting differences can be pointed out. For example, the absence of Pedaliodes uniformis the fifth most abundant species overall, and Lymanopoda inaudita along the Trocha Unión transect most likely reflects their south-eastern distribution limit, which is relevant from a zoogeographical perspective as it uncovers a discreet geographical pattern. On the other hand, the absence of Panyapedaliodes phila, a widespread Andean species common at 2600-3000 m along the Qurqurpampa transect is difficult to explain. For comparison, the faunas of Pronophilina of two sites separated by 50 km in the Venezuela Cordillera de Merida were also studied (Pyrcz & Wojtusiak, 2002; Pyrcz & Garlacz, 2012). In that case, the Sørensen similarity index was significantly higher, reaching 0.96. The two samples in Venezuela differed by one species only. Our results indicate much higher beta diversity, understood as species turn-over on a geographical scale, in the Peruvian Andes. Also, the species richness in Venezuela, whereas the altitudinal span covered was much wider, 700 m compared to 400 m in this study, was considerably lower with 22 and 23 species respectively. Higher diversity of two Peruvian localities was also reflected by Shannon and Fisher alpha indices.
Faunal structure of Pronophilina evidenced in this study is similar with other sampled Andean localities, such as Monte Zerpa and El Baho in the Venezuelan Cordillera de Merida (Pyrcz & Wojtusiak, 2002; Pyrcz & Garlacz, 2012), Alto del Zuque-Bogotá (Mahecha-Jiménez et al. 2011) and Frailejonal- La Calera (Díaz-Suárez et al. 2022) in the Colombian Eastern Cordillera; Belmira (Pyrcz et al. 2018) in the Colombian Central Cordillera; Tambito (Pyrcz & Wojtusiak, 1999), and Golondrinas (Pyrcz et al. 2009) on the Pacific slopes in Colombia and Ecuador, and Chachapoyas in northern Peru (Pyrcz, 2004). All the communities of Pronophilina are characterized by the dominance, in terms of species-richness and abundance, of Pedaliodes¸ with one to three strong dominant or co-dominant species, and a comparative under-representation of other genera in the sample. Such a faunal structure is not an artefact of the method of sampling nor seems to be related with the geographic setting of sampling locality or altitudinal span covered. The species dominance of Pedaliodes in the sample is not surprising given that it is also the most species-rich genus of Pronophilina accounting for nearly 50% of all known species in this subtribe (Pyrcz, 2004). However, the dominance of Pedaliodes in terms of abundance is less straightforward to interpret. Also, in the less diverse communities the dominance of a single species of Pedaliodes is more accentuated, which is particularly striking in the case of El Baho sample where Pedaliodes minabilisPyrcz, 2008 constitutes as much as 53% of the sample (Pyrcz & Garlacz, 2012). Interestingly, dominant species of Pedaliodes in each locality belong to completely unrelated clades of the genus (Pyrcz et al. in prep.).
Our results suggest good preservation of the patches of uppermost forest where both samplings were carried out. This is shown, first, by the presence of species indicators of well-preserved habitats, in particular Corades sp. nov., Apexacuta astoreth (Thieme, 1907), Lymanopoda inauditaPyrcz, 2010, or Pedaliodes phaeinomorpha Viloria, Lamas & Pyrcz, 2008, the latter one of the co-dominant species in the study area, elsewhere extremely localized and rare, for example along the nearby Acjanaco - Manu Road due to high disturbance of forest cover at 3000-3200 m. At the same time, very few species related with disturbed forest habitats were found in the study area, in particular only one individual of Panyapedaliodes drymaea (Hewitson, 1858) which is one of the most prominent indicators of high anthropogenic disturbance, very few specimens of Steremnia umbracina (Butler, 1873), another species related with secondary habitats, and none of Pedaliodes palaepolis (Hewitson, 1878), again a secondary forest indicator species (Pyrcz, 2004; Mahecha-Jiménez et al. 2011; Díaz-Suárez et al. 2022). Second, the RAD models fit in Trocha Unión (Zipf-Mandelbrot) and Qurqurpampa (log-normal) are also indicative of undisturbed, natural, varied and mature communities (may, 1975; Hill et al. 1995; Hill & Hamer, 1998; Marimon et al. 2015; Harterreiten-Souza et al. 2020; Nallis, 2021). In fact, the Zipf-Mandelbrot model assumes that, in the succession processes, late colonizing species have a specialized niche and thus are more difficult to find, while early colonizing species are more generalist and dominate in abundance regardless of the time of succession (Magurran, 2013; Harterreiten-Souza et al. 2020; Nallis, 2021). This model also assumes that colonization is initially stochastic depending on the regional abundance of generalist species, but that mature communities are determined according to the niche of late species (Nallis, 2021). The log-normal model assumes that ecological communities will have many species with intermediate abundance and few rare or common species (Schluter & Ricklefs, 1993; Harterreiten-Souza et al. 2020; Nallis, 2021). It has been argued that the use of RAD is a much more reliable method in comparing communities than non-parametric indices of diversity (Kim et al. 2013; Sæther et al. 2013). The latter, frequently used in the studies of biodiversity are shown to oversimplify the complex structural nature of communities and are not independent of sampling intensity, thus on sample size as well (Mouillot & Lepretre, 2000; Dornelas et al. 2011). Interestingly, this does not seem to be the case in our study. Most indices show closely similar values despite the fact that the Trocha Unión sample contains two times as many records as Qurqurpampa.
In sum, our study confirms that Pronophilina, thanks to their ecological and also behavioural qualities, such as habitat fidelity coupled with low adult vagility, and high sensibility to human made disturbance, which immediately express in their community structure, can be explored in the assessment of cloud forest state of preservation, and thus in formulating future conservation policies. In addition, to their other features, such as easy high species richness, standard sampling procedures, rapid data gathering protocols, and good knowledge of taxonomy among others, they surpass as potential habitat preservation indicators any other group of Andean Lepidoptera.
This research was supported by NCN grant Harmonia-10 2018/30/M/NZ8/00293 “Evolutionary biogeography and diversification of the predominantly Andean butterfly subtribe Pronophilina (Nymphalidae, Satyrinae) based on phylogenetic data generated using modern molecular methods”. Taxonomical studies, genital dissections and barcoding was carried out in the laboratory of the Nature Education Centre, Jagiellonian University, Kraków, Poland (CEPUJ). Field work was performed under authorization R. J. Nº 0010-2012 SERNANP-SNM issued by the “Servicio Nacional de Áreas Naturales Protegidas por el Estado” (SERNANP), Ministerio del Ambiente, Peru.
* Autor para la correspondencia / Corresponding author: oscarmahecha23@gmail.com