Artículos
Evaluation of the type of bait in catching Nymphalidae in Van Someren-Rydon traps in the National Park Yanachaga-Chemillén-Paujil sector, Pasco, Peru (Lepidoptera: Papilionoidea)
Evaluación del tipo de cebo-atrayentes en la captura de Nymphalidae en trampas tipo Van Someren-Rydon en el Parque Nacional Yanachaga-Chemillén-Sector Paujil, Pasco, Perú (Lepidoptera: Papilionoidea)
Evaluation of the type of bait in catching Nymphalidae in Van Someren-Rydon traps in the National Park Yanachaga-Chemillén-Paujil sector, Pasco, Peru (Lepidoptera: Papilionoidea)
Shilap Revista de Lepidopterología, vol. 52, no. 206, pp. 355-367, 2024
Sociedad Hispano-Luso-Americana de Lepidopterología
Received: 10 December 2022
Accepted: 14 August 2023
Published: 30 June 2024
Abstract: The use of baited traps for the study of Lepidoptera has increased in recent decades, offering advantages and disadvantages compared to direct methods of capture. However, studies evaluating the attractiveness of different types of different baits are scarce, and published works are limited to using the standard bait consisting of fermented fruit with sugar. This study evaluates the variation in the attractiveness and effectiveness of three types of bait (fermented fruit, rotting fish, and human feces) in the capture of Lepidoptera in Van Someren-Rydon traps (VSR) in the National Park Yanachaga-Chemillén (PNYCh), sector Paujil, by the use of VSR located in sites with different canopy cover. Certain species were found to visit all three types of bait, e.g. Archaeoprepona demophon (Linnaeus, 1758) and Diaethria clymena (Cramer, 1775), others only two of these, e. g. Memphis glauce (C. Felder & R. Felder, 1862), Memphis moruus (Fabricius, 1775), Adelpha mesentina (Cramer, 1777), Temenis laothoe (Cramer, 1777), and some were unique to one type of bait, e. g. Zaretis itys (Cramer, 1777), Ancyluris etias (Saunders, 1859), Catoblepia berecynthia (Cramer, 1777), Catonephele numilia (Cramer, 1775). Furthermore, it was found that the fruit bait presents significant differences concerning the rotting fish and feces baits, however, it presented the least number of individuals and species attracted. Meanwhile, rotting fish and feces presented a similar attractiveness in species number and quantity of individuals. The results suggest the bait’s importance in capturing Lepidoptera and, consequently, their potential relevance in the elaboration of subsequent ecological studies.
Keywords: Lepidoptera, Papilionoidea, Nymphalidae, biological monitoring, diversity, faunistic studies, frugivorous, sampling techniques, tropical forest, Peru.
Resumen: El uso de trampas con cebo para el estudio de los Lepidoptera se ha incrementado en las últimas décadas, ofreciendo ventajas e inconvenientes en comparación con los métodos directos de captura. Sin embargo, los estudios que evalúan el atractivo de los distintos tipos de cebos son escasos, y los trabajos publicados se limitan a utilizar el cebo estándar consistente en fruta fermentada con azúcar. Este estudio evalúa la variación en el atractivo y la eficacia de tres tipos de cebo (fruta fermentada, pescado podrido y heces humanas) en la captura de Lepidoptera en trampas Van Someren-Rydon (VSR) en el Parque Nacional Yanachaga-Chemillén (PNYCh), sector Paujil, mediante el uso de VSR situadas en lugares con diferente cobertura de copas. Se encontró que algunas especies visitaban los tres tipos de cebo, por ejemplo Archaeoprepona demophon (Linnaeus, 1758) y Diaethria clymena (Cramer, 1775), otras sólo dos de ellos, por ejemplo Memphis glauce (C. Felder & R. Felder, 1862), Memphis moruus (Fabricius, 1775), Adelpha mesentina (Cramer, 1777), Temenis laothoe (Cramer, 1777), y algunos eran exclusivos de un tipo de cebo, p. ej. Zaretis itys (Cramer, 1777), Ancyluris etias (Saunders, 1859), Catoblepia berecynthia (Cramer, 1777), Catonephele numilia (Cramer, 1775). Además, se comprobó que el cebo de fruta presenta diferencias significativas respecto a los cebos de pescado podrido y heces, sin embargo, presentó el menor número de individuos y especies atraídos. Mientras, el pescado podrido y las heces presentaron un atractivo similar en número de especies y cantidad de individuos. Los resultados sugieren la importancia de los cebos en la captura de Lepidoptera y, en consecuencia, su relevancia potencial en la elaboración de estudios ecológicos posteriores.
Palabras clave: Lepidoptera, Papilionoidea, Nymphalidae, diversidad, estudios faunísticos, frugívoros, monitoreo biológico, técnicas de muestreo bosque tropical, Perú.
Introduction
Protected areas have a fundamental role in the conservation of interactions and populations of various organisms, as well as in the provision of ecosystem services essential for society. (Figgis et al. 2015). In this way, it is evident the need for and importance of monitoring the state of these areas and studying their internal ecological dynamics to understand how they respond to the constant changes caused by the continuous growth of the human population. However, monitoring the entire diversity of a tropical forest is an impossible task, leading to certain groups being selected based on their response to environmental changes, ease of capture, short generation times, identification, or other desirable characteristics, facilitating the monitoring (Whitworth et al. 2018). Because Lepidoptera are sensitive to changes in their habitat and for their diversity of ecological roles, they have been considered good bioindicators (Bonebrake et al. 2010; Whitworth et al. 2018), thus becoming ideal organisms for numerous ecological studies, for example, have been extensively studied in topics about fragmentation (Ockinger et al. 2009; Jew et al. 2015; Filgueiras et al. 2019), edge effect (Bossart & Opuni-Frimpong, 2009; Sung-Soo & Tae-Sung, 2018), in comparison to habitats with different level of disturbance (Devries et al. 1997; Ribeiro & Freitas, 2012; Barbosa et al. 2017) and in monitoring programs (Pozo et al. 2008; Costa-Pereira et al. 2013).
The study of Lepidoptera may be carried out through several methodologies, where the most frequent is the establishment of linear transects in open areas (grassland, heathlands, dunes, clearings, and less dense areas in general) capturing adult individuals, storing them, and processing them as reported by Van Swaay et al. (2015). However, this methodology has several disadvantages, for example, it is strongly influenced by the researcher’s experience in detecting and capturing individuals (Ebert, 1969; Brown, 1972; Iserhard et al. 2013). Furthermore, the vertical gradient of diversity cannot be effectively sampled, and sometimes independent samples cannot be established for comparison in ecological studies (Freitas et al. 2014). In this way, in recent decades the use of barley traps for the study of Lepidoptera has increased since they do not suffer from the same inconveniences (Freitas et al. 2014).
For ecological and comparative studies, the most used bait traps are the Van Someren-Rydon (VSR), which consist of a cylinder of black or white tulle fabric with the top sealed and the bottom open where the bait is held by a base attached to the trap (Rydon, 1964; Andrade-C et al. 2013; Freitas et al. 2014; Purwanto et al. 2015; Bokelaar, 2017). The principle of operation of the trap lies in the attraction of Lepidoptera by the bait they feed on the bottom, due to the small diameter of the cylinder that limits the maneuverability to fly, the Lepidoptera ascends and is trapped inside (Bokelaar, 2017). It should be mentioned that not all Lepidoptera can be caught using traps, constituting a major disadvantage for diversity and composition studies (Devries et al. 1997).
Various types of baits can be used in VSR traps, including fermented fruits, rotting fish, decaying seafood, and feces (Andrade-C et al. 2013; Freitas et al. 2014; Checa et al. 2018; Olarte-Quiñonez et al. 2021). Preferably fermented bananas mixed with sugar are used and it is this type of bait that predominates in diversity studies in the Neotropical region (Hughes et al. 1998; Aduse-Poku et al. 2012; Martins et al. 2017; Panjaitan et al. 2019). The other baits are usually used in a complementary way to the fruit bait, in addition, they are less used by its strong and unpleasant odor, by the difficulty of standardization in the preparation, because it attracts other groups of unwanted insects, but the negative results in the use of alternative baits are rarely reported by the authors (Freitas et al. 2014). The baits effectiveness lies in supplying salts and minerals from the diet of Lepidoptera, which are relatively limited under natural conditions and affect key aspects of reproduction (number of eggs, the viability of the pups, etc.) and survival of the individual (Geister et al. 2008). Moreover, each type of bait is preferred by certain groups of Lepidoptera and even certain genera are only attracted by a particular type of bait, e. g. the genus Adelpha Hübner, [1819] and the pronophilines in general (Olarte-Quiñonez et al. 2021), is attracted by rotting fish, the feces attract many Charaxinae and nectarivores Lepidoptera (Molleman et al. 2005; Freitas et al. 2014; Fucilini, 2014). Nevertheless, the only study found where a comparison was made on the attractiveness of the baits was carried out by Bokelaar (2017), finding that there are no significant differences in the preference between baits, but it is noted that in terms of abundances that the rotting fish as bait was the least effective bait and that the effectiveness of the traps showed variations between the different types of plant cover (Bokelaar, 2017).
In this study, the variation in attractiveness and effectiveness of three types of baits (rotting fish, human feces, and fermented fruit) in the capture of Lepidoptera in VSR traps in the YanachagaChemillén National Park (PNYCh) - Paujil sector, was evaluated. In this way, with our study, we may contribute to the study about using bait traps for future ecological studies and monitoring of protected areas and consequently provide relevant information for conservation plans and strategies.
Materials and method
Study Area
Figure 1.
Location of the study área.
The work was carried out in the Yanachaga-Chemillén National Park (PNYCh), located in the department of Pasco, Peru. Specifically, the study was carried out in the Paujil sector (Figure 1). The area is typically a tropical humid forest and is located at an altitude between 300 and 800 a.s.l. with average temperatures of 25ºC and the rainy season from November to April. The Paujil sector has several types of vegetation, for example, gallery forests where trees of the species Ficus spp. (Linnaeus), Inga marginate (Willd.), Zygia longifolia ((Humb. & Bonp. ex Willd.) Britton & Rose) dominate, alluvial terraces with relatively flat terrain, characterized by primary canopy-dominated forests of the Fabaceae, Lauraceae, Lecythidaceae, Moraceae, Myristicaceae and Malvaceae families, in the middle stratum are found Annonaceae, Fabaceae, Malvaceae, Moraceae, Myristicaceae and Rubiaceae. In the shrub layer predominate Melastomataceae, Piperaceae, and Rubiaceae. Finally, in the area there are low and medium hills where a transition from the change of vegetation to the montane forest is observed (Laura, 2007).
Methods of Capture and Identification
Three linear transects were established in three types of vegetation cover in the Paujil sector: 1) in the paths to the lookout, 2) near the banks of the Iscozacín river, and 3) in the surrounding primary forest near a small watercourse, following the methodology proposed by Andrade-C et al. (2013). The sampling period comprised 8 effective days and was carried out in the rainy season in the sector in the month of Februar.
In the transect near the small watercourse, Pourouma guianensis (Aubl.), P. minor (Benoist), Cedrelinga sp. and Socratea salazarii (H. E. Moore) predominated in the tree stratum and easily exceeded the 15 m of height, the shrub layer was predominantly represented by Schizocalyx sp. and Zygia sp.; and, finally, the herbaceous stratum was represented by Maranthaceae and Cyclanthaceae families. In the transect of the lookout, the vegetation was scrubbier with respect to the surrounding premontane forest, dominating species of Wettinia sp., Chrysophyllum sp., and several species of Ericaceae. It is emphasized that in the herbaceous stratum dominated by Iriartella sp. and Pariana (Aubl.), the height of the canopy was between 8 to 15 m and the canopy was more open. Finally, the transect located on the banks of the Iscozacín river presented open areas, numerous bare rocks, and exposed sand regions, features of a gallery forest, where Calliandra angustifolia (Spruce ex Beth.) was the dominant species reaching the 5 to 8 m of height.
In this way, one VSR trap was placed at a height between 2 and 5 m from the ground, depending on the architecture of the vegetation in the area, with a different bait for every two traps, i. e. two traps had fermented fruit bait, two traps had rotting fish, and two traps had human feces. Each trap was checked every 10 to 30 minutes to minimize the risk of Lepidoptera escaping, from 9:00 to 15:30. The captured individuals were stored on parchment paper marked with the date, time, and bait of the trap. Later they were kept in a cold chain until their assembly and the identification was made with the taxonomic keys from Le Crom et al. (2004), Willmott (2003), Pinzón-C (2006), Garwood & Jaramillo (2017 a, b, c, d), Rengifo & Montero (2010), Gallice (2016) and with the database of type specimens Butterflies of America (Warren et al. 2019).
Data Analysis
Abundance histograms were made of the number of individuals caught and of the species recorded in relation to the type of bait in the R software (R Core Team, 2020). Additionally, a linearized correspondence analysis (DCA) was performed to evaluate the association of the species with the type of bait. Because the data is not normal, a Kruskal-Wallis analysis was performed to assess whether there are significant differences between bait types. Subsequently, a post hoc test of Dunn was conducted to determine which baits presented these differences (Sheskin, 2003). These tests were performed on the PAST software (Hammer et al. 2001) (Figure 2).
Results
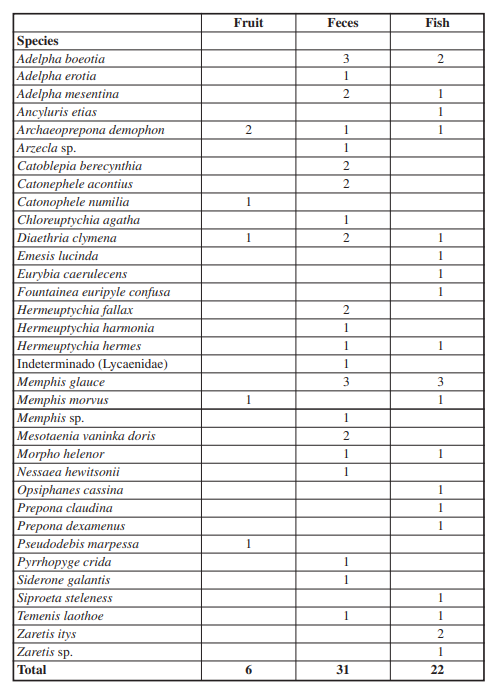
In the 8 days of sampling, 60 individuals were captured with an effort of 54 hours/trap. The individuals were distributed in four families: Hesperiidae, Lycaenidae, Riodinidae, and Nymphalidae. The family Nymphalidae had the most records (54 individuals and 26 species), followed by Riodinidae (3 individuals, 3 species), Lycaenidae (2 individuals, 2 species) and finally Hesperiidae (1 individual, 1 species) (Figure 2C). For the lockout site (premontane forest), 37 specimens were captured, for the transect near the small watercourse (primary forest) 9 specimens were captured, and for the site located on the banks of the river Iscozacín (gallery forest) 14 specimens were captured. In the lockout transect, the feces bait was the more effective with 19 specimens collected, followed by rotting fish with 14 specimens collected, and finally the fermented fruits bait with 4 specimens collected. Besides, for the banks of the river Iscozacín transect, the feces bait was the more effective with 8 specimens collected, followed by rotting fish with 4 specimens collected, and finally the fermented fruits bait with 2 specimens collected. Finally, for the transect near the small watercourse, the rotting fish was the more effective bait with 5 specimens collected, followed by feces bait with 4 specimens collected, and the fermented fruits bait did not show specimens collected. In total, 33 species were collected in the VSR traps, representing 32.7% of the diversity as found by Vásquez (in prep.) and 25.6% according to the study by Grados et al (2008). It should be noted that 17 species are new records for the PNYCh-Sector Paujil (Figure 4), which are not included in the work of Grados et al. (2008).
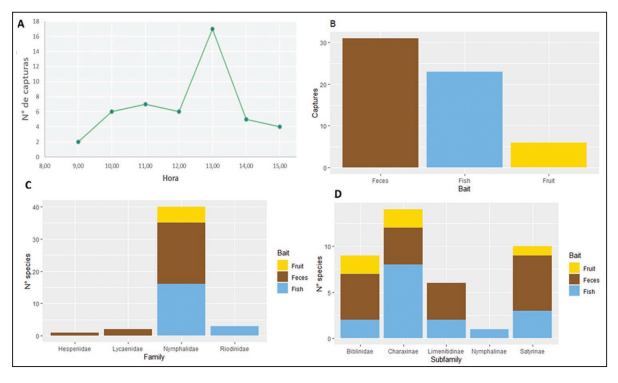
Figure 2.
Effectiveness of different types of baits in Van Someren-Rydon traps. A) Variation in catch rate throughout the day. B) Number of individuals caught relative to bait type. C) Number of species from each family caught in each bait type. D) Number of species from each Nymphalidae subfamily caught in each bait type
The most abundant genera were Adelpha Hübner, [1819] with 8 individuals captured and Memphis Hübner, [1819] with 9 individuals. The species captured for these two genera were 3 species for each, highlighting that Adelpha capucinus (Walch, 1775) and Memphis glauce (C. Felder & R. Felder, 1862) are the species with the highest abundance, with 5 and 6 individuals, respectively. It is important to note that most individuals of these two genera were not caught using fermented fruit bait, except for one individual from Memphis moruus (W. Comstock, 1961). Moreover, catches of the genus Adelpha were more frequent in the viewing sector (premontane forest). Other frequent species were Archaeoprepona demophon (Linnaeus, 1758) and Diaethria clymena (Cramer, 1775) with 4 individuals each, where this late species was collected only in the banks of the Iscozacín river (gallery forest).
To respect to the effectiveness of the baits, it was found that there is an increase in the catch rate in the VSR traps from noon for the three sites, extending for one hour and decreasing in the subsequent hours (Figure 2A). By discriminating the effectiveness of baits by families it is evident that individuals captured of Hesperiidae and Lycaenidae are attracted exclusively by feces, while individuals of Riodinidae are attracted by decaying fish. On the other hand, the family Nymphalidae is attracted by the three types of bait, however, more individuals are recorded in fish baits and feces (Figure 2C). At the subfamily level within Nymphalidae, it was observed that baits attracted individuals from 5 subfamilies, with feces being the bait that recorded the highest number of species within Biblidinae, Limenitidinae, and Satyrinae, while rotting fish recorded the highest number of species within Charaxinae (Figure 2D). It is worth mentioning that the fruit recorded species within Biblininae, Charaxinae, and Satyrinae, but did not attract species from the subfamily Limenitidinae; the decomposing fish attracted species from the 5 subfamilies caught in the VSR traps; the fish and fruit recorded the same number of catches in the subfamily Biblininae. Finally, only one species, Siproeta stelenes (Linnaeus, 1758), of Nymphalinae was collected and this one was attracted by the decomposing fish (Figure 2D).
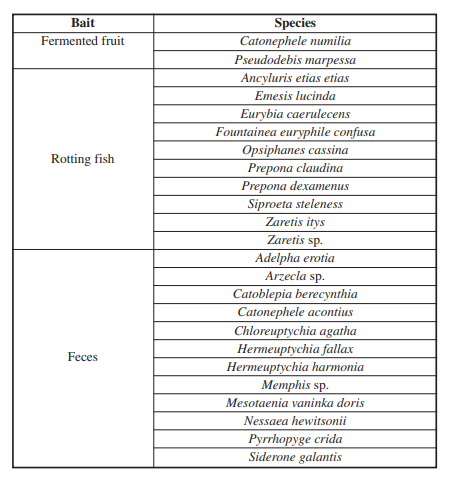
Also, a difference was observed in the capture of Lepidoptera depending on the type of bait used (Kruskal-Wallis p-value=7,5*10-5; α=0,05), where the largest number of individuals collected corresponds to feces bait, followed by decomposing fish and finally fermented fruit, with 31, 23 and 5 individuals captured, respectively (Fig. 2B). However, Dunn’s post hoc test showed that the differences occurred between the fruit and the other two types of bait (Dunn fruit-feces: 7,91*10-5; Dunn fruit-fish: 0,006); in other words, rotting fish bait does not differ significantly from feces in terms of the number of individuals caught (Dunn’s post hoc fish-feces: p-value= 0,81). On the other hand, it was found that the species attracted by bait are not the same, even reporting exclusive species for each type of bait, e.g. Adelpha erotia (Hewitson, 1847), Catoblepia berecynthia (Cramer, 1777), Catonephele acontius (Linnaeus, 1771), Chloreuptychia agatha (A. Butler, 1867), Hermeuptychia harmonia (A. Butler, 1867), Nessaea hewitsonii (C. Felder & R. Felder,1859), Siderone galantis (Cramer, 1775) were exclusive for the feces bait, Fountainea euripyle confuse (A. Hall, 1929), Opsiphanes cassina (C. Felder & R. Felder,1862), Prepona claudina (Godart, [1824]), Prepona dexamenusdexamenus (Hopffer, 1874), Siproeta steleness, Zaretis itys (Cramer, 1777) were exclusive for the rotting fish bait, and Catonophele numilia (Cramer, 1775), Pseudodebis marpessa (Hewitson, 1862) were exclusive for the fermented fruit bait (Figure 3; Table 1). This observation is supported by the result of the DCA, in which it is evident that there are species strongly associated with a type of bait (Figure 3). Note that rotting fish and feces baits has a similar number in unique species, 10 and 12 species, respectively, while fermented fruit bait have only two unique species. It is important to mention that there were Lepidoptera that were caught on more than one type of bait, for example, Archaeoprepona demophon visited all three types of bait with the same frequency, as Diaethria clymena, but one more individual was caught in the bait of feces, other species were common to two types of bait, mostly between feces and fish. Likewise, no attracted species shared between fermented fruit and feces baits were recorded, only one species (Memphis moruus) was reported shared between fermented fruit and rotting fish baits, and the the largest number of species shared between baits was between rotting fish and feces, e.g. A. capucinus capucinus, A. mesentina (Cramer, 1777), Hermeuptychia hermes (Fabricius, 1775), M. glauce, Morpho helenor (Cramer, 1776), Temenis laothoe (Cramer, 1777) (Tabla 2).
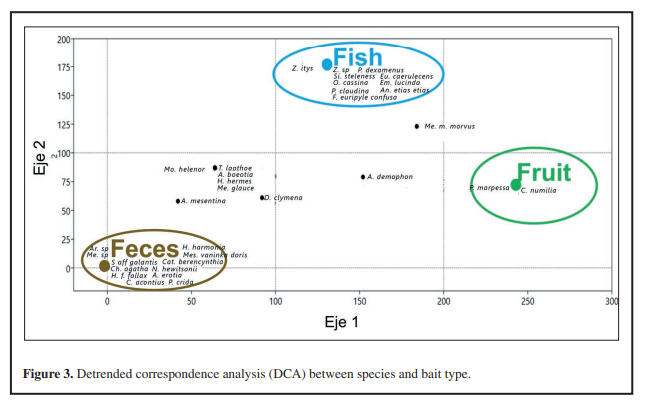
Figure 3.
Detrended correspondence analysis (DCA) between species and bait type.
Discussion
Although it is a low percentage compared to other capture methods, it was indispensable to record rare species such as Prepona claudina, Prepona dexamenes, Siderone galantis, which are usually not captured by performing transects with the entomological network. This high number of new reports can be explained due to the methodology used by the authors, which was limited exclusively to the use of entomological networks and therefore causes canopy species and/or species with low abundances to be unlikely to collect (Devries et al. 1997; Schulze et al. 2001). Another possible explanation may be that the Lepidoptera community presents differences according to the time of year as found in other sites (Hamer et al. 2005; Checa et al. 2009; Bandini et al. 2010). Thus, the Lepidoptera community may differ in the rainy season in which the present study was conducted during 8 days with respect to the periods immediately after the rains and the dry season in which the study was conducted Grados et al. (2008). However, Grados et al. (2008) only a difference of 3 species between the times evaluated, leading to the unlikely occurrence of this pattern in the Paujil sector.
Baits showed significant differences in catch rates and a slight preference of certain subfamilies to certain types of baits is observed, a trend that is more evident at the species level, although this was only evident between the fruit and the other two types of bait. To surprise and contrary to predictions made a priori, fermented fruit bait was the least effective in attracting the lowest number of individuals and species. This result differs from that found by Bokelaar (2017) and Checa et al. (2018), who found this type of bait to be the most efficient in primary forest and in any season, when considering abundance as an indicator. Possibly the flowering and fruiting season of several plant species in the sector will cause this type of resource to be more widely distributed in the area, thus altering the dispersion of the species (Stevens et al. 2010) and consequently the effectiveness of the bait. An alternative to explain the low effectiveness of fermented fruit as bait is the effect of climate. Bokelaar (2017) records that the effectiveness of sugar-macerated bananas decreases under rainy conditions, being predominant during the sampling days in this study. Furthermore, it seems that the dry and rainy season does have an impact on the attractiveness of the baits (Checa et al. 2018), so it is conjectured that, if this study were conducted in the dry season in the Paujil sector, it would present differences with the results found in the present stud.
It is noted that the rotting fish in the Bokelaar study (2017) was one of the baits with the lowest number of collects, contrasting with the results obtained. In this last study he used coffee baits, macerated fruit, or cut with sugar, rotting fish in different environments with different degree of disturbance, causing the results to be affected by the configuration of the landscape, microclimatic conditions, and fragmentation as suggested Rochat et al. (2017) and Orlandín et al. (2019). As opposed to the findings of Bokelaar (2017), Freitas et al. (2014) mentions that this type of bait has a high effectiveness and can attract several groups of Lepidoptera, coinciding with what was found. Likewise, a similar pattern is reported by Checa et al. (2018) using shrimp as bait, where they found a higher species richness compared to fermented banana bait.
With respect to feces as bait, it is evident that their effectiveness is comparable to that of rotting fish and even recorded the highest number of individuals in the study, also attracted some species of nectarivores Lepidoptera (Hesperiidae, Lycaenidae). However, this bait did not attract all groups of frugivorous Lepidoptera, possibly limiting their potential to record Lepidoptera diversity. It should be noted that feces were more effective than rotting fish to attract species of the genus Adelpha Hübner, [1819], but less effective in attracting species of Charaxinae. The above differs partially with what Freitas et al. (2014) mentioned, where it indicates that feces are most effective in attracting Charaxinae and rotting fish as the best attractant for the genus Adelpha. Moreover, several species of the Satyrini tribe were attracted exclusively by feces bait, for example, Chloreuptychia agatha, Hermeuptychia fallax fallax, H. harmonia y H. hermes, contrasting with what is reported in literature where it is mentioned that one of the disadvantages is its ineffectiveness in attracting Lepidoptera in this tribe. Nevertheless, this difference can be caused by the type of cover where the traps were installed and the height at which they were placed, leading to the assessment of both the type of cover and the height of the traps in the assessment of the attractiveness of the baits.
On the contrary, several of the results mentioned by Freitas et al. (2014), Garwood & Jaramillo (2017d), agree with the observations made, for example, feces more effectively attracted individuals of the species Memphis glauce, Nessaea hewitsonii was only caught on this kind of bait, Catonephele numilia (Cramer, 1775) was collected in fermented fruits bait, and the rotting fish was good attractant for Emesis lucinda (Cramer, 1775), Eurybia caerulescens (H. Druce, 1904) of the family Riodinidae. In addition, some individuals of nectarylvorous Lepidoptera such as Heliconius (Kluk, 1780) and some Hesperiidae were observed flying over the traps with this bait; although they did not enter the trap, that might suggest that feces also attract these species. Other findings complement the records of Freitas et al. (2014), highlighting the fact that genus Prepona (Boisduval,1836) was caught exclusively on rotting fish bait and not on feces bait. It should be noted that this exclusivity in attracting certain taxa to specific baits may be influenced by the short sampling time. Additionally, it has been reported that species composition varies with the season and years (Checa et al. 2018), causing them to respond differentially to being attracted by a specific bait.
Added to the above and as additional result outside to the study aim, the three types of baits were also efficient attracting other arthropods, especially flies, crickets, and bees. In this last group, the presence of numerous females and some males of the tribe Euglossini of the family Apidae stands out. Individuals who visited the baits were observed feeding on the exudate and exhibited a pattern like that previously discussed with Lepidoptera, more abundance in fish baits and feces and less abundance in fruit bait. Similarly, it was observed that females of Eulaema mocsaryi (Friese, 1899) collected feces in the neckties, possibly to make their nests, and, equivalently, Eufriesea sp. collected fish possibly to feed their broods. These records complement the work of Günther Gerlach for the sector, highlighting that species of Euglossinii were collected that were not recorded using scent bait. These findings may be useful for later comparative studies on the effectiveness of baits, obtaining more data with the same sampling effort, as suggested by Freitas et al. (2014).
In conclusion, our results suggest the importance of bait in the capture of Lepidoptera and even reflect a preference of certain species for a specific type of bait. These preferences should be considered in further research to maximize sampling effectiveness to know the diversity and community of Lepidoptera, for the to assess the conservation status at a site, and to develop conservation strategies involving other organisms.
Acknowledgments
The authors thank Missouri Botanical Garden for supporting research in the natural areas of Peru, to Christopher Davidson and Sharon Christoph for the opportunity offered by funding the Amazonian Andean ecosystems course. To Rocio Rojas for her availability and advice during this research. To Rodolfo Vásquez and Luis Valenzuela for their suggestions in the formulation and elaboration of this study. To Thibaud Aronson for his help in collecting certain specimens, for his admiration, respect, and devotion to the Colombian delegation. To Jaime Flores and Roger Zehnder for their support in the field. To Günter Gerlach for his help in determining the specimens of Euglossini. To Pamela Vasquez for working with Lepidoptera. Finally, to cohort 2020 of the EAA course, to Andrea Martínez, and to Argenis Bonilla for her continued support.
References
Aduse-Poku, K., William, O., Oppong, S., Larsen, T., Ofori-Boateng, C., & Molleman, F. (2012). Spatial and temporal variation in butterfly biodiversity in a West African forest: Lessons for establishing efficient rapid monitoring programmes. African Journal of Ecology, 50, 326-334. https://doi.org/10.1111/j.1365-2028.2012.01328.x
Andrade-C, M. G., Henao, E. R., & Triviño, P. (2013). Técnicas y procesamiento para la recolección, preservación y montaje de Mariposas en estudios de biodiversidad y conservación. (Lepidoptera: Hesperioidea Papilionoidea). Revista de la Academia Colombiana de Ciencias Exactas, Físicas y Naturales, 37(144), 311325. https://doi.org/10.18257/raccefyn.12
Bandini, D., Prado, P., Brown, K., & Freitas A. (2010). Temporal Diversity Patterns and Phenology in Fruit-feeding Butterflies in the Atlantic Forest, Biotropica 42(6), 710-716. https://doi.org/10.1111/j.1744-7429.2010.00648.x
Barbosa, E., Nogueira, De Vasconcelos R., Mariano-Neto, E., Viana, B., & Zikán Cardoso, M. (2017). Positive forestry: The effect of rubber tree plantations on fruit feeding butterfly assemblages in the Brazilian Atlantic Forest. Forest Ecology and Management, 397, 150-156. https://doi.org/10.1016/j.foreco.2017.04.043
Bokelaar, J. (2017). Scrumptious butterfly bait: A study of bait trapping techniques & a butterfly inventory in Un poco del Chocó (Ecuador). Hogeschool, 6-33. https://doi.org/10.13140/RG.2.2.34071.06564
Bonebrake, T. C., Ponisio, L. C., Boggs, C. L., & Ehrlich, P. R. (2010). More than just indicators: A review of tropical butterfly ecology and conservation. Biological Conservation, 143,1831-1841. https://doi.org/10.1016/j.biocon.2010.04.044
Bossart, J. L., & Opuni-Frimpong, E. (2009). Distance from edge determines fruit-feeding butterfly community diversity in Afrotropical Forest fragments. Environmental Entomology, 38(1), 43-52. https://doi.org/10.1603/022.038.0107 PMid:19791597
Brown, K. S. (1972). Maximizing daily butterfly counts. Journal of the Lepidopterists’ Society, 26(3), 183-196.
Checa, M., Barragán, A., Rodríguez, J., & Christman, M. (2009). Temporal abundance patterns of butterfly communities (Lepidoptera: Nymphalidae) in the Ecuadorian Amazonia and their relationship with climate. Annales de la Société entomologique de France (N.S.), 45(4), 470-486. https://doi.org/10.1080/00379271.2009.10697630
Checa, M. F., Donoso, D. A., Rodriguez, J., Levy, E., Warren, A., & Willmott, K. (2018). Combining sampling techniques aids monitoring of tropical butterflies. Insect Conservation and Diversity, 12(4), 362-372. https://doi.org/10.1111/icad.12328
Costa-Pereira, R., Roque, F. O., Constantino, P. A. L., Sabino, J., & Uehara-Prado, M. (2013). Monitoramento in situ da biodiversidade: Proposta para um sistema brasileiro de monitoramento da biodiversidade. ICMBio.
Devries, P., Murray, D., & Lande, R. (1997). Species diversity in vertical, horizontal, and temporal dimensions of a fruit-feeding butterfly community in an Ecuadorian rainforest. Biological Journal of the Linnean Society, 62(3), 343-364. https://doi.org/10.1111/j.1095-8312.1997.tb01630.x
Ebert, H. (1969). On the frequency of butterflies in eastern Brazil, with a list of the butterfly fauna of Poços de Caldas. Minas Gerais. Journal of the Lepidopterists’ Society, 23 (Supplement 3), 1-48.
Figgis, P., Mackey, B., Fitzsimons, J., Irving, J., & Clarke, P. (2015). Valuing Nature: Protected Areas and Ecosystem Services. Australian Conservation Foundation.
Filgueiras, B., Melo, D., Uehara-Prado, M., Freitas, A. V. L., Leal, I., & Tabarelli, M. (2019). Compensatory dynamics on the community structure of fruit-feeding butterflies across hyper-fragmented Atlantic Forest habitats. Ecological Indicators, 98, 276-284. https://doi.org/10.1016/j.ecolind.2018.11.005
Freitas, A. V. L., Agra Iserhard, C., Pereira Santos, J., Oliviera, J. Y., Bandini, D., Alves, D.H., Batista, A.H., Marini-Filho, O. J., Mattos, G., & Uehara-Prado, M. (2014). Studies with butterfly bait traps: an overview. Revista Colombiana de Entomología, 40(2), 203-212.
Gallice, G. (2016). Clearwing butterflies (Nymphalidae: Ithomini). Florida Museum of Natural History.
Garwood, K., & Jaramillo, V. J. G. (2017a). Catálogo de mariposas Papilionidae de Colombia y del neotrópico Catalog of Swallowtails of Colombia and the neotropics. http://www.butterflycatalogs.com.
Garwood, K., & Jaramillo, V. J. G. (2017b). Catálogo de mariposas Nymphalidae de Colombia y del neotrópico Catalog of Brushfoots of Colombia and the neotropics. http://www.butterflycatalogs.com.
Garwood, K., & Jaramillo, V. J. G. (2017c). Catálogo de mariposas Pieridae de Colombia y del neotrópico Catalog of Whites and Sulphurs of Colombia and the neotropics. http://www.butterflycatalogs.com.
Garwood, K., & Jaramillo, V. J. G. (2017d). Catálogo de mariposas Riodinidae de Colombia y del neotrópico Catalog of Colombian and the neotropical Metalmarks. http://www.butterflycatalogs.com.
Grados, J., Lamas, G., Azorsa, F., & Carbonel, S. (2008). Diversidad de Papilionoidea, Hesperioidea, Noctuoidea: Arctiidae (Lepidoptera) y Formicidae (Hymenoptera) del Parque Nacional Yanachaga-Chemillén [Informe].
Hamer, K., Hill, J., Mustaffa, N., Benedick, S., Sherratt, N., Chey, V., & Maryati, M. (2005). Temporal Variation in Abundance and Diversity of Butterflies in Bornean Rain Forests: Opposite Impacts of Logging Recorded in Different Seasons. Journey of Tropical Ecology, 21(4), 411-425. https://doi.org/10.1017/S0266467405002361
Hammer, Ø., Harper, D. A. T., & Ryan, P. D. (2001). PAST: Paleontological statistics software package for education and data analysis. Palaeontologia Electronica, 4(1), 9. http://palaeo-electronica.org/2001_1/past/issue1_01.htm
Hughes, J. B., Daily, G., & Ehrlich, P. R. (1998). Use of fruit bait traps for monitoring of butterflies (Lepidoptera: Nymphalidae). Revista de Biología Tropical, 46(3), 697-704. https://doi.org/10.15517/rbt.v46i3.20199
Iserhard, C. A., & Freitas, A. V. L. (2013). Maximized sampling of butterflies to detect temporal changes in tropical communities. Journal of Insect Conservation, 17(3), 615-622. https://doi.org/10.1007/s10841-013-9546-z
Jew, E., Loos, J., Dougill, A., Sallu, S., & Benton, T. (2015). Butterfly communities in miombo woodland: Biodiversity declines with increasing woodland utilization. Biological Conservation, 192, 436-444. https://doi.org/10.1016/j.biocon.2015.10.022
Laura, C. R. (2007). Parque Nacional Yanachaga-Chemillén. Universidad de Guadalajara.
Lecrom, J. F., Constantino, L. M., & Salazar, J. A. (2002). Mariposas de Colombia. Pieridae (Vol. 2). Carlec Ltda.
Martins, L., Araujo, J., Martins, A., Duarte, M., & Azevedo, G. (2017). Species diversity and community structure of fruit-feeding butterflies (Lepidoptera: Nymphalidae) in an eastern Amazonian Forest. Papéis Avulsos de Zoologia, 57(38), 481-489. https://doi.org/10.11606/0031-1049.2017.57.38
Öckinger, E., Dannestam, A., & Smith, H. (2009). The importance of fragmentation and habitat quality of urban grasslands for butterfly diversity. Landscape and Urban Planning, 93(1), 31-37. https://doi.org/10.1016/j.landurbplan.2009.05.021
Olarte-Quiñonez, C. A., Carrero-Sarmiento, D., Viloria, A. L., & Ríos-Malaver, I. C. (2021). Patrones de diversidad de las mariposas de la subtribu Pronophilina (Lepidoptera: Nymphalidae: Satyrinae) en un gradiente altitudinal del Cerro de Tierra Negra, Cordillera Oriental, Norte de Santander, Colombia. Boletín Científico Centro De Museos Museo De Historia Natural, 25(2), 197-218. https://doi.org/10.17151/bccm.2021.25.2.12
Orlandin, E., Piovesan, M., D’agostini, F., & Carneiro, E. (2019). Use of microhabitats affects butterfly assemblages in a rural landscape. Papéis Avulsos de Zoologia, 59, 1-23. https://doi.org/10.11606/1807-0205/2019.59.49
Pozo, C., Llorente, J. B., Salas, N., Maya, A., & Vargas, I. F. (2008). Seasonality and phenology of butterflies (Lepidoptera: Pieridae, Papilionidae, Nymphalidae, Lycaenidae and Hesperiidae) in Calakmul, biosphere reserve. Florida Entomologist, 91(3), 407-422. https://doi.org/10.1653/0015-4040(2008)91.
Panjaitan, R., Hidayat, P., Buchori, D., Peggie, D., Sakti, I., Drescher, J., & Scheu, J. (2019). Diversity of butterflies (Lepidoptera) caught by using fruit traps in Bukit Duabelas and Harapan forest landscape, Jampi. AIP Conference Proceedings, 2120, 1-7. https://doi.org/10.1063/1.5115732
Purwanto, A., Harsanto, F. A., Marchant, N. C., Houlihan, P. R., Ross, K., Tremlett, C., & Harrison, M. E. (2015). Good Practice Guidelines: Butterfly Canopy Trapping. Orangutan Tropical Peatland Project.
R Core Team. (2020). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Rengifo, H., & Montero, P. (2010). Diversidad de mariposas diurnas (Lepidoptera: Rophalocera) en los bosques de tierra firme adyacentes a la comunidad campesina San Rafael, Loreto-Perú [Tesis pregrado]. Universidad Nacional de la Amazonía Peruana.
Ribeiro, D. B., & Freitas, A. V. L. (2012). The effect of reducedimpact logging on fruit-feeding butterflies in Central Amazon, Brazil. Journal of Insect Conservation, 16(5), 733-744. https://doi.org/10.1007/s10841-012-9458-3
Rochat, E., Manel, S., Deschamps-Cottin, M., Widmer, I., & Joost, S. (2017). Persistence of butterfly populations in fragmented habitats along urban density gradients: motility helps. Heredity, 119(5), 328-338. https://doi.org/10.1038/hdy.2017.40 PMid:28792492 PMCid:PMC5637364
Rydon, A. (1964). Notes on the use of butterfly traps in East Africa. Journal of the Lepidopterists’ Society, 18(1), 51-58.
Schulze, C. H., Linsenmair, K. E., & Fiedler, K. (2001). Understory versus canopy: patterns of vertical stratification and diversity among Lepidoptera in a Bornean rain forest. Plant Ecology, 153(1-2), 133-152. https://doi.org/10.1023/A:1017589711553
Sheskin, D. J. (2003). Handbook of Parametric and Nonparametric Statistical Procedures. CRC Press. https://doi.org/10.1201/9781420036268 PMid:12636158
Stevens, V. M., Turlure, C., & Baguette, M. (2010). A meta-analysis of dispersal in butterflies. Biological Reviews, 85, 625-642. https://doi.org/10.1111/j.1469-185X.2009.00119.x PMid:20055815
Sung-Soo, K., & Tae-Sung, K. (2018). Changes in butterfly assemblages and increase of open-land inhabiting species after forest fires. Journal of Asia-Pacific Biodiversity, 11(1), 39-48. https://doi.org/10.1016/j.japb.2017.11.004
Van Swaay, C., Regan, E., Ling, M., Bozhinovska, E., Fernandez, M., Marini-Filho, O. J., Huertas, B., Phon, C.-K., Körösi, Á., Meerman, J., Peer, G., Uehara-Prado, M., Sáfián, S., Sam, L., Shuey, J., Taron, D., Terblanche, R., & Underhill, L. G. (2015). Guidelines for Standardised Global Butterfly Monitoring. GEO BON Technical Series, 1, 1-32.
Willmott, K. (2003). The genus Adelpha: its systematics, biology, and biogeography (Lepidoptera: Nymphalidae: Limenitidini). Scientic Publishers.
Whitworth, A., Huarcaya, R. P., Mercado, H. G., Braunholtz, L. D., & Macleod, R. (2018). Food for thought. Rainforest carrion-feeding butterflies are more sensitive indicators of disturbance history than fruit feeders. Biological Conservations, 217, 383-390. https://doi.org/10.1016/j.biocon.2017.11.030
Author notes
* Autor para la correspondencia / Corresponding author: E-mail: yevegag@unal.edu.co
