Abstract: Almost 90 years have passed after the last publication of a catalogue of the family Zygaenidae of the world (Bryk, 1936). At that time there was no correct understanding of the taxonomic, genetic and ecological framework of the subfamily Procridinae. Many species of this subfamily were included into the Chalcosiinae and Zygaeninae. A new contemporary annotated catalogue consisting of five tribes, 94 genera and 570 species of Procridinae is provided. This is the first ever published complete worldwide catalogue of the subfamily. All together, 1111 taxa (including synonyms) are discussed. Three new tribes (Thyrassiini, tribus nov., Pollanisini tribus nov. and Cleleini tribus nov.), two new genera ( Afromalamblia gen. nov. and Psudohedina gen. nov.) as well as four new subgenera ( Longiterna subgen. nov., Afroterna subgen. nov., Obscuriterna subgen. nov., and Eurasiterna subgen. nov.) are described. 36 comb. nov., 14 stat. nov. and 3 syn. nov. are established.
Keywords: Lepidoptera, Zygaenidae, Procridinae, Thyrassiini, Pollanisini, Artonini, Cleleini, Procridni, systematic catalogue, taxonomy, distribution, larval host-plants, World.
Resumen: Han pasado casi 90 años desde la última publicación de un catálogo de la familia Zygaenidae del mundo (Bryk, 1936). En aquella época no existía una comprensión correcta del marco taxonómico, genético y ecológico de la subfamilia Procridinae. Muchas especies de esta subfamilia se incluyeron en los Chalcosiinae y Zygaeninae. Se proporciona un nuevo catálogo contemporáneo anotado que consta de cinco tribus, 94 géneros y 570 especies de Procridinae. Se trata del primer catálogo mundial completo publicado de la subfamilia. En total, se analizan 1111 taxones (incluidos los sinónimos). Se describen tres nuevas tribus ( Thyrassiini, tribus nov., Pollanisini tribus nov. y Cleleini tribus nov.), dos nuevos géneros ( Afromalamblia gen. nov. y Psudohedina gen. nov.) y cuatro nuevos subgéneros ( Longiterna subgen. nov., Afroterna subgen. nov., Obscuriterna subgen. nov. y Eurasiterna subgen. nov.). Se establecen 36 comb. nov., 14 stat. nov. y 3 syn. nov.
Palabras clave: Lepidoptera, Zygaenidae, Procridinae, Thyrassiini, Pollanisini, Artonini, Cleleini, Procridni, catálogo sistemático, taxonomía, distribución, plantas nutricias de larvas, Mundo.
Artículos
An annotated catalogue of the Procridinae of the World (Lepidoptera: Zygaenidae)
Catálogo anotado de los Procridinae del mundo (Lepidoptera: Zygaenidae)
Received: 23 January 2024
Accepted: 28 February 2024
Published: 30 September 2024
Introduction...............................................................410
Historical background......................................................411
The concept of Alberti (genera, subgenera, species groups).................413
A new concept to use tribes in Procridinae.................................413
The concept for species and subspecies in this catalogue...................414
Catalogue..................................................................415
The tribe Thyrassiini......................................................415
The tribe Pollanisini......................................................416
The tribe Artonini.........................................................421
The tribe Cleleini.........................................................429
The tribe Procridini.......................................................427
Comments...................................................................487
Acknowledgements...........................................................500
References.................................................................501
Index.......................................................................52
Plates.................................................................... 537
The family Zygaenidae is an important model group for ecological, environmental, zoogeographic, biochemical, karyological, genetic, morphological and taxonomic investigations in Lepidoptera (see e.g. Can et al. 2018, 2019; Can Cengiz et al. 2018; Drouet & Tarmann, 1989; Drouet et al. 2021; Gernaat et al. 2022; Huang & Efetov, 2021; Efetov, 1996e, 2012b, 2016, 2018, 2019; Efetov et al. 2004, 2010a, 2010b, 2010c, 2010d, 2011, 2012a, 2012b, 2013, 2014a, 2014b, 2014c, 2014d, 2015a, 2015b, 2016a, 2016b, 2016c, 2016d, 2017, 2018, 2019a, 2019b, 2019c, 2019d, 2020, 2021, 2022, 2023a, 2023b; Efetov & Gorbunov, 2016; Efetov & Knyazev, 2014; Efetov & Kucherenko, 2020, 2021; Efetov & Savchuk, 2009, 2013; Efetov & Tarmann, 2012, 2014b, 2017a, 2020a, 2020b, 2022a, 2022b; Knyazev et al. 2015a, 2015b; Landoldt et al. 1991; Marianelli et al. 2020; Markl et al. 2022; Mutanen et al. 2016; Nahirnic-Beshkova et al. 2021; Nazari & Efetov, 2023; Nazari et al. 2019; Parshkova & Efetov, 2014; Razov et al. 2017; Subchev et al. 2016, 2012, 2013; Tarmann, 1975, 1979, 1984a, 1984b, 1992a, 1992b, 1995, 1998, 2004, 2016, 2019a, 2019b, 2019c; Tarmann & Efetov, 2021; Tarmann et al. 2019; Vrenozi et al. 2019; Zahiri et al. 2021). Its systematics is well established. According to the contemporary classification the family Zygaenidae consists of five subfamilies: Inouelinae Efetov & Tarmann, 2017; Zygaeninae Latreille, 1809; Callizygaeninae Alberti, 1954; Chalcosiinae Walker, 1865, and Procridinae Boisduval, 1828 ( Efetov, 1999c, 2001d, 2004d; Efetov & Tarmann, 2014a, 2017a; Tarmann, 1994; Miric et al. 2023).
The Procridinae is the only subfamily that has a worldwide distribution and which occurs also in America and Australia. Many species have been described during the last years (see e.g. Efetov, 1998a, 1998b, 2010, 2012a; Efetov & Tarmann, 2013a, 2013b, 2014a, 2014b, 2016a, 2016b, 2017a, 2017b; Keil, 1998, 1999, 2016b, 2020; Mollet, 2008, 2015, 2016a, 2016b, 2016c, 2016d, 2017, 2018, 2020a, 2020b; Mollet & Tarmann, 2007, 2010, 2023; Owada, 2021; Owada et al. 2021, 2022a, 2022b; Owada & Inada, 2005; Shih & Owada, 2022; Sondhi et al. 2023; Tarmann & Cock, 2019; Tarmann & Drouet, 2015). However, newer checklists of taxa exist only for the Palaearctic and Australian regions ( Tarmann, 2004; Efetov & Tarmann, 2012).
As the last catalogue of the Procridinae of the world was published 88 years ago ( Bryk, 1936) it is urgently time for an update. Although there are several papers in preparation where more species will be described, we think this catalogue should be published now. We have postponed this work already for many years, always in the hope of making it more complete by waiting for the next publication series about Procridinae. Further new species must be summarized as an addition at a later time.
Illustrations of Zygaenidae can be found on the paintings of the Dutch Masters of 17th century where they join Papilionoidea and other moths on and around flower bouquets ( Efetov & Tarmann, 2008a). The first Zygaenidae were described in the 10th edition of Carolus Linnaeus’ Systema Naturae in the year 1758, viz. Sphinx filipendulaeLinnaeus, 1758, and Sphinx statices Linnaeus, 1758. Soon there followed numerous descriptions by other authors such as Eugen Johann Christoph Esper, Michael Denis & Ignaz Schiffermüller, Jacob Hübner, Ferdinand Ochsenheimer and others. However, a first comprehensive summary of the so far known names was not available before the publication of Jean- Baptiste Alphonse DéChauffour de Boisduval’s work Monographie des Zygaenides (1828). A significant step forward in our knowledge about Zygaenidae followed slightly later in the 19th century with authors like Gottlieb August Wilhelm Herrich-Schäffer ( 1843-1855, 1843-1856, [1853]-[1858], [1856]-1861), Christian Friedrich Freyer ( 1833-1836), Francis Walker ( 1854, 1856), Otto Staudinger ( 1862, 1871, 1878-1879, 1887a, 1887b), George Francis Hampson (1893), Charles Oberthür ( 1880, 1893, 1894, 1916), John-Henry Leech ( 1889a, 1889b, 1898), Charles Swinhoe ( 1890, 1891, 1892, 1894, 1903), Edward Meyrick ( 1886-1888), Herbert Druce ( 1881-1900, 1889, 1896, 1899, 1806, 1910) and others. These authors did not only deal with the Palaearctic species but were working in a world- wide context. An impressive overview of all known species of Zygaenidae of the world with short descriptions of the species and, if known, also of their distribution and biology, with colour illustrations of all mentioned species can be found in Adalbert Seitz’s books Die Gross-Schmetterlinge der Erde. In these series of books the Zygaenidae were treated by Karl Jordan ( 1907a, 1907c- 1908, 1913) and Max Gaede ( 1926).
A list of all known taxa in the world with synonyms, citations of original descriptions and geographical notes on the distribution was published by Embrik Strand in Lepidopterorum Catalogus in volumes 33 (by Helmut Burgeff, 1926, all Zygaeninae) and 71 (by Felix Bryk, 1936, all other Zygaenidae groups). The fascination about the variability of Zygaenidae and their geographical differences especially of the Palaearctic species of Zygaena Fabricius, 1775, resulted in a huge number of descriptions of subspecies, forms and aberrations. Helmut Burgeff, Franz Daniel, Francis Dujardin, Otto Holik, Manfred Koch, Hans Rauch, Hugo and Günther Reiss, Ubaldo Rocci, Ruggero Verity, Karl-Heinz Wiegel, etc. described hundreds of taxa.
Only Burchard Alberti ( 1954, 1958-1959) saw the familiy Zygaenidae as a whole and in a global context. He summarised their characters, especially those of their genitalia and tried the first phylogenetic analysis. Interesting new ideas came from Ernst Rudolf Reichl (1964) who implemented for the first time biometry and biostatistics into the discussion about populations, subspecies and species. He also created the first electronic database for Zygaenidae. The phylogenetic approach of Alberti, based on the studies of more and new characters, was further developed by Clas Naumann (morphology, ultrastructures, biochemistry, and biogeography) and many of his students, e.g. Oliver Niehuis (molecular biology), Axel Hille (biology, biochemistry, biometry), Harald Fänger (morphology, ultrastructure), also by Konstantin Efetov & Gerhard Tarmann (morphology, biology, behaviour, biochemistry, molecular biology, genetics) and recently by Mirela Miric and Jadranka Rota (genetics).
We also have to mention Walter Gerald Tremewan (genetics, bionomics, bibliography, systematics, taxonomy, and phylogeny), Axel Hofmann (biology, biogeography, systematics, taxonomy, and phylogeny), Hiroshi Inoue (morphology, systematics, and taxonomy), Bernard Mollet and Thomas Keil (morphology, biology, systematics) and Shen-Horn Yen (morphology, systematics, molecular biology, and phylogeny) as other important contributors to the knowledge of the family.
By all these works more lepidopterologists were stimulated to study Zygaenidae. Many of them compiled large collections. Here we have to mention the rich special collections of Zygaenidae of Eyjolf Aistleitner (Palaearcic region, especially Iberian Peninsula), Francis Dujardin (Palaearctic region), Ulf Eitschberger (Palaearcti region), Helmut und Ruth Holzinger (Palaearctic region), Predrag Jaksic (Balkans), Thomas Keil (especially Palaearctic region), Hans Rauch (Alps, Italy, Balkans), and especially the huge collection of Thomas Witt (mainly Palaearctic and Oriental region). In some countries collectors have formed very active working groups that contributed significantly especially to the knowledge of the Zygaenidae fauna of their country, e.g. in France the GIRAZ with members like Louis Faillie, Jean-Marie Desse, Eric Drouet, Marc Nicolle, Bruno Lambert and others.
A number of impressive monographies in the form of books have been published on Zygaenidae during the last years (e.g. Efetov, 2001f, 2004a, 2005a; Efetov & Tarmann, 1999a, 2008b, 2012; Guenin, 2023; Keil, 2014; Hofmann & Tremewan, 2017, 2020a, 2020b; Naumann, Tarmann & Tremewan, 1999; Tarmann, 2004) and more will follow.
Extended material was compiled during various collecting expeditions by persons who deposited their material in private collections and in museums. Here we have to mention especially the collectors of the British, Dutch, German, Austrian and Russian Empires that worked between the end of the 19th century up to the Second World War. Extraordinary persons like Lord Walter Rothschild and the Grand Duke Nikolay Mikhaylovich Romanov, and later Adalbert Seitz, invested lots of money in compiling material and in the publication of richly illustrated book series. For those private people but also for larger national museums a number of collectors travelled around the world and brought material of Zygaenidae from almost all countries where they are distributed. Moreover, members of the navy of the British Empire and employees of powerful commercial companies (e.g. the Dutch East Indian Company) were sent to foreign countries and many of them were collectors of insects. As examples for some of these almost “professional collectors” we mention Hugo Theodor Christoph (Central Asia), J. Z. Kannegieter (South-East Asia) and as an example of well-known people who were based far awayfrom their home countries Rudolph van Eecke (South-East Asia), Hermann Höne (China), Edi Diehl(Indonesia), Georg Brückner (Guatemala) and J. F. Zikân (Brazil).
In Europe Leo Sheljuzhko (Eastern Europe, Caucasus, Central Asia), Guy Barrague (North Africa), Josef J. de Freina (Western Palaearctic and Africa), Wolfgang Eckweiler (especially western Asia), Ernst Görgner (western Asia), and Jirˇi Klir (Asia) collected valuable material also of Zygaenidae that is now mainly deposited in other collections.
In Asia especially the Japanese scientists and collectors like Hiroshi Inoue, Mamoru Owada, Yasunori Kishida, Kiyoshi Horie and Toshitsugo Endo established large collections that contain important material of Zygaenidae. Others such as Masanao Nakamura, Kayoko Nishihara und Chiharu Koshio contributed significantly to our knowedge of morphology and biology.
In spite of all these efforts we still have large gaps in the knowledge of Zygaenidae for many parts of the world, where newer comprehensive summaries are missing and the identification of species is therefore difficult. Many undescribed species are already known and waiting to be described. A very substantial contribution to the knowledge of the Asian and Australian Zygaenidae fauna, mainly based on his own field observations and rearing experiments, has recently been carried out by Bernard Mollet (e.g. Mollet, 1995, 2003a, 2003b, 2003c, 2019a; Mollet & Tarmann, 2023).
Revisions on generic level are permanently published but the progress is slow. Revisions on Zygaenidae dealing with whole continents have only been published for parts of the Palaearctic ( Naumann et al. 1999; Efetov, 2001d, 2005a, 2005b; Efetov & Tarmann, 1995b, 1999a; Keil, 2014), America ( Tarmann, 1984b) and Australia ( Tarmann, 2004; Mollet & Tarmann, 2023) during the last 50 years.
The authors of this catalogue have already summarised their total knowledge on Zygaenidae and undertook an attempt to provide a hypothetical ground plan of the family in 2017 ( Efetov & Tarmann, 2017a).
Scientific work with Zygaenidae has been often controversial. While in one case a character can be extremely useful for classification, the same character can be completely irrelevant in another case. This problem is not new. Already scientists like Karl Jordan (e.g. Jordan, 1907a, 1907c) and Martin Hering (e.g. Hering 1922, 1926), who summarised parts of the World’s Zygaenidae in several books and papers, stepped into some of the tricky traps that have been laid by an unpredictable evolutionary history especially in the subfamily Procridinae ( Tarmann, 1984b, 2004).
Alberti (1954) published the first comprehensive revision of the Zygaenidae of the world. It was based on extended studies, especially on the genitalia morphology of males and females. He recognised seven subfamilies for Zygaenidae: Zygaeninae, Phaudinae (now family Phaudidae within Zygaenoidea), Chalcosiinae, Anomoeotinae (now family Anomoeotidae within Zygaenoidea), Himantopterinae (now family Himantopteridae within Zygaenoidea), Charideinae (moved to Thyrididae by Minet, 1991) and Procridinae.
Especially for his treatment of the Procridinae Alberti (1954) used genera and also subgenera and within these subgenera species groups. In the same way, he also treated the subfamily Zygaeninae.
This concept has been widely accepted, especially in Procridinae. However, the discovery of new characters and more knowledge on their variability has made it necessary to describe a new subfamily, viz. Inouelinae ( Efetov & Tarmann, 2017a), new genera and to divide some genera into more subgenera.
There is a common consensus that species can be grouped together with closely related species to form so-called species groups. However, there is no consensus if it makes sense to give a group of species or a group of species groups a taxonomic status in the form of the description of a subgenus. The fact that many of these described subgenera have suddenly appeared as genera in various publications and have confused users of scientific names like taxonomists, applied entomologists, bio- geographers etc. led to a widely distributed rejection of subgeneric names. Opinions about the pros and cons of subgeneric names have been strongly held between entomologists and it is mainly dependent on the education and the tradition of an entomological society in which direction these feelings go (see e.g. Hofmann & Tremewan, 2009).
The authors of this catalogue have studied the family Zygaenidae for almost half a century. Nevertheless, they could not avoid following Alberti’s concept throughout all their works as it was in most cases clearly the best solution for taxonomic and systematic work in Zygaenidae and especially in the Procridinae, which are treated in this catalogue. This concept enables good grouping of monophyletic units, the subgeneric names are based on types with a designated type species and there is a responsible authorship for each of them. Moreover, in many cases clear monophyletic species groups are found within the subgenera which enables further good subgrouping. Examples are genera like Artona Walker, 1854, IlliberisWalker, 1854, ZygaenoprocrisHampson, 1900, AdscitaRetzius, 1783, or JordanitaAgenjo, 1940, each with a number of well differentiated subgenera ( Figures 1-2).
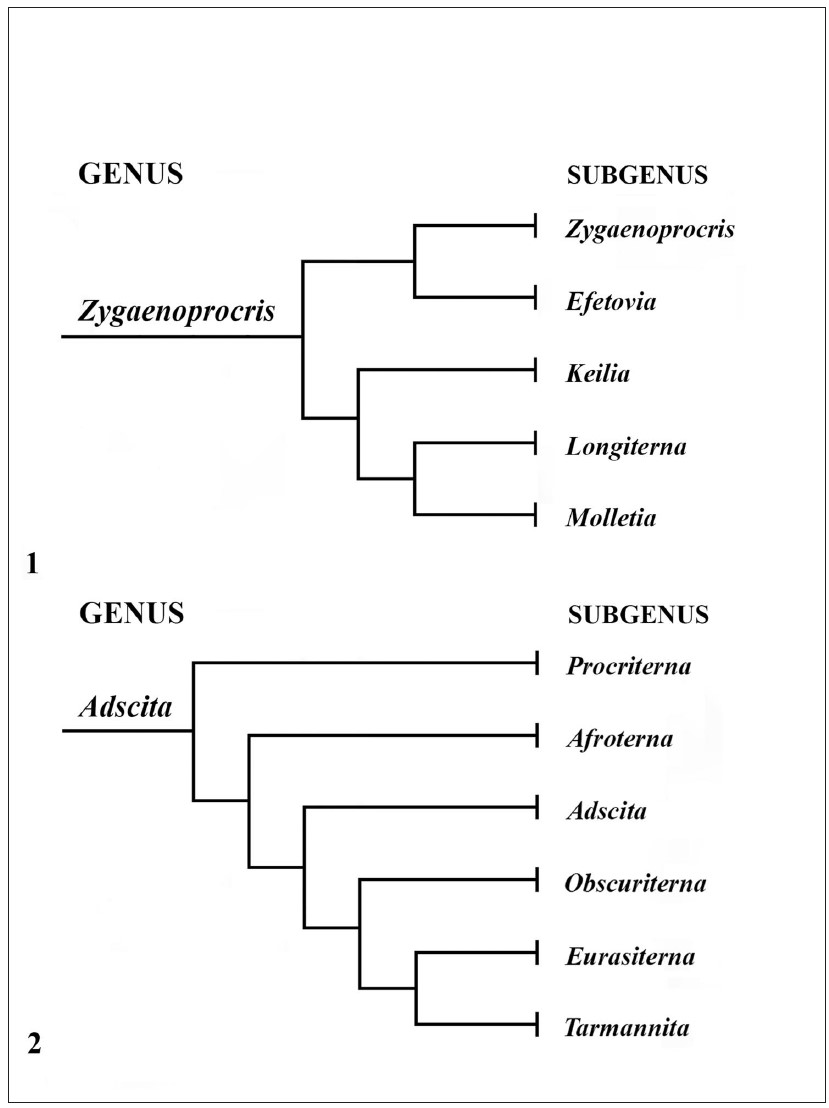
Since 1994 the subfamily Procridinae has been divided into two tribes: Artonini Tarmann, 1994 and Procridini Boisduval, 1828 ( Tarmann, 1994; Efetov & Tarmann, 2012, 2017a). This concept was based on a large number of significant morphological, biological and other characters. All these relevant characters were listed, compared and discussed in detail in Efetov & Tarmann (2017a). However, some characters that have earlier been thought to belong to Artonini only have later also been found in Procridini. The concept that Artonini are monophyletic and Procridini paraphyletic has not been confirmed as valid in recent years. The final confirmation that the concept used so far was wrong is based on the newest DNA results using 32 nuclear and mitochondrial genes (Miric et al. 2023). Taking into account these new results and combining them with the so-far known earlier results on morphology and biology we see that the subfamily Procridinae consists of five monophyletic traits.
Consequently, we divide here the Procridinae into Thyrassiini tribus nov., Pollanisini tribus nov., Artonini Tarmann, 1994, Cleleini tribus nov., and Procridini Boisduval, 1828.
Since the times of Linnaeus it has been an unsolvable problem to find a common agreement between authors of what is a species and, of course, also what is a subspecies. Populations with common characters have been grouped together and named but the moment when species level is reached has never been completely clear. There are many zoological species concepts. Wilkins (2006) mentions in a short summary at least 26 existing species concepts. It is not the aim of this catalogue to contribute to this discussion but we have to agree to a pragmatic solution for the list of taxa given below. The subfamily Procridinae of Zygaenidae that is treated in this catalogue shows especially rich character variability and character combinations. We are confronted with the situation that sometimes characters are very constant in one group but are completely variable in another. Moreover, many species interbreed without problems and produce hybrid populations that are also fertile in further generations. Therefore, it has often been difficult to decide whether a taxon has to be treated as a species or included in a known taxon as a subspecies. According to Alberti’s concept ( Alberti, 1954) (see above) most taxa that show unique genitalic characters have to be treated as species in Procridinae. However, there are species groups that do not show significant differences in genitalia but are, without doubt, different species (e.g. Pollanisus in Australia, Harrisina in America). Even characters like DNA distances cannot solve this problem (e.g. Jordanita in the Palaearctic and Pollanisus in Australia). Moreover, even if small genitalic differences are present the existence of broad hybrid belts show that it is sometimes better to treat geographically separated population groups as subspecies and not as valid species even if they also have small genetic differences.
Based on the current knowledge about the variability of characters in Procridinae the authors strongly support the opinion that for the subfamily Procridinae subspecies only make sense and can be accepted if they represent a group of populations that have important constant characters that separate them clearly from the nominotypical populations without exceptions. As examples we mention Adscita (Adscita) statices statices and A. ( A.) statices drenowskii (constant genitalic differences in male, no genitalic differences in female, small genetic differences in the COI gene within the populations but clustering into two well-separated groups, similar biology, vicariant distribution, broad hybrid belt where the two subspecies meet), Adscita (Adscita) italica italica and A. ( A.) italica storaiae (no constant genitalic differences in male, small but also not constant genitalic differences in female, clear genetic distance in the COI gene, clearly isolated geographic range, no geographical meeting points and therefore no hybrids known in nature), A. ( A.) geryon geryon and A. ( A.) geryon orientalis (no constant genitalic differences but large genetic distances in the COI gene, vicariant distributional ranges, no proof of hybrids in nature so far) and A. ( A.) obscura obscura and A.( A.) obscura maxima (no differences in genitalia, constant differences only in habitus and clearly isolated geographic ranges, small genetic differences in the COI gene).
Many species and subspecies described earlier do not fit into this concept. They often were based on insufficient characters and have shown to be synonyms. Especially subspecies were often based only on habitus characters like the different size and colour of the specimens and on an isolated geographic occurrence. The lack of a profound knowledge of the overall variability of these characters in Procridinae has led to this situation. The authors have therefore already synonymized most of the so-far described subspecies in Procridinae ( Efetov & Tarmann, 1995b, 2012; Efetov, 2001d). However, recently a number of new taxa have been described where other criteria have been used by the authors as accepted by us for this catalogue (e.g. Keil, 2016b, 2020; Zolotuhin, 2020). All these taxa are here included, discussed in detail and mainly placed according to our above mentioned concept.
C - comments that follow immediately after the checklist
Family Zygaenidae Latreille, 1809, 189, 211 (as Zygaenides)
[Type genus: ZygaenaFabricius, 1775, 550]
Subfamily ProcridinaeBoisduval, 1828, 38 (as Procridae)
[Type genus: Procris [Fabricius in Illiger], 1807, 289] (see Taeger & Gaedike, 2001, 87)
Tribe Thyrassiini Efetov & Tarmann, tribus nov. (C01)
[Type-genus: ThyrassiaButler, 1876, 355]
Larval host-plants: Vitaceae.
Distribution: Southern and southeastern Asia, Australia.
Genus ThyrassiaButler, 1876, 355
[Type species: Syntomis subcordata Walker, 1854, 132, by original designation]
MonoschalisHampson, 1893 (“1892”), 238
[Type species: Monoschalis virescens Hampson, 1893 (“1892”), 238, by original designation]
AtuciaWatson, 1980, 20
[Type species: Acutia bidens Kaye, 1919, 89, by original designation], a junior homonym of Acutia Ragonot, 1891, 539 - Insecta: Lepidoptera, Pyralidae. The objective replacement is AtuciaWatson, 1980, 20
Larval host-plants: Vitaceae.
T. subcordata ( Walker, 1854, 132) ( Syntomis)
Distribution: Noth India (ssp. subcordata), South India, Sri Lanka (ssp. aurodisca).
T. subcordata subcordata ( Walker, 1854, 132) ( Syntomis)
subcaudata [sic] Swinhoe, 1892, 55, misspelling
T. subcordata aurodisca Hampson, 1891, 44
T. virescens ( Hampson, 1893, 238) ( Monoschalis)
Distribution: Sri Lanka.
T. penangae ( Moore, 1859, 198) ( Syntomis)
Distribution: southern China, Myanmar (Burma), Malaysia, Singapore, Indonesia (Sumatra) (ssp. penangae), Java, Bali (ssp. rafflesi).
T. penangae penangae ( Moore, 1859, 198) ( Syntomis)
diversa (Walker, 1864, 31) ( Hydrusa) (synonymised by Bryk, 1936, 271)
penanga [sic] ( Hampson, 1896, 466) ( Syntomis), misspelling
T. penangae rafflesi ( Moore, 1859, 198) ( Syntomis)
T. bidens (Kaye, 1919, 89) ( Acutia)
Distribution: Malaysia (Borneo), Indonesia (Borneo).
T. philippina Jordan, 1908, 51
penangae (sensu Semper, 1898, 433) (nec Moore, 1859, 198) (see Bryk, 1936, 271)
Distribution: Philippines (Luzon).
T. aprepes Swinhoe, 1905, 144
Distribution: Indonesia (Sumba. Kai Islands).
T. inconcinna Swinhoe, 1892, 55 ( Figure 3)


mimetica ( Turner, 1902, 200) ( Monoschalis)
Distribution: Australia (Queensland, Lizard Island).
Tribe Pollanisini Efetov & Tarmann, tribus nov. (C02)
[Type-genus: PollanisusWalker, 1854, 114]
Larval host-plants: Dilleniaceae, Myrtaceae, Vitaceae, Arecaceae. Distribution: Australia, Fiji, South Africa.
Genus PollanisusWalker, 1854, 114 (C03)
[Type species: Procris viridipulverulenta Guérin-Méneville, 1839, pl. 11, by subsequent designation by Kirby, 1892, 87]
Larval host-plants: Dilleniaceae (two species on Urticaceae and one on Fabaceae).
P. acharon ( Fabricius, 1775, 556) ( Zygaena)
eumetopus Turner, 1926, 443
eungellaeTarmann, 2004, 95
Distribution: Australia (Queensland).
Pollanisus sp. 7 (discussed and figured in Tarmann, 2004, 103; Mollet & Tarmann, 2023, 9, 11)
Distribution: Australia (Northern Territory).
P. yugambeh Mollet & Tarmann, 2023, 30
Distribution: Australia (Queensland, New South, Wales).
P. angustifrons Tarmann, 2004, 100
Pollanisus sp. 4 (discussed and figured in Tarmann, 2004, 102)
Pollanisus sp. 5 (discussed and figured in Tarmann, 2004, 102)
Distribution: Australia (Queensland).
P. jumbun Mollet & Tarmann, 2023, 13
Distribution: Australia (Queensland).
P. horakaeMollet & Tarmann, 2023, 33
Distribution: Australia (Queensland).
P. trimacula ( Walker, 1854, 110) ( Procris)
Distribution: Australia (Queensland, New South Wales).
P. worimi Mollet & Tarmann, 2023, 36
Distribution: Australia (New South Wales).
P. kalliesi Mollet & Tarmann, 2023, 37
Distribution: Australia (New South Wales).
P. subdolosa ( Walker, 1865, 32) ( Procris)
Distribution: Australia (Queensland, New South Wales (subsp. clara), Victoria (subsb. subdolosa)).
P. subdolosa subdolosa ( Walker, 1865, 32) ( Procris)
P. subdolosa clara Tarmann, 2004, 89
P. edwardsi Tarmann, 2004, 86
Distribution: Australia (Queensland, New South Wales).
Pollanisus sp. 2 (discussed and figured in Tarmann, 2004, 102; Mollet & Tarmann, 2023, 20)
Distribution: Australia (New South Wales).
P. commoni Tarmann, 2004, 98 ( Figure 15)

Distribution: Australia (Queensland).
Pollanisus sp. 8 (discussed and figured in Tarmann, 2004, 104; Mollet & Tarmann, 2023, 22)
Distribution: Australia (Queensland).
P. contrastus Tarmann, 2004, 91
Distribution: Australia (Queensland, New South Wales).
Pollanisus sp. 3 (discussed and figured in Tarmann, 2004, 102; Mollet & Tarmann, 2023, 26)
Distribution: Australia (Queensland).
Pollanisus sp. 6 (discussed and figured in Tarmann, 2004, 103; Mollet & Tarmann, 2023, 26)
Distribution: Australia (Queensland).
P. incertus Tarmann, 2004, 99
Distribution: Australia (Queensland).
P. jirrbal Mollet & Tarmann, 2023, 45
Distribution: Australia (Queensland).
P. viridipulverulenta ( Guérin-Méneville, 1839, pl. 11) ( Procris) ( Figure 21) adelaide Turner, 1926,444

Distribution: Australia (Queensland, New South Wales, Victoria, South Australia, Tasmania).
P. cupreusWalker, 1854, 115
Distribution: Australia (Western Australia).
P. nielseni Tarmann, 2004, 75 ( Figure 22)
Distribution: Australia (Western Australia).
P. empyrea ( Meyrick, 1888, 927) ( Procris) (C04)
Distribution: Australia (Western Australia).
P. amethystina ( Meyrick, 1888, 927) ( Procris) (C04)
Distribution: Australia (Western Australia).
P. apicalis ( Walker, 1854, 111) ( Procris)
sequensWalker, 1854, 115
novaehollandiae ( Wallengren, 1860, 39) ( Procris)
Distribution: Australia (Queensland, New South Wales, Victoria, South Australia, Tasmania).
P. nocturna Mollet & Tarmann, 2023, 50
Distribution: Australia (Queensland, New South Wales).
P. modestus Tarmann, 2004, 76
Distribution: Australia (New South Wales).
P. lithopastusTurner, 1926b, 443
Distribution: Australia (New South Wales, Victoria, Tasmania).
P. marriotti Kallies & Mollet, 2011
Distribution: Australia (Victoria).
Pollanisus sp. 1 (discussed and figured in Tarmann, 2004, 79; Mollet & Tarmann, 2023, 18)
Distribution: Australia (Australian Capital Territory).
P. cyanota ( Meyrick, 1886, 793) ( Procris)
Distribution: Australia (Queensland, New South Wales, Victoria).
P. hyacinthusKallies & Mollet, 2018
Distribution: Australia (Kangaroo Island).
P. isolatusTarmann, 2004, 105
Distribution: Australia (Victoria).
P. callicerosTurner, 1926a, 117
Distribution: Australia (southern New South Wales, Victoria, Tasmania (subsp. calliceros); central east and north of New South Wales (subsp. azurea).
P. calliceros callicerosTurner, 1926a, 117
P. calliceros azurea Tarmann, 2004, 107
Genus SaliuncellaJordan, 1907b, 124 (C29)
[Type species: Saliuncella marshalli Jordan, 1907b, 124, by monotypy]
Larval host-plants: Vitaceae.
S. marshalli Jordan, 1907b, 124 (Figure 45)
Distribution: South Africa (Natal, Swasiland).
Genus OnceropygaTurner, 1906, 137
[Type species: Onceropyga anelia Turner, 1906, 137, by monotypy]
Larval host-plants: Vitaceae.
O. anelia Turner, 1906, 137 ( Figure 4)
Distribution: Australia (Queensland, New South Wales).
O. pulchra Tarmann, 2004, 135
Distribution: Australia (Queensland, New South Wales).
Genus HestiochoraMeyrick, 1886, 788
[Type species: Procris tricolor Walker, 1854, 111, by subsequent designation by Turner, 1926b, 441]
Larval host-plants: Myrtaceae (in one species also Proteaceae).
H. erythrota Meyrick, 1886, 789 ( Figure 23)
Distribution: Australia (Queensland, New South Wales).
H. tricolor ( Walker, 1854, 111) ( Procris) ( Figure 5)
Distribution: Australia (Victoria, Tasmania).
H. continentalis Tarmann, 2004, 145
Distribution: Australia (Western Australia, Queensland, New South Wales, Victoria, South Australia).
H. furcata Tarmann, 2004, 148
Distribution: Australia (Queensland, New South Wales, Victoria, South Australia).
H. queenslandensis Tarmann, 2004, 149
Distribution: Australia (Queensland, New South Wales).
H. xanthocoma Meyrick, 1886, 788
Distribution: Australia (Northern Territory, Queensland).
H. occidentalis Tarmann, 2004, 151
Distribution: Australia (Western Australia).
H. intermixta Tarmann, 2004, 152
Distribution: Australia (Queensland, New South Wales, South Australia).
Genus TurneriprocrisBryk, 1936, 304
[Type species: Procris dolensWalker, 1854, 112 , by original designation]
NeoprocrisTurner, 1926b, 445 (a junior homonym of NeoprocrisJordan, 1915, 300 - Lepidoptera, Zygaenidae)
The objective replacement name is TurneriprocrisBryk, 1936, 304
[Type species: Neoprocris saltuaria Jordan, 1915, 300, by original designation]
Larval host-plants: Myrtaceae.
T. dolens ( Walker, 1854, 112) ( Procris)
Distribution: Australia (Queensland, New South Wales, Victoria, South Australia, Tasmania).
Genus MyrtartonaTarmann, 2004, 169
[Type species: Procris rufiventris Walker, 1854, 110, by original designation]
Larval host-plants: Myrtaceae
M. coronias ( Meyrick, 1886, 792) ( Procris)
Distribution: Australia (Queensland, New South Wales, Victoria, South Australia, Tasmania).
M. leucopleura ( Meyrick, 1886, 792) ( Procris)
Distribution: Australia (Queensland, New South Wales).
M. rufiventris ( Walker, 1854, 110) ( Procris) ( Figure 24)
Distribution: Australia (Western Australia, South Australia, western Victoria).
M. mariannae Tarmann, 2004, 176
Distribution: Australia (Queensland).
Genus LevuanaBethune-Baker, 1906, 343(C05)
[Type species: Levuana iridescens Bethune-Baker, 1906, 344, by original designation]
Larval host-plants: Arecaceae (former pest on Cocos nucifera).
L. iridescens Bethune-Baker, 1906, 344
Distribution: Fiji Islands (Viti Levu, Ovalau, Cagalai).
Tribe ArtoniniTarmann, 1994, 120(C06)
[Type-genus: ArtonaWalker, 1854, 439]
Larval host-plants: Poaceae, Zingiberaceae, Pittosporaceae, Lauraceae, Musaceae, Arecaceae. Distribution: Australia, eastern, southern and southeastern Asia, tropical Africa.
Genus ArtonaWalker, 1854, 439(C07)
[Type-species: Artona discivitta Walker, 1854, 440, by monotypy]
Larval host-plants: Poaceae (mainly bamboo) (subgenera Artona, Zeuxippa, Balataea, Fuscartona).
Subgenus ArtonaWalker, 1854, 439(C07)
[Type-species: Artona discivitta Walker, 1854, 440, by monotypy]
Larval host-plants: Poaceae (bamboo).
A. (A). discivittaWalker, 1854, 440(C08) ( Figure 25)
Distribution: southern India.
Subgenus ZeuxippaHerrich-Schäffer, 1855, 87, stat. nov. (C07)
[Type-species: Sphinx pulchra Drury, 1773, 52, by monotypy]
Larval host-plants: Poaceae (bamboo).
A. (Z.). phaeoxanthaHampson, 1920a, 274(C09)
Distribution: southern India (Madras).
A. (Z). zebraicaButler, 1876, 356(C09)
Distribution: northern India, Myanmar (Burma), Thailand, Malaysia.
A. (Z). fulvidaButler, 1876, 356(C09)
diffusaOberthür, 1894, 30 ( Artona fulvida var. diffusa)
Distribution: northern India, Myanmar (Burma), Thailand, Malaysia.
A. (Z). flaviciliataHampson, 1920a, 273(C09)
Distribution: northern India (Sikkim).
A. (Z). confusaButler, 1876, 357(C09)
Distribution: northern India.
A. (Z). digitata Hampson, 1920a, 273 (C09)
Distribution: Myanmar (Burma) (Tenasserim).
A. (Z). khasianaJordan, 1908, 43(C09)
Distribution: NE India (Assam).
A. (Z). sikkimensisElwes, 1890, 379(C09)
Distribution: north India (Sikkim).
A. (Z). nigrescens Butler, 1876, 356, stat. nov. (C10)
Distribution: northern India.
A. (Z). guttata ( Snellen, 1892, 32) ( Syntomis), stat. rev. (C10)
Distribution: Indonesia (Sumatra).
A. (Z). neglecta Hering, 1925, 175, stat. rev. (C10)
Distribution: Myanmar (Burma).
A. (Z). walkeri ( Moore, 1859), 199 (“? Syntomis”) (C11) ( Figure 26)
Distribution: Indonesia (Java) (ssp. walkeri), Indonesia (Bali) (ssp . baliensis).
A. (Z). walkeri walkeri ( Moore, 1859), 199 (“? Syntomis”)
A. (Z). walkeri baliensisJordan, 1908, 43
A. (Z). hainanaButler, 1876, 357
walkeri (sensu auct.) (nec Moore, 1859, 199)
fulvida (sensu auct.) (nec Butler, 1876, 356)
Distribution: China (Hainan).
A. (Z). flavipunctaHampson, 1900, 225
Distribution: northeastern India (Assam).
A. (Z). hypomelasJordan, 1908, 43
Distribution: northern India (NW India, Sikkim).
A. (Z). superba Alphéraky, 1897, 124
Distribution: western China.
A. (Z). pulchra ( Drury, 1773, pl. 29, fig. 3) ( Sphinx)
Distribution: western China.
A. (Z). cuneonotataLeech, 1898, 328
Distribution: western China.
A. (Z). lucasseni( Snellen, 1903), 234 ( Brachartona)
Distribution: Indonesia (Java).
A. (Z). pluristrigataHampson, 1907, 328(C12)
Distribution: Indonesia (Borneo, Pulo Laut).
A. (Z). refulgens( Hampson, 1893, 232) ( Chrysartona)
[systematic position of this taxon needs verification]
Distribution: Myanmar (Burma).
Subgenus BalataeaWalker, 1865, 110, stat. rev. (C07)
[Type-species: Balataea aegerioides Walker, 1865, 111, by monotypy]
RhaphidognathaFelder & Felder, 1862, 31, a junior homonym of RhaphidognathaMurray, 1857, 316 - Insecta: Coleoptera. There is no objective replacement name but the type species of RhaphidognathaFelder & Felder, 1862, is considered to be conspecific with Balataea aegerioides Walker, 1865, the type species of BalataeaWalker, 1865. The latter is therefore available for use as a subjective replacement name.
[Type-species: Rhaphidognatha sesiaeformis Felder & Felder, 1862, 32, by monotypy]
BinthaWalker, 1865, 127
[Type-species: Bintha gracilis Walker, 1865, 127, by monotypy]
Larval host-plants: Poaceae.
A. ( B.) octomaculata( Bremer, 1861, 476) ( Euchromia), stat. rev. ( Figure 27)


sesiaeformis ( Felder & Felder, 1862, 32) ( Rhaphidognatha)
aegerioides Walker, 1865, 111 ( Balataea)
Distribution: Russia (Far East (Amur Region, Khabarovsk Territory, Pimorye Territory)), eastern China, Korea, Japan.
A. ( B.) gracilis( Walker, 1865, 127) ( Bintha), stat. rev.
Distribution: Russia (Far East (Sakhalin, southern Kuril Islands)), eastern China, Korea, Japan.
A. ( B.) taiwanaWileman, 1911, 174 ( Artona “(?)”) (see Owada & Inada, 2005, 5), stat. nov.
Distribution: China (Taiwan).
A. ( B.) kimurai( Owada & Inada, 2005, 2) ( Balataea), stat. nov.
Distribution: Japan (Okinawa).
A. ( B.) angustaAlberti, 1954, 269, stat. rev.
Distribution: eastern China.
A. ( B.) intermedianaAlberti, 1954, 270, stat. rev.
Distribution: eastern China.
A. ( B.) elegantiorAlberti, 1954, 270, stat. rev.
Distribution: eastern China.
Subgenus PseudosesidiaAlberti, 1954, 271, stat. rev. (C07)
[Type-species: Balataea (Pseudosesidia) aegeriaeformis Alberti, 1954, 271, by original designation and monotypy]
Larval host-plants: unknown.
A. ( P.) aegeriaeformis( Alberti, 1954, 271) ( Balataea), stat. rev.
Distribution: southeastern China.
Subgenus FuscartonaEfetov & Tarmann, 2012, 13, 38(C07)
[Type-species: Artona martini Efetov, 1997a, 170, by original designation]
Larval host-plants: Poaceae (bamboo).
A. ( F.) martini Efetov, 1997a, 170 ( Figures 6, 16)
funeralis (sensu Alberti, 1954, 267 ( Balataea) et sensu auct.) (nec Butler, 1879, 351) (see Efetov, 1997a, 166)
Distribution: eastern and southeastern China (including Taiwan), Vietnam, Japan, introduced also to Ogasawara Islands (Japanese Micronesia), New Zealand and even in Europe, viz. Italy ( Marianelli et al. 2020).
A. ( F.) funeralis( Butler, 1879a, 351) ( Procris), stat. rev.
tokyonella (sensu Alberti 1954, 267 ( Balataea)) (nec Matsumura, 1927, 76) (see Efetov, 1997a, 166)
Distribution: Russia (Far East (Sakhalin, southern Kuril Islands)), eastern and southeastern China, Japan.
A. ( F.) uniformis( Alberti, 1954, 268) ( Balataea), stat. rev.
Distribution: China (Shanxi).
A. ( F.) parilisEfetov, 1997a, 175, stat. rev.
Distribution: China (Jiangxi).
Genus Procotes Butler, 1896, 355
[Type-species: Euchromia diminuta Walker, 1854, 230, by original designation and monotypy]
Larval host-plants: unknown.
P. diminuta ( Walker, 1854, 230) ( Euchromia)
Distribution: Sri Lanka.
Genus StriartonaEfetov & Tarmann, 2012, 14, 39
[Type-species: Bintha clathrata Poujade, 1886a, 117, by original designation and monotypy]
Larval host-plants: unknown.
S. clathrata( Poujade, 1886a, 117) ( Bintha)
Distribution: China (Sichuan, Shaanxi).
S. nanlingOwada & Wang, 2021, 123(C13)
Distribution: southern China.
Genus AllobremeriaAlberti, 1954, 277
[Type-species: Allobremeria plurilineata Alberti, 1954, 277, by original designation and monotypy]
Larval host-plants: Poaceae ( Xue & Han, 2003).
All. plurilineataAlberti, 1954, 277
Distribution: southeastern China.
All. maoershanOwada & Wang, 2021, 122
Distribution: southern China.
Genus Amuria Staudinger, 1887b, 172
[Type-species: Amuria cyclopsStaudinger, 1887b, 172, by monotypy]
BrachartonaHampson, 1891, 44
[Type-species: Artona quadrimaculata Moore, 1879a, 390, by original designation]
Larval host-plants: Zingiberaceae (known for A. (Am.) chorista and A. (Am.) trisignata), Pittosporaceae (known for A. (Am.) trisignata), and Lauraceae (known for A. (Am.) trisignata), Musaceae (known for two undescribed species from India and New Guinea).
Am. cyclops Staudinger, 1887b, 172
Distribution: Russia (Far East (Khabarovsk Territory, Primorye Territory), China, Korea.
Am. microstigma ( Jordan, 1908, 44) ( Artona) (C14)
Distribution: northeastern India (Assam).
Am. flavigula ( Hampson, 1896, 477) ( Chrysartona) (C14)
Distribution: northeastern India (Assam).
Am. celebensis ( Jordan, 1908, 45) ( Artona) (C14)
Distribution: Indonesia (Sulawesi).
Am. annulipes ( Jordan, 1908, 47) ( Homophylotis) (C14)
Distribution: Indonesia (Natuna island group, Riau island province: Pulo Laut).
Am. sciara ( Jordan, 1908, 47) ( Homophylotis) (C14)
Distribution: Indonesia (Amboina).
Am. xanthosoma ( Jordan, 1908, 47) ( Homophylotis) (C14)
Distribution: Sula Islands (Sula Mangoli).
Am. chorista ( Jordan, 1908, 44) ( Artona) (C14)
Distribution: northeastern India.
Am. lugubris ( Jordan, 1908, 44) ( Artona) (C14)
Distribution: northeastern India.
Am. postvitta ( Moore, 1879b, 13) ( Artona) (C14)
Distribution: northeastern India (Sikkim).
Am. quadrimaculata ( Moore, 1879a, 390) ( Brachartona), comb. nov. (C14)
Distribution: northern India, Myanmar (Burma).
Am. trisignata ( Snellen, 1903, 235) ( Brachartona), comb. nov. (C14) (Figure 28)
quadrisignata ( Snellen, 1903, 234) ( Brachartona), comb. nov., syn. nov. (C15)
Distribution: Indonesia (Sumatra, Java).
Am. sythoffi ( Snellen, 1903, 236) ( Brachartona), comb. nov. (C14)
Distribution: Indonesia (Java).
Am. melaleuca ( Jordan, 1908, 46) ( Homophylotis), comb. nov. (C17)
Distribution: New Guinea (both subspecies).
Am. melaleuca melaleuca ( Jordan, 1908, 46) ( Homophylotis)
Am. melaleuca postica ( Jordan, 1908, 46) ( Homophylotis melaleuca ssp.)
Am. assimilis ( Jordan, 1908, 47) ( Homophylotis), comb. nov. (C16)
Distribution: Malaysia (Malakka).
Am. aenea ( Jordan, 1925, 231) ( Homophylotis), comb. nov. (C18)
Distribution: Papua New Guinea (Feni Island (Ambitle island) east of New Ireland (Latangai island)).
Am. chalcosoma (Jordan, 1926, 366) ( Artona), comb. nov. (C19)
Distribution: Indonesia (Kai Ketjil).
Am. albicilia ( Hampson, 1900, 222) ( Artona), comb. nov. (C20)
Distribution: northeastern India (Naga Hills).
Am. nigra ( Hampson, 1893, 237) ( Tasema), comb. nov. (C20)
Distribution: Myanmar (Burma).
Am. purpurata ( Jordan, 1908, 47) ( Homophylotis), comb. nov. (C20)
Distribution: Papua New Guinea.
Am. neglecta ( Tarmann, 2004, 203) ( Pseudoamuria), comb. nov. (C20)
Distribution: Australia (Queensland).
Genus PalmartonaTarmann, 2004, 207(C16)
[Type species: Brachartona catoxantha Hampson, 1893 (“1892”), 233, by original designation]
Larval host-plants: Arecaceae (pest on Cocos nucifera) L.
Palm. catoxantha ( Hampson, 1893, 233) ( Brachartona)
Distribution: Myanmar (Tenasserim), Malaysia, Singapore, Indonesia (Sumatra, Nias, Java, Bangka, Kalimantan, Sulawesi), Philippines (Palawan), Papua New Guinea, Australia (Queensland).
Genus AustralartonaTarmann, 2004, 185
[Type species: Australartona mirabilis Tarmann, 2004, 186, by original designation and monotypy]
Larval host-plants: Poaceae.
Austr. mirabilis Tarmann, 2004, 186
Distribution: Australia (Queensland, New South Wales).
Genus HomophylotisTurner, 1904, 243
[Type species: Homophylotis thyridota Turner, 1904, 243, by monotypy]
Homopylotis [sic] Alberti, 1954, 408, misspelling
Larval host-plants: unknown.
H. thyridota Turner, 1904, 243
Distribution: Australia (Queensland).
H. pseudothyridota Tarmann, 2004, 194
Distribution: Australia (Queensland).
H. artonoides Tarmann, 2004, 195
Distribution: Australia (Queensland).
H. doloides ( Pagenstecher, 1900, 25) ( Procris)
Distribution: Papua New Guinea (Bismarck Archipelago).
Genus PseudoamuriaTarmann, 2004, 201
[Type species: Pseudoamuria uptoni Tarmann, 2004, 202, by original designation]
Larval host-plants: unknown.
Pseud. uptoni Tarmann, 2004, 202
Distribution: Australia (Queensland).
Genus ArachotiaMoore, 1879b, 14
[Type-species: Arachotia flaviplagaMoore, 1879b, 14 , by monotypy]
Larval host-plants: unknown.
Arach. flaviplaga Moore, 1879b, 14
Distribution: northern and northeastern India (West Bengal to Assam).
Arach. piaoac Owada & Pham, 2022, 203
Distribution: Vietnam.
Arach. euglenia Jordan, 1908, 50
Distribution: northeastern India (Assam).
Arach. tamdao Owada & Pham, 2021, 40
Distribution: Vietnam (Vinh Phuc, Tam Dao).
Arach. sapa Owada & Pham, 2021, 43
Distribution: Vietnam (Lao Cai, Sa Pa).
Arach. nanling Owada & Wang, 2021, 43
Distribution: China (Guangdong, Shaoguan, Nanling).
Arach. dadongshan Owada & Wang, 2021, 43
Distribution: China (Guangdong, Lianzhou, Dadongshan).
Arach. hohuanshanensis Shih & Owada, 2022, 136
Distribution: China (Taiwan).
Arach. vespoides Moore, 1879a, 390
Distribution: northern India.
Arach. quadricolor ( Semper, 1898, 426) ( Lophosoma)
aeneaJordan, 1908, 50
Distribution: Philippines.
Arach. xeniaetamara Bryk, 1936, 304
hyalinaHering, 1925, 175 (nec Leech, 1889b, 123)
Distribution: Philippines (Luzon).
Genus ChalconyclesJordan, 1907b, 123(C29)
[Type species: Chalconycles vetulina Jordan, 1907b, 124, by monotypy]
Larval host-plants: Arecaceae ( Cocos nucifera L. (coconut), Elaeis guineensis Jacq. (oil palm), palms).
C. vetulinaJordan, 1907b, 124
velutina [sic] Jordan, 1907b, pl. 2k, misspelling
Distribution: Eastern and Central Africa (Uganda).
C. chloauges ( Holland, 1893, 374) ( Adscita)
Distribution, Africa (Cameroon, Gaboon, Congo)
C. catori ( Jordan, 1907b, 125) ( Homophylotis)
Distribution: Africa (Sierra Leone, Ivory Coast, Togo, Gabon).
C. anhyalea Hampson, 1920a, 276
Distribution: Africa (Gabon).
C. albipalpis Hampson, 1920a, 275
Distribution: Africa (Ivory Coast).
C. chalybeia Rebel, 1914, 290
Distribution: Central Africa.
Genus NeobalataeaAlberti, 1954, 306(C29)
[Type species: Neobalataea nigriventris Alberti, 1954, 307, by original designation]
Larval host-plants: unknown.
N. nigriventris Alberti, 1954, 307
Distribution: Africa (Tanzania).
N. leptis ( Jordan, 1907b, 125) ( Homophylotis)
Distribution: Africa (Angola).
Tribe Cleleini Efetov & Tarmann, tribus nov. (C21)
[Type-genus: Clelea Walker, 1854, 465 ]
Larval host-plants: Vitaceae, Hydrangeaceae, Fagaceae, Rosaceae, Poaceae. Distribution: eastern, southern and southeastern Asia, tropical Africa.
Genus CleleaWalker, 1854, 465
[Type-species: Clelea sapphirina Walker, 1854, 465, by monotypy]
Larval host-plants: Vitaceae.
C. sapphirina Walker, 1854, 465
Distribution: southern China, Myanmar (Burma), India [comment: due to a lot of misidentifications in literature the real distribution of Clelea sapphirina is not known.]
C. discriminis Swinhoe, 1891, 474
Distribution: northeastern India (Assam).
C. simplexJordan, 1908, 45
Distribution: northeastern India (Assam).
C. nigroviridis Elwes, 1890, 380 ( Figure 32)
Distribution: northeastern India, Myanmar (Burma).
C. cyanescens Alberti, 1954, 289
Distribution: southern China (Hunan, Guandong) (subsp. cyanescens); eastern China (subsp. monotona) (Jiangsu, Zhejiang).
C. cyanescens cyanescens Alberti, 1954, 289
C. cyanescens monotona Alberti, 1954, 289
C. formosana Strand, 1915, 119 (as nigroviridis v. formosana) ( C22)
Distribution: China (Taiwan), Japan (Sakishima).
C. formosana formosanaStrand, 1915, 119
C. formosana simplicior Bryk, 1926, 255, stat. nov.
C. formosana kanoi Owada, 2021, 205, stat. nov.
C. melli Hering, 1925a, 174
Distribution: China (Guangdong).
C. esakiiInoue, 1958, 238 ( Figure 7)
Distribution: Japan.
C. yuennana Alberti, 1954, 290 (as yünnana)
Distribution: China (Yunnan).
C. chala ( Moore, 1859, 311) ( Procris)
Distribution: Indonesia (Java, Borneo?).
C. refulgens Hampson, 1905, 193
Distribution: northeastern India (Assam), Myanmar (Burma).
C. metacyanea Hampson, 1896, 467
Distribution: northeastern India (Assam).
C. plumbeola Hampson, 1893, 240
Distribution: northeastern India (Assam), Bhutan, Myanmar (Burma).
C. albofascia ( Leech, 1898, 340) ( Arbudas)
albifascia [sic] Bryk, 1936, 247, misspelling
Distribution: China (Sichuan).
Genus ThibetanaEfetov & Tarmann, 1995b, 74
[Type-species: Artona sieversi Alphéraky, 1892, 5, by original designation]
Larval host-plants: unknown.
Th. sieversi ( Alphéraky, 1892, 5) ( Artona)
dejeani ( Oberthür, 1894, 29) ( Artona)
gephyra ( Hering, 1936, 1) ( Artona)
Distribution: China (Qinghai, Sichuan).
Th. delavayi ( Oberthür, 1894, 29) ( Artona)
Distribution: China (Yunnan).
Th. witti Efetov, 1997c, 509
Distribution: China (eastern Tibet).
Th. zebra ( Elwes, 1890, 379) ( Artona) (C23)
Distribution: northern India (Sikkim).
Th. postalba ( Elwes, 1890, 379) ( Artona) (C23)
Distribution: northern India (Sikkim).
Th. keili Efetov & Tarmann, 2017b(C23)
Distribution: China (eastern Tibet).
Genus BremeriaAlphéraky, 1892, 7
[Type-species: Bremeria manza Alphéraky, 1892, 7, by monotypy]
SubcleleaAlberti, 1954, 292 ( Clelea subg.)
[Type-species: Clelea (Subclelea) parabella Alberti, 1954, 293, by original designation]
Larval host-plants: unknown.
B. manza Alphéraky, 1892, 7
Distribution: northeastern China.
B. parabella ( Alberti, 1954, 293) ( Clelea)
Distribution: eastern China (Zhejiang).
B. aurulenta ( Poujade, 1886a, 116) ( Bintha)
Distribution: China (Sichuan) (subsp. aurulenta); China (Zhejiang) (subsp. bella).
B. aurulenta aurulenta ( Poujade, 1886a, 116) ( Bintha)
B. aurulenta bella ( Alberti, 1954, 292) ( Clelea)
B. albomacula ( Leech, 1898, 329) ( Artona)
Distribution: China (Sichuan).
B. sinica Alphéraky, 1897, 122
Distribution: China (Sichuan).
B. cyanicornis ( Poujade, 1886a, 116) ( Bintha) (see Efetov, 2000a, 23)
Distribution: China (Sichuan).
Genus CerodendraTremewan, 1973, 122
[Type-species: Dendrocera quadripunctata Hampson, 1893 (“1892”), 231, by original designation (for
DendroceraHampson, 1893 (“1892”)]
DendroceraHampson, 1893 (“1892”), 231, a junior homonym of DendroceraLamarck, 1817, 422 - Insecta: Coleoptera. The objective replacement name is CerodendraTremewan, 1973
Larval host-plants: unknown.
C. bipuncta ( Hampson, 1895, 283) ( Clelea)
bipunctataJordan, 1908, (explicatio tabulae)
Distribution: south India (Nilgiris).
C. quadripunctata ( Hampson, 1893, 231) ( Dendrocera)
Distribution: Sri Lanka.
Genus HagiangaMollet, 2020a, 106
[Type-species: Hagianga tieni Mollet, 2020a, 107, by original designation and monotypy]
Larval host-plants: unknown.
H. tieni Mollet, 2020a, 107
Distribution: northern Vietnam.
Genus ChrysartonaSwinhoe, 1892, 57 (see Efetov, 1996; Efetov & Tarmann, 2008b, 2008c) [Type-species: Procris stipata Walker, 1854, 114, by original designation and monotypy]
Larval host-plants: Vitaceae (known for one species that has been misidentified as Ch. stipata Walker, 1854, from Taiwan) (see Yen & Fan, 1995; Efetov, 2006).
Subgenus ChrysartonaSwinhoe, 1892, 57
[Type-species: Procris stipata Walker, 1854, 114, by original designation and monotypy]
Ch. ( Ch.) stipata( Walker, 1854, 114) ( Procris)
Distribution: northern and northeastern India, Myanmar (Burma). All records from other regions (e.g. China, Indonesia) refer to other species.
Ch. ( Ch.) efetoviParshkova, 2007, 143
Distribution: northeastern India (Assam).
Ch. ( Ch.) hausmanniEfetov, 2006, 27
Distribution: Indonesia (Java, Borneo). (C24)
Ch. ( Ch.) murziniMollet, 2016a, 59(C25)
Distribution: Thailand.
Ch. ( Ch.) stueningiEfetov, 2006, 29
stipata partim (sensu Alberti, 1954, 294 ( Clelea)) (nec Walker, 1854, 114)
Distribution: eastern China.
Ch. ( Ch.) tremewaniEfetov, 2006, 31
Distribution: northeastern India.
Ch. (Ch.) sineviEfetov, 2006, 33
stipata partim (sensu Alberti, 1954, 294, pl. 28, figs 9a-9c ( Clelea)) (nec Walker, 1854, 114)
Distribution: China, Myanmar (Burma).
Ch. ( Ch.) sylvianaeMollet, 2016a, 62
Distribution: Myanmar (Burma).
Ch. ( Ch.) dangvaniMollet, 2018, 80 ( Figure 31)
Distribution: southern Vietnam.
Subgenus ChrystremewanaEfetov, 2006, 36
[Type-species: Chrysartona birmana Efetov, 2006, 37, by original designation]
Ch. ( Chrystrem.) birmanaEfetov, 2006, 37
Distribution: northeastern India (Assam), Myanmar (Burma), Laos.
Ch. ( Chrystrem.) honeyiEfetov, 2006, 39
Distribution: northeastern India (Assam).
Subgenus ChrystarmannaEfetov, 2006, 41
[Type-species: Chrysartona sikkima Efetov, 2006, 42, by original designation]
Ch. ( Chrystarm.) sikkimaEfetov, 2006, 42
stipata (sensu Wang, 1995, 23 ( Clelea)) (nec Walker, 1854, 114)
Distribution: northeastern India (Sikkim).
Ch. ( Chrystarm.) meyiEfetov, 2006, 44
stipata partim (sensu Alberti, 1954, 294 ( Clelea)) (nec Walker, 1854, 114)
Distribution: Indonesia (Java).
Ch. ( Chrystarm.) margarita Efetov, 2006, 47
Distribution: northeastern India (Assam).
Ch. (Chrystarm.) mineti Efetov & Tarmann, 2013b, 198
Distribution: northern Vietnam.
Ch. ( Chrystarm.) antenorMollet, 2018, 79 (C26)
Distribution: southern Vietnam.
Ch. ( Chrystarm.) pravata(Moore, 1859, 199) ( Syntomis)
Distribution: Indonesia (Sumatra, Java, southeastern Borneo), Malaysia (northern Borneo), Philippines (Luzon).
Ch. ( Chrystarm.) variata( Swinhoe, 1892, 58) ( Phacusa)
Distribution: Indonesia (New Guinea, Irian Jaya) (subsp. variata); Indonesia (island Mefor NW. of New Guinea) (subsp. fumosa); Indonesia (Sumba, islands Kepulauan Kai, New Guinea (Irian Jaya)), Papua New Guinea (subsp. separata); Indonesia (island Ambon) (subsp. amboinensis); Indonesia (island Bacan) (subsp. guttigera).
Ch. ( Chrystarm.) variata variata ( Swinhoe, 1892, 58) ( Phacusa)
Ch. ( Chrystarm.) variata fumosa ( Jordan, 1908, 46) ( Clelea)
Ch. ( Chrystarm.) variata separata ( Jordan, 1908, 46) ( Clelea)
Ch. ( Chrystarm.) variata amboinensis ( Jordan, 1908, 46) ( Clelea)
Ch. ( Chrystarm.) variata guttigera ( Jordan, 1908, 46) ( Clelea)
Ch. ( Chrystarm.) explorata( Hering, 1925a, 175) ( Clelea)
Distribution: Papua New Guinea.
Genus AraeoceraHampson, 1893 (“1892”), 244
[Type-species: Araeocera cyanescens Hampson, 1893 (“1892”), 244, by original designation]
Larval host-plants: Poaceae (bamboo) (known for A. compta according to a rearing note by R. W. Paine on paratype label).
A. compta Jordan, 1908, 48
Distribution: Indonesia (South Flores).
A. cyanescensHampson, 1893, 244
Distribution: Myanmar (Burma).
A. posthyalina Hampson, 1893, 244
Distribution: India (Sikkim).
A. prasina Jordan, 1931, 277
Distribution: Indonesia (Java).
Genus PlatyzygaenaSwinhoe, 1892, 57
[Type-species: Soritia moelleri Elwes, 1890, 385, by original designation and monotypy]
Larval host-plants: unknown.
P. moelleri ( Elwes, 1890, 385) ( Soritia)
Distribution: northeastern India (Sikkim, Assam).
P. melaleuca ( Jordan, 1907a, 17) ( Clelea) (comb.: Efetov & Tarmann, 2012, 17)
Distribution: China (Sichuan).
Genus PiarosomaHampson, 1893 (“1892”), 243 ( C27)
[Type species: Piarosoma albicinctum Hampson, 1893 (“1892”), 243, fig. 160, by original designation and monotypy]
HysterosceneHering, 1925, 177
[Type-species: Hysteroscene extravagans Hering, 1925a, 177, by original designation]
Hysteroscena [sic] Horie & Wang, 2011, 44, pl. 3, fig. 2, misspelling)
Larval host-plants: Hydrangeaceae ( Hydrangea macrophylla (Thunb.) Ser.) (known for P. fushan Owada & Shih, 2022) ( Owada et al. 2022b).
P. arunachalensis Sondhi, Efetov, Tarmann & Kunte, 2023, 140.
Distribution: northeastern India.
P. albicinctum Hampson, 1893, 243
Distribution: Myanmar (Burma).
P. hyalina ( Leech, 1889, 123) ( Arachotia)
univittataStrand, 1915, 122 ( Piarosoma hyalina ab.) (unavailable)
Distribution: southeastern and southern China.
P. thibetana ( Oberthür, 1894, 31) ( Phacusa)
hyalina partim (sensu Leech 1898, 336 ( Piarosoma)) (nec Leech, 1889, 123)
thibetana (sensu Jordan, 1907, 17, pl. 3-i ( Piarosoma hyalina f. thibetana))
thibetana partim (sensu Bryk, 1936, 269 ( Piarosoma hyalina subsp. thibetana))
hyalina partim (sensu Alberti, 1954, 223 ( Hysteroscene hyalina)) (nec Leech, 1889, 123)
Distribution: western China.
P. guangdong Owada & Wang, 2022, 214
thibetana (sensu Hering, 1925b, 82, fig. 3 (male genitalia) ( Hysteroscene thibetana)) (nec Oberthür, 1894, 31)
thibetana partim (sensu Bryk, 1936, 269 ( Piarosoma hyalina subsp. thibetana)) (nec Oberthür, 1894, 31)
thibetana (sensu Alberti, 1954, 223, pl. 17, fig. 1 ( Hysteroscene hyalina)) (nec Leech, 1889, 123))
thibetana (sensu Horie & Wang, 2011, 45, pl. 3, fig. 5 ( Piarosoma thibetana)) (nec Oberthür,1894, 31)
thibettana (sensu Horie & Wang, 2011, pl. 3, caption of figure 5, misspelling ( Piarosoma thibettana [sic])) (nec Oberthür, 1894, 31)
Distribution: China (Guangdong).
P. tamdao Owada & Wang, 2022, 215
Distribution: Vietnam (Vinh Phuc, Tam Dao).
P. bachma Owada & Pham, 2022, 217
Distribution: Vietnam (Thua Thien Hue, Bach Ma).
P. oquyho Owada & Pham, 2022, 207
Distribution: Vietnam (Lao Cai, Sa Pa, O Quy Ho).
P. melli ( Hering, 1925b, 82) ( Hysteroscene)
Distribution: southern China.
P. sapa Owada & Pham, 2022, 210
Distribution: Vietnam (Lao Cai, Sa Pa).
P. fushan Owada & Shih, 2022, 220
hyalina partim (sensu Matsumura, 1931, 991, fig. ( Piarosoma)) (nec Leech, 1889, 123)
univittata (sensu Inoue, 1987, 299 ( Piarosoma)) (nec Hering, 1925a, 178)
hyalina partim (sensu Tarmann, 1992b, 98 ( Hysteroscene)) (nec Leech, 1889, 123)
hyalina partim (sensu Efetov & Tarmann, 1995, 77 ( Hysteroscene)) (nec Leech, 1889, 123)
hyalina partim (sensu Wang, 1995, 9-10 (( Hysteroscene)) (nec Leech, 1889, 123)
hyalina partim (sensu Efetov & Tarmann, 2012, 17 ( Hysteroscene)) (nec Leech, 1889, 123)
Distribution: China (Taiwan: Taipei, Taoyuan, Ilan, Hsinchu).
P. extravagans ( Hering, 1925a, 177) ( Hysteroscene)
annulatissimaStrand, 1915, 122 ( Piarosoma hyalina ab.) (unavailable)
univittata ( Hering, 1925a, 178) ( Hysteroscene sp.)
Distribution: China (Taiwan).
P. sizala ( Swinhoe, 1894, 441) ( Phacusa) (sensu Bryk, 1936, 269) (C 28)
Distribution: northeastern India (Assam).
Genus TasemaWalker, 1856, 1597
[Type-species: Tasema bipars Walker, 1856, 1597, by monotypy]
Larval host-plants: Fagaceae.
T. bipars Walker, 1856, 1597
Distribution: northern and northeastern India.
T. viridescens Alberti, 1954, 282
Distribution: China (Yunnan).
T. longipennis Hampson, 1893, 236
Distribution: Myanmar (Burma)
Genus EphemeroideaHampson, 1893 (“1892”), 242
[Type-species: Ephemeroidea ariel Hampson, 1893 (“1892”), 242, by original designation]
Larval host-plants: Poaceae (bamboo) (known for E. viridescens).
E. ariel Hampson, 1893, 242
Distribution: northeastern India (Assam), Myanmar (Burma).
E. cyanea Jordan, 1908, 50
Distribution: northeastern India (Assam).
E. flavocincta Hampson, 1893, 242
Distribution: Myanmar (Burma).
E. virescens Snellen, 1903, 222 ( Figure 29)
Distribution: Indonesia (Sumatra, Java).
Genus LophosomaSwinhoe, 1892
[Type-species: Syntomis cupreaWalker, 1856, 1596, by original designation]
Larval host-plants: unknown.
L. cuprea ( Walker, 1856, 1596) ( Syntomis) ( Figure 30)
Distribution: northern India, Andaman islands.
L. quadricolor ( Walker, 1856, 1596) ( Syntomis)
Distribution: northern India.
Genus MorioniaJordan, 1910, 256
[Type-species: Morionia sciaraJordan, 1910, 256 , by monotypy]
Larval host-plants: unknown.
M. sciara Jordan, 1910, 256 ( Figure 33)
sciaria [sic] Efetov & Tarmann, 1995b, 76, misspelling
Distribution: China (Taiwan).
Genus PseudoinopeEfetov & Tarmann, 1999b, 165
[Type-species: Procris fuscaLeech, 1889a, 595 , by original designation and monotypy]
Larval host-plants: Hydrangeaceae.
P. fusca ( Leech, 1889a, 595) ( Procris)
syriaca ( Hampson, 1920a, 275) ( Clelea) (synonymized by Efetov & Tarmann, 1999b, 167)
albicilia ( Inoue, 1976a, 159) ( Clelea) (synonymized by Efetov & Tarmann, 1999b, 167)
Distribution: Japan.
Genus InopeStaudinger, 1887b, 170 (see Efetov & Tarmann, 1996)
[Type-species: Inope heterogyna Staudinger, 1887b, 170, by monotypy]
AglainoStaudinger, 1887b, 171
[Type-species: Aglaino maerens Staudinger, 1887b, 171, by monotypy]
PollanistaStrand, 1915, 118
[Type-species: Pollanista inconspicuaStrand, 1915, 118 , by original designation]
Larval host-plants: Rosaceae.
I. heterogyna Staudinger, 1887b, 170
impellucidaGraeser, 1888, 108
Distribution: Russia (Far East), Japan.
I. maerens ( Staudinger, 1887b, 171) ( Aglaino)
microphaea ( Hampson, 1920a, 275) ( Clelea) (synonymized by Efetov & Tarmann, 1999b, 167)
tokyonella ( Matsumura, 1927, 76) (Artona) (synonymized by Efetov, 1997a, 166)
sachalinensis ( Matsumura, 1927, 77) ( Artona) (synonymized by Efetov, 1999a, 235)
fusca (sensu Inoue, 1976a, 160 ( Clelea)) (nec Leech, 1889a, 595) (synonymized by Efetov & Tarmann, 1999b,167)
moerens [sic] ( Jordan, 1907a, pl. 3) ( Artona), misspelling
Distribution: Russia (Far East), northeastern China, Korea, Japan.
I. inconspicua ( Strand, 1915, 118) ( Pollanista)
Distribution: China (Taiwan).
I. fuliginosa ( Moore, 1879a, 14) ( Artona)
Distribution: northeastern India.
Genus AlloprocrisHering, 1925b, 84
[Type-species: Alloprocris draesekei Hering, 1925b, 84; by original designation]
Larval host-plants: unknown.
A. adusta Draeseke, 1926, 44
Distribution: Western China.
A. draesekei Hering, 1925b, 84
Distribution: Western China.
A. draesekei draesekei Hering, 1925b, 84)
A. draesekei ellenae Alberti, 1954, 286
A. draesekei hoeneiAlberti, 1954, 286 (as hönei)
A. augustaeAlberti, 1940, 99
Distribution: Central China.
A. spielhagenae Alberti, 1954, 286
Distribution: Western China.
LeptozygaenaJordan, 1907c, 13
[Type species: Leptozygaena gracilis Jordan, 1907c, 13, by monotypy]
Larval host-plants: unknown.
L. gracilisJordan, 1907c, 13
Distribution: New Guinea.
Genus Metanycles Butler, 1876, 425(C29)
[Type species: Aclytia contracta Walker, 1865 (“1864”), 102, by original designation]
M. contracta ( Walker, 1865), 102) ( Aclytia)
Distribution: Africa (Sierra Leone).
M. flavibasis Hampson, 1920a, 274
Distribution: Eastern Africa (Malawi (Mount Mulanje)).
M. sachtlebeni Alberti, 1954, 304
Distribution: Africa (Ecuatorial Guinea, Gabon).
Genus Aethioprocris Alberti, 1954, 305(C29)
[Type species: Aethioprocris togoensis Alberti, 1954, 306, by original designation and monotypy]
Larval host-plants: unknown.
A. togoensis Alberti, 1954, 306
Distribution: Africa (Togo).
A. congoensis Alberti, 1957, 294
Distribution: Africa (Congo).
Genus SthenoprocrisHampson, 1920a, 275(C29)
[Type species: Sthenoprocris malgassica Hampson, 1920a, 275, by original designation]
Larval host-plants: unknown.
S. malgassica Hampson, 1920a, 275
Distribution: Madagascar.
S. meinickei Hering, 1928, 280
Distribution: Eastern Africa (Tanzania).
S. brondeli Viette, 1978, 59
Distribution: Eastern Madagascar.
Genus AnkasocrisViette, 1965, 122(C29)
[Type species: Ankasocris striatus Viette, 1965, 123, by original designation and monotypy]
Larval host-plants: unknown.
A. striatus Viette, 1965, 123
Distribution: Madagascar.
Genus IschnusiaJordan, 1928, 133(C29)
[Type species: Syntomis culiculina Mabille, 1878, 85, by monotypy]
Pseudonacta (partim) Kirby, 1892, 107
[Type species: Syntomis culiculina Mabille, 1878, 85, by monotypy]
Larval host-plants: unknown.
I. culiculina ( Mabille, 1878, 85) ( Syntomis)
Distribution: Madagascar.
Genus AlteramenelikiaAlberti, 1971, 239(C29)
[Type species: Menelikia jordani Alberti, 1954, 309, by original designation (for MenelikiaAlberti, 1954)]
MenelikiaAlberti, 1954, 308
[Type species: Menelikia jordani Alberti, 1954, 309, by original designation], a junior homonym of MenelikiaArambourg, 1941, 341 (Mammalia)
Larval host-plants: unknown.
A. jordani Alberti, 1954, 309
Distribution: Eastern Africa (Ethiopia).
Genus TriacanthiaRomieux, 1937, 124(C29)
[Type species: Triacanthia filictorum Romieux, 1937, 126, by original designation]
Larval host-plants: unknown.
T. filictorum Romieux, 1937, 126
Distribution: Africa (Congo).
Genus XenoprocrisRomieux, 1937, 127 (C29)
[Type species: Xenoprocris jordani Romieux, 1937, 129, by original designation]
Larval host-plants: unknown.
X. jordani Romieux, 1937, 129
Distribution: Africa (Congo).
Genus MadaprocrisViette, 1978, 61(C29)
[Type species: Madaprocris minetorum Viette, 1978, 62, by original designation and monotypy]
Madaproctis [sic] Viette, 1978, 60, fig.2, misspelling
Larval host-plants: unknown.
M. minetorum Viette, 1978, 62
Distribution: Central Madagascar.
Genus Afromalamblia Efetov & Tarmann, gen. nov. (C30)
[Type species: Malamblia flavipalpis Hampson, 1910, 488, here designated]
A. flavipalpis ( Hamspon, 1910, 488) ( Malamblia), comb. nov.
Distribution: Southern Africa (Zimbabwe).
A. titoea ( Druce, 1896, 351) ( Brachartona), comb. nov.
Distribution: Southern Africa (Transvaal).
A. unxia ( Druce, 1896, 351) ( Brachartona), comb. nov.
Distribution: Nigeria (Lagos).
Tribe ProcridiniBoisduval, 1828, 38 (as Procridae) (C31)
[Type genus: Procris [ Fabricius in Illiger], 1807, 289] (see Taeger & Gaedike, 2001, 87)
Larval host-plants: Vitaceae, Rosaceae, Fagaceae, Polygonaceae, Asteraceae, Plumbaginaceae, Cistaceae, Geraniaceae and some others, a few species are polyphagous.
Distribution: Europe, Asia, Africa, America.
Genus Pseudoilliberis Efetov & Tarmann, 2012, 19, 42
[Type-species: Illiberis kuprijanovi Efetov, 1995a, 237, by original designation and monotypy]
Larval host-plants: Fagaceae ( Quercus).
Ps. kuprijanovi ( Efetov, 1995a, 237)
Distribution: Russia (Far East (Amur Region, Khabarovsk Territory, Pimorye Territory)), nothern China, Korea.
Genus IlliberisWalker, 1854, 280
[Type-species: Illiberis sinensis Walker, 1854, 280, by monotypy]
Larval host-plants: Rosaceae, Fagaceae ( Quercus), Corylaceae ( Corylus).
Subgenus NikilliberisEfetov & Tarmann, 2012, 19, 43
[Type-species: Illiberis kardakoffiAlberti, 1951, 143 , by original designation and monotypy]
Larval host-plants: Fagaceae ( Quercus) (Efetov, 2000b), Corylaceae ( Corylus) ( Efetov, 2005b).
I. ( N.) kardakoffiAlberti, 1951, 143
Distribution: Russia (Far East (Amur Region, Khabarovsk Territory, Pimorye Territory)), northern China.
Subgenus PrimilliberisAlberti, 1954, 230
[Type-species: Illiberis laeva Püngeler, 1914, 53, by original designation]
Larval host-plant: Rosaceae.
I. ( P.) laevaPüngeler, 1914, 53
glaucosquamataStrand, 1915, 120
Distribution: China (eastern part and Taiwan).
I. ( P.) yeniEfetov, 1997b, 231(Illiberis)
Distribution: China (Taiwan).
I. ( P.) rotundataJordan, 1907a, 15 ( Illiberis)
fujisanaMatsumura, 1927, 78 ( Illiberis)
fumataAlberti, 1954, 231 (see Efetov, 2005a, 200) ( Illiberis)
kaszabi Alberti, 1970b: 194 (see Efetov, 2005a, 200) ( Illiberis)
ononicaDubatolov, 2002: 109 (see Efetov, 2005a, 200) ( Illiberis)
psychina (sensu Alberti, 1951, 134) ( Illiberis) (nec Oberthür, 1880, 28)
rotundifolia [sic] Hofmann & Kia-Hofmann, 2011, 66, misspelling ( Illiberis)
Distribution: Russia (southeastern Siberia (Zabaykalye Territory), Far East (Amur Region)) ( Efetov, 2005, 61, 210), Mongolia, China, Korea ( Kim, Sohn & Cho, 2004, 237), Japan.
I. ( P.) pruniDyar, 1905a, 954 ( Illiberis)
Distribution: Russia (southeastern Siberia (Zabaykalye Territory), Far East (Amur Region, Khabarovsk Territory, Pimorye Territory)), China, Korea (subsp. pseudopsychina); Japan (subsp. pruni).
I. ( P.) pruni pruni Dyar, 1905a, 954
aomoriensisMatsumura, 1927, 77
japonica Alberti 1951, 140 ( Illiberis pseudopsychina subsp.)
elegans (sensu Jordan, 1907a, 7 ( Procris)) (nec Poujade, 1886, 143) (see Alberti, 1954, 232)
nigra (sensu auct.) (nec Leech, 1889a, 595 ( Procris))
I. ( P.) pruni pseudopsychinaAlberti, 1951, 139 ( Illiberis)
sinensis auct. (nec Walker, 1854, 280) ( Illiberis)
Subgenus IlliberisWalker, 1854, 280
[Type-species: Illiberis sinensis Walker, 1854, 280, by monotypy]
Larval host-plants: Rosaceae ( Kim, Sohn & Cho, 2004; Efetov, Klir & Tarmann, 2010a).
I. ( I.) sinensis Walker, 1854, 280
formosana ( Matsumura, 1927, 76) ( Procris)
Distribution: China, Korea. Literature data on the presence of I. sinensis in the Russian Far East are based on misidentifications of I. pruni Dyar, 1905a (see Efetov, 2005a, 200).
I. ( I.) assimilisJordan, 1907a, 15
Distribution: Russia (Far East (Pimorye Territory)), eastern China, Korea, Japan.
I. ( I.) hoeneiAlberti, 1954, 234 (as hönei)
Distribution: China (Yunnan).
I. ( I.) ellenaeAlberti, 1954, 235
Distribution: China (Yunnan).
I. ( I.) crispoidesMollet, 2020a, 62
Distribution: Vietnam (North Vietnam).
I. ( I.) crispaMollet, 2016b, 162
Distribution: China (Hainan).
Subgenus EuphacusaMatsumura, 1927, 79
[Type-species: Euphacusa taikozana Matsumura, 1927, 79, by original designation and monotypy]
Larval host-plants: Vitaceae ( Kim, Sohn & Cho, 2004).
silvestris-group
I. ( E.) silvestris( Strand, 1915, 121) ( Phacusa)
taikozana ( Matsumura, 1927, 79) ( Euphacusa)
Distribution: China (Taiwan).
cybele-group
I. ( E.) phacusanaStrand, 1915, 120
Distribution: China (Taiwan).
I. ( E.) dirce( Leech, 1889a, 596) ( Northia)
Distribution: eastern China, Korea.
I. ( E.) formosensisStrand, 1915, 120
horishanaMatsumura, 1927, 78
Distribution: China (Taiwan).
I. ( E.) inermis Alberti, 1954, 238
Distribution: China (Hunan).
I. ( E.) cybele( Leech, 1889a, 596) ( Northia)
contrariaAlberti, 1954, 239 (see Efetov, 2005a, 201)
Distribution: Russia (Far East (Pimorye Territory)), China (including Taiwan).
I. (E.) paracybeleAlberti, 1954, 239 ( Illiberis)
Distribution: China (Taiwan).
Subgenus AlterasveniaAlberti, 1971a, 239
[Type-species: Northia ulmivora Graeser, 1888, 107, by original designation]
SveniaAlberti, 1954, 246 (a junior homonym of Svenia Brotzen, 1937, 66 - Protozoa. The objective replacement name is AlterasveniaAlberti, 1971a, 239)
[Type-species: Northia ulmivora Graeser, 1888, 107, by original designation]
Distribution: Trinidad.
Larval host-plants: Ulmaceae.
ulmivora-group
I. ( A.) ulmivora( Graeser, 1888, 107) ( Northia)
pekinensis ( Draeseke, 1926, 44) ( Procris)
Distribution: Russia (Far East (Khabarovsk Territory, Pimorye Territory)), Mongolia, northern China.
I. ( A.) yuennanensisAlberti, 1951, 139 (as yünnanensis)
Distribution: central and southern China.
ochracea-group
I. ( A.) ochraceaLeech, 1898, 335
Distribution: China (including Taiwan).
I. ( A.) kislovskyiEfetov & Tarmann, 2016a, 138
Distribution: northern Myanmar (Burma).
I. ( A.) habaensisMollet, 2015, 224
Distribution: southwestern China.
I. ( A.) wuzhiensisMollet, 2015, 227
Distribution: China (Hainan).
banmauka-group
I. ( A.) banmaukaEfetov & Tarmann, 2014a, 63
Distribution: northern Myanmar (Burma).
I. ( A.) cernyiEfetov & Tarmann, 2013a, 33 ( Figure 34)
Distribution: northern Thailand.
Genus Pseudohedina Efetov & Tarmann, gen. nov. (C32)
[Type species: Illiberis paradistincta Alberti, 1954, 246, here designated, type-species also by monotypy]
Larval host-plants: unknown.
P. paradistincta ( Alberti, 1954, 246) ( Illiberis .Svenia)), comb. nov.
Distribution: eastern China (Jiangsu, Zhejiang).
Genus HedinaAlberti, 1954, 249 (see Efetov & Tarmann, 2012, 22)
[Type-species: Northia tenuisButler, 1877a, 394, by original designation]
ThyrinaPoujade, 1886b, 143
[Type-species: Thyrina elegans Poujade, 1886b, 143, by monotypy]
Larval host-plants: Vitaceae.
H. nigra ( Leech, 1889a, 595) ( Procris) ( Figure 35)
Distribution: China, Japan ( Horie, 2012, 23).
H. psychina ( Oberthür, 1880, 28) ( Procris)
sinensis partim ( Kirby, 1892, 88) ( Illiberis) (nec Walker, 1854, 280)
ussuriensis ( Alberti, 1951, 137) ( Illiberis)
Distribution: Russia (Far East (Pimorye Territory)), northeastern China (Shanxi, Hebei, Beijing) ( Xue & Han, 2003, 263), Korea ( Kim, et al, 2004, 246), Japan
H. consimilis( Leech, 1898, 334) ( Illiberis)
hyalina partim ( Jordan, 1907a, 15) ( Illiberis) (nec Staudinger, 1887b, 169)
distinctus ( Kardakoff, 1928, 415) ( Illiberis)
Distribution: Russia (Far East (Khabarovsk Territory, Pimorye Territory)), northeastern China, Korea ( Kim, Sohn & Cho, 2004, 247), Japan.
H.hyalina( Staudinger, 1887b, 169) ( Northia (Ino))
transvena ( Jordan, 1907a, 16) ( Illiberis)
coreana ( Matsumura, 1927, 77) ( Illiberis)
Distribution: Russia (Far East (Khabarovsk Territory, Pimorye Territory)), China, Korea.
H. sinyaeviMollet, 2016b, 28
Distribution: China (Shaanxi).
H. tenuis ( Butler, 1877a, 394) ( Northia) ( Figure 8)
khasiana ( Moore, 1879b, 12) ( Northia)
Distribution: Russia (Far East (Khabarovsk Territory, Pimorye Territory)), northeastern India, China, Korea, Japan.
H. elegans ( Poujade, 1886b, 143) ( Thyrina)
Distribution: China (Sichuan, Guizhou, Hunan, Zhejiang). Literature data on the presence of H. elegans in the Russian Far East (e.g. Xue & Han, 2003, 262) are based on a misidentification of Iliiberis pruni Dyar, 1905a, by Jordan (in Seitz) ( 1907a, 7) (see Alberti, 1954, 232, 252).
H. serrata ( Alberti, 1954, 254) ( Illiberis (Hedina))
Distribution: southern China (Yunnan).
H. albiventris( Alberti, 1954, 254) ( Illiberis .Hedina))
Distribution: central China (Shaanxi).
H. louisi( Efetov, 2010, 235) ( Illiberis (Hedina))
Distribution: China (Shaanxi).
H. translucida( Poujade, 1884, 136) ( Procris) ( Figure 36)
Distribution: China (Sichuan, Hunan, Hubei, Zhejiang).
H. taiwana( Efetov, 1997b, 236) ( Illiberis (Hedina))
Distribution: China (Taiwan).
H. annamitaMollet, 2017b, 135
Distribution: Vietnam.
H. vietnama( Efetov, 1997b, 240) ( Illiberis (Hedina))
Distribution: southern China (Yunnan), northern Vietnam ( Efetov, 2003, 27).
Genus MaculaiaMollet, 2019b, 141
[Type-species: Maculaia danhi Mollet, 2019b, 142, by original designation and monotypy]
Larval host-plants: unknown.
M. danhi Mollet, 2019b, 142
Distribution: southern Vietnam.
Genus DubernardiaAlberti, 1954, 257
[Type-species: Phacusa djreuma Oberthür, 1893, 21, by original designation and monotypy]
Larval host-plants: unknown.
D. djreuma ( Oberthür, 1893, 21) ( Phacusa) ( Figure 37)
Distribution: southern China.
Genus GoazreaMollet, 2016c, 33
[Type-species: Goazrea lao Mollet, 2016c, 35, by original designation and monotypy]
Larval host-plants: unknown.
G. lao Mollet, 2016c, 34 ( Figure 38)
Distribution: northern Thailand, Laos.
Genus GerryaMollet, 2017, 63
[Type-species: Gerrya radiatus Mollet, 2017a, 64, by original designation and monotypy]
Larval host-plants: unknown.
G. radiatus Mollet, 2017, 64
Distribution: China (Hainan).
Genus GoeHampson, 1893 (“1892”), 242 (as Goë) (see Efetov, 1998a, 50)
[Type-species: Goe diaphanaHampson, 1893 (“1892”), 242 , by original designation and monotypy]
KublaiaAlberti, 1954, 255 ( Illiberis subg.) (see Efetov, 1998a, 60)
[Type-species: Illiberis heringi Draeseke, 1926, 45, by original designation and monotypy]
Larval host-plants: unknown.
G. tarmanni Efetov, 1998a, 52
Distribution: China (Sichuan).
G. heringi ( Draeseke, 1926, 45) ( Illiberis)
Distribution: China (Sichuan).
G. diaphana Hampson, 1893, 242 ( Goë)
Distribution: Myanmar (Burma).
G. dentata Efetov, 1998a, 57
heringi partim (sensu Alberti, 1954, 256 ( Illiberis .Kublaia))) (nec Draeseke, 1926, 45)
Distribution: China (Shaanxi, Hunan).
Genus Zama Herrich-Schäffer, 1855, 87 (see Efetov & Tarmann, 2012, 23)
[Type-species: Zama cyaneculaHerrich-Schäffer, 1855, 87, by monotypy]
NorthiaWalker, 1854, 141 (a junior homonym of Northia Gray, 1847, 140 - Mollusca).
[Type-species: Glaucopis nigrigemmaWalker, 1854, 141, by monotypy]
Larval host-plants: Vitaceae.
Z. shensiensis ( Alberti, 1954, 242) ( Illiberis (Zama))
Distribution: central China (Shaanxi).
Z. nigrigemma ( Walker, 1854, 141) ( Glaucopis)
cyaneculaHerrich-Schäffer, 1855, 87
Distribution: Myanmar (Burma), Laos, northern Vietnam, China.
Z. horni ( Strand, 1915, 121) ( Phacusa)
Distribution: China (Taiwan).
Z. arisana ( Matsumura, 1927, 79) ( Phacusa)
Distribution: China (Taiwan).
Z. endocyanea ( Hampson, 1920a, 273) ( Illiberis)
Distribution: India (Assam), Bhutan.
Z. cyanocera ( Hampson, 1893, 241) ( Phacusa)
ignea ( Oberthür, 1894, 29) ( Northia)
Distribution: Myanmar (Burma).
Genus PhacusaWalker, 1854, 150
[Type-species: Glaucopis tenebrosa Walker, 1854, 150, by monotypy]
Notioptera Butler, 1876, 355
[Type-species: Syntomis dolosaWalker, 1856, 1594]
Larval host-plants: Vitacae, Dilleniaceae ( Dillenia) ( Küppers & Buchsbaum, 2015).
Ph. tenebrosa ( Walker, 1854, 150) ( Glaucopis)
siamensisOberthür, 1894, 31 ( Phacusa sp.)
Distribution: northeastern India, Thailand.
Ph. nicobarica Hampson, 1920a, 272, stat. rev. (C33)
Distribution: Nicobare Islands.
Ph. crawfurdi ( Moore, 1859, 327) ( Syntomis sp.) (see Holloway, 2011, 16)
subtilis Hering, 1925a, 176
Distribution: Indonesia (Java, Borneo).
Ph. birmana ( Oberthür, 1894, 30) ( Northia)
Distribution: Myanmar (Burma), Thailand, Laos.
Ph. discoidalis ( Swinhoe, 1903, 500) ( Illiberis)
tonkinensisAlberti, 1954, 259
Distribution: Vietnam.
Ph. chalcobasis Hampson, 1920a, 272
Distribution: Indonesia (Sumatra).
Ph. dolosa ( Walker, 1856, 1594) ( Syntomis)
Distribution: northeastern India.
Ph. properta ( Swinhoe, 1890, 400) ( Notioptera)
dohertyi ( Oberthür, 1894, 36) ( Northia sp.)
Distribution: Myanmar (Burma).
Ph. janicornaeKüppers & Buchsbaum, 2016, 148
Distribution: Thailand.
Ph. manilensis Hampson, 1920a, 272
Distribution: Philippines (Luzon).
Ph. strigosa ( Walker, 1865, 69) ( Syntomis)
Distribution: Indonesia (Sulawesi).
Genus PseudophacusaEfetov & Tarmann, 2016, 82
[Type-species: Pseudophacusa multidentata Efetov & Tarmann, 2016, 82, by original designation and monotypy]
Larval host-plants: unknown.
P. multidentata Efetov & Tarmann, 2016, 82
Distribution: northern Myanmar (Burma).
Genus FuneraliaAlberti, 1954, 264
[Type-species: Funeralia transiens Alberti, 1954, 264, by original designation and monotypy]
Larval host-plants: unknown.
F. transiens Alberti, 1954, 264
Distribution: eastern China (Zhejiang).
Genus ErythrocleleaEfetov & Tarmann, 1995b, 70
[Type-species: Laurion syfanicum Oberthür, 1894, 25, by original designation and monotypy]
Larval host-plants: unknown.
E. syfanicum ( Oberthür, 1894, 25) ( Laurion)
Distribution: China (Sichuan).
Genus PraeprocrisAlberti, 1954, 315 ( Rhagades subg.) (see Efetov & Tarmann, 1999a, 17)
[Type-species: Rhagades (Praeprocris) pseudomaerens Alberti, 1954, 315, by original designation andmonotypy]
Larval host-plants: unknown.
P. pseudomaerens ( Alberti, 1954, 315) ( Rhagades)
Distribution: China (Beijing).
Genus TheresimimaStrand, 1917, 137
[Type-species: Zygaena ampellophaga Bayle-Barelle, 1809, 2, by monotypy (of Theresia Spuler, 1906)]
Theresia Spuler, 1906, 165 (a junior homonym of Theresia Robineau-Desvoidy, 1830, 325 - Insecta, Diptera. The objective replacement name is TheresimimaStrand, 1917)
[Type-species: Zygaena ampellophaga Bayle-Barelle, 1809, 2, by monotypy]
Larval host-plants: Vitaceae: Vitis vinifera L., Parthenocissus quinquefolia (L.) Planch. (Embacher & Tarmann, 2002), hybrid P. inserta (A. Kerner) Fritsch x P. quinquefolia (L.) Planch. (Crimea: Efetov, 1990), P.tricuspidata (Siebold & Zucc.) Planch. (Crimea: Efetov, 1998a).
Th. ampellophaga ( Bayle-Barelle, 1809, 2) ( Zygaena)
ampelophaga [sic] (Hübner, 1822, figs 153, 154) ( Sphinx), misspelling (see Efetov et al. 2023a)
vitis ( Freyer, 1834, 48) ( Sphinx)
astrapta ( Dannehl, 1933, 147) ( Ino ampelophaga [sic] “ab (?) rasse (?)”)
Distribution: southern France, Italy (including Sicily), southeastern Austria, Slovenia, Croatia, Bosnia and Herzegovina (?), Serbia, Montenegro, Albania, Macedonia Greece (mainland and islands including Rhodes and Crete), Slovakia, Hungary, Romania (Dobrogea), Bulgaria, southern Ukraine, Moldova, Russia (southern European part including Crimea, Northern Caucasus), Georgia, Turkey, Cyprus, Syria, Lebanon, Israel. The historical occurrence in Algeria may have its origin in introduced specimens as they all come from Alger and surrounding places (no newer records known). No other records known from Africa. The occurrence in Spain is also doubtful and needs confirmation.
Genus Rhagades Wallengren, 1863, 110
[Type-species: Sphinx pruni [ Denis & Schiffermüller], 1775, 308, by monotypy]
Larval host-plants: Rosaceae. In one species, Rh. (Rh.) pruni, larval host-plants can also belong to other families: Fagaceae (e.g. subsp. chinensis), Ericaceae, Cistaceae, Rhamnaceae, Salicaciae.
Subgenus NaufockiaAlberti, 1954, 317
[Type-species: Procris brandti Alberti, 1938b, 398, by original designation and monotypy]
Larval host-plants: Rosaceae ( Prunus, Amygdalus, Cotoneaster).
Rh. ( N.) brandti( Alberti, 1938b, 398) ( Procris)
Distribution: Iran (Zagros, southern Kuh-Rud).
Subgenus WiegeliaEfetov & Tarmann, 1995b, 66
[Type-species: Procris amasina Herrich-Schäffer, 1851, 42, by original designation]
Larval host-plants: Rosaceae ( Pyrus (e.g. Pyrus bourgaeana Decne.), Prunus (e.g. Prunus ramburii Boiss., Prunus spinosa L., Prunus cerasifera Ehrh.) ( Vives Moreno & Huertas-Dionisio, 1985; Murria-Beltrán, 2007; Efetov & Tarmann, 2020b), Amygdalus, Cotoneaster, Crataegus, Malus).
Rh. (W.) amasina( Herrich-Schaffer, 1851, 42) ( Procris) ( Figure 17)
Distribution: Greece (Islands of the Dodekanes and Rhodos), Bulgaria, Turkey, Syria, Lebanon.
Rh. (W.) predotae( Naufock, 1930, 107) ( Procris) ( Figure 9)
Distribution: Spain.
Rh. (W.) tarmanniKeil, 1999, 73
Distribution: Iran (Zagros).
Subgenus Rhagades Wallengren, 1863, 110
[Type-species: Sphinx pruni [ Denis & Schiffermüller], 1775, 308, by monotypy]
Larval host-plants: Rosaceae ( Crataegus, Fragaria, Malus, Padellus, Potentilla, Poterium, Prunus, Pyrus, Rosa, Rubus), Ericaceae ( Calluna, Erica), Cistaceae ( Helianthemum), Fagaceae ( Fagus, Quercus), Rhamnaceae ( Rhamnus), Salicaceae ( Populus, Salix).
Rh. ( Rh.) pruni([ Denis & Schiffermüller], 1775, 308) ( Sphinx) (C34) ( Figure 10)
Distribution: northeastern Spain, France, Belgium, Luxembourg, Netherlands, Denmark, Germany, Switzerland, Austria, northern Italy, Slovenia, Croatia, Bosnia and Herzegovina, Serbia, Montenegro, Albania, Macedonia, Greece, Poland, Czech Republic, Slovakia, Hungary, Romania, Bulgaria, Finland, Estonia, Latvia, Lithuania, Belarus, Ukraine, Moldova, Russia (northern, central and southern European part including Crimea, Northern Caucasus, Siberia), European Turkey, Georgia, Azerbaijan, Kyrgyzstan, Kazakhstan (subsp. pruni); Russia (Far East), China, North Korea (subsp. chinensis); Russia (Kuril Islands), Japan (subsp. esmeralda).
Rh. ( Rh.) pruni pruni ([ Denis & Schiffermüller], 1775, 308) ( Sphinx)
callunae Spuler, 1906, 166 ( Rhagades pruni “v. ? ”)
Rh. ( Rh.) pruni chinensis ( Felder & Felder, 1862, 31) ( Ino sp.)
tristis ( Bremer, 1865, 97) ( Procris sp.)
Rh. ( Rh.) pruni esmeralda ( Butler, 1877a, 394) ( Procris sp.)
Genus ZygaenoprocrisHampson, 1900, 225 (see Efetov, 2001b, 41) (C35) (Figure 1)
[Type-species: Zygaenoprocris chalcochlora Hampson, 1900, 225, by original designation and monotypy]
Larval host-plants: Polygonaceae ( Efetov, 1991b, 1994c; Keil, 2014, 2016b, 2020), Plumbaginaceae ( Mollet & Tarmann, 2007; Keil, 2014, 2016b, 2020), Asteraceae ( Mollet & Tarmann, 2007; Keil, 2014, 2016b, 2020).
Subgenus ZygaenoprocrisHampson, 1900, 225(C35)
[Type-species: Zygaenoprocris chalcochlora Hampson, 1900, 225, by original designation and monotypy]
Larval host-plants: Plumbaginaceae ( Acantholimon) ( Mollet & Tarmann, 2007; Keil, 2014, 2016b, 2020), Asteraceae ( Cousinia) ( Mollet & Tarmann, 2007; Keil, 2014, 2016b, 2020).
Z. ( Z.) chalcochloraHampson, 1900, 225
Distribution: Afghanistan, Pakistan.
Z. ( Z.) mystrocera( Püngeler, 1914, 52) ( Ino) (comb.: Efetov, 2001b) ( C35)
Distribution: northern central, northern and northeastern Iran (ssp. mystrocera), central Iran (Yazd) (ssp. molleti), and southern Iran (Kerman) (ssp. valii).
Z. ( Z.) mystrocera mystrocera ( Püngeler, 1914), stat. rev.
Z. ( Z.) mystrocera molleti Keil, 2020, 89, stat. nov.
Z. ( Z.) mystrocera valii Keil, 2020, 90, stat. nov.
Z. ( Z.) khorassana( Alberti, 1939a, 3) ( Procris) (see Efetov & Tarmann, 2012, 26, 46)
Distribution: northeastern Iran, Turkmenistan (Kopetdag).
Z. ( Z.) hofmanniMollet & Tarmann, 2007, 71
Distribution: northeastern Iran (Kuh-e Shavar).
Z. ( Z.) efetoviMollet & Tarmann, 2007, 69
Distribution: eastern Iran (Kuh-e Mirza Arab).
Subgenus EfetoviaMollet, 2001, 51
[Type-species: Procris fredi Alberti, 1939a, 4, by original designation and monotypy]
Larval host-plants: unknown.
Z. ( E.) fredi( Alberti, 1939a, 4) ( Procris) (comb.: Efetov, 2001b)
Distribution: northeastern Iran (Kuh-e Binaloud).
Subgenus Longiterna Efetov & Tarmann, subgen. nov. (C36)
[Type-species: Procris rjabovi Alberti, 1938c, 94, here designated]
Larval host-plants: Polygonaceae ( Polygonum) ( Keil, 2014).
Z. ( L.) rjabovi( Alberti, 1938c, 94) ( Procris) (comb., Efetov, 2001b)
Distribution: northeastern Iran (Shah-Kuh).
Z. ( L.) eberti( Alberti, 1968, 249) ( Procris) (comb.: Efetov, 2001b)
Distribution: Afghanistan (Koh-i-Baba).
Subgenus Keilia Efetov, 2001b, 47
[Type-species: Adscita minna Efetov, 1991b, 155, by original designation]
Larval host-plant: Polygonaceae ( Atraphaxis ) ( Efetov, 1991b, 1994c; Efetov & Daricheva, 1992; Keil, 2014, 2016a).
Z. ( K.) minna( Efetov, 1991b, 155) ( Adscita) (comb.: Efetov, 2001b)
Distribution: Turkmenistan (Kopetdag), Iran (northern Khora¯sa¯ n).
Z. ( K.) albertii( Efetov, 1991b, 57) ( Adscita) (comb.: Efetov, 2001b)
Distribution: Turkmenistan, Iran (northern Khora¯sa¯ n-e Razaw¯ı).
Z. ( K.) naumanni( Efetov, 1994a, 53) ( Adscita (Zygaenoprocris)) (comb.: Efetov, 2001b)
Distribution: northern Afghanistan.
Subgenus MolletiaEfetov, 2001b, 45(C35)
[Type-species: Procris taftana Alberti, 1939a, 4, by original designation]
Larval host-plant: Polygonaceae ( Atraphaxis, Polygonum) ( Efetov, 1997d; Mollet, 2000; Keil, 2020).
Z. (M.) taftana( Alberti, 1939a, 4) ( Procris) (comb.: Efetov, 2001b) ( Figures 18, 39)

Distribution: Armenia, Iran.
Z. (M.) persepolis( Alberti, 1938b, 399) ( Procris) (comb.: Efetov, 2001b)
Distribution: Iran: southern Zagros Mountains (subsp. persepolis); northern and central Zagros Mountains (subsp. puschmanni); central Quohrud Mountains (subsp. schahdadiani); southern Iran (Kerman) (subsp. hasarani).
Z. ( M.) persepolis persepolis ( Alberti, 1938b, 399) ( Procris)
Z. ( M.) persepolis puschmanni Keil, 2020, 94, stat. nov.
Z. ( M.) persepolis schahdadiani Keil, 2020, 94, stat. nov.
Z. ( M.) persepolis hasarani Keil, 2020, 96, stat. nov.
Z. ( M.) duskei( Grum-Grshimailo, 1902, 197) ( Ino) (comb.: Efetov, 2001b)
Distribution: Iran: central parts between Teheran and Esfahan (subsp. kliri); central (south of 33oN) and southern parts (subsp. kermana); Kuh-e Taftan (subsp. duskei); Mekran (subsp. aerea).
Z. (M.) duskei kliri Keil, 2002, 55 (Zygaenoprocris sp.) (see Efetov, 2004a, 113)
Z. ( M.) duskei kermana ( Alberti, 1967, 99) ( Procris sp.) (see Efetov, 2001d, 154)
Z. ( M.) duskei duskei ( Grum-Grshimailo, 1902, 197) ( Ino)
sengana ( Alberti, 1939a, 28) ( Procris sp.) (see Efetov, 1992b, 147)
Z. ( M.) duskei aerea ( Grum-Grshimailo, 1902, 198) ( Ino duskei var.) (see Efetov & Tarmann, 1999a, 32, 71)
mekrana ( Alberti, 1939a, 29) ( Procris “sp. ?”) (see Efetov & Tarmann, 1999a, 32)
Genus AdscitaRetzius, 1783, 35 (Figure 2)
[Type-species: Adscita turcosa Retzius, 1783, 35, by subsequent designation by Kirby, 1892, 84]
ChrysaorHübner, 1806, [1] (included in a work rejected for nomenclatural purposes by theInternational Commission on Zoological Nomenclature, 1926, Opinion 97, 19)
[Type-species: Sphinx statices Linnaeus, 1758, 495, by monotypy]
Procris [Fabricius in Illiger], 1807, 289 (see Taeger & Gaedike, 2001, 87)
[Type-species: Sphinx statices Linnaeus, 1758: 495, by subsequent designation by Latreille, 1810, 441]
AtychiaOchsenheimer, 1808, [9], [10], 11
[Type-species: Sphinx statices Linnaeus, 1758, 495, by subsequent designation by Tremewan, 1973, 119]
Ino Leach, 1815, 131
[Type-species: Sphinx statices Linnaeus, 1758, 495, by monotypy]
BradyptesisSodoffsky, 1837, 83 (unnecessary objective replacement name for AtychiaOchsenheimer, 1808)
Larval host-plants: Polygonaceae, Cistaceae, Geraniaceae. In some Cistaceae-feeding species host-plants can also be Rosaceae and Fabaceae. Some larvae can be leaf-mining but only in early instars.
Subgenus ProcriternaEfetov & Tarmann, 2004a, 184
[Type-species: Ino subtristis Staudinger, 1887a, 68, by original designation]
ProcritaEfetov & Tarmann, 1999a, 31, 63 (a junior homonym of ProcritaHendel, 1908, 59 - Insecta, Diptera. The objective replacement name is ProcriternaEfetov & Tarmann, 2004a, 184)
[Type-species: Ino subtristis Staudinger, 1887a, 68, by original designation]
Larval host-plants: Polygonaceae ( Atraphaxis spp.). Larva not leaf-mining (biology only known for A. subtristis).
A. ( P.) subtristis( Staudinger, 1887a, 68) ( Ino) ( Figure 40)
dolosa ( Staudinger, 1887a, 69) ( Ino) (see Efetov & Tarmann, 1999a, 31)
Distribution: Uzbekistan, Tajikistan, Kyrgyzstan, southern Kazakhstan.
A. (P.)amaura( Staudinger, 1887a, 70) ( Ino)
banghaasi ( Alberti, 1938a, 119) ( Procris amaura subsp.) (see Efetov & Tarmann, 1999a, 31)
Distribution: Uzbekistan, Tajikistan.
A. (P.)subdolosa( Staudinger, 1887a, 70) ( Ino dolosa var.)
pamirensis ( Hampson, 1920b, 433) ( Procris)
Distribution: Uzbekistan, Tajikistan, Kyrgyzstan, southern Kazakhstan, Afghanistan.
A. ( P.) pligoriEfetov, 2012, 99
Distribution: Afghanistan.
Subgenus Afroterna Efetov & Tarmann, subgen. nov. (C37)
[Type-species: Procris mauretanica Naufock, 1932, 77, here designated]
Larval host-plants: Polygonaceae, Cistaceae. Larva leaf-mining only in early instars.
A. ( Afr.) mauretanica( Naufock, 1932, 77) ( Procris), comb. nov.
Distribution: Morocco (Middle Atlas and Rif), northwestern Algeria (subsp. mauretanica); Morocco (High Atlas) (subsp. wiegeli).
A. ( Afr.) mauretanica mauretanica ( Naufock, 1932, 77) ( Procris)
bohigasi ( Agenjo, 1940, 105) ( Procris mauretanica var.)
meson Dujardin, 1973, 160
A. ( Afr.) mauretanica wiegeli ( Alberti, 1973a, 12) ( Procris)
atlasicaDujardin, 1973, 159
Larval host-plants: Polygonaceae ( A. mauretanica wiegeli), Cistaceae ( A. mauretanica mauretanica).
Subgenus AdscitaRetzius, 1783, 35
[Type-species: Adscita turcosa Retzius, 1783, 35, by subsequent designation by Kirby, 1892, 84]
ChrysaorHübner, 1806, [1] (see above)
Procris [ Fabricius in Illiger], 1807, 289 (see above)
AtychiaOchsenheimer, 1808, [9], [10], 11 (see above)
Ino Leach, 1815, 131 (see above)
BradyptesisSodoffsky, 1837, 83 (see above)
Larval host-plants: Polygonaceae ( Rumex spp.). Larva leaf-mining only in early instars.
jordani-group (see Efetov & Tarmann, 2012, 29)
Larval host-plants: Polygonaceae ( Rumex spp.)
A. (A.) jordani( Naufock, 1921, 63) ( Procris)
Distribution: Portugal, Spain.
statices-group (see Efetov & Tarmann, 2012, 29)
Larval host-plants: Polygonaceae ( Rumex spp.).
A. (A). krymensisEfetov, 1994b, 267
Distribution: Crimea.
A. (A). schmidti( Naufock, 1933b, 61) ( Procris)
ariasae ( Agenjo, 1975, 9) ( Procris schmidti subsp.)
Distribution: Portugal, Spain.
A. ( A.) alpina( Alberti, 1937a, 435) ( Procris)
oblita ( Rocci, 1937, 146) ( Procris sp.)
viridisVerity, 1946, 148 ( Adscita alpina “forma”)
caerulea Verity, 1946, 148 ( Adscita alpina “forma”)
minusculaVerity, 1946, 151 ( Adscita alpina alpina “sottorazza”) (see Efetov, 2001c, 128)
bellissimaVerity, 1946, 151 ( Adscita alpina “razza”)
Distribution: Alps (southeastern France, southern Switzerland, western Austria, northern Italy).
A. (A.) italica( Alberti, 1937a, 438) ( Procris)
Distribution: central and southern Italy (including Sicily) (subsp. italica); eastern Turkey (subsp. storaiae).
A. (A). italica italica ( Alberti, 1937a, 438) ( Procris) ( Figures 11, 19)
A. (A). italica storaiae ( Tarmann, 1977a, 97) ( Procris)
A. (A). statices ( Linnaeus, 1758, 495) ( Sphinx) ( Figure 41)
Distribution: northeastern Spain, Andorra, Ireland, Scotland, England, Wales, France, Belgium, Luxembourg, Netherlands, Germany, Switzerland, Austria, northern Italy, Slovenia, Croatia, Bosnia and Herzegovina, Serbia, Montenegro, Albania, Macedonia, Greece, Denmark, Norway, Sweden, Finland, Poland, Czech Republic, Slovakia, Hungary, Romania, Bulgaria, Estonia, Latvia, Lithuania, Belarus, Ukraine (without Crimea), Moldova, Russia (European part, Northern Caucasus, Siberia), Georgia, Kyrgyzstan, Kazakhstan, Turkey, northwestern China.
Subsp. statices. Absent from most parts of the Iberian Peninsula where it is replaced by A. (A). schmidti; absent from most parts of northern Italy where it is replaced by A. (A). alpina; absent from central and southern Italy where it is replaced by A. (A). italica italica; absent from the Crimea where it is possibly replaced by A. (A). krymensis; absent from southern Balkans and western, central and southern Turkey where it is replaced by A. (A). statices drenowskii (see below); absent from eastern Turkey where it is replaced by A. (A). italica storaiae.
Subsp. drenowskii. Southern Balkans to central and southern Turkey.
A. (A.) statices statices ( Linnaeus, 1758, 495) ( Sphinx)
turcosa Retzius, 1783, 8 ( Adscita sp.) (unnecessary objective replacement name for Sphinx statices Linnaeus, 1758)
micans ( Freyer, 1833, 27) ( Sphinx sp.)
uralensis ( Grum-Grshimailo, 1893, 385) ( Ino statices var.)
viridisTutt, 1899, 390 ( Adscita statices “ab.”)
griseonigra ( Hoffmann & Klos, 1923, 44) (Ino statices f.)
grisea ( Niepelt, 1924, 50) ( Procris statices f.)
extensa ( Alberti, 1937b, 100) ( Procris)
anomalaVerity, 1946, 152 ( Adscita statices “razza”)
lutrinensis ( Heuser, 1960, 28) ( Procris sp.)
heuseri ( Reichl, 1964, 100) ( Procris sp.)
albis ( Heuser, 1964, 68) ( Procris sp.)
palatis ( Heuser, 1964, 68) ( Procris sp.)
talis ( Heuser, 1964, 68) ( Procris sp.)
A. (A). statices drenowskii ( Alberti, 1939b, 43) ( Procris sp.)
Subgenus Obscuriterna Efetov & Tarmann, subgen. nov. (C38)
[Type-species: Procris obscura Zeller, 1847a, 15, here designated]
Larval host-plants: Cistaceae, Rosaceae and Fabaceae. Larva leaf-mining only in early instars.
A. (A.) obscura ( Zeller, 1847a, 15) ( Procris)
Distribution: Albania, Macedonia, Greece, Romania, Bulgaria, most parts of Turkey, Iraq, Iran (Elburs, Zagros) (subsp. obscura); southern Turkey (Mersin, Adana, Hatay), Syria, Lebanon, Israel, Jordan, northeastern Egypt (subsp. pallida); Russia (Northern Caucasus (Dagestan)), Georgia, Armenia, Azerbaijan, northeastern Turkey, northwestern Iran (subsp. maxima).
A. ( A.) obscura obscura ( Zeller, 1847a, 15) ( Procris)
anceps ( Staudinger, 1862, 355) ( Ino sp.)
balcanica ( Staudinger, 1862, 356) ( Ino obscura “Localform”)
pallida ( Alberti, 1938a, 122) ( Procris)
A. ( A.) obscura maxima ( Alberti, 1938a, 122) ( Procris)
Subgenus Eurasiterna Efetov & Tarmann, subgen. nov. (C39)
[Type-species: Adscita dujardini Efetov & Tarmann, 2014b, 182, here designated]
Larval host-plants: Cistaceae, Geraniaceae. Larva leaf-mining only in early instars.
geryon-group (see Efetov & Tarmann, 2012, 30)
Larval host-plants: Cistaceae ( A. geryon sometimes also on Geraniaceae).
A. (Eur.) capitalis ( Staudinger, 1879, 317) ( Ino)
Distribution: Macedonia, Greece, Turkey.
A. ( Eur). geryon(Hübner, 1813, pl. 28, figs 130, 131) ( Sphinx)
Distribution: Spain, Andorra, England, Wales, France, Belgium, Germany, Switzerland, Austria, Italy (including Sicily), Greece (except north-west), Poland, Czech Republic, Slovakia, Hungary, Romania, Bulgaria, southern Ukraine, Moldova, Russia (southern European part, southern Crimea), northwestern Turkey (subsp. geryon); northeastern Italy, Slovenia, Croatia, Bosnia and Herzegovina, Serbia, Montenegro, Albania, Macedonia, northwestern Greece (subsp. orientalis); high mountains of eastern Switzerland, northern Italy, and central Austria (subsp. chrysocephala); central Italy (subsp. acutafibra).
A. ( Eur). geryon geryon (Hübner, 1813, pl. 28, figs 130, 131) ( Sphinx)
minor (sensu Jordan, 1907a, 9 ( Procris)) (nec Eversmann, 1844, 91)
caeruleaTutt, 1899, 401 ( Adscita geryon “ab.”)
viridisTutt, 1899, 401 ( Adscita geryon “ab.”)
virescens ( Agenjo, 1937, 311) ( Procris geryon “forma”)
aerisVerity, 1946, 154 ( Adscita geryon “razza”)
A. ( Eur). geryon chrysocephala ( Nickerl, 1845, 93) ( Atychia sp.)
A. ( Eur). geryon acutafibra Verity, 1946, 149 ( Adscita alpina “forma”) (see Efetov, 2001c, 128)
A. ( Eur). geryon orientalis ( Alberti, 1938d, 54) ( Procris)
hyalicolorVerity, 1946, 150 ( Adscita alpina “razza”) (see Efetov, 2001c, 128)
albanica-group (see Efetov & Tarmann, 2012, 31)
Larval host-plants: Geraniaceae ( Efetov, 1992a, 1999b).
A. (Eur.) dujardini Efetov & Tarmann, 2014b, 182
Distribution: southeastern France, Switzerland, Italy, Slovenia.
A. (Eur.) albanica ( Naufock, 1926, (126)) ( Procris)
jegorowi ( Alberti, 1971, 76) ( Procris) [nomen nudum]
accolaZolotuhin & Nedoshivina, 2020, 144 ( Adscita albanica ssp.) (see Efetov & Tarmann, 2022)
Distribution: Serbia, Albania, Macedonia, Greece, Bulgaria, Ukraine, Russia (European part, Crimea, Northern Caucasus).
Subgenus Tarmannita Efetov, 2000f, 169
[Type-species: Ino mannii Lederer, 1853, 103, by original designation]
Larval host-plants: Cistaceae; for A. mannii also Rosaceae and Fabaceae, for A. antoniovivesi also Rosaceae.
A. ( T.) mannii( Lederer, 1853, 103) ( Ino) ( Figure 42)
heydenreichii ( Lederer, 1853, 103) ( Ino sp.)
crassicornis ( Staudinger, 1862, 358) ( Ino heydenreichii “v.”)
prasina ( Rothschild, 1917, 345) ( Procris bellieri subsp.)
superba ( Rocci, 1937, 145) ( Procris micans “f. p.”)
atlantica ( Alberti, 1937b, 98) ( Procris mannii subsp.)
denticulataVerity, 1946, 140 ( Adscita mannii “forma”)
caerulea Verity, 1946, 143 ( Adscita mannii bellieri “forma”)
glaucaVerity, 1946, 144 ( Adscita mannii “razza”)
gracilisVerity, 1946, 145 ( Adscita mannii crassicornis “sottorazza”)
pseudostaticesVerity, 1946, 146 ( Adscita mannii “razza”)
heliocausta Dujardin, 1975, 39
micans (sensu Jordan, 1907a, 9 ( Procris)) (nec Freyer, 1833, 27)
Distribution: northeastern Spain, France, southwestern Germany, southern Switzerland, southern Austria, Italy (including Sicily), Slovenia, Croatia, Bosnia and Herzegovina, Serbia, Montenegro, Albania, Macedonia, Greece, Romania, Bulgaria, northwestern Turkey.
A. (T.) antoniovivesi Efetov & Tarmann, 2024, 10
Distribution: Spain.
A. ( T.) bolivari( Agenjo, 1937, 314) ( Procris)
Distribution: Portugal, Spain.
Genus JordanitaVerity, 1946, 134
[Type-species: Sphinx chloros Hübner, 1813, pl. 28, figs 128, 129; by original designation, name made available by designation of type species]
JordanitaAgenjo, 1940, 46 (without designation of type species; unavailable under Code, Article 13.3)
Larval host-plants: Asteraceae. Larva leaf-mining, even in last instars.
Subgenus Roccia Alberti, 1954, 326
[Type-species: Ino budensis Speyer & Speyer, 1858, 466, by original designation]
Larval host-plants: Asteraceae. Larva leaf-mining, even in last instars.
budensis-group (see Efetov & Tarmann, 2012, 32)
J. ( R.) budensis( Speyer & Speyer, 1858, 466) ( Ino)
Distribution: Spain, France, Austria, Italy, Slovenia, Croatia, Bosnia and Herzegovina, Serbia, Montenegro, Albania, Macedonia, Greece, Slovakia, Hungary, Romania, Bulgaria, Russia (southern European part, southern Crimea, Northern Caucasus,), Georgia, Armenia, Azerbaijan, Turkey (subsp. budensis); Russia (southern Siberia, Far East (Amur Region)), Kazakhstan, Mongolia (subsp. centralasiae).
J. (R.) budensis budensis ( Speyer & Speyer, 1858, 466) ( Ino)
cuprea ( Rambur, 1866, 186) ( Procris)
J. (R.) budensis centralasiae ( Alberti, 1937c, 87) ( Procris)
J. (R.) paupera ( Christoph, 1887, 162) ( Ino) (C40)
hamifera ( Jordan, 1907a, 8) ( Procris)
tamerlana ( Alberti, 1937c, 86) ( Procris hamifera subsp.)
minor ( Alberti, 1937c, 87) ( Procris hamifera “f. (ssp. ?)”) (a junior primary homonym of Atychia statices var. minor Eversmann, 1844, 91)
scintillosaZolotuhin, 2020, 8 (replacement name for Procris hamifera minor Alberti, 1937c) (see Efetov et al. 2022; Efetov & Tarmann, 2022)
smaragdonnaZolotuhin, 2020, 9 (see Efetov et al. 2022; Efetov & Tarmann, 2022)
Distribution: Russia (southern European part, Northern Caucasus), Azerbaijan (including Nakhichevan), Turkmenistan, Uzbekistan, Tajikistan, Kyrgyzstan, Kazakhstan, Turkey, Jordan, northern Iran.
J. (R.) mollis ( Grum-Grshimailo, 1893, 385) ( Ino budensis var.) (C41)
Distribution: eastern Russia (southern Siberia and Far East), China, Korea.
volgensis-group (see Efetov & Tarmann, 2012, 32)
J. ( R.) volgensis( Möschler, 1862, 139) ( Ino) ( Figure 12)
Distribution: Ukraine (eastern part), Russia southern (European part, Crimea ( Efetov & Savchuk, 2009), Northern Caucasus, southern Siberia), Kazakhstan (subsp. volgensis), Georgia, Armenia, Azerbaijan, northwestern Iran (subsp. muelleri), Turkey, Syria (subsp. grandis).
J. ( R.) volgensis volgensis ( Möschler, 1862, 139) ( Ino)
J. ( R.) volgensis muelleri ( Alberti, 1973b, 387) ( Procris)
J. ( R.) volgensis grandis ( Alberti, 1974, 49) ( Procris)
monotona ( Alberti, 1937c, 91) ( Procris volgensis subsp. hector f.)
J. ( R.) suspecta( Staudinger, 1887a, 71) ( Ino cognata “var.?”)
globulariae partim (sensu Jordan, 1907a, 8 ( Procris)) (nec Hübner, 1793, pl. 67)
Distribution: Uzbekistan, Kyrgyzstan.
naufocki-group (see Efetov & Tarmann, 2012, 32)
J. ( R.) tianshanica( Efetov, 1990, 8) ( Adscita)
Distribution: Kyrgyzstan.
J. ( R.) naufocki( Alberti, 1937c, 88) ( Procris)
Distribution: Kyrgyzstan, Kazakhstan, northwestern China.
J. ( R.) almatiensisMollet, 2008, 57
Distribution: southeastern Kazakhstan.
J. ( R.) kurdica( Tarmann, 1987, 1) ( Adscita)
Distribution: southeastern Turkey, western and southern Iran.
hector-group (see Efetov & Tarmann, 2012, 33)
J. ( R.) hector ( Jordan, 1907a, 8) ( Procris)
staudingeri ( Alberti, 1954, 328) ( Procris)
Distribution: southern Turkey (Mersin, Nigde, Gaziantep, Hatay), Syria, Lebanon, Israel.
Subgenus LucasiternaAlberti, 1961, 59
[Type-species: Procris cirtana Lucas, 1849, 374, by original designation]
Lucasia Alberti, 1954, 319 (a junior homonym of Lucasia Robineau-Desvoidy, 1863, 409 - Insecta, Diptera. The objective replacement name is LucasiternaAlberti, 1961, 59)
[Type-species: Procris cirtana Lucas, 1849, 374, by original designation]
Larval host-plants: Asteraceae ( Mollet, 2003).
J. ( L.) cirtana( Lucas, 1849, 374) ( Procris)
orana ( Austaut, 1880, 284) ( Ino)
orana ( Bethune-Baker, 1888, 117) ( Ino) (a junior primary homonym of Ino orana Austaut, 1880) bakeri ( Kirby, 1892, 82) ( Adscita) (objective replacement name for Ino orana Bethune-Baker, 1888)
Distribution: Northern Algeria, Tunisia.
Subgenus TremewaniaEfetov & Tarmann, 1999a, 42
[Type-species: Atychia notata Zeller, 1847b, 294, by original designation]
Larval host-plants: Asteraceae. Larva leaf-mining, even in last instars.
J. ( T.) notata( Zeller, 1847b, 294) ( Atychia)
soror ( Rambur, 1866, 187) ( Procris)
chlorotica ( Agenjo, 1937, 291) ( Procris globulariae var.)
cyanotica ( Agenjo, 1937, 291) (Procris globulariae var.)
superior ( Rocci, 1937, 130) (Rhagades notata “f. p.”)
globulariae partim (sensu Jordan, 1907a, 8 ( Procris)) (nec Hübner, 1793, pl. 67)
globulariae (sensu Agenjo, 1937, 291 ( Procris)) (nec Hübner, 1793, pl. 67)
globulariae (sensu Verity, 1946, 130 ( Procris)) (nec Hübner, 1793, pl. 67)
Distribution: Portugal, Spain, France, Germany, Switzerland, Austria, Italy (including Sicily), Slovenia, Croatia, Bosnia and Herzegovina, Serbia, Montenegro, Albania, Macedonia, Greece (including Crete), Czech Republic, Slovakia, Hungary, Romania, Bulgaria, Ukraine, Russia (soouthern European part, Crimea, Northern Caucasus), Georgia, Armenia, Azerbaijan, Turkey, Iran.
J. ( T.) splendens ( Staudinger, 1887a, 68) ( Ino) ( Figure 43)
incerta ( Staudinger, 1887a, 72) ( Ino)
heringi ( Alberti, 1937c, 78) ( Procris splendens subsp.)
globulariae suspecta (sensu Jordan, 1907a, 8 ( Procris)) (nec Staudinger, 1887a, 71)
acroptilon ( Stshetkin & Stshetkin, 1993, 139) ( Procris) [nomen nudum: unavailable under Code, Article 13.1]
Distribution: Turkmenistan, Uzbekistan, Tajikistan, Kyrgyzstan, Kazakhstan.
J. ( T.) ambigua( Staudinger, 1887a, 71) ( Ino)
Distribution: Turkmenistan, southern Uzbekistan, western Tajikistan (subsp. ambigua); eastern Uzbekistan, Kyrgyzstan (subsp. asiatica); Iran (subsp. schakuhensis); Afghanistan, southeastern Tadjikistan (Pamirs) (subsp. omotoi).
J. (T.) ambigua ambigua ( Staudinger, 1887a, 71) ( Ino)
J. (T.) ambigua asiatica ( Staudinger, 1887a, 73) ( Ino budensis var.)
J. (T.) ambigua schakuhensis ( Alberti, 1954, 328) ( Procris)
J. (T.) ambigua omotoi ( Alberti, 1965, 1) ( Procris sp.)
Subgenus Gregorita Povolný & Smelhaus, 1951, 159
[Type-species: Procris hispanica Alberti, 1937b, 87, by original designation]
Larval host-plants: Asteraceae. Larva leaf-mining, even in last instars.
hispanica-group (see Efetov & Tarmann, 2012, 34)
J. ( G.) hispanica( Alberti, 1937b, 87) ( Procris)
danieli ( Alberti, 1937b, 89) ( Procris hispanica subsp.)
soror (sensu Agenjo, 1937, 295 ( Procris)) (nec Rambur, 1866, 187)
soror (sensu Povolný & Smelhaus, 1951, 187 ( Procris)) (nec Rambur, 1866, 187)
Distribution: Portugal, Spain, southern France.
algirica-group (see Efetov & Tarmann, 2012, 34)
J. ( G.) algirica( Rothschild, 1917, 345) ( Procris orana subsp.)
reisseri ( Naufock, 1932, 75) ( Procris)
azrouica ( Barragué, 1986, 324) ( Adscita algirica subsp.)
taon ( Barragué, 1986, 324) ( Adscita algirica subsp.)
stena ( Barragué, 1986, 325) ( Adscita algirica subsp.)
intermedia ( Barragué, 1986, 325) ( Adscita algirica subsp.)
Distribution: Morocco, northern Algeria, Tunisia, Sicily (?).
J. (G.) minutissima ( Oberthür, 1916, 240) ( Procris tenuicornis “morphe”)
Distribution: northern Algeria.
J. (G.) carolae ( Dujardin, 1973, 157) ( Adscita rungsi subsp.)
Distribution: Morocco (southern High Atlas).
J. (G.) rungsi ( Dujardin, 1973, 155) ( Adscita)
Distribution: Morocco (Middle Atlas).
cognata-group (see Efetov & Tarmann, 2012, 35)
J. ( G.) cognata(Herrich-Schäffer, 1847, pl. 13, figs 94, 95) ( Procris) ( Figure 44)
cognata ( Lucas, 1849, 373) ( Procris) (a junior primary homonym)
gigantea ( Naufock, 1933a, 96) ( Procris)
Distribution: northern Algeria, Tunisia.
J. ( G.) benderi( Tarmann, 1985a, 17) ( Adscita)
koriflana ( Rungs, 1980, 140) ( Adscita cognata subsp.) [nomen nudum: unavailable under Code, Article 13.1]
Distribution: Morocco (Middle Atlas, High Atlas).
J. ( G.) maroccana( Naufock, 1937, 30) ( Procris)
Distribution: Morocco (Middle Atlas, High Atlas).
Subgenus JordanitaVerity, 1946, 134
[Type-species: Sphinx chloros Hübner, 1813, pl. 28, figs 128, 129; by original designation]
Larval host-plants: Asteraceae. Larva leaf-mining, even in last instars.
graeca-group (see Efetov & Tarmann, 2012, 35)
J. (J.) syriaca ( Alberti, 1937c, 94) ( Procris)
Distribution: southern Turkey (?), Syria, Lebanon, Israel, Jordan.
J. ( J.) graeca( Jordan, 1907a, 9) ( Procris)
Distribution: eastern Austria, Croatia, Bosnia and Herzegovina (?), Serbia, Montenegro, Albania, Macedonia, Greece, Slovakia, Hungary, Romania, Bulgaria, Ukraine, Moldova, Russia (European part, Crimea, Northern Caucasus), Georgia, Azerbaijan, Turkey, Cyprus, northern Iraq (subsp. graeca); Armenia, Iran (subsp. persica).
J. ( J.) graeca graeca ( Jordan, 1907a, 9) ( Procris)
sultana ( Alberti, 1937c, 96) ( Procris) (see Efetov, 2001d, 156)
J. ( J.) graeca persica ( Alberti, 1938a, 125) ( Procris “ssp. ?”)
chloros-group (see Efetov & Tarmann, 2012, 35)
J. ( J.) chloros(Hübner, 1813, pl. 28, figs 128, 129) ( Sphinx)
Distribution: southern France, Germany, eastern Switzerland, eastern Austria, northern Italy, Slovenia, Croatia, Bosnia and Herzegovina, Serbia, Montenegro, Albania, Poland, Czech Republic, Slovakia, Hungary, Romania, Bulgaria, Ukraine, Russia (European part, Crimea, Northern Caucasus, southern Siberia), Georgia, eastern Kazakhstan, Turkey, northern Syria, northern Iraq (subsp. chloros); southern Macedonia, southwestern Bulgaria, Greece (subsp. hades).
J. ( J.) chloros chloros (Hübner, 1813, pl. 28, figs 128, 129) ( Sphinx)
sepium ( Boisduval, 1834, 81) ( Procris sp.)
minor ( Eversmann, 1844, 91) ( Atychia statices var.) (see Efetov & Tarmann, 1999a, 25)
haegeri ( Alberti, 1973b, 386) ( Procris chloros subsp.)
J. ( J.) chloros hades ( Alberti, 1970a, 82) ( Procris)
J. ( J.) chloronota( Staudinger, 1871, 100) ( Ino chloros var.)
minima ( Alberti, 1937c, 93) ( Procris chloronota f.)
Distribution: Southern Turkey (Mersin, Adana).
globulariae-group (see Efetov & Tarmann, 2012, 36)
J. ( J.) tenuicornis( Zeller, 1847b, 293) ( Atychia)
Distribution: central Italy (subsp. tenuicornis); southern Italy (including Sicily) (subsp. turatii).
J. ( J.) tenuicornis tenuicornis ( Zeller, 1847b, 293) ( Atychia)
bellieri ( Rambur, 1866, 184) ( Procris sp.)
J. ( J.) tenuicornis turatii ( Bartel, 1906, 178) ( Ino sp.)
translucensVerity, 1946, 136 ( Jordanita tenuicornis “razza”)
J. ( J.) globulariae( Hübner, 1793, pl. 67) ( Sphinx)
caerulea ( Tutt, 1899, 408) ( Rhagades globulariae var.)
viridis ( Tutt, 1899, 408) ( Rhagades globulariae “ab.”)
azurea ( Vorbrodt, 1914, 248) ( Procris globulariae “Form”) (see Efetov & Tarmann, 1999a, 26)
acanthophora ( Agenjo, 1937, 302) ( Procris)
bosniaca ( Alberti, 1937b, 99) ( Procris globulariae subsp.)
stricta ( Verity, 1946, 134) ( Procris cognata “forma”)
aureoviridis ( Verity, 1946, 134) ( Procris cognata “forma”)
caerulea ( Verity, 1946, 134) ( Procris cognata “forma”)
urbis ( Verity, 1946, 134) ( Procris cognata “razza”)
cognata (sensu Jordan, 1907a, 8 ( Procris)) (nec Herrich-Schäffer, 1847, pl. 13)
cognata (sensu Agenjo, 1940, 48 ( Procris)) (nec Herrich-Schäffer, 1847, pl. 13)
cognata (sensu Verity, 1946, 132 ( Procris)) (nec Herrich-Schäffer, 1847, pl. 13)
Distribution: central and northern Spain, Andorra, southern England, France, Belgium, Luxembourg, central and southern Germany, Switzerland, Austria, northern and central Italy, Slovenia, Croatia, Bosnia and Herzegovina, Serbia, Montenegro, Albania, Macedonia, Greece, Poland, Czech Republic, Slovakia, Hungary, Romania, Bulgaria, Belarus, Ukraine, Russia (European part, Crimea, Northern Caucasus), northwestern Turkey.
J. (J.) fazekasi Efetov, 1998c, 183
Distribution: southern Hungary.
J. (J.) vartianae ( Malicky, 1961, 216) ( Procris)
Distribution: southeastern Spain.
Subgenus PravielaAlberti, 1954, 329
[Type-species: Procris anatolica Naufock, 1929, 94, by original designation]
Larval host-plants: Asteraceae. Larva leaf-mining, even in last instars.
J. (P.) anatolica ( Naufock, 1929, 94) ( Procris) (C42)
Distribution: Greece (Dodekanes), Turkey, Cyprus, Syria, Lebanon, Israel, Jordan, Iraq, Iran (Zagros and Elburs), Armenia, Azerbaijan (Nakhichevan) (subsp. anatolica); northeastern Libya, northern Egypt (subsp. kruegeri)
J. ( P.) anatolica anatolica ( Naufock, 1929, 94) ( Procris)
levantina ( Jordan, 1931, 277) ( Procris sp.)
pfeifferi ( Naufock, 1935, 7) ( Procris sp.)
J. ( P.) anatolica kruegeri ( Turati, 1930, 50) ( Ino sp.)
J. ( P.) rietzschiKeil, 2016b, 201(C42)
Distribution: Iran (Kuh-e-Gebal Bares).
J. ( P.) christinaeKeil, 1998, 113(C42)
Distribution: Iran (central Zagros).
Subgenus SolaniternaEfetov, 2004a, 33, 119
[Type-species: Ino subsolana Staudinger, 1862, 352, by original designation]
Larval host-plants: Asteraceae. Larva leaf-mining until hibernation, in last instars boring in stems of the host plant (only known for J. subsolana).
J. ( S.) subsolana( Staudinger, 1862, 352) ( Ino cognata “var. ?”)
cognata ( Rambur, 1858, pl. 3, fig. 1) ( Procris) (a junior primary homonym of Procris cognata
Herrich-Schäffer, 1847, pl. 13, figs 94, 95)
incognita ( Staudinger, 1862, 359) ( Ino cognata “fragliche Varietät”) [nomen nudum]
ramburi ( Praviel, 1938, 113) ( Procris subsolanasubsp.)
schuetzei ( Alberti, 1940, 313) ( Procris subsolana subsp.)
modesta ( Verity, 1946, 129) ( Procris subsolana “razza”)
venusta ( Verity, 1946, 129) ( Procris subsolana “razza”)
globulariae partim (sensu Jordan, 1907a, 8 ( Procris)) (nec Hübner, 1793, pl. 67)
Distribution: Morocco (Rif), Portugal, Spain, France, Belgium, Germany, Switzerland, Austria, Italy (including Sicily), Slovenia, Croatia, Bosnia and Herzegovina, Serbia, Montenegro, Albania, Macedonia, Greece, Czech Republic, Slovakia, Hungary, Romania, Bulgaria, Ukraine, Moldova, Russia (European part, Crimea, Northern Caucasus, southern Siberia), Georgia, Armenia, Azerbaijan, Kazakhstan, Turkey, northern Iran.
J. ( S.) solana( Staudinger, 1887a, 72) ( Ino subsolana “var. ?”)
gouldschaensis ( Alberti, 1937ca 81) ( Procris solana subsp.) (see Efetov & Tarmann, 1999a, 25)
Distribution: Uzbekistan, Kyrgyzstan, southern Kazakhstan.
Subgenus RjaboviaEfetov & Tarmann, 1995b, 70
[Type-species: Procris horni Alberti, 1937c, 93, by original designation and monotypy]
Larval host-plants: Asteraceae. Larva leaf-mining, even in last instars.
J. (R.) horni ( Alberti, 1937c, 93) ( Procris)
armena ( Alberti, 1970a, 79) ( Procris) [nomen nudum]
Distribution: Armenia, Azerbaijan, Turkey, Iran.
Genus SaliuncaWalker, 1865 (“1864”), 108 (C29)
[Type species: Tipulodes thoracica Walker, 1865 (“1864”), 108, by original designation]
Larval host-plants: unknown.
S. styx ( Fabricius, 1775, 556) ( Zygaena)
Distribution: Africa (Gulf of Guinea: Sierra Leone, Togo, Cameroon, Ecuatorial Guinea).
S. styx styx ( Fabricius, 1775, 556) ( Zygaena. thoracica ( Walker, 1865, 108) ( Tipulodes. latipennis Strand, 1913, 57
S. styx ealaensis Alberti, 1957, 299
S. pallida Alberti, 1957, 299
Distribution: Central Africa (Congo).
S. ventralis Jordan, 1907b, 123
analogaAlberti, 1957, 300 ( Saliunca ventralis “mod. ”), unavailable (infrasubspecific)
Distribution: Eastern Africa (Uganda).
S. rufidorsis ( Plötz, 1880, 80) (“ Antichloris ?”)
Distribution: western central Africa (Cameroon).
S. cyanea Hampson, 1920a, 276
Distribution: Eastern Africa (Uganda).
S. vidua Rebel, 1914, 290
Distribution: Central Africa (Congo).
S. aurifrons Walker, 1864, 109
Distribution: western and Central Africa (subsp. aurifrons) and Eastern Africa (subsp. ugandana).
S. aurifrons aurifrons Walker, 1864, 109
aitcha ( Vuillot, 1892, CXL) (as aïtcha) ( Naclia)
S. aurifrons ugandana Jordan, 1909, 258
S. flavifrons ( Plötz, 1880, 80) (“ Antichloris ?”)
Distribution: western Central Africa (Cameroon, Ecuatorial Guinea).
S. flavifrontis Bryk, 1936, 279
flavifrons Bethune-Baker, 1927: 333 (nec Plötz, 1880, 80)
Distribution: western Central Africa (Cameroon).
S. fulviceps Hampson, 1920a, 276
fulvicepes [sic] Bryk, 1936, 279, misspelling
Distribution: Eastern Africa.
S. cyanothorax Hampson, 1920a, 277
Distribution: Eastern Africa (Uganda).
S. solora ( Plötz, 1880, 80) (“ Antichloris ?”) ( Figure 47)

Distribution: western Central Africa (Cameroon, Ecuatorial Guinea).
S. sapphirina Hampson, 1920a, 2
saphirina [sic] Gaede, 1926, 31, misspelling
Distribution: Eastern Africa (Uganda).
S. meruana Aurivillius, 1910, 48
Distribution: Central and Eastern Africa (Congo, Ethiopia, Kenia, Uganda, Tanzania) (subsp. meruana), western Central Africa (Ecuatorial Guinea) (subsp. tessmanni).
S. meruana meruana Aurivillius, 1910, 48
S. meruana tessmanni Alberti, 1957, 302
S. anhyalina Alberti, 1957, 302
Distribution: Eastern Africa (Ethiopia).
S. chalconota Hampson, 1920a, 277
Distribution: Eastern Africa (Uganda).
S. homochroa ( Holland, 1897, 409) (“ Charidea ?”) ( Figure 46)
Distribution: Eastern Africa (Ethiopia, Kenya, Mozambique, Somalia, Uganda, Zimbabwe).
S. metacyanea Hampson, 1920a, 277
Distribution: Eastern Africa.
S. aenescens Hampson, 1920a, 277
Distribution: Eastern Africa (Uganda, Kenia).
S. mimeticaJordan, 1907, 123
nkolentangensis Strand, 1912, 57
Distribution: Africa (Nigeria to Congo).
S. kamilila Bethune-Baker, 1911, 575
Distribution: Eastern Africa.
S. egeria Bethune-Baker, 1913, 67
Distribution: Eastern Africa (Uganda).
S. ignicincta Andreini, 1912, 141
Distribution: Eastern Africa (Eritrea).
S. rubriventris Holland, 1919, 324
Distribution: Central Africa (Congo).
S. orphina Hering, 1931, 114
Distribution: Eastern Africa (Zaire).
Genus SyringuraHolland, 1893, 394(C29)
[Type species: Syringura uranopetes Holland, 1893, 394, by original designation]
Larval host-plants: unknown.
S. pulchra ( Butler, 1876, 358) ( Tascia) ( Figure 48)
Distribution: Central and Eastern Africa.
S. triplex ( Plötz, 1880, 79) ( Tascia)
triplax [sic] ( Kirby, 1892, 169), misspelling ( Tascia)
uranopetesHolland, 1893, 394
triguttata ( Aurivillius, 1925, 1347) ( Saliunca)
Distribution: Africa (Togo, Sierra Leone, Cameroon).
Genus TasciaWalker, 1856, 1600 (C29)
[Type species: Tascia chrysotelus Walker, 1856, 1600, by monotypy]
ParasyntomisDistant, 1897, 15
[Type species: Parasyntomis aethiops Distant, 1897, 15, by monotypy]
Tassia [sic] Druce, 1910, 392, misspelling
Larval host-plants: unknown.
T. amata Druce, 1910, 402
Distribution: Eastern Africa (Ethiopia).
T. finalis ( Walker, 1854, 245) ( Euchromia) ( Figure 49)
chrysotelusWalker, 1856, 1600
virescens Butler, 1876, 357
aethiops ( Distant, 1897) ( Parasyntomis)
Distribution: South Africa (Transvaal, Natal).
T. instructa ( Walker, 1854, 254) ( Anycles (Euchromia)) erythrogaster ( Mabille, 1892, CXXXIX) (“ Naclia ?”) erythropygaGaede, 1926, 33
Distribution: Africa (Sierra Leone).
T. rhabdophora Vari, 2002, xi, pl. 1 ( Figure 13)
Distribution: Africa (Zimbabwe).
Genus AstyloneuraGaede, 1914, 53(C29)
[Type species: Astyloneura trefurthi Gaede, 1914, 53, by monotypy]
Larval host-plants: unknown.
A. trefurthi Gaede, 1914, 53
cyanopis ( Hampson, 1920a, 278) ( Saliunca)
Distribution: Southern Africa (Zimbabwe).
A. meridionalis ( Hampson, 1920a, 278) ( Saliunca) ( Figure 50)
Distribution: eastern Central and Eastern Africa (from Kenia, Uganda, Tanzania, southwards to northern South Africa) (subsp. meridionalis), western Central Africa (Congo) (subsp. centralis).
A. meridionalis meridionalis ( Hampson, 1920a, 278) ( Saliunca)
A. meridionalis centralis Alberti, 1957, 306
A. incerta Alberti, 1957, 306
Distribution: Central Africa (Congo).
A. difformis ( Jordan, 1907b, 122) ( Saliunca)
Distribution: Central and Eastern Africa (Congo, Uganda, Tanzania).
A. cupreotincta ( Hampson, 1920a, 279) ( Saliunca)
Distribution: Eastern Africa.
A. esmeralda ( Hampson, 1920a, 279) ( Saliunca)
Distribution: Africa (from Nigeria to Uganda).
A. glennia ( Jordan, 1907, 123) ( Saliunca)
Distribution: Southern Africa (Zimbabwe).
A. biplagata ( Bethune-Baker, 1911, 575) ( Saliunca)
biplagiata [sic] Gaede, 1926, 33, misspelling
Distribution: Southern Africa (Zimbabwe).
A. assimilis ( Jordan, 1907b, 122) ( Saliunca)
Distribution: Southern Africa (Zimbabwe).
A. nitens ( Jordan, 1907b, 122) ( Saliunca)
Distribution: Eastern Africa (Uganda).
A. ostia ( Druce, 1896, 350) ( Saliunca)
ostea [sic] Gaede, 1926, 36, misspelling
Distribution: Eastern Africa.
A. chlorotica ( Hampson, 1920a, 280) ( Saliunca)
Distribution: Central Africa (Congo).
A. monotona Hering, 1931, 115
Distribution: Central Africa (Congo).
A. gaedei Alberti, 1957, 310
Distribution: Central Africa (Congo (W. Ruwenzori)).
A. bicolora Röber, 1929, 327
Distribution: Eastern Africa (Ethiopia).
Genus AcoloithusClemens, 1861, 539
[Type species: Acoloithus falsarius Clemens, 1861, 539, by monotypy]
Larval host-plants: Vitaceae ( Vitis spp.) (known for A. falsarius, A. novaricus, A. rectarius).
A. falsarius Clemens, 1861, 539
sanborni ( Packard, 1864, 32) ( Harrisina)
Distribution: eastern USA.
A. novaricus Barnes & McDunnough, 1913, 295
Distribution: eastern USA.
A. rectarius Dyar, 1898, 44 ( Figure 51)

Distribution: western USA.
A. ruficollis (Druce, 1884, 37) ( Urodus)
Distribution: Guatemala.
A. dyraspes ( Druce, 1896, 330) ( Urodus)
Distribution: Mexico.
A. rubrojugulatus ( Alberti, 1954, 340) ( Gonioprocris)
Distribution: eastern USA.
A. totusniger Alberti, 1954, 339
Distribution: Mexico.
A. opacusJordan, 1913, 25
Distribution: Colombia.
Genus GaedeaHering, 1924, 272
[Type species: Gaedea separata Hering, 1924, 273, by original designation and monotypy]
Larval host-plants: unknown.
G. separata Hering, 1924, 273
Distribution: Bolivia.
Genus NeoilliberisTarmann, 1984b, 55
[Type species: Pyromorpha fusca H. Edwards, 1885, 43, by original designation]
Larval host-plants: Fabaceae ( Acacia spp.) (known for N. fusca).
N. martenii ( French, 1884, 13) ( Triprocris)
barnea ( Druce, 1896, 329) ( Procris)
Distribution: southwestern USA, northern Mexico.
N. tarahumara Tarmann, 1984b, 58
Distribution: northern Mexico.
N. fusca (H. Edwards, 1885, 43) ( Pyromorpha) ( Figure 52)
landia ( Druce, 1896, 329) ( Procris)
Distribution: southwestern USA, northern Mexico.
N. mas ( Dyar, 1918a, 137) ( Gingla)
Distribution: northern Mexico.
N. raconica ( Dyar, 1913, 316) ( Gingla) Mexico
purpusi ( Hering, 1924, 270) ( Malthaca)
Distribution: northern Mexico.
N. thyesta ( Druce, 1884, 37) ( Procris)
Distribution: Guatemala, Costa Rica.
N. puebla Tarmann, 1984b, 59
Distribution: central Mexico.
N. kendalli Tarmann, 1984b, 60
Distribution: southern USA.
N. arizonica Tarmann, 1984b, 64
Distribution: southern USA.
N. ignorata ( Hering, 1925, 157) ( Malthaca)
Distribution: southern Mexico, Guatemala.
Genus NeofelderiaTarmann, 1984b, 65
[Type species: Acreagris correbioides Felder, 1874, pl. 83, by monotypy]
FelderiaKirby, 1892, 163 (a junior homonym of Felderia Walsingham 1887, 165 - Insecta, Lepidoptera. Tineidae. The objective replacement name is NeofelderiaTarmann, 1984b, 65)
AcreagrisFelder, 1874, 83 (a junior homonym of AcreagrisKoch & Berendt, 1854, 123 - Insecta,Collembola. The objective replacement name is Felderia Kirby, 1892, 163)
[Type species: Acreagris correbioides Felder, 1874, pl. 83, by monotypy]
Larval host-plants: Fabaceae.
N. rata ( H. Edwards, 1882, 124) ( Lycomorpha) ( Figure 53)
xanthogramma ( Hering, 1924, 268) ( Malthaca)
Distribution: southern USA, Mexico.
N. correbioides ( Felder, 1874, [83]) ( Acreagris)
aurora ( Dyar, 1918b, 366) ( Pyromorpha)
Distribution: Mexico.
N. mexicana ( Druce, 1884, 41) ( Lycomorpha)
venadiocola ( Dyar, 1920, 198) ( Triprocris)
Distribution: Mexico.
N. eromena ( Hering, 1924, 267) ( Malthaca)
analoga ( Hering, 1924, 268) ( Malthaca)
Distribution: Mexico.
N. hoerwertneri Tarmann, 1984b, 67
Distribution: Guatemala.
Genus NeoalbertiaTarmann, 1984b, 68
[Type species: Lycomorpha constans H. Edwards, 1881, 81]
Larval host-plants: unknown.
N. constans ( H. Edwards, 1881, 81) ( Lycomorpha) ( Figure 54)
sancta ( Neumoegen & Dyar, 1894, 64) ( Triprocris constans “forma”)
Distribution: Southern USA.
N. brunnea ( Alberti, 1954, 388) ( Malthaca)
Distribution: Mexico.
N. variabilis Tarmann, 1984b, 70
Distribution: Mexico.
N. guatemalteca Tarmann, 1984b, 72
Distribution: Guatemala.
N. anacreon ( Druce, 1884, 41) ( Procris)
Distribution: Costa Rica.
Genus ProcrisimilisTarmann, 1984b, 73
[Type species: Procrisimilis columbianaTarmann, 1984b, 74, by original designation and monotypy]
Larval host-plants: unknown.
P. columbiana Tarmann, 1984b, 74
Distribution: Colombia.
Genus Neoprocris Jordan, 1915, 300
[Type species: Neoprocris saltuaria Jordan, 1915, 300, by original designation]
Neoproctis [sic] Bryk, 1936, 302, 308, misspelling
Larval host-plants: Rosaceae (known for N. aversa, N. floridana and N. prunivora).
N. aversa ( H. Edwards, 1884a, 13) ( Triprocris) ( Figure 55)
huachuca ( Dyar, 1905b, 186) ( Parasa chloris var.)
Distribution: southwestern USA.
N. floridana Tarmann, 1984b, 77
Distribution: southeastern USA (Florida).
N. prunivora Tarmann, 1984b, 80
Distribution: southern USA (Texas).
N. basalis ( H. Edwards, 1887, 91) ( Triprocris)
Distribution: Mexico.
N. flora ( Schaus, 1911, 183) ( Propyria)
Distribution: Costa Rica.
N. isochroa ( Jordan, 1913, 25) ( Acoloithus)
pusilla (sensu Druce, 1884, 37 ( Procris)) (nec Walker, 1854, 112)
Distribution: Panama.
N. thyana ( Druce, 1884, 37) ( Procris)
Distribution: Guatemala.
N. seva ( Hering, 1938, 432) ( Seryda)
Distribution: Guatemala.
N. satanas ( Hering, 1938, 432) ( Acoloithus)
Distribution: Guatemala.
N. mirabilis Tarmann, 1984b, 82
Distribution: Costa Rica.
N. trismegistos ( Hering, 1926, 112) ( Acoloithus)
Distribution: Brazil.
N. viridis ( Druce, 1906, 86) ( Procrimima)
saltuariaJordan, 1915, 300
Distribution: Brazil, Bolivia, Paraguay, Argentina.
N. gigantea ( Hering, 1924, 272) ( Acoloithus)
Distribution: Bolivia.
N. metallica ( Schaus, 1892, 284) ( Ardonea)
Distribution: Peru.
N. mimetica ( Hering, 1928, 280) ( Tetraclonia)
Distribution: Bolivia.
Genus HoerwertneriaTarmann, 1984b, 84
[Type species: Malthaca rubriventris Hering, 1938, 406, by original designation and monotypy]
Larval host-plants: unknown.
H. rubriventris ( Hering, 1938, 406) ( Malthaca)
Distribution: Guatemala.
Genus PseudoprocrisDruce, 1884, 38
[Type species: Pseudoprocris gracilis Druce, 1884, 38, by subsequent designation by Kirby, 1892, 86]
Larval host-plants: Rosaceae (probably, see Tarmann 1984, 88).
P. dolosa Druce, 1884, 38
Distribution: Guatemala.
P. gracilis Druce, 1884, 38 ( Figure 56)
morelia ( Dyar, 1912a, 99) ( Adscita)
Distribution: southern Mexico, Guatemala.
Genus MalambliaJordan, 1907b, 124 (see Tarmann, 1985b, 341)
[Type species: Malamblia durbanica Jordan, 1907b, 125, by monotypy]
ChilioprocrisJordan, 1913, 24
[Type species: Procris melas Guérin-Méneville, 1839, 2, by original designation and monotypy]
Larval host-plants: Rosaceae (probably, see Tarmann 1984, 92).
M. melas ( Guérin-Méneville, 1839, 2) ( Procris) ( Figure 57)

durbanicaJordan, 1907b, 125
Distribution: Chile.
Genus PyromorphaHerrich-Schäffer, 1854, pl. 43
[Type species: Pyromorpha dimidiata Herrich-Schäffer, 1854, pl. 43, by monotypy]
MalthacaClemens, 1861, 540
[Type species: Malthaca perlucidula Clemens, 1860, 541, by monotypy]
CoementaDruce, 1885, 123
[Type species: Coementa timon Druce, 1885, 123, by subsequent designation by Kirby, 1892, 287]
Larval host-plants: Fagaceae ( Quercus).
Subgenus Gingla Walker, 1864, 128
[Type species: Gingla radialis Walker, 1864, 128, by monotypy]
Larval host-plants: unknown.
P. (G.) radialis (Walker, 1864, 128) ( Gingla)
Distribution: Mexico (subsp. radialis); Guatemala (subsp. drucei); Guatemala (subsp. crypta).
P. (G.) radialis radialis (Walker, 1864, 128) ( Gingla)
P. (G.) radialis drucei ( Jordan, 1913, 22) ( Malthaca)
P. (G.) radialis crypta ( Hering, 1924, 270) ( Malthaca)
P. (G.) synecha ( Hering, 1924, 271) ( Malthaca)
Distribution: Costa Rica, Panama, Colombia, Peru.
P. (G.) contermina ( H. Edwards, 1884a, 13) ( Lycomorpha)
aequalis ( Druce, 1885, 123) ( Gingla)
Distribution: Mexico.
P. (G.) perezi Tarmann, 1984b, 97
Distribution: Mexico.
P. (G.) josialis ( Druce, 1885, 123) ( Gingla)
Distribution: Guatemala
P. (G.) timon ( Druce, 1885, 123) ( Coementa)
Distribution: Costa Rica.
P. (G.) tristeza Tarmann, 1984b, 99
Distribution: Colombia.
P. (G.) xanthura ( Jordan, 1913, 23) ( Malthaca)
Distribution: Ecuador.
P. (G.) semifulva ( Druce, 1896, 332) ( Lycomorpha) (C43)
basirei ( Druce, 1896, 331) ( Lycomorpha)
Distribution: Mexico.
P. (G.) morio (Druce, 1885, 123) ( Coementa)
Distribution: Guatemala, Costa Rica.
P. (G.) monochroma ( Hering, 1924, 270) ( Malthaca)
Distribution: Bolivia.
Subgenus EuclimaciopsisTremewan, 1973, 126
[Type species: Gingla tortricalis Druce, 1885, 120, by monotypy]
Euclimacia Jordan, 1913, 21 (a junior homonym of Euclimacia Enderlein, 1910, 342 - Insecta, Neuroptera. The objective replacement name is EuclimaciopsisTremewan, 1973, 126)
[Type species: Gingla tortricalis Druce, 1885, 120, by monotypy]
Larval host-plants: unknown.
P. (E.) tortricalis (Druce, 1885, 120) ( Gingla)
Distribution: Costa Rica.
Subgenus Pyromorpha Herrich-Schäffer, 1854, pl. 43
[Type species: Pyromorpha dimidiata Herrich-Schäffer, 1854, pl. 43, by monotypy]
Larval host-plants: Fagaceae ( Quercus) (known for P. (P). dimidiata, P. (P). cinniana, P. (P). dyari, P. (P.) brueckneri)
P. (P.) dimidiata Herrich-Schäffer, 1854, pl. 43 ( Figure 59)
perlucidula ( Clemens, 1861, 541) ( Malthaca)
Distribution: eastern USA.
P. (P.) caelebs Blanchard, 1972, 79
Distribution: southern USA (Texas).
P. (P.) marginata ( H. Edwards, 1884a, 13) ( Lycomorpha)
Distribution: Mexico.
P. (P.) teos ( Schaus, 1889, 88) ( Lycomorpha)
Distribution: Mexico.
P. (P.) astora ( Dyar, 1912a, 99) ( Gingla)
Distribution: Mexico.
P. (P.) cinniana Druce, 1884, 40
tristrigata ( Hering, 1926, 110) ( Tetraclonia)
Distribution: southern Mexico, Guatemala.
P. (P.) dyari ( Jordan, 1913, 24) ( Tetraclonia) ( Figure 60)
laterculae ( Dyar, 1900, 32) ( Gingla) (a junior primary homonym of Lycomorpha latercula H. Edwards, 1882, 124)
Distribution: southern USA, Mexico.
P. (P.) forreri (Druce, 1884, 37) ( Procris)
Distribution: Mexico.
P. (P.) jordani ( Hering, 1924, 226) ( Malthaca)
Distribution: Mexico.
P. (P.) latercula ( H. Edwards, 1882, 124) ( Lycomorpha. flavescens (Hering, 1924, 272) ( Tetraclonia latercula “forma”) beovava ( Dyar, 1918b, 366) ( Gingla)
erythromelas ( Jordan, 1913, 22) ( Malthaca)
Distribution: southern USA, Mexico.
P. (P.) brueckneri ( Hering, 1926, 109) ( Tetraclonia)
mimica ( Hering, 1926, 111) ( Seryda)
Distribution: Guatemala.
P. (P.) centralis ( Walker, 1854, 288) ( Lycomorpha)
notha ( H. Edwards, 1885, 128) ( Lycomorpha)
Distribution: Mexico.
P. (P.) ursula ( Hering, 1924, 267) ( Malthaca)
Distribution: Mexico.
P. (P.) monotona ( Hering, 1924, 266) ( Malthaca)
Distribution: Mexico.
P. (P.) myrla ( Dyar, 1912a, 99) ( Gingla)
Distribution: Mexico.
P. (P.) flavibasalis ( Hering, 1938, 431) ( Tetraclonia)
Distribution: Guatemala.
P. (P.) brueckneriana ( Hering, 1938, 405) ( Malthaca)
Distribution: Guatemala.
P. (P.) costaricensisTarmann, 1984b, 102
Distribution: Costa Rica.
P. (P.) cuchumatanaTarmann, 1984b, 100
Distribution: Guatemala.
P. (P.) ripena ( Druce, 1906, 86) ( Ptychoglene)
tetraclonioides ( Hering, 1924, 269) ( Malthaca)
Distribution: Bolivia, Peru.
Genus TetracloniaJordan, 1913, 24
[Type species: Tetraclonia sauciaJordan, 1913, 2 4, by original designation]
Larval host-plants: unknown.
T. nigrivena Hering, 1928, 280
Distribution: Peru.
T. zischkai Tarmann, 1984b, 105
Distribution: Bolivia.
T. saucia Jordan, 1913, 24
Distribution: Peru.
Genus GonioprocrisJordan, 1913, 23
[Type species: Gonioprocris xena Jordan, 1913, 23, by original designation]
Larval host-plants: Fagaceae ( Quercus) (probably, see Tarmann 1984, 110).
G. xena Jordan, 1913, 23
Distribution: Guatemala.
G. siruna Hering, 1938, 406 ( Figure 58)
Distribution: Guatemala.
G. megalops (Druce, 1884, 38) ( Seryda)
Distribution: Guatemala.
Genus TriprocrisGrote, 1873, 35
[Type species: Procris smithsoniana Clemens, 1861, 540, by original designation and monotypy]
Larval host-plants: Nyctaginaceae (known for T. smithsoniana, T. yampai, T. cyanea).
T. smithsoniana ( Clemens, 1861, 540) ( Procris)
Distribution: western USA.
T. yampai Barnes, 1905, 215
Distribution: western USA.
T. cyanea Barnes & McDunnough, 1910, 162 ( Figure 61)
Distribution: western USA.
T. lustrans Beutenmüller, 1894, 367
Distribution: western USA.
T. ruemelii (Druce, 1884, 40) ( Harrisina)
Distribution: Mexico.
T. rosetta Dyar, 1918b, 366
Distribution: Mexico.
T. similissima Tarmann, 1984b, 112
Distribution: Mexico.
T. auchenochrysa ( Dyar, 1912a, 99) ( Harrisina)
flavithorax ( Hering, 1925, 165) ( Harrisina)
T. flavipuncta Tarmann, 1984b, 114
Distribution: Brazil.
Genus Pampa Walker, 1854, 238
[Type species: Euchromia mystica Walker, 1854, 239. by subsequent designation by Kirby, 1892, 112]
NesaceKirby, 1892, 112 (unnecessary objective replacement name for Pampa Walker, 1854, 238)
[Type species: Euchromia mystica Walker, 1854, 239, by subsequent designation by Kirby, 1892,112]
Larval host-plants: Vitaceae.
P. anisa ( Hering, 1924, 275) ( Harrisina)
Distribution: Bolivia.
P. boliviensis ( Hering, 1924, 274) ( Harrisina)
Distribution: Bolivia.
P. pseudovenata Tarmann & Drouet, 2015, 215
Distribution: French Guiana.
P. approximata ( Hering, 1924, 275) ( Harrisina)
prava ( Hering, 1925, 166) ( Harrisina)
Distribution: Columbia.
P. tersa ( Druce, 1899, 232) ( Harrisina)
Distribution: Mexico.
P. mystica ( Walker, 1854, 239) ( Euchromia)
Distribution: Honduras.
P. venata ( Jordan, 1913, 28) ( Harrisina)
Distribution: Brazil.
P. peritta ( Hering, 1924, 274) ( Harrisina)
Distribution: Brazil.
P. erythrogramma ( Hering, 1924, 273) ( Harrisina)
Distribution: Uruguay.
P. virescens ( Hampson, 1907, 328) ( Nesace)
Distribution: Brazil.
P. erroris Tarmann, 1984b, 119
Distribution: Brazil.
P. rubroventralis ( Hering, 1932, 154) ( Harrisina)
Distribution: Brazil.
P. hermieri Tarmann & Drouet, 2015, 214
Distribution: French Guiana.
P. splendens ( Jordan, 1913, 29) ( Harrisina)
Distribution: Peru.
P. smaragdina ( Hering, 1941, 111) ( Harrisina)
Distribution: Columbia, French Guiana.
P. janeira ( Schaus, 1892, 272) ( Harrisina)
Distribution: Brazil.
P. lepta ( Jordan, 1913, 28) ( Harrisina)
Distribution: Columbia.
P. mephisto ( Jones, 1921, 356) ( Harrisina)
Distribution: Brazil.
P. aidae Tarmann, 1984b, 123
Distribution: Brazil.
P. eminens ( Schaus, 1892, 272) ( Harrisina)
Distribution: Brazil.
P. incredibilis Tarmann, 1984b, 124
Distribution: Brazil.
P. esperanzae Tarmann, 1984b, 127
Distribution: Brazil.
P. zikani ( Hering, 1932, 154) ( Harrisina)
Distribution: Brazil.
P. anamariae Tarmann, 1984b, 132
Distribution: Brazil.
P. pseudoeminens Tarmann, 1984b, 133
Distribution: Paraguay.
P. proeminens ( Jörgensen, 1932, 49) ( Harrisina)
Distribution: Brazil.
P. fulvinota ( Butler, 1876, 361) ( Harrisina)
Distribution: Brazil.
P. brevistrigata ( Hering, 1924, 276) ( Harrisina)
Distribution: Brazil.
P. seitzi ( Hering, 1932, 155) ( Harrisina)
Distribution: Brazil.
P. innocens ( Hering, 1925, 165) ( Harrisina)
Distribution: Brazil.
P. ricara ( Jörgensen, 1932, 49) ( Harrisina)
Distribution: Paraguay.
Genus StyluraBurmeister, 1878, 391
[Type species: Laemocharis forficula Herrich-Schäffer, 1855, pl. 54, by monotypy]
Larval host-plants: Vitaceae.
S. forficula ( Herrich-Schäffer, 1855, pl. 54) ( Laemocharis)
Distribution: Brazil.
S. brasiliensis Costa Lima, 1928, 25 ( Figure 14)
Distribution: Brazil.
S. cirama ( Druce, 1896, 330) ( Harrisina)
Distribution: Guatemala, Costa Rica.
S. guyanensis Tarmann & Drouet, 2015, 225
Distribution: French Guiana.
S. lamonti Tarmann & Cock, 2019, 155
Distribution: Trinidad.
Genus HarrisinaPackard, 1864, 31
[Type species: Aglaope americana Guérin-Méneville, 1844, 501, by subsequent designation by Dallas, 1865, 499]
HarrisinulaHering, 1925, 158
[Type species: Harrisinula infernalis Hering, 1925, 153, 158, by original designation]
Larval host-plants: Vitaceae (known for H. americana, H. coracina, H. metallica, H. guatemalena).
H. americana ( Guérin-Méneville, 1844, 501) ( Aglaope)
texana Stretch, 1872, 181
australis Stretch, 1885, 102
Distribution: eastern and southern USA.
H. coracina ( Clemens, 1861, 539) ( Aglaope)
nigrinaGraef, 1887, 41
Distribution: southern USA, northern Mexico.
H. metallica Stretch, 1885, 102 ( Figure 62)
brilliansBarnes & McDunnough, 1910, 162
coracina (sensu Neumoegen & Dyar, 1894, 65) (nec Clemens 1861, 539)
elongata Druce, 1896, 330
tessacansDyar, 1912a, 100
Distribution: western USA, northern Mexico.
H. charax Druce, 1896, 330
Distribution: Mexico.
H. draudti Hering, 1925, 166
Distribution: Mexico.
H. chalcinaJordan, 1913, 28
Distribution: Mexico.
H. mexicana Schaus, 1889, 87, stat. rev. (C44)
Distribution: Mexico.
H. guatemalena (Druce, 1884, 40) ( Aglaope)
Distribution: Guatemala, Costa Rica.
H. terginaJordan, 1913, 28
Distribution: Colombia.
H. infernalis ( Hering, 1925, 158) ( Harrisinula)
Distribution: Brazil.
H. longicaulis Hering, 1925, 165
Distribution: Venezuela.
Genus HarrisinopsisJordan, 1913, 26
[Type species: Harrisinopsis robusta Jordan, 1913, 26, by original designation and monotypy]
Monalita Tremewan, 1973, 134 (see Gernaat et al. 2022, 164) [Type species: Lamontia calibana Kaye, 1923, 997, by monotpy]
LamontiaKaye, 1923, 997 (a junior homonym of Lamontia Kirk, 1895, 289 - (Calcarea, Spongidae (now Baeriidae)).
The objective replacement name is MonalitaTremewan, 1973, 134) [Type species: Lamontia calibanaKaye, 1923, 997, by monotpy]
Larval host-plants: Chrysobalanaceae ( Hirtella paniculata) ( Gernaat et al. 2022).
H. robusta Jordan, 1913, 26 ( Figure 20)
tessmanniHering, 1928, 281
Distribution: Peru, Brazil, French Guiana, Surinam.
H. calibana ( Kaye, 1923, 997) ( Lamontia)
Distribution: Trinidad.
H. faurei ( Tarmann & Drouet, 2015, 227) ( Monalita)
Distribution: French Guiana.
H. laguerrei ( Tarmann & Drouet, 2015, 228) ( Monalita)
Distribution: French Guiana.
Genus SetiodesHerrich-Schäffer, 1866, 106
[Type species : Setiodes nana Herrich-Schäffer, 1866, 106, by monotypy]
FormiculusGrote, 1866, 184
[Type species : Formiculus pygmaeus Grote, 1866, 185, by monotypy]
Larval host-plants: Vitaceae ( Vitis spp.).
S. nana Herrich-Schäffer, 1866, 106
Distribution: Cuba (subsp. nana); Bahamas (subsp. bahamensis).
S. nana nana Herrich-Schäffer, 1866, 106
pygmaeus ( Grote, 1866, 185) ( Formiculus)
S. nana bahamensis Dyar, 1899, 100
albimacula ( Hampson, 1904, 180) ( Nesaca)
Genus ZikanellaHering, 1932, 153
[Type species: Zikanella rubrivitta Hering, 1932, 153, by original designation and monotypy]
Larval host-plants: unknown.
Z. rubrivitta Hering, 1932, 153
Distribution: Brazil.
Genus Urodopsis Jordan, 1913, 29
[Type species: Urodus subcaeruleus Dognin, 1910, 43, by original designation]
Larval host-plants: unknown.
U. subcaerulea ( Dognin, 1910, 43) ( Urodus)
Distribution: Colombia.
U. dryas Jordan, 1915, 301
Distribution: Brazil.
U. pusilla ( Walker, 1854, 112) ( Procris)
Distribution: Venezuela, Brazil.
U. melaenella (Hampson, 1919, 272) ( Pycnoctena)
hyalinaHering, 1932, 155
Distribution: Brazil.
Genus PycnoctenaFelder, 1874, pl. 83
[Type species: Pycnoctena angustula Felder, 1874, pl. 83, by monotypy]
Larval host-plants: unknown.
P. angustula Felder, 1874, pl. 83
Distribution: Brazil, French Guiana.
P. invaria ( Walker, 1854, 240) ( Euchromia)
Distribution: Brazil.
P. tristis Hering, 1932, 153
Distribution: Brazil.
P. dantasi ( Schaus, 1892, 272) ( Harrisina)
uniformis ( Hering, 1932, 153) ( Seryda)
Distribution: Brazil.
Genus SerydaWalker, 1856, 1598
[Type species: Seryda cincta Walker, 1856, 1598, by monotypy]
Larval host-plants: unknown.
S. cincta Walker, 1856, 1598
Distribution: Brazil.
S. actinota Jordan, 1913, 26
Distribution: Colombia.
S. isa Jordan, 1913, 26
Distribution: Ecuador.
S. glaucotis ( Hampson, 1907, 328) ( Caementa)
Distribution: Guatemala.
S. gallardi Tarmann & Drouet, 2015, 231
Distribution: French Guiana.
S. confusa Tarmann & Drouet, 2015, 232
Distribution: French Guiana.
S. kairi Tarmann & Cock, 2019, 159
C01 The data of morphology and last DNA results showed that the genus Thyrassia forms an isolated monophyletic group within the subfamily Procridinae. We are describing here a new tribe Thyrassiini to reflect this situation.
Tribe Thyrassiini Efetov & Tarmann, tribus nov.
[Types-genus: ThyrassiaButler, 1876, 355]
Description. Habitus ctenuchid like, forewings triangular, brown, with transparent, white or yellow spots; hindwings extremely short, oval, brown, transparent or yellow with dark brown margin; head rounded, not compressed dorso-ventrally, with large compound eyes and a chaetosema that extends slightly but not completely between the compound eyes and the ocellus; chaetosema extended dorsomedially, the two ends of each side almost touching each other medially. The free space between the base of antenna, ocellus, compound eye and chaetosema and a narrow ring around the compound eye not smoothly scaled (as in all other Procridinae) but covered with a series of tiny hair-like scales ( Tarmann 2004, p. 21, fig. 70). Antenna shortly bipectinate proximally, very shortly biserrate distally in both sexes ( Tarmann, 2004, p. 22, figs 71-74). Legs short and robust; foretibia with epiphysis, tibial spurs absent. Frenulum a single spine in male and female. Abdomen without lateral evaginations on segments two and seven.
Male genitalia with short, stout, triangular, distally rounded uncus that is completely fused with the large tegumen; valva almost rectangular, without processes, central translucent part slightly fan- shaped; a long finger-like process inserted at the dorsal base of valva; this process forked basally, the short lateral process obviously representing an apodeme for a muscle to move the finger, the latter bent distally and bearing a brush of strong, short, backward-pointing spines. Aedeagus short, stout, strongly sclerotized, everted vesica without cornuti ( Tarmann, 2004, p. 217, figs 423-424).
Female genitalia with the papillae anales fused to a short ovipositor; ostium and ductus bursae translucent, without antrum; praebursa absent. The insertion of the ductus seminalis into the bag-like translucent corpus bursae is situated at the very centre of the corpus. This indicates that a possibly earlier present praebursa may be fused with the corpus bursae as otherwise the ductus seminalis must insert at the point where the ducus bursae widens into the corpus ( Tarmann, 2004, p. 217, fig. 425). See also Tarmann ( 2004, pp. 215-216). Larval host-plants: Vitaceae. Distribution. Southern and southeastern Asia, Australia.
C02 Most of Australian genera form a separate monophyletic branch of Procridinae that cannot be attributed to any hitherto known tribe. Therefore we are providing a description of a new tribe with the aim to accommodate this group.
Tribe Pollanisini Efetov & Tarmann, tribus nov.
[Type-genus: Pollanisus Walker, 1854, 114]
Description. Habitus variable, head dorsoventrally compressed, chaetosema extended between compound eye and ocellus (like in Artonini); foreleg without epiphysis, hindleg without medial spurs; genitalia male - valva fan-shaped without “ Artona-finger”; genitalia female - praebursa absent or if present, translucent, without double row of spines; larval host-plants: Dilleniaceae, Myrtaceae, Vitaceae, Arecaceae. Distribution. Australia, Fiji (1 species), South Africa (1 species).
C03 The genus Pollanisus was revised recently by Mollet & Tarmann (2023). The taxonomy and systematic order given here is following this revision.
C04 The taxon Pollanisus amethystina ( Meyrick, 1888) was synonymized with P. empyrea ( Meyrick, 1888) by Tarmann (2004) on the base of genitalia similarity and the fact that both taxa occur in the same regions of Western Australia and colour variations between more greenish, coppery or bluish shiny scales on the forewing upperside are not a significant character to distinguish species in the genus Pollanisus. However, new studies by Mollet (2019) have shown that these two taxa are in fact good species. Mollet reared both species from eggs to imagines, found that they live on different larval host-plants and have different habitats and distribution patterns. Consequently, he correctly reinstated Pollanisus amethystina as valid species.
C05 The phylogenetic position of LevuanaBethune-Baker, 1906, was cleared up by Nazari et al. (2019) by obtaining New Generation Sequencing (NGS)-results of historical material of L. iridescens, the type-species of Levuana. The DNA results and the morphological results were combined and show that this genus is close to the Australian genus MyrtartonaTarmann, 2004.
C06 After the exclusion of Thyrassiini, Pollanisini and Cleleini (see below) Artonini form a monophyletic group with important apomorphic character, viz. the presence of a movable process near the apex of sacculus (“ Artona-finger”). All species with this character are included within Artonini Tarmann, 1994, for which redescription can be found below.
Tribe ArtoniniTarmann, 1994, 120
[Type-genus: ArtonaWalker, 1854, 439]
Description. Habitus variable, head dorsoventrally compressed, chaetosema extended between compound eye and ocellus; foreleg with epiphysis, hindleg with single medial spur; genitalia male - valva with “ Artona-finger”, genitalia female - praebursa present, sclerotized, with double row of pointed spines; larval host-plants: Poaceae, Zingiberaceae, Pittosporaceae, Lauraceae, Musaceae, Arecaceae. Distribution. Australia, eastern, southern and southeastern Asia, tropical Africa.
C07 The genus ArtonaWalker, 1854, is still not sufficiently revised (see comment “C1” in Efetov & Tarmann 2012, p. 38) and the taxonomic situation at species level has not changed since. Artona still needs revising based on the entire type material but also with the inclusion of all the already known new species that have to be described and accommodated. The genus Artona is divided here into 5 subgenera: ArtonaWalker, 1854, Zeuxippa Herrich-Schäffer, 1855, stat. nov., Balataea Walker, 1865, stat. rev., PseudosesidiaAlberti, 1954, stat. rev., and FuscartonaEfetov & Tarmann, 2012.
C08 The type-species of the genus Artona, viz. A. discivitta Walker, 1854, originates from the Nilgiris (Western Ghat Mountains, Tamil Nadu, southern India) and is a geographically very isolated species. It shows genitalic characters that are different from all other species that are currently included into the genus Artona. All these species share with Artona discivitta the characteristic black and yellow habitus, the characteristic sexual dimorphism in pattern and the larval host-plants from the family Poaceae. However, the “brush” at the end of the “ Artona-finger” in the male genitalia is much stronger developed in all these species (except A. martini Efetov, 1997a, and A. uniformis (Alberti, 1954)) than in A. discivitta where it is almost absent and only represented by a few setae. The “ Artona-finger” in A. discivitta is only weakly developed, shorter and more slender than in all other species. There are also significant differences in the female genitalia (i.e. different form of praebursa in A. discivitta). The authors are well aware of this situation but prefer not to separate all other black and yellow species that are currently included into the genus Artona from its type-species A. discivitta. However, we leave A. discivitta as only one species in the subgenus Artona. Other species of the genus are included in the subgenera Zeuxippa, Balataea, Pseudosesidia and Fuscartona.
C09 Artona zebraica and A. fulvida are described in the same paper by Butler (1876, p. 356). These two taxa show very well how problematic the taxonomic situation within the genus Artona is. There are several species known with the habitus of A. zebraica (i.e. black ground colour with yellow spots on the forewing and some dark stripes in the hindwing and an abdomen that is narrowly ringed with yellow and black). These species are: A. digitata, A. nigrescens, A. phaeoxantha, A. khasiana (male), A. hypomelas (male), A. walkeri (male), A. hainana (male). However, most specimens with the habitus of A. zebraica are males whereas all specimens with the habitus of A. fulvida are females.
A. fulvida has the hindwing yellow with a dark margin and the abdomen always uniformly yellow with the first segment dark and the abdominal end also dark at least laterally. Species with A. fulvida habitus are: A. confusa, A. diffusa, A. flaviciliata, A. hypomelas (female), A. khasiana (female), A. walkeri (female), A. hainana (female). Species from the subgenera Artona and Zeuxippa (genus Artona) are sexually dimorphic. It is therefore possible that Butler in 1876 described males and females of the same species as two different taxa. However, most species of Artona (Zeuxippa) show this characteristic sexual dimorphism and this has already been mentioned by Jordan ( 1907a- 1908) (e.g. for A. khasiana Jordan, 1908, A. hypomelas Jordan, 1908). There are good genitalic differences between the different taxa. However, we often do not know which males belong to which females. Moreover, there are also females with almost “male habitus” known and it seems that the sexual dimorphism is not equally strong in all species. The whole situation is pretty hopeless as long as we are dependent on historical museum material. Rearing experiments from egg would solve this problem. In addition, the comparison of DNA sequences of new material could bring us further. However, species of Artona (subgenera Artona and Zeuxippa) have rarely been collected in the last decades and little new material is available. Moreover, the early stages and the life histories of most species are unknown.
C10 Artona nigrescensButler, 1876 , is distributed in northern India. It has been treated as a subspecies of A. walkeri Butler, 1876, by Jordan (1908, p. 43). Bryk (1936, p. 252) followed this opinion. However, A. (Zeuxippa) walkeri (Moore, 1859) is endemic to Java and Bali (see comment
C11). Therefore we have to raise Artona nigrescens Butler, 1876, back to species level until more information is available. The new status is: Artona (Zeuxippa) nigrescens Butler, 1876, stat. rev.
Artona neglecta Hering, 1925, is based on one single female (holotype). Hering describes it as “nahestehend Artona walkeri” (near to A. walkeri) and states that the pattern looks like that of A. walkeri hainana. However, we must take in account that A. (Z). walkeri is endemic to Java and Bali (see comment C11) and A. (Z). hainana is considered to represent a good species. A. neglecta is described from northern India and is almost certainly not conspecific with A. (Z). walkerior A. (Z). hainana. We therefore treat A. (Z). neglecta in this checklist again as a separate species as long as we cannot prove that it belongs to any other so far described species. The status is now: Artona (Zeuxippa) neglecta, stat. rev.
Artona guttata Snellen, 1892, has also been treated as a subspecies by Jordan ( 1908, p. 43) (like A. nigrescens) but of A. zebraica Butler, 1876. It occurs in central Sumatra. The female type deposited in NNML (Leiden, NL) and additional material from the BMNH (London, UK) show that this taxon is different from all Artona (Zeuxippa) species that are known from India, the SE Asian mainland and from the other Sunda Islands. We therefore treat Artona guttata Snellen, 1892, herewith again as a valid species. The status is now: Artona (Zeuxippa) guttata, stat. rev.
C11 Moore (1859) described Syntomis walkeri after male and female specimens from Java collected by Dr Horsefield. One male specimen in the BMNH has a round pin-label with red ring with the printed inscription “Type”. As far as we know, a lectotype has never been designated. The attached type label must have been put on the pin later, presumably by Karl Jordan who treated the Zygaenidae in the volumes of Seitz (1907a, 1907c, 1908). We therefore designate this specimen herewith as the male lectotype and figure it ( Figure 26).
Subsequently, Artona walkeri was mentioned from various south and southeast Asian countries between the Himalayas, southern China and the Sunda Islands and several taxa have been attached to this species as subspecies ( Hampson, 1893; Jordan, 1907; Bryk, 1926; Alberti, 1854). However, as far as examined by us, Artona (Zeuxippa) walkeri ( Moore, 1859) is restricted to the islands of Java and Bali. All specimens that are mentioned in the literature as A. walkeri but not originating from these two islands have been misidentified. The genitalia of A. (Z). walkeri are very characteristic (especially of the female) and cannot be mixed with other Artona species.
C12Artona pluristrigata is only known from its female holotype. The habitus is different from most other species that are currently included into the genus Artona. Its generic position has to be verified and has already been doubted by Holloway ( 2011, p. 24, tab. 1, fig. 6).
C13 The recently described three female specimens from China, Guangdong as a new species S. nanling Owada & Wang, 2021, have no strong differences in characters from those of the holotype of Bintha clathrata Poujade, 1886a, a species known so far only from two female specimens ( Huang & Efetov, 2021). It is difficult to judge whether the specimens described as S. nanling really belong to a species different from S. clathrata. More material is necessary to evaluate the character variability in this genus.]
C14 A number of species that have so far been treated as ArtonaWalker, 1854, and HomophylotisTurner, 1904, belong to the genus Amuria Staudinger, 1887, as they are congeneric with its type species Amuria cyclops Staudinger, 1887. The larvae of none of them feed on Poaceae, as far as the biology is known. The habitus is dominated by dark (mainly brown, purplish or bluish) colors. Some of these species are almost uniformly dark and if there is a pattern the dark colors contrast with white or light yellow markings. The typical black and yellow wing pattern of Artona s. str. (now subgenera Artona and Zeuxippa) is absent and the body is never ringed with yellow and black like in most Artona species. In the male genitalia the apex of the valva bears spiny prolongations (found also in Balataea but absent in the subgenera Artona and Zeuxippa). There are also differences in the female genitalia. In habitus, Amuria species are also similar to species of the subgenus PseudoamuriaTarmann, 2004, and species of the genus HomophylotisTurner, 1904. At the moment we have to admit that we still have a great deficit in the knowledge of many of the existing species. More careful morphological examinations are necessary to give us an idea on the variability of characters. Fresh material would be important for DNA analyses. The knowledge of the biology may also give us important further information. However, most of the known species are only available in single specimens and many of them are more than 100 years old. See also comment C16.
C15 Brachartona quadrisignata Snellen, 1903 (on p. 234) and Brachartona trisignata Snellen, 1903 (on p. 235) are female and male of the same species as can be seen from extended reared material from Java that is deposited in “Naturalis, Natural History Museum, Leiden, Netherlands”. The habitus described by Snellen for B. quadrisignata is that of the female, that described for B. trisignata that of the male. Consequently, we have to accept that these two names are synonyms. However, the name Brachartona trisignata Snellen, 1903, is much more often used in literature than Brachartona quadrisignata Snellen, 1903, especially in the literature on applied entomology (e.g. Tothill, et al. 1930). We therefore prefer to keep the name Brachartona trisignata Snellen, 1903, and synonymise Brachartona quadrisignata Snellen, 1903, syn. nov., with B. trisignata. This species is currently accommodated in the genus AmuriaStaudinger, 1887.
C16 PseudoamuriaTarmann, 2004, was described to accommodate the species Pseudoamuria uptoni Tarmann, 2004. There was a second species included in Tarmann (2004), viz. Pseudoamuria neglecta Tarmann, 2004. Both have similar habitus as HomophylotisTurner, 1904, but they lack the typical genitalia structures of Homophylotis (description see below). However, when Tarmann described this genus he had three males and one female of the type species P. uptoni Tarmann, 2004, with well- preserved genitalia but only one very bad female genitalia of the holotype of Pseudoamuria neglecta Tarmann, 2004. This specimen had been attacked by Anthrenus beetles earlier. Since then more material has become known from P. neglecta and also a well preserved female. Now we can see that the female of P. neglecta has a well-developed praebursa with a prominent crest with dentations such as we find in AmuriaStaudinger, 1887.
While examining more material from the Indo-Pacific region with the habitus of Amuria, Pseudoamuria and Homophylotis we found that there are more species involved where the praebursa lacks the characteristic crest with dentations. However, there is also more material available of species with genitalia of the “ Pseudoamuria neglecta type”. They cannot be mixed with Homophylotis as in this genus the female has a very characteristic autapomorphy, i.e. the translucent praebursa and the corpus bursae are fused to a single structure and the ductus seminalis inserts in the middle of this structure (at the place where the fusion has taken place) (see Tarmann, 2004, pp. 197-200, figs 385- 401).
We therefore find three genitalia types in the females of Amuria, Pseudoamuria and
Homophylotis:
1, praebursa well developed, strongly sclerotized, with a prominent crest that bears dentations ( Amuria-type);
2, praebursa well-developed, translucent, without a prominent crest that bears dentations ( Pseudoamuria; but not P. neglecta - with strongly sclerotized praebursa with dentations!);
3, praebursa and corpus bursae fused to a single, translucent structure ( Homophylotis-type).
Only after an examination of all the type material and the additional material that is deposited in museum collections can we decide whether it will be necessary to describe more genera to accommodate all the known species (see comment C07). This work could not be completed so far.
Moreover, it seems highly uncertain that all species can be summarized in one genus or subgenus (e.g. Pseudoamuria) where the typical praebursa of the Amuria-type is not found and which cannot be placed under Homophylotis because their genitalia characters do not agree with the characters (see above) of that genus. The reduction of the sclerotized praebursa with the characteristic crest with dentations has most probably taken place several times independently in evolution.
Therefore, for the moment, as a compromise, we accommodate all Indo-Pacific species with the habitus of Amuria, Pseudoamuria or Homophylotis with a well-developed praebursa with a prominent crest that bears dentations into the genus AmuriaStaudinger, 1887, but also all species that have this habitus and lack the praebursa with the dentate crest. All species that have praebursa and corpus bursae fused to a single structure (as described above) are accommodated under the genus HomophylotisTurner, 1904. An exception is PalmartonaTarmann, 2004, that has very special antennal and genitalic characters. This taxon is left separately and treated here as a genus.
C17Homophylotis melaleucaJordan, 1908 , is treated as Pseudoamuria melaleuca ( Jordan, 1908) in Tarmann ( 2004, p. 294, figs 403, 409) and its male genitalia is figured. However, the specimen taken for the figures has now shown to be an undescribed new species from New Guinea. Its larva lives on banana trees. (A description is postponed as a dissection of the type of Homophylotis melaleucaJordan, 1908, deposited in BMNH (London) is essential for a proper differential diagnosis!).
C18 Homophylotis aeneaJordan, 1925, lacks the characters that are described as typical for HomophylotisTurner, 1904, by Tarmann ( 2004, p. 191) based on the examination of type material. This is (in the male) the very small and translucent valva without ventral processes distally and a stout aedeagus (phallus) without any cornuti or eversible spines but with a characteristic spiny surface on the vesica. H. aenea has a female genitalia with a well-developed praebursa that contains a slightly curved spiny crest with long dentations. We therefore transfer Homophylotis aenea Jordan, 1925, to the genus AmuriaStaudinger, 1887. The new combination is: Amuria aenea ( Jordan, 1925), comb. nov.
C19 Homophylotis chalcosoma Jordan, 1926, has also a well-developed praebursa with a row of dentations in the female genitalia. This species is therefore also transferred into the genus Amuria Staudinger, 1887 (see also comment C18). The new combination is: Amuria chalcosoma (Jordan, 1926), comb. nov.
C20 Tasema nigra Hampson, 1893, Artona albicilia Hampson, 1900, Homophylotis purpurata Jordan, 1908, and Pseudoamuria neglecta Tarmann, 2004, are, for the moment, transferred to AmuriaStaudinger, 1887, based on the arguments explained under C16. The new combinations are: Amuria nigra ( Hampson, 1893), comb. nov., Amuria albicilia ( Hampson, 1900), comb. nov., Amuria purpurata (Jordan, 1908), comb. nov., Amuria neglecta ( Tarmann, 2004), comb. nov.
C21 All the remaining species that were hitherto included into Artonini and cannot be attributed to Thyrassiini, Pollanisini and Artonini (sensu stricto, see definition above) form a group that is described here as the tribe Cleleini.
Tribe Cleleini Efetov & Tarmann, tribus nov.
[Type-genus: CleleaWalker, 1854, 465]
Description. Habitus variable, wings often of “ Procris-type” sensu Alberti, 1954; foreleg with epiphysis (sometimes secondarily reduced), hindleg with single medial spur (sometimes secondarily reduced); genitalia male - valva without “ Artona-finger”, genitalia female – praebursa absent or if present without double row of pointed spines; larval host-plants: Vitaceae, Hydrangeaceae, Fabaceae, Rosaceae, Poaceae. Distribution. eastern, southern and southeastern Asia, tropical Africa.
C22 Owada (2021) published a study on the Clelea formosana complex from Taiwan and comes to the conclusion that C. formosanaconsists of the three different species: C. formosana Strand, 1915, C. simplicior Bryk, 1936, and C. kanoi Owada, 2021. In addition to habitus differences Owada found also small genitalic differences between these three taxa. All three taxa have a vicariant distribution. Based on the “concept for species and subspecies in this catalogue” (see introduction), we see these three taxa as good subspecies with geographical vicariant distribution and small genitalic differences and treat them in this catalogue as subspecies of C. formosana. C. formosana formosanaStrand, 1915, C. formosana simplicior Bryk, 1936, stat. nov. and C. formosana kanoi Owada, 2021, stat. nov.
C23 After the description of Thibetana witti Efetov, 1997c, it became clear that the two taxa Artona zebraElwes, 1890, and Artona postalba Elwes, 1890, do not belong to the genus ArtonaWalker, 1854. They belong to ThibetanaEfetov & Tarmann, 1995 ( Efetov & Tarmann, 2017b).
C24 We studied a specimen figured by Holloway, 2011, plate 1, fig. 26 as a male of “ Chrysartona pravata” and found that it is a female of Ch. hausmanni.
C25 Ch. ( Ch.) murziniMollet, 2016a, was described on the base of one male holotype. Only female holotype is known from Ch. (Ch.) efetoviParshkova, 2007, distributed in northeastern India. The possible conspecificity of these taxa could be proven on the basis of additional material. Moreover, Ch. (Ch.) hausmanniEfetov, 2006, is also known only from females.
C26 Ch. (Chrystarm.) mineti Efetov & Tarmann, 2013b, and Ch. ( Chrystarm.) antenorMollet, 2018, are very similar and both originate from Vietnam. Character variability in these two species in habitus as well as in the genitalia cannot be excluded. More material has to be examined to clear this.
C27 The genus Piarosoma was revised by Efetov & Tarmann (unpublished) and Owada et. al. (2022) simultaneously. Owada et al. (2022) finished their studies earlier and have therefore priority. However, we list here additional facts from our typescript to give the reader a more complete picture on this interesting group.
HysterosceneHering, 1925 is a junior synonym to PiarosomaHampson, 1893 ( Owada et al. 2022).
At the time of Hering (1925), Bryk (1936) and Alberti (1954) the family Zygaenidae was separated into seven subfamilies: Himantopterinae Rogenhofer, 1884 (now a separate family within the Zygaenoidea); Phaudinae Kirby, 1892 (now a separate family within the Zygaenoidea); Chalcosiinae Walker, 1865; Charideinae Butler, 1876 (now a separate subfamily within the Thyrididae); Anomoeotinae Hering, 1937 (now a separate family within the Zygaenoidea); Procridinae Boisduval, 1828; and Zygaeninae Latreille. Based on the contemporary classification, after the exclusion of the subfamily Phaudinae Kirby, 1892 (now it is the family Phaudidae) and description of a new subfamily Inouelinae Efetov & Tarmann, 2017a, the family Zygaenidae includes five subfamilies: Inouelinae Efetov & Tarmann, 2017a; Procridinae Boisduval, 1828; Chalcosiinae Walker, 1865; Callizygaeninae Alberti, 1954; and Zygaeninae Latreille, 1809 ( Efetov, 1999, 2001b; Efetov et al. 2014b; Efetov & Tarmann, 2017a). The Procridinae are represented by five tribes: Thyrassiini Efetov & Tarmann, tribus nov., Pollanisini Efetov & Tarmann, tribus nov., Artonini Tarmann, 1994, Cleleini Efetov & Tarmann, tribus nov., and Procridini Boisduval, 1828 ( Efetov, 1996, 1997a, 1997b, 1998, 2001a, 2006, 2010; Efetov et al. 2004, 2014a, 2014c, 2016; Efetov & Hayashi, 2008; Efetov & Tarmann, 1995, 2013a, 2013b, 2014a, 2014b, 2016a, 2016b, 2017b, this publication, Tarmann, 1994).
PiarosomaHampson, 1893, as well as HysterosceneHering, 1925, were arranged in Zygaeninae by Bryk (1936). However, Alberti (1954) in his world revision of Zygaenidae, transferred many genera from Zygaeninae and Chalcosiinae to Procridinae Boisduval, 1828, amongst them Piarosoma and Hysteroscene. In the meantime the subfamily Procridinae of Zygaenidae has been proved to represent a well-separated monophyletic group divided into the five tribes (see above). PiarosomaHampson, 1893 (syn. HysterosceneHering, 1925) is a Procridinae based on the fact that in the female the spermatheca (= receptaculum seminis) is not divided into a tube-like utriculus and a bulb-like lagena as in most other ditrysian Lepidoptera and all other Zygaenidae subfamilies but developed as one structure, the bursa utricularis ( Efetov & Tarmann, 2017a).
Piarosoma has a spherical head capsule and a triangular chaetosema that is placed very dorsally and is only slightly extended anteriorly and does not fill the space between the dorsal edge of the compound eye and the ocellus - it therefore should belong to the tribe Procridini. A fan-like valva in the male genitalia is slightly developed and can be seen in some male genitalia preparations but only under special illumination. The protuberances on abdominal segments 2 and 7 are absent. A single third medial spur on hind tibia is developed. Summarising all these characters we include here Piarosoma into the tribe Cleleini Efetov & Tarmann, tribus nov.
Redescription of Piarosoma
Body with blackish ground colour with a bluish tinge in fresh specimens; thorax with white orcreamy yellow patagia, forming a collar and a white or creamy yellow pattern on abdomen.
Head. Spherical, frons only slightly rounded, not proceeding dorsally; space between compound eye and ocellus without scales anteriorly but covered by a short extension of the triangular chaetosema posteriorly, labial palps short, slightly upcurved, not exceeding frons; antenna bipectinate in both sexes, pointed distally, with the length of the rami tapering towards apex. Proboscis well developed yellow or light brown.
Thorax. Patagia and tegulae well developed, patagia often coloured. Legs long, mid and hindleg almost as long as length of body. Foreleg with tibial epiphysis, hindleg with a pair of apical spurs and a single medial spur.
Wings with transparent areas, forewings one third longer than hindwing. Wing venation on forewing with R.+R. stalked together, all other veins free from cell, medial stem only developed as a groove and not as a full vein, the posterior margin of cell is formed by the basal part of vein CuA. which is then running from the posterior edge of the cell in an upcurved way to the wing border, vein CuA. is not fully developed, the basal part is completely reduced and the visible vein starts from a curved cross vein between CuA. and CuP that forms an edge where CuA. arises, CuP is fully developed and also 1A+2A; hindwing with Sc fused with the anterior part of cell for a short distance and free distally, all other veins free from cell, with three medial veins, medial stem only developed as a vein distally, CuP and the three anal veins 1A, 2A and 3A fully developed. Wing pattern very characteristic, with blackish and densely scaled parts and clear, completely scaleless, transparent “windows” that are arranged in groups on the wings and correspond in their margins with the exceptional wing venation of Piarosoma.
Abdomen without exceptional structures.
Genitalia male. Uncus strongly sclerotized, base triangular, apex short or long. Valva subquadrate or rounded, with or without process at the distal part of ventral margin (at the distal end of sacculus). Juxta triangularly rounded; transtilla prominent, consisting of a basal plate of different shape and a pair of “horns” dorsally. The translucent central part of the valva with smooth surface basally, but fan- shaped with parallel folds distally (not visible in all dissections). Aedeagus (outer sclerotized part of phallus) broad anteriorly, tapering towards the posterior end, at this distal end a heart-shaped, folded structure is present that is covered with hundreds of little spines. This structures is connected by a translucent band with the juxta. Vesica (translucent eversible part of phallus) with a long band-like structure that bears many small cornuti; if the vesica is not everted this structure appears as a serpentine-like band within the aedeagus, but if everted the vesica is a long, very narrow tube of ca. 4- times the length of the aedeagus and the spines are arranged in a long band with a single small plate of different shape at the distal end.
Genitalia female. Papillae anales small, oval, covered with short setae. Apophyses posteriores with a broad base and a short, very slender distal part that ends round and not pointed distally. 8th sternite and 8th tergite fused to a broad band-like ring. Apophyses anteriores short, developed as translucent, triangular edges. Ostium developed as a narrow sclerotized, oval ring; no sclerotized antrum developed and the translucent, narrow, folded and extremely long ductus bursae starts directly from the ostium ring and ends without forming a praebursa into the translucent, oval corpus bursae that has a large lateral oval appendix. Inner female genitalia with a receptaculum typical for Procridinae with a long ductus spiralis, a bag-shaped translucent utriculus, without a lagena (as is present in most Lepidoptera) and with a long and narrow accessory gland. The two “horns” of the glandula sebacea are long and tube-like, slightly asymmetrical, one longer and broader, the other shorter and more slender. Petersen’s gland is well visible and developed as a very small translucent organ with two narrow ducts arising from the terminal end of the oviduct and each of them ending into a globular bulb (very difficult to see due to the fragility of this structure).
Differential diagnosis. The genus PiarosomaHampson, 1893, is externally similar to PhacusaWalker, 1854. The exceptional wing venation in Piarosoma is unique. Piarosoma has a medial spur on the hind tibia that is absent in Phacusa. In Phacusa males the vesica bears between two to four cornuti, and at least two of them are hook-shaped whereas in Piarosoma many small cornuti are arranged along a very long band-like vesica seminalis. In the female genitalia Phacusa has a prominent praebursa with strongly sclerotized structures developed while in Piarosoma a praebursa is absent.
C28 Phacusa sizala Swinhoe, 1894, was removed from Piarosoma by Owada et al. (2022). However, as the systematic position of this species is unclear until the type investigation, we provisionally leave it in the genus Piarosoma.
C29 The afrotropical Zygaenidae had to be compiled without major revisional work. Based on the treatments of the group by Gaede (1926), Bryk (1936) and Alberti ( 1954, 1957), with the inclusion of some smaller contributions, e.g., Viette ( 1965, 1978), Tarmann ( 1985b, 2003), Vari, Kroon & Krüger (2002) etc. and after having undertaken only some preliminary own examinations this list has to be seen as provisional. The following examinations are responsible for basic changes compared with earlier treatments:
1. Examination of the female genitalia proved that the receptaculum seminis consists of the utriculus only and lacks a lagena. However, the tube-like form of the utriculus (at the position where the lagena is situated in non procridine zygaenids and other Lepidoptera) is always slightly or prominently broadened in Procridinae, forming a bag-like structure. This unique structure represents a basic autapomorphy of the Procridinae and is described as “bursa utricularis” ( Efetov & Tarmann, 2017a). Gaede (1926) placed the afrotropical Zygaenidae in Zygaeninae, Bryk (1936) in Chalcosiinae and Zygaeninae. Alberti (1954) treated the group as Procridinae but included the genera Saliunca Walker, 1864, SyringuraHolland, 1893, TasciaWalker, 1856, and AstyloneuraGaede, 1914, within his tribe Callizygaenini (raised to Callizygaeninae by Tarmann, 1994) that has a well-developed lagena in receptaculum seminis like all other Zygaenidae. Our examinations showed that the African Procridinae do not belong to Callizygaeninae but are true Procridinae.
2. The examination of the head capsule, abdomen, legs and genitalia show that the genera ChalconyclesJordan, 1907, and NeobalataeaAlberti, 1954, belong to the tribe Artonini Tarmann, 1994, the genera MetanyclesButler, 1876, AethioprocrisAlberti, 1954, Sthenoprocris Hampson, 1920, AnkasocrisViette, 1965, IschnusiaJordan, 1928, AlteramenelikiaAlberti, 1971, Triacanthia Romieux, 1937, XenoprocrisRomieux, 1937, MadaprocrisViette, 1978, Afromalambliagen. nov. belong to the tribe Cleleini Efetov & Tarmann, tribus nov., whereas Saliunca Walker, 1864, Syringura Holland, 1893, TasciaWalker, 1856, and AstyloneuraGaede, 1914, are here placed in Procridini Boisduval, 1828.
3. Saliuncella Jordan, 1907, with the only known species S. marshalliJordan, 1907, is extraordinary as this species is the only African taxon that is closely related to the Australian Pollanisus-Onceropyga-group. The habitus is that of a Pollanisus in male and female. The female has an abdominal hairtuft. The genitalia are close to Onceropyga. The larval host- plants are Vitaceae like in Onceropyga. S. marshalli was reared from Rhoicissus sp. (Vitaceae) by N. J. Duke in 1994 from Swasiland (South Africa) (reference specimens in BMNH) ( Fig. 45). Consequently, this species is here placed into the tribe Pollanisini Efetov & Tarmann, tribus nov.
Redescription of Saliuncella marshalli Jordan, 1907.
Body densely scaled, with dark greyish green ground colour and shiny blue scales that give the body and legs a strong bluish sheen or only a tinge of blue depending from the angle of the incoming light.
Head. Dorsoventrally compressed (like in Pollanisus and Artona), space between compound eye and ocellus covered with scales anteriorly, chaetosema triangular, anteriorly extended into the space between compound eye and ocellus but not exceeding this space. Labial palps well developed, curved upwards, slightly exceeding frons; maxillary palps very small; antenna with broad shaft, basal part in male strongly bipectinate, the last 5 segments biserrate, apex bluntly pointed, antenna in female biserrate, covered with shiny bluish scales at shaft (in fresh specimens). 28 antennal segments. Proboscis well developed, yellow. Compound eyes small, frons broader than compound eyes in frontal view. Distance between dorsal margin of compound eye and ocellus approximately as broad as diameter of ocellus.
Genitalia male. The male genitalia of Saluncella marshalli is similar to that of the eastern Australian species of Onceropyga ( Tarmann, 2004, figs 273-282), although the habitus is very different. Uncus slender, 1/3 shorter than length of valva; tegumen and vinculum very translucent, slender; pulvinus very small, with a very long bundle of hairs that is coming off easily during preparation; valva translucent, strongly fan-shaped, without sclerotization dorsally, with narrow, straight ventral sclerotisation that exceeds the length of the ventral margin of the valva like in Onceropyga anelia, but is almost as slender as that of O. pulchra; aedeagus very long and slender, tube-like, with a very narrow, needle-like, slightly twisted cornutus that has almost the length of the aedeagus.
Genitalia female. Ostium narrow, oval; ductus bursae slender, with a double spiral in the middle that is slightly sclerotised, inserting in a very translucent, bag-like corpus bursae; papillae anales weakly sclerotized, with very short setae, apophyses posteriors short and slender; apophyses anteriores also short and slender.
C30 The genus Malamblia Jordan, 1907, with its type-species M. durbanica Jordan, 1907, is removed from the African zygaenid list. Its type-species is a synonym to Procris melasGuérin-Méneville, 1839 , for which Jordan described the genus ChilioprocrisJordan, 1913. This species is restricted to Chile (South America). The error had its origin in a misreading of the original label of the type specimen by Jordan (see Tarmann, 1985b). The other species included in Malamblia by Hampson (1910), Gaede (1926) and Bryk (1936) cannot be placed into a so far described genus. We therefore describe here the new genus Afromalambliagen. nov. to accommodate these species.
Afromalamblia Efetov & Tarmann, gen. nov.
Type species: Malamblia flavipalpis Hampson, 1910, 488, here designated.
Diagnosis (based on the female holotype and one male of Malamblia flavipalpis Hampson, 1910, Brachartona unxia Druce, 1896, and Brachartona titoea Druce, 1896, and one more undescribed species from Madagascar in the collection of the Natural History Museum, London).
Head dorsoventrally flattened, frons round, protruding dorsally, maxillary palps rudimentary, invisible, labial palps porrect, proboscis yellow, compound eyes small, black, frons 1.5 times broader than compound eye in frontal view, breadth of frons equal in male and female (no sexual dimorphism in this character), ocelli small, distance from compound eye 1.5 times the diameter of ocellus, chaetosema long, oval, strongly protruding forwards (as in Pseudoamuria). Foretibia with epiphysis, mid- and hindlegs with one pair of spurs (no third medial spur present!). Frenulum consisting of only one spine in female, retinaculum between base of CuP and posterior margin of cell visible as a small aggregation of specialized long, forward pointing scales.
The genitalia of Malamblia flavipalpis was dissected by Vari (No. 617) and one wing was also prepared by Vari on a slide. For more than two years a request to photograph the genitalia and the wing preparation on the slide has been sent to London. However, due to lack of resources this could not be done so far. A description of the genitalia has therefore to be postponed.
Derivatio nominis. The name is a construction compiled from Malamblia (now a genus valid for a South American species) and the term “Africa”.
C31 A redescription of the tribe Procridinae is provided below.
Tribe ProcridiniBoisduval, 1828, 38
[Type genus: Procris [Fabricius in Illiger], 1807, 289]
Description. Head capsule spherical, not compressed dorsoventrally, chaetosema not extended between compound eye and ocellus; foreleg with or without epiphysis, hindleg without medial spur; genitalia male - valva not fan-shaped, without “ Artona-finger”; genitalia female - with or without praebursa; larval host-plants: Vitaceae, Rosaceae, Fagaceae, Polygonaceae, Asteraceae, Plumbaginaceae, Cistaceae, Geraniaceae and some others, a few species are polyphagous. Distribution. Europe, Asia, Africa, America.
C32 The genitalia of Illiberis (Alterasvenia) paradistinctaAlberti, 1954, differs significantly from those of other species of Alterasvenia, including the type-species of this subgenus, viz. Northia ulmivora Graeser, 1888. The aedeagus in the male genitalia resembles that of species of the genus HedinaAlberti, 1954, but the female genitalia of Illiberis paradistincta has no huge praebursa with rows of sclerotized spines as in Hedina. As this combination of characters is unique, we describe here a new genus Pseudohedinagen. nov. to accommodate Illiberis paradistincta (type-species of Pseudohedina by monotypy).
Pseudohedina Efetov & Tarmann, gen. nov.
Type species: Illiberis paradistincta Alberti, 1954, 246, by original desigantion and monotypy.
Diagnosis: Adult with typical Illiberis-habitus with transparent wings and well visible veins. In male genitalia valvae without any processes on the ventral margin of sacculus; aedeagus broader distally, with a group of large, pointed, strongly sclerotized cornuti (like in Hedina). Female genitalia without strongly dilated praebursa, lacking rows of sclerotized spines. Corpus bursae globular, not asymmetric (as in Hedina) and not double lobed (as in Alterasvenia).
C33 Revisional work on PhacusaWalker, 1854, by the authors has shown that the genitalia of Phacusa nicobarica Hampson, 1920, are different from those of Ph. tenebrosa ( Walker, 1854) and Ph. nicobaricais not a synonym of Ph. tenebrosa (synonymised by Efetov & Tarmann (2012)). As a consequence, Phacusa nicobarica Hampson, 1920, stat. rev. is here accepted as a good species.
C34 New material collected in European Turkey (Thrace) by Feza Can with the help of newly synthesised attractant from the series EFETOV-2 shows that Rhagades pruni ([ Denis & Schiffermüller], 1775) occurs also in Turkey ( Can Cengiz et al. 2016).
C35 In agreement with the concept of this catalogue how to treat species and subspecies (see chapter “The concept for species and subspecies in this catalogue” above) we feel obliged to change the status of some recently described taxa of ZygaenoprocrisHampson, 1900. This was not an easy decision as this genus has been extensively investigated in recent years by Keil ( 2002, 2014, 2016a, 2016b, 2016c, 2020) and most descriptions of new taxa are accompanied by biological and ecological data. Moreover, also first genetic results are available.
However, in comparison with other well-separated taxa that are treated in this catalogue as subspecies (e.g. Adscita statices statices, A. statices drenowskii, A. geryon geryon, A. geryon orientalis), some of the new taxa within Zygaenoprocris cannot be treated as separate species as they do not differ more from their nearest related taxa than in many other cases where we have accepted subspecific rank. We accept therefore here all taxa described by Keil as valid, but we group some together and treat only those taxa as species that have relevant genitalic differences (even if these differences can be small). Also considering our DNA results from the ZYGMO project in BOLD a clear picture is shown in two species, viz. Z. ( Molletia. persepolis ( Alberti, 1938) with four subspecies and Z. ( M.) duskei ( Grum-Grshimailo, 1902) with also four subspecies. In Zygaenoproris (Zygaenoprocris) the situation is more difficult as several taxa have been observed occurring syntopically (e.g. Z. ( Z.) mystrocera and Z. ( Z.) efetovi) ( Keil, 2020), in addition, these taxa have stable genitalic differences) and have to be treated as species. Moreover, the taxonomic situation regarding Z. ( Z.) khorassana in relation to Z. ( Z.) mystrocera seems not completely clear. We therefore treat the latter for the moment as an independent species and Z. ( Z.) mystrocera together with Z. ( Z.) molleti and Z. ( Z) valii as one species with three well separated subspecies.
As was mentioned above, Z. ( M.) duskei ( Grum-Grshimailo, 1902) includes four subspecies. One of these subspecies, viz. Z. ( M.) duskei kliri Keil, 2002, has been raised to species level by Keil (2023) without any comments. Here we consider this taxon as a subspecies of Z. ( M.) duskei. Moreover, Keil (2023) mentioned erroneously two more species in the genus Zygaenoprocris, viz. Zygaenoprocris statices and Z. obscura, both correctly belonging to the genus Adscita.
C36 Zygaenoprocris rjabovi ( Alberti, 1938c) and Zygaenoprocris eberti ( Alberti, 1968) were placed in the subgenus Zygaenoprocris until now. However, they have good morphological differences from Zygaenoprocris chalcochlora Hampson, 1900, the type species of this subgenus. New DNA data (Miric et al. 2023) confirmed the isolated position of Z. rjabovi and Z. eberti. It has therefore been necessary to erect a new subgenus of the genus Zygaenoprocris to accommodate these two taxa (Figure 1).
Subgenus Longiterna Efetov & Tarmann, subgen. nov. (Figure 1)
[Type-species: Procris rjaboviAlberti, 1938c, 94, here designated]
Zygaenoprocris (Longiterna) rjabovi and Zygaenoprocris (Longiterna) eberti have forewings green with strong metallic sheen, long aedeagus with single strongly sclerotized cornutus in male and extremely long ductus bursae in female ( Efetov & Tarmann, 1999, figs 109, 167, 168; Efetov et al. 2014a).
Differentional diagnosis. All remaining species of the subgenus Zygaenoprocris have the ductus bursae and the aedeagus much shorter and a weakly sclerotised cornutus. All species of the other subgenera of the genus Zygaenoprocris have strongly different genitalia structures ( Efetov & Tarmann, 1999).
Etymology. The word “Longiterna” consists of two parts: “Longi” shows that these species have very long ductus bursae in female genitalia; the ending “terna” has been used already by B. Alberti to create a subgeneric name Lucasiterna (later the same principle was used for Procriterna Efetov & Tarmann and Solaniterna Efetov).
C37 Adscita mauretanica ( Naufock, 1932) is the only Adscita species in northwestern Africa. Although this species is in habitus, antennal structures and biology a clear Adscita, its genitalia are strongly different from all Adscita known so far. Moreover, in the COI gene (barcoding tree) this species is far away from all other Adscita species. Its nearest neighbour Adscita bolivari shows ca. 7% genetic difference. All these characters show the unique position of A. mauretanica in Adscita. We therefore describe a new subgenus for this isolated species. Moreover, new genetic studies including nuclear genes confirm the position of A. mauretanica between Adscita and Jordanita (Miric et al. 2023).
Subgenus Afroterna Efetov & Tarmann, subgen. nov. ( Figure 2)
Type-species: Procris mauretanicaNaufock, 1932, 77, here designated.
Diagnosis. Males and females are externally very similar to Adscita statices, the type species of the genus AdscitaRetzius, 1783, with shiny green body and forewing upperside and with clubbed antennae. Antenna with pectinations in male, the last segments fused to plates; female without antennal pectinations. Male genitalia. Uncus slender, as long as tegumen. Valva long and slender, ventral margin of sacculus without process, apex of sacculus rounded. Phallus short and broad, ca. 3.5 times longer than broad, vesica with two strongly sclerotized, curved cornuti. Female genitalia. Caudal part of ductus bursae strongly dilated, forming a spherical or ovoid praebursa that has a ventral pocket-like evagination and bears sclerotized dentations ( Efetov & Tarmann, 1999, p. 108, fig. 95, p. 123, fig. 157, Efetov, 2001f, p 161, plate 9, fig. 12, p. 209, plate 32, fig. 12).
The morphology of the early instars and their biology is not strongly different from those of other Adscita species. The larval host-plants are Polygonaceae and Cistaceae.
Etymology. The word “Afroterna” consists of two parts: “Afro” shows that this species is only found in Africa (North African distribution); for meaning of “terna” see above (comment C36).
C38 According to morphological, biological and molecular characters the subgenus Adscita Retzius, 1783, is a paraphyletic subgenus clearly consisting of three monophyletic units, viz. ( jordani-group + statices-group), ( obscura-group) and ( geryon-group + albanica-group). In this catalogue we are describing two new subgenera with the aim to have three monophyletic subgenera.
Subgenus Obscuriterna Efetov & Tarmann, subgen. nov. (C38) ( Figure 2)
[Type-species: Procris obscuraZeller, 1847a, 15, here designated]
Adscita (Obscuriterna) obscura has an isolated position in the genus. It has extraordinary genitalia structures and larvae feeding on at least three plant families. The characters of the new subgenus are an extremely long aedeagus (ratio of length to breadth is approximately 20) and also extremely long ductus bursae (ratio of length to breadth more than 60) ( Efetov & Tarmann, 1999, figs 94, 156; Efetov, 2001f, plates 9, 33), the larvae feed on Cistaceae, Rosaceae and Fabaceae (and not on Polygonaceae).
Differentional diagnosis. All remaining species of the subgenus Adscita are oligophagous and have much shorter aedeagus and ductus bursae (Efetov & Tarmann, 1999, figs. 89-93. 95-107, 151-155, 157-165; Efetov, 2001f, plates 3-12, 28-32, 34-36).
Etymology. The word “Obscuriterna” consists of two parts: “Obscuri” shows that this subgenus is monotypic with one species A. obscura. For meaning of “terna” see above (comment C36).
C39
Subgenus Eurasiterna subgen. nov. ( Figure 2)
[Type-species: Adscita dujardini Efetov & Tarmann, 2014b, 182, here designated]
Included species. Adscita (Eurasiterna) capitalis( Staudinger, 1879), A. ( Eur.. geryon (Hübner, 1813), A. (Eur.) dujardini Efetov & Tarmann, 2014b, and A. (Eur.) albanica ( Naufock, 1926).
All four species form one branch on a DNA-tree and have a much smaller ratio of length to breadth of aedeagus (less than 15) and also much smaller ratio of length to breadth of ductus bursae (less than 15) than in A. (O). obscura. Larval host-plants belong to Geraniaceae and/or Cistaceae. Species of this subgenus form two groups, viz. geryon-group (ductus bursae tubular, without praebursa) and albanica-group (ductus bursae strongly delated formig a praebursa) ( Efetov & Tarmann, 1999).
Differentional diagnosis. All remaining species of the subgenus Adscita have another combination of characters. The larvae of the representatives of the subgenus Adscita are feeding on Polygonaceae ( Rumex species). For characters of Afroterna and Obscuriterna see above.
Etymology. The word “Eurasiterna” consists of two parts: “Eurasi” shows western Palaearctic distribution of all four included species (Europe and western Asia); for meaning of “terna” see above (comment C36).
C40 Different populations of Jordanita (Roccia) paupera ( Christoph, 1887) (sometimes strongly geographically isolated from one another) can have DNA barcode distances. However, as there are no constant genitalic differences between these populations, we consider them as one species. Recently, Zolotuhin (2020) described two new taxa: Jordanita (Roccia) scintillosaZolotuhin, 2020, and J. ( R.) smaragdonna Zolotuhin, 2020. These two taxa are considered to be synonyms of Jordanita (Roccia) paupera ( Christoph, 1887) ( Efetov et al. 2022; Efetov & Tarmann, 2022).
C41 As shown in Efetov et al, (2019)Ino budensis var. mollis Grum-Grshimailo, 1893, described from China, and so far treated as a synonym of Jordanita (Roccia) paupera ( Christoph, 1887) ( Efetov & Tarmann, 2012), is a good species: Jordanita (Roccia) mollis ( Grum-Grshimailo, 1893). This opinion is based on the presence of stable differences in the male genitalia and a significant difference in the DNA barcodes. This species is recorded for eastern Russia (Southern Siberia and Far East), China and Korea.
C42 Jordanita (Praviela) anatolica ( Naufock, 1929) and Jordanita (Praviela) christinae Keil, 1998, are treated as two different species in Keil (2014). In Efetov & Tarmann (2012, p. 36)J. anatolica christinae is treated as a subspecies. The authors always had strong doubts that these two taxa are really different species. The reason for these doubts was the well-known variability in the male genitalia of J. anatolica ( Efetov & Can, 2010) where combinations and forms of cornuti could be found in various populations with the characters of those described and figured by Keil ( 1998, p. 113, fig. 1b) for J. christinae. Moreover, specimens with the habitus of J. christinae (reddish wings and body) could also be found as aberrations within other Iranian populations of J. anatolica. In his original description even Keil himself had doubts and described therefore the new taxon only as a subspecies of J. anatolica ( Keil, 1998). However, Keil was secretly always convinced that the two taxa are different species and continued searching for arguments. He visited Iran and the type locality of J. christinae many times. Finally he found both colour forms syntopic in the type locality of J. christinae with a significant difference in the time of flight activity. Specimens with the characters of J. anatolica(green wings and body) were always earlier on the wing in the year than those with the characters of J. christinae. Keil reared both taxa from egg to imago and described their biology ( Keil, 2014, pp. 140-147). In addition to the syntopic occurrence he observed oviposition of females of both taxa on different larval host- plants at the same locality. The larvae are also slightly different in coloration. The latter character was also known by the authors for several years but color variation in larvae of Procridinae are common and also this character did not convince us that both taxa are really good species. Keil (2014, pp. 140-147), in his work on the Zygaenidae of Iran, again summarizes all arguments and publishes figures on the biologies of J. anatolica and J. christinae.
Based on these data but also on our genetic data from our ZYGMO project in BOLD (the genetic distance between J. christinae and J. anatolica is larger than between all other J. anatolica populations that are so far examined) we now accept the specific status of J. christinae. However, specimens with intermediate characters in habitus and genitalia have been found around the type-locality of J. christinae in the Zagros mountains. This is a clear sign that both species are very closely related.
Jordanita (Praviela) rietzschiKeil, 2016, can be only a colour form of J.(P). anatolica because these two “taxa” have no genitalic differences.
Further molecular and biologal investigations are necessary in the subgenus PravielaAlberti, 1954, and it also would be very important to find newer material of J. ( P.) anatolica kruegeri ( Turati, 1930) from North Africa which we here treat as a subspecies to J. anatolica based on its genitalic characters.
C43 In Tarmann (1984, p. 36) the date behind the author of Pyromorpha .Gingla. semifulva (Druce, 1896) is wrongly cited as “Druce, 1884”. This error was discovered by Steve Nanc (New York, USA) in December 2016. It was an error by G. M. Tarmann in 1984 when he mixed Dycladia semifulvaDruce, 1884 (now Cosmosoma semifulva (Druce, 1884), Erebidae, Ctenuchinae) with Lycomorpha semifulva Druce, 1896 (Zygaenidae, Procridinae). It would not make sense to change now also the synonymy between Lycomorpha semifulva Druce, 1896: 332, and Lycomorpha basirei Druce, 1896: 331, just because L. basirei was described one page earlier in the same paper. According to the principle of the first revising author we leave Pyromorpha .Gingla. semifulva ( Druce, 1896) as valid species and L. basirei Druce, 1896 as its synonym.
C44 In Tarmann (1984, p 40)Harrisina mexicana Schaus, 1889, was synonymized with Aglaope coracina Clemens, 1861, due to almost no genitalic differences. More material has now shown that this was wrong and both taxa represent different spercies. H. mexicana is smaller and has longer pectination in the male antenna. The male genitalia are, as in many Harrisina, not significantly different. This species was described from one male from Mexico (Veracruz, Paso de San Juan). Females can only be attached to this species if they are reared and if also males from the same rearing are available. This has not been done so far. Sufficient DNA data are not available so far.
Names which are accepted as valid in this Catalogue are printed in bold.








For providing type specimens or help in the examination of material, help with literature, photos and for fruitful discussions we are indebted to our colleagues Dr. S. Gaal (Austria), Prof. Dr V. V. Anikin (Russia), Dr S. Beshkov (Bulgaria), Mr L. V. Bolshakov (Russia), Dr M. J. W. Cock (Great Britain), Dr J. De Pins (Belgium), Mr J.-M. Desse (France), Mr U. Drechsel (Paraguay), Mr E. Drouet (France), Dr O. G. Gorbunov (Russia), Dr A. Hausmann (Germany), Mrs E. Hayashi (Japan), Prof. Dr P. D. N. Hebert (Canada), Mr A. Hofmann (Germany), Mr M. R. Honey (Great Britain), Dr K. Horie (Japan), the late Prof. Dr H. Inoue (Japan), Prof. Dr P. Jaksic (Serbia), Mr Th. Keil (Germany), Dr M. A. Klepikov (Russia), Mr J. Klír (Czech Republic), Mr S. A. Knyazev (Russia), Dr C. Koshio (Japan), Mr I. Yu. Kostjuk, Dr A. V. Krupitsky (Russia), Mr A. V. Kuprijanov (Russia), Dr M. Lödl (Austria), Prof. Dr V. A. Lukhtanov (Russia), Dr A. L. Lvovsky (Russia), Dr W. Mey (Germany), Prof. Dr J. Minet (France), Mr B. Mollet (France), Mrs A. Nahirnic-Beshkova (Serbia), Dr M. Nakamura (Japan), Mr Steve Nanc (USA), the late Prof. Dr C. M. Naumann (Germany), Dr M. Owada (Japan), Dr I. G. Pljushtch (Ukraine), Dr A. S. Prosvirov (Russia), Ms S. Renneberg (Australia), Dr R. Rougerie (France), Prof. Dr S. A. Sachkov (Russia), Mr V. V. Savchuk (Russia), Dr V. I. Shchurov (Russia), Dr S. Yu. Sinev (Russia), Dr D. Stüning (Germany), the late Prof. Dr M. A. Subchev (Bulgaria), the late Dr A. V. Sviridov (Russia), Dr T. B. Toshova (Bulgaria), Dr J. Van Den Heuvel (Belgium), the late Dr Th. Witt (Germany), Dr B. Würsten (Belgium), Prof. Dr F.-S.Xue (China), Prof. Dr S.-H. Yen (Taiwan), the late Prof. Dr V. V. Zolotuhin (Russia), Dr A. Zwick (Australia). For technical editing of the manuscript we are indebted to Dr E. E. Kucherenko, Dr Z. S. Lazareva and Dr E. V. Parshkova (all Crimea). Last but not least we acknowledge the late Dr W. G. Tremewan (Great Britain) and Dr A. Spalding (Great Britain) for editing a part of the original typescript and for correcting the English.
* Autor para la correspondencia / Corresponding author shysh1981@mail.ru




















