Artículos
Gelechiidae of the Canary Islands (Spain). Part 1. Anacampsinae (Insecta: Lepidoptera)
Gelechiidae de las Islas Canarias (España). Parte 1. Anacampsinae (Insecta: Lepidoptera)
Gelechiidae of the Canary Islands (Spain). Part 1. Anacampsinae (Insecta: Lepidoptera)
Shilap Revista de Lepidopterología, vol. 53, no. 209, pp. 5-41, 2025
Sociedad Hispano-Luso-Americana de Lepidopterología
Received: 30 December 2024
Accepted: 30 December 2024
Abstract: The Gelechiidae subfamily Anacampsinae in the Canary Islands is revised. We recognize 20 species and one subspecies of Anacampsini and three species of Chelariini. Seven species and two subspecies are described as new: Stomopteryx palmella Falck & Karsholt, sp. nov. (Spain: La Palma), Stomopteryx cuestaella Falck & Karsholt, sp. nov. (Spain: Gran Canaria and Tenerife), Stomopteryx minimella Falck & Karsholt, sp. nov. (Spain: Gran Canaria), Stomopteryx variabilis Falck & Karsholt, sp. nov. (Spain: Gran Canaria, Tenerife, La Gomera, La Palma and El Hierro), Aproaerema ayacatella Falck & Karsholt, sp. nov. (Spain: Gran Canaria), Aproaerema pseudomercedella Falck & Karsholt, sp. nov. (Spain: La Gomera and El Hierro), Aproaerema angustumella Falck & Karsholt, sp. nov. (Spain: Tenerife and La Palma), Stomopteryx schizogynae grancanariella Falck & Karsholt, ssp. nov. (Spain: Gran Canaria) and Nothris congressariella tenerifensis Falck & Karsholt, ssp. nov. (Spain: Tenerife). The status of Gelechia elachistella (Stainton, 1859) is changed into a synonym of the nominotypical subspecies of Aproaerema anthyllidella (Hübner, [1813]), stat. nov. The use of subspecies in the Gelechiidae is discussed. Mesophleps silacella (Hübner, 1796), Mesophleps ochracella (Turati, 1926) and Anarsia lineatella Zeller, 1839 are recorded as new to the Canary Islands. We also present several records of species new to single Canary Islands. The paper is illustrated with photographs of adults and genitalia of the new species as well as of other species when relevant. Analyses of DNA barcodes show that the identifications and distinctiveness of each species as well-supported and genetically isolated.
Keywords: Insecta, Lepidoptera, Anacampsini, Chelariini, new species, new records, subspecies, endemic, DNA barcodes, Canary Islands, Spain.
Resumen: Se revisa la subfamilia Gelechiidae Anacampsinae en las Islas Canarias. Se reconocen 20 especies y una subespecie de Anacampsini y tres especies de Chelariini. Siete especies y dos subespecies se describen como nuevas: Stomopteryx palmella Falck & Karsholt, sp. nov. (España: La Palma), Stomopteryx cuestaella Falck & Karsholt, sp. nov. (España: Gran Canaria y Tenerife), Stomopteryx minimella Falck & Karsholt, sp. nov. (España: Gran Canaria), Stomopteryx variabilis Falck & Karsholt, sp. nov. (España: Gran Canaria, Tenerife, La Gomera, La Palma y El Hierro), Aproaerema ayacatella Falck & Karsholt, sp. nov. (España: Gran Canaria), Aproaerema pseudomercedella Falck & Karsholt, sp. nov. (España: La Gomera y El Hierro), Aproaerema angustumella Falck & Karsholt, sp. nov. (España: Tenerife y La Palma), Stomopteryx schizogynae grancanariella Falck & Karsholt, ssp. nov. (España: Gran Canaria) y Nothris congressariella tenerifensis Falck & Karsholt, ssp. nov. (España: Tenerife). El estatus de Gelechia elachistella (Stainton, 1859) pasa a ser sinónimo de la subespecie nominotípica de Aproaerema anthyllidella (Hübner, [1813]), stat. nov. Se discute el uso de subespecies en los Gelechiidae. Mesophleps silacella (Hübner, 1796), Mesophleps ochracella (Turati, 1926) y Anarsia lineatella Zeller, 1839 se registran como nuevas para las Islas Canarias. También se presentan varios registros de especies nuevas para las Islas Canarias. El artículo está ilustrado con fotografías de adultos y genitalia de las nuevas especies, así como de otras especies cuando es pertinente. Los análisis de los códigos de barras de ADN muestran que las identificaciones y los caracteres distintivos de cada especie están bien fundamentados y aislados genéticamente.
Palabras clave: Insecta, Lepidoptera, Anacampsini, Chelariini, nuevas especies, nuevos registros, subespecies, endemismo, códigos de barras de ADN, Islas Canarias, España.
Introduction
Gelechiidae is a diverse family of very small to medium sized Microlepidoptera comprising worldwide about 5000 described species in about 500 genera, and with numerous species - probably at least equally as many - still awaiting description. They are distributed in all continents except Antarctica.
Whereas there is general agreement about which taxa belong to the Gelechiidae, the higher-level taxonomy of the family has undergone many changes, but a review of these is outside the scope of the present paper. A genetic study by Karsholt et al. (2013) has won general acceptance, although subsequent studies (e.g. Sohn et al. 2016; Wang & Li, 2020), based on more restricted taxon samplings, have suggested alternative relationships between the subfamilies. One can probably expect further changes in the higher-level taxonomy of the Gelechiidae when more southern hemisphere genera are analysed.
This is the first part of a planned series of papers on the Gelechiidae of the Canary Islands (Spain). It follows Karsholt et al. (2013) for the higher systematics and the European checklist of Gelechiidae (Huemer & Karsholt, 2020) for the sequence of genera and species. We here deal with the subfamily Anacampsinae, which is a medium-sized group of about 60 genera falling into two almost equal-sized tribes: Anacampsini and Chelariini, both of which are distributed over a larger part of the world. In the Canary Islands the former is by far the largest and especially the genera Stomopteryx Heinemann, 1870 and Aproaerema Durrant, 1897 have diversified into a number of endemic species, most of which we describe below.
The first three species of Gelechiidae from the Canary Islands were reported by Rebel (1892). In 1908 Walsingham recorded 28 species of Gelechiidae (a few of them now synonyms) mostly from Tenerife, including descriptions of several new species, mainly based on his own field work. Walsingham (1908) also gave host-plant records and other biological details for some of the species, and presented small, but accurate paintings of several of the species. During most of the 20th century the number of Gelechiidae grew very slowly, until 1984 when Klimesch publish a paper devoted to the Gelechiidae of the Canary Islands. This paper (Klimesch, 1984) listed 49 species (four of them identified only to genus) and had illustrations of the adults and genitalia of a number of species, especially those that are endemic species. During the present century some checklists have been published, enumerating the Gelechiidae of the Canary Islands. Baéz & Martín (2001) listed 53 species, Baéz & Martín (2004) 54 species, and Baéz & Oromi (2010) 57 species of Gelechiidae. The comprehensive catalogue by Vives Moreno (2014) listed also 57 species, and in the online source Fauna Europaea (Karsholt, 2004-2024), with the last update of Gelechiidae in 2011, 63 species of Gelechiidae are listed from the Canary Islands.
Material and methods
Most of the specimens were collected by the first author and attracted to an 8-watt super actinic light, and some were caught during the day. Specimens from other collectors were mainly attracted to stronger light sources.
Male and female genitalia were dissected and prepared using standard methods (Huemer & Karsholt, 2010). However, male genitalia of Anacampsinae are formed differently from those of Gelechiinae, and therefore the unrolling technique described by, e.g., Pitkin (1986) does not always give the best result for showing differences between closely related species. Instead, we have arranged the male genitalia according to how we can best show diagnostic characters.
For all genera and species synonyms are only listed if they have been used in literature dealing with Gelechiidae of the Canary Islands. Additional synonyms are listed by Vives Moreno (2014) and Huemer & Karsholt (2020).
Details of all examined specimens are listed for newly described species and subspecies. For other species only specimens used for DNA barcoding or for genitalia dissections are listed.
Adults were photographed with a Canon EOS 700D camera equipped with a Canon EF 100 mm objective. The genitalia slides were photographed using a Soptop CX40T Trinocular microscope in conjunction with a Toup Tek P10500AE3 / E3ISPM05000KPA-E3 / 5.0MP USB3 camera.
We examined the morphology of all species and the DNA barcodes from new and cryptic species. DNA samples were prepared as described by Falck & Karsholt (2023: 271). Details of successfully sequenced voucher specimens are publicy available through the dataset DS-ANACAMP at https://www.boldsystems.org. and at https://doi.org/10.5883/DS-ANACAMP.
Plant names are according to World Flora Online (2024).
Abbreviations used
AW Collection of Andreas Werno, Nunkirchen, Germany
MB Collection of Marcos Báez, La Laguna, Tenerife, Spain
PF Collection of Per Falck, Neksø, Denmark
MNCN Collection of Antonio Vives, Museo Nacional de Ciencias Naturales, Madrid, Spain
MZH Finnish Museum of Natural History, Helsinki, Finland
RMNH Naturalis Biodiversity Center, Leiden, The Netherlands
TL Type locality
WS Collection of Willibald Schmitz, Bergisch Gladbach, Germany
ZMUC Zoological Museum, Natural History Museum of Denmark, Copenhagen, Denmark
Checklist
Only synonyms used in literature on Gelechiidae of the Canary Islands are included. For additional synonyms see Vives Moreno (2014).
GELECHIIDAE ANACAMPSINAE ANACAMPSINI
Stomopteryx detersella (Zeller, 1847)
Stomopteryx remissella (Zeller, 1847)
Stomopteryx schizogynae schizogynae (Walsingham, 1908)
Stomopteryx schizogynae grancanariella Falck & Karsholt, ssp. nov.
Stomopteryx palmella Falck & Karsholt, sp. nov.
Stomopteryx cuestaella Falck & Karsholt, sp. nov.
Stomopteryx minimella Falck & Karsholt, sp. nov.
Stomopteryx variabilis, Falck & Karsholt, sp. nov.
Aproaerema polychromella (Rebel, 1902)
Aproaerema genistae Walsingham, 1908
adenocarpella Rebel, 1927
Aproaerema thaumalea Walsingham, 1905
Aproaerema anthyllidella (Hübner, [1813])
elachistella (Stainton, 1859), stat. nov.
psoralella (Millière, 1865)
infestella (Rebel, 1896)
Aproaerema mercedella Walsingham, 1908
Aproaerema ayacatella Falck & Karsholt, sp. nov.
Aproaerema pseudomercedella Falck & Karsholt, sp. nov.
Aproaerema angustumella Falck & Karsholt, sp. nov.
Mesophleps corsicella Herrich-Schäffer, [1856]
Mesophleps silacella (Hübner, 1796)
Mesophleps oxycedrella (Millière, 1871)
Mesophleps trinotella Herrich-Schäffer, [1856]
Mesophleps ochracella (Turati, 1926)
CHELARIINI
Nothris congressariella tenerifensis Falck & Karsholt, ssp. nov.
Anarsia lineatella Zeller, 1839
Anarsia acaciae Walsingham, 1896
Results
Stomopteryx Heinemann, 1870
Stomopteryx Heinemann, 1870. Schmett. Dtl. Schweiz, (2)2(1), 324
A genus with more than 60 described, and many undescribed, species, distributed in Africa and Eurasia. Karsholt & Šumpich (2018, p. 237) gave a list of North African species; none of which occurs in the Canary Islands. The adults are very variable in size and colouration, but they have rather similar genitalia. The best diagnostic characters are found in the phallus. In order to examine it, it is necessary to cut it free from the very setose juxta lobes and place it laterally in the genitalia slide.
The larvae live inside stems and roots of various plants. In the Canary Islands only the hostplant of S. schizogynae is known.
Stomopteryx detersella (Zeller, 1847)
Gelechia detersella Zeller, 1847. Isis von Oken, 1847, 846
Diagnosis: A rather large species with cream-white forewings having more or less distinct brown veins and some brown patches, especially at base, and distinct black spots in fold and at end of cell. The labial palps are upcurved, long and slender. It is not similar to other Gelechiidae in the Canary Islands (Spain). The genitalia resemble those of the other Stomopteryx species.
Biology: The biology is imperfectly known, and the larva has not been found in the Canary Islands (Spain). From continental Europe it has been reported to feed in the leafstalk of Eryngium campestre L. The few adults are recorded from February to April, at low altitudes.
Distribution in the Canary Islands (Spain): First recorded by Arenberger (1999, p. 13) from Fuerteventura. New island record. Lanzarote: Puerto del Carmen, 3-II-1994, leg. J. P. Baungaard (ZMUC).
General distribution: Widely distributed in the Mediterranean countries and southern part of central Europe to Turkey.
Remarks: S. detersella is very variable both in size and forewing markings. We have examined specimens from continental Europe with wingspans from 13 to 25 mm, and some specimens have the forewings plain cream-white.
Stomopteryx remissella (Zeller, 1847)
Gelechia remissella Zeller, 1847. Isis von Oken, 1847, 854
Aristotelia remissella rufobasella Rebel, 1916. Verh. zool.-bot. Ges. Wien, 66, (10)
Diagnosis: A rather small species with brown head, thorax and basal part of forewing, middle and distal part blackish grey with a couple of black dots and whitish costal and tornal spots. Should not be confused with other gelechiids in the Canary Islands.
DNA barcode: See Remarks below.
Biology: The early stages are unknown, and even the host plant is uncertain.
Distribution in the Canary Islands (Spain): Only recorded from a single specimen from Tenerife, Güímar, 3-X-1966, leg. J. Klimesch (Klimesch, 1984, p. 161).
General distribution: Widely distributed in Europe, especially in the Mediterranean countries and Balkans, northwards to Sweden and eastwards to Russia (Karsholt et al. 2023, p. 103).
Remarks: S. remissella seems to represent a species complex. Specimens from continental Europe are variable in size and forewing colour and markings, and also their DNA barcode shows a high degree of variation, falling into eight different BINs (Huemer & Karsholt, 2020, p. 120). We have not had the opportunity to examine the single specimen collected in Tenerife.
Stomopteryx schizogynae schizogynae (Walsingham, 1908) (Figures 1, 41, 41a, 41b, 54, 54a)

Figures 1-8.
1.Stomopteryx schizogynae schizogynae (Walsingham, 1908), ♂, Tenerife, 13 mm. 2.Stomopteryx schizogynae ssp. grancanariella Falck & Karsholt, ssp. nov., ♂, Gran Canaria, 13 mm. 3.Stomopteryx palmella Falck & Karsholt, sp. nov., ♂, La Palma, 11.5 mm. 4.Stomopteryx palmella Falck & Karsholt, sp. nov., ♀, La Palma, 12 mm. 5. Stomopteryx cuestaella Falck & Karsholt, sp. nov., ♂, Gran Canaria, 14.5 mm. 6.Stomopteryx minimella Falck & Karsholt, sp. nov., ♂, Gran Canaria, 8 mm. 7.Stomopteryx variabilis Falck & Karsholt, sp. nov., ♀, Gran Canaria, 15 mm. 8.Stomopteryx variabilis Falck & Karsholt, sp. nov., ♀, Tenerife, 15 mm.
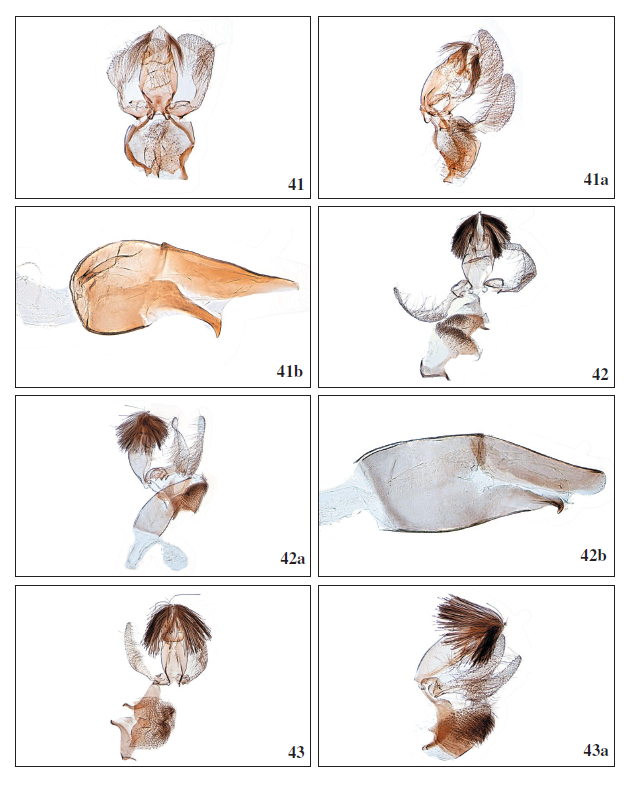
Figures 41-43a.
41.Stomopteryx schizogynae schizogynae (Walsingham, 1908), ♂, Tenerife, GP4016PF. 41a.Stomopteryx schizogynae schizogynae (Walsingham, 1908), ♂, lateral view, GP4015PF. 41b.Stomopteryx schizogynae schizogynae (Walsingham, 1908), ♂, phallus, GP4016PF. 42.Stomopteryx palmella Falck & Karsholt sp. nov., ♂, La Palma, GP4058GP. 42a. Stomopteryx palmella Falck & Karsholt sp. nov., ♂, lateral view, GP4042GP. 42b.Stomopteryx palmella Falck & Karsholt sp. nov., ♂, phallus, GP4058GP. 43.Stomopteryx cuestaella Falck & Karsholt, sp. nov., ♂, Gran Canaria, GP3936PF. 43a. Stomopteryx cuestaella Falck & Karsholt, sp. nov., ♂, lateral view, GP4020PF.
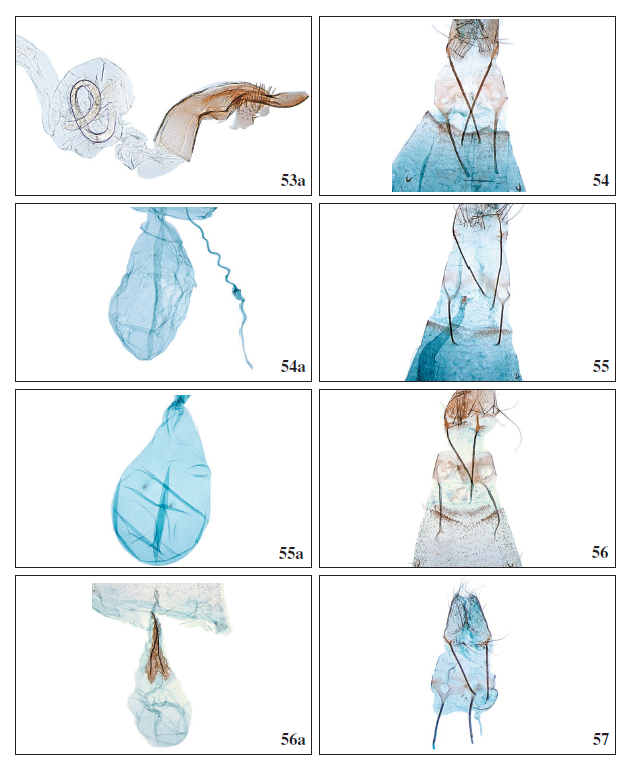
Figures 53a-57.
53a.Anarsia acaciaeWalsingham, 1896, ♂, phallus, GP3956PF. 54.Stomopteryx schizogynaeschizogynae (Walsingham, 1908), ♀, antrum complex, Tenerife, GP4017PF. 54a.Stomopteryx schizogynae schizogynae (Walsingham, 1908), ♀, corpus bursae, GP4017PF. 55.Stomopteryx palmella Falck & Karsholt sp. nov., ♀, antrum complex, La Palma, GP4006GP. 55a.Stomopteryx palmella Falck & Karsholt sp. nov., ♀, corpus bursae, GP4009GP. 56.Stomopteryx cuestaella Falck & Karsholt, sp. nov., ♀, antrum complex, Tenerife, GP4023PF. 56a.Stomopteryx cuestaella Falck & Karsholt, sp. nov., ♀, corpus bursae, GP4023PF. 57.Stomopteryx minimella Falck & Karsholt, sp. nov., ♀, antrum complex, Gran Canaria, ZM4081PF.
Telphusa schizogynae Walsingham, 1908, Proc. zool. Soc. Lond., 1907, 936
Material examined: SPAIN, TENERIFE, Faro de Rasca, 1 ♂, 17-IV-1998, leg. K. Larsen (ZMUC); Armeñime, 50 m, 1 ♂, 1 ♀, 25-XI-2-XII-2012, leg. P. Falck, genitalia slide 4014PF (PF); El Médano, 20 m, 4 ♂, 1 ♀, 18-XI-8-XII-2018, leg. P. Falck, genitalia slide 4013PF, 4016PF, 4017PF, DNA samples Lepid Phyl 0837PF/CILEP836-21, 0838PF/CILEP837-21, 1428PF/CILEP1427-24, 1429PF/CILEP1428-24 (PF); Puertito de Güímar, 20 m, 3 ♂, 18-XI-8-XII-2018, leg. P. Falck, genitalia slide 4015PF, 4034PF (PF).
Diagnosis: Stomopteryx schizogynae is a very characteristic species because of its colour and wing pattern. It resembles no other known Stomopteryx species. In the male genitalia the abruptly narrowing, bent and pointed lateral projection of the phallus is characteristic. In the female genitalia the lack of signa is characteristic.
Description Adult (Figure 1): Wingspan 12-15 mm. Labial palp slender, strongly upturned; segment 2 white, with outer surface basally dark grey; segment 3 longer than segment 2, white, lower surface with a black streak, upper surface blackish. Antenna blackish with few scattered white scales, from scapus to about 1/3 a posteriorly white stripe. Head, neck, thorax and tegula as forewing. Forewing whitish grey with a reddish brown tinge, becoming darker grey in apical third; two distinct black plical spots before two distinct black discal spots, one in the middle and one at the end of cell; costa black basally, before apex to tornus a whitish diffuse post-median fascia; fringe grey mottled with few dark grey scales. Hindwing grey; fringe grey.
Variation: The inner plical spot sometimes forming an outwardly oblique streak.
Male genitalia (Figures 41, 41a, 41b): Uncus short, triangular, covered with long setae reaching middle of tegumen. Gnathos laterally broadly rounded, flattened anteriorly. Tegumen elongate, elliptical. Valva narrow, before middle angled and broadened, apex rounded. Vinculum subtriangular with two flaps densely covered with setae covering the phallus. Phallus almost straight, broad and rounded basally, posterior half narrowing towards rounded apex, lateral projection abruptly narrowing, straight, apex bent and pointed.
Female genitalia (Figures 54, 54a): Papillae anales rounded. Posterior apophysis slender, about 1.5 times longer than anterior apophysis. Ostium bursae broadly rounded, weakly sclerotized. Antrum semi-rectangular, rounded anteriorly. Ductus bursae membranous, narrow and relatively short, about the length of segment VII. Corpus bursae oval without signa.
DNA barcodes (Figure 66): We obtained DNA barcode fragments of 500 bp and 473 bp. The intraspecific p-distance is 0%. The barcodes fall within Barcode Index Number (BIN) BOLD: AEI3051. The minimum p-distance between the populations (Tenerife vs. Gran Canaria) is 1.32 %. The minimum p-distance to nearest neighbour, an unnamed Gelechiidae species, is 5.67%.
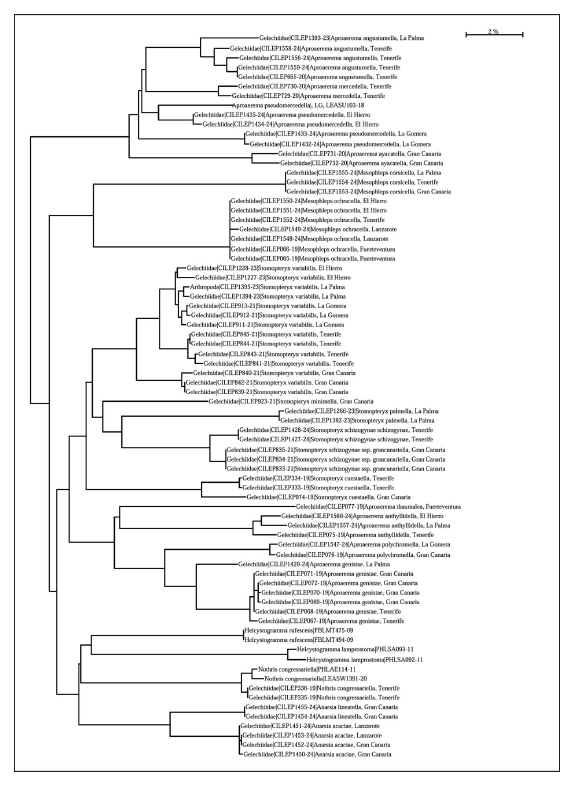
Figure 66.
Neighbor-joining tree of DNA barcodes of twenty species of Anacampsinae.
Biology: The larva lives in galls on stems of Schizogyne sericea (L. f.) DC. (Walsingham, 1908, p. 936). The adults were collected at light from the middle of November to the middle of April in coastal areas at low altitudes.
Distribution: Known only from a few coastal locations on the island of Tenerife (Spain). The nominotypical subspecies of S. schizogynae is probably endemic to Tenerife.
Stomopteryx schizogynae grancanariella Falck & Karsholt, ssp. nov. (Figure 2)
Holotype ♂: SPAIN, GRAN CANARIAValle de Agaete, 200 m, 1 ♂, 21-III-1979, leg. P. Stadel Nielsen (ZMUC).
Paratypes: SPAIN, GRAN CANARIA, Puerto Rico, 50 m, 2 ♂, 17-30-IX-2018, leg. P. Falck, genitalia slide 4018PF, DNA sample Lepid Phyl 0834PF/CILEP833-21 (PF); 8 km N Pie de la Cuesta, 895 m, 4 ♂, 17-30-IX-2018, leg. P. Falck, genitalia slide 4019PF, 4041PF, DNA samples Lepid Phyl 0835PF/CILEP834-21, 0836PF/CILEP835-21 (PF).
Diagnosis: Stomopteryx schizogynae grancanariella ssp. nov. differs from the nominotypical subspecies in the reduced markings of the forewing, having only a distinct black outwardly oblique streak from near base of dorsum to the cell, while the distinct black plical spots before two distinct black discal spots are missing or only faintly indicated. The genitalia do not differ between these two subspecies.
Description Adult (Figure 2): Wingspan 11.5-15.5 mm. Labial palp slender, strongly upturned; segment 2 white, with outer surface basally dark grey; segment 3 longer than segment 2, white, lower surface with a black streak, upper surface blackish. Antenna blackish with few scattered white scales, from scapus to about 1/3 an anterior white stripe. Head, neck, thorax and tegula as forewing. Forewing whitish grey with a reddish brown tinge, becoming darker grey in apical third; a distinct black outwardly oblique streak from near base of dorsum to the cell; costa black basally, before apex to tornus a whitish diffuse post-median fascia; fringe grey mottled with few dark grey scales. Hindwing grey; fringe grey.
Male genitalia: Identical to those of the nominotypical subspecies.
Female genitalia: Unknown.
DNA barcodes (Figure 66): We obtained full length DNA barcode (658 bp) from one specimen and DNA barcode fragments of 614 bp and 632 bp from two specimens. The intraspecific p-distance is 0%. The barcodes fall within Barcode Index Number (BIN) BOLD: AEI3051. The minimum p-distance to nearest neighbour BIN BOLD: ADZ1582, an unnamed Gelechidae species from South Africa, is 5.67%.
Biology: Early stages unknown. All the specimens were collected at light during March and September at altitudes from 50 m to 895 m.
Distribution: Known from a few locations on the island of Gran Canaria (Spain). Probably endemic to Gran Canaria.
Etymology: The subspecies is named after the island of Gran Canaria (Spain).
Remarks: See also our discussion about subspecies in the discussion chapter at the end of this paper.
Stomopteryx palmella Falck & Karsholt, sp. nov. (Figures 3, 4, 42, 42a, 42b, 55, 55a)
Holotype ♂: SPAIN, LA PALMA, Barranco del Río, 450 m, 29-VII-11-VIII-2023, leg. P. Falck, genitalia slide 4062PF (ZMUC).
Paratypes: SPAIN, LA PALMA; 3 km NE El Paso, Pista de Valencia, 800 m, 1 ♂, 7-II-2002, leg. H. van der Wolf (RMNH); Los Llanos de Aridane, Barranco de las Angustias, 228 m, 1 ♀, 29-V-2016, leg. A. Werno (AW); Tigalate, 670 m, 1 ♂, 1 ♀, 9-30-III-2023, leg. P. Falck, DNA samples Lepid Phyl 1267PF/CILEP1266-23, 1268PF/CILEP1267-24 (PF), same data but, 4 ♂, 4 ♀, 29-VII-11-VIII-2023, leg. P. Falck, genitalia slide 3950PF, 4006PF, 4007PF, 4009PF, DNA sample Lepid Phyl 1393PF/CILEP1392-23 (PF), same data but, 18 ♂, 28 ♀, 10-30-XI-2023, leg. P. Falck, genitalia slide 4058PF, 4060PF (PF, MNCN); Los Cancajos, 10 m, 1 ♂, 1 ♀, 10-30-XI-2023, leg. P. Falck, genitalia slide 4042PF (PF); Barranco del Río, 450 m, 3 ♂, 29-VII-11-VIII-2023, leg. P. Falck (PF); Barranco de la Madera, 500 m, 2 ♂, 29-VII-11-VIII-2023, leg. P. Falck (PF), same data but, 1 ♂, 5 ♀, 10-30-XI2023, leg. P. Falck (PF); Los Sauces, 200 m, 1 ♀, 10-30-XI-2023, leg. P. Falck; Los Llanos, 750 m, 1 ♀, 5-25-I-2024, leg. P. Falck (PF).
Diagnosis: Stomopteryx palmella sp. nov. is characterized by the dark grey colour and the lighter grey, often reddish brown (in fresh specimens) coloration towards the costa, three distinct black spots in the fold, at 1/3 and at end of the cell, basal part of costa black. In the male genitalia the angled valva and the short hook-like lateral projection of the phallus are characteristic. In the female genitalia the long and relatively broad ductus bursae and the small rounded signa are characteristic.
Description Adult (Figures 3, 4): Wingspan 10.5-14 mm. Labial palp slender, strongly upturned; segment 2 dark grey, with whitish inner and upper surface; segment 3 longer than segment 2, white, lower surface with a black streak. Antenna grey and ringed dark brown, scape black. Head, neck and thorax dark grey with lighter tipped scales. Forewing dark grey, lighter grey from the fold towards costa, mottled with black in apical part; an indistinct black spot near base on dorsum; a distinct black plical spot at 1/3; two distinct black discal spots at each end of the cell; costa black basally, at 3/4 an indistinct yellowish spot; fringe grey with few black-tipped scales. Hindwing grey; fringe grey.
Variation: The lighter grey part of the forewing sometimes has a reddish brown tinge, or it may be absent.
Male genitalia (Figures 42, 42a, 42b): Uncus triangular, covered with long strong setae reaching middle of tegumen. Gnathos broadly rounded. Tegumen elongate, elliptical. Valva narrow, angled and broadened before middle, apex rounded. Vinculum subtriangular with two flaps densely covered with setae covering the phallus. Phallus almost straight, posterior half beak-like, distally rounded, lateral projection with small pointed, sclerotized hook.
Female genitalia (Figures 55, 55a): Papillae anales rounded. Posterior apophysis slender, twice as long as anterior apophysis. Antrum small, rectangular and sclerotized. Ductus bursae membranous, about three times longer than corpus bursae and relatively broad, slightly narrowing towards antrum. Corpus bursae oval with two small rounded signa, covered with very small spines.
DNA barcodes (Figure 66): We obtained full length DNA barcodes (658 bp) from two specimens. The barcodes fall within Barcode Index Number (BIN) BOLD: AFG5360. The intraspecific p-distance is 0.16%. The minimum p-distance to nearest neighbour Stomopteryx sp. (from Kyrgyzstan) is 7.21%.
Biology: Early stages unknown. All the specimens were collected at light from January to late November, at altitudes from 10 m to 800 m.
Distribution: Known from several scattered locations on the island of La Palma (Spain). Probably endemic to La Palma.
Etymology: The species is named after its place of occurrence, the island of La Palma.
Stomopteryx cuestaella Falck & Karsholt, sp. nov. (Figures 5, 43, 43a, 43b, 56, 56a)
Holotype ♀: SPAIN, GRAN CANARIA, Pie de la Cuesta, 500 m, 9-22-VI-2021, leg. P. Falck, genitalia slide 4021PF (ZMUC).
Paratypes: SPAIN, GRAN CANARIA, Mogán, 5 km E Soria, 800 m, 16 ♂, 3 ♀, 22-30-VII -1995, leg. K. Larsen; 2 km S Andrés, Mogán, El Baranquillo, 3 ♂ 24-VII-1995, leg. K. Larsen; Inagua, Tasarte, 600 m, 8 ♂, 2 ♀, 28-VII-1995, leg. K. Larsen; Cueva de la Ninas, 800 m, 2 ♀, 29-VII-1995, leg. K. Larsen (all ZMUC, MNCN); Pie de la Cuesta, 500 m, 8 ♂, 11-24-VI-2018, leg. P. Falck, genitalia slide 3936PF, DNA sample Lepid Phyl 0074PF/CILEP74-19 (PF), same data but, 15 ♂, 2 ♀, 21-VIII-4-IX 2020, leg. P. Falck (PF), same data but, 6 ♂, 1 ♀, 9-22-VI-2021, leg. P. Falck (PF); Ayacata, 1400 m, 1 ♂, 9-22-VI-2021, leg. P. Falck (PF); Barranco de Guayadeque, 800 m, 1 ♂, 1 ♀, 9-22-VI-2021, leg. P. Falck, genitalia slide 4040PF (PF); Fontanales, 1100 m, 1 ♂, 1 ♀, 8-20-VIII-2020, leg. P. Falck (PF). TENERIFE, Aguamansa, 1300 m, 1 ♂, 1-VIII-1979, leg. P. Stadel Nielsen (ZMUC); Teide, La Crucita, 2190 m, 1 ♂, 8-VIII-2018, leg. K. Larsen (ZMUC); 4 km S Aguamansa, 1400 m, 1 ♂, 1 ♀, 13-26-VIII2019, leg. P. Falck, genitalia slide 4022PF, 4023PF, DNA samples Lepid Phyl 0334PF/CILEP333-19, 0335PF/CILEP334-19 (PF).
Diagnosis: Stomopteryx cuestaella sp. nov. is characterized by having dark to blackish brown forewings with indistinct black markings and yellowish spots at costa before apex and in the fold. It resembles S. hungaricella Gozmány, 1957 from continental Europe, which is without a black spot followed by yellowish in the fold. S. lusitaniella Corley & Karsholt, 2014, also from continental Europe, is a more slender-winged species without marking in the forewings. S. alpinella Nel & Varrene, 2016 from the European Alps is characterized by more distinct black markings in the forewing and white-tipped scales in the apical part of the wing; the lateral process of the phallus is distinctly larger than in S. cuestaella sp. nov. The rather variable North African S. nigricella (Chrétien, 1915) is smaller and has more markings in the forewing. In the male genitalia of S. cuestaella sp. nov. the short, straight, lateral projection of the phallus with a small tooth-like apex is characteristic. They mostly resemble S. hungaricella, which has a shorter posterior part of phallus. In the female genitalia the large signa with longitudinal ridges are characteristic.
Description Adult (Figure 5): Wingspan 9.5-16 mm. Labial palp slender, strongly upturned; segment 2 dark brown, inner surface whitish; segment 3 slightly shorter than segment 2, whitish, lower surface heavily mottled with blackish brown. Antenna blackish; scape black. Head, neck, thorax and tegula dark brown. Forewing dark brown; two black, indistinct discal spots, and two black, very indistinct plical spots; distal plical spot followed by small indistinct orange spot; costal spot yellowish, rather distinct opposite indistinct tornal spot; fringe dark grey with a mixture of black-tipped scales. Hindwing dark grey-brown; fringe dark grey.
Variation: There is slight variation in the colour of the forewings, from dark brown to blackish brown.
Male genitalia (Figures 43, 43a, 43b): Uncus subrectangular, covered with long strong setae reaching beyond middle of tegumen. Gnathos laterally broadly rounded, flattened anteriorly. Tegumen elongate, elliptical. Valva narrow, evenly rounded, slightly broadened before middle, apex rounded. Vinculum subtriangular with two flaps densely covered with setae covering the phallus. Phallus almost straight, posterior half slightly tapering, distally rounded, lateral projection short, straight, apex with a small tooth-like process.
Female genitalia (Figures 56, 56a): Papillae anales rounded. Posterior apophysis slender, as long as anterior apophysis. Antrum small, rounded, cup-shaped and membranous. Ductus bursae membranous, about 1.5 times longer than corpus bursae. Corpus bursae membranous, oval with two long ridged, sclerotized signa, edge irregular.
DNA barcodes (Figure 66): We obtained full length DNA barcodes (658 bp) from two specimens and DNA barcode fragments of 637 from one specimen. The barcodes fall within two Barcode Index Numbers (BIN) BOLD: ADU1068 (one specimen from Gran Canaria) and BOLD: AEC4744 (two specimens Tenerife), the minimum p-distance between the BINs is 2.88%. The maximum intraspecific p-distance is high 3.04%. The nearest neighbour is Stomopteryx flavipalpella Jäckh, 1959 with a 4.74% divergence.
Biology: Early stages and hostplant are unknown. Adults have been collected at light from June to the beginning of September, at altitudes from 800 m to 1400 m.
Distribution: Endemic to the Canary Islands (Spain) and only known from the mountain areas on the islands of Gran Canaria and Tenerife.
Etymology: The species is named after the small-town Pie de la Cuesta situated near the typelocality.
Stomopteryx minimelia Falck & Karsholt, sp. nov. (Figures 6, 44, 44a, 57, 57a)
Holotype ♂: SPAIN, GRAN CANARIA, Cueva de las Niñas, 800 m, 29-VII-1995, leg. K. Larsen (ZMUC).
Paratypes: SPAIN, GRAN CANARIA, San Bartolomé de Tirajana, 1000 m, 1 ♂, 5-VIII-1979, leg. P. Stadel Nielsen; Mogán 5 km E Soria, 800 m, 1 ♂, 1 ♀, 22-30-VII. 1995, leg. K. Larsen (ZMUC); 2 km S Andrés, Mogán El Baranquillo, 1 ♀ 24-VII-1995, leg. K. Larsen, genitalia slide ZM4081PF; Inagua, Tasarte, 600 m, 2 ♂, 28-VII-1995, leg. K. Larsen; Cueva de las Niñas, 800 m, 3 ♂, 11 ♀, 29-VII-1995, leg. K. Larsen (ZMUC, MNCN); Ayacata, 1400 m, 1 ♂, 9-22-VI-2021, leg. P. Falck, genitalia slide 4055PF, DNA sample Lepid Phyl 0924PF/CILEP923-21 (PF).
Diagnosis: Stomopteryx minimella sp. nov. is characterized by its small wingspan and having grey, towards apex blackish, mottled forewings with two rather distinct and large black discal spots and a cream-white post-median fascia. It may resemble very small specimens of S. remissella, but that species has brownish (not greyish) forewings. In the male genitalia the triangular juxta lobes and the medial tooth-like projection of the slightly bent, pointed lateral projection of the phallus are characteristic. In the female genitalia the signa are characteristic.
Description Adult (Figure 6): Wingspan 8 mm. Labial palp slender, strongly upturned; segment 2 dark whitish, outer surface basally dark grey; segment 3 slightly longer than segment 2, white, lower surface with a black streak. Antenna blackish, distinctly white ringed. Head and neck lead grey. Thorax grey. Forewing grey, mottled with dark-tipped scales, blackish apically; two black discal spots, one small spot at 1/4 and one large spot at 1/2; from costa to tornus a rather distinct, cream-white post-median fascia edged by black; fringe grey, fringe-line with black-tipped scales. Hindwing grey; fringe grey.
Male genitalia (Figures 44, 44a): Uncus subrectangular, covered with long strong setae reaching more than middle of tegumen. Gnathos laterally broadly rounded, flattened anteriorly. Tegumen elongate, elliptical. Valva narrow, evenly rounded, slightly broadened before middle, apex rounded. Vinculum subtriangular with two, setae covered, triangular flaps covering the phallus. Phallus almost straight, posterior half slightly tapering, distally rounded, lateral projection slightly bent, near apex a medial tooth-like process, apex pointed.
Female genitalia (Figures 57, 57a): Papillae anales slightly elongate. Posterior apophysis slender, 1/5 longer than anterior apophysis. Antrum membranous, laterally with two small, sclerotized areas. Ductus bursae membranous. Corpus bursae membranous, oval; signa rounded with a tooth-like projection.
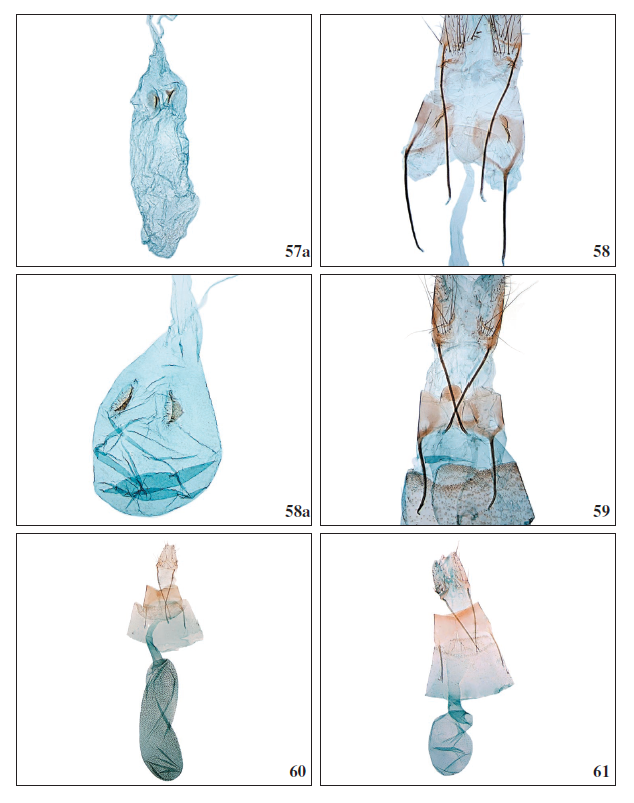
Figures 57a-61.
57a.Stomopteryx minimella Falck & Karsholt, sp. nov., ♀, corpus bursae, ZM4081PF. 58.Stomopteryx variabilis Falck & Karsholt, sp. nov., ♀, antrum complex, La Palma, GP3945PF. 58a.Stomopteryx variabilis Falck & Karsholt, sp. nov., ♀, corpus bursae, La Gomera, GP4049PF. 59.Aproaerema genistae Walsingham, 1908, ♀, antrum complex, Tenerife, GP3989PF. 60.Aproaerema mercedellaWalsingham, 1908, ♀, Tenerife, GP3968PF. 61.Aproaerema ayacatella Falck & Karsholt, sp. nov., ♀, Gran Canaria, GP3975PF.
DNA barcode (Figure 66): We obtained full length DNA barcode (658 bp) from one specimen. The barcode falls within Barcode Index Number (BIN) BOLD: AEM8857. The minimum p-distance to nearest neighbour, an unnamed Gelechiidae from Pakistan, with BIN BOLD: ACW9764 is 4.79%.
Biology: Early stages and hostplant are unknown. Adults have been collected at light from late June to the beginning of August, at altitudes from 600 m to 1400 m.
Distribution: Known from several scattered locations on the island of Gran Canaria (Spain). Probably endemic to Gran Canaria.
Etymology: The species is named because of its small size, from the Latin adjective minimus meaning smallest.
Remarks: S. minimella sp. nov. is probably related to the similar small S. biangulata Meyrick, 1921 from Mozambique, but differs in details of wing markings and genitalia (see Janse, 1951: 244- 245, pls CVIII-CVIX).
Stomopteryx variabilis Falck & Karsholt, sp. nov. (Figures 7-12, 45, 45a, 45b, 58, 58a)

Figures 9-16.
9.Stomopteryx variabilis Falck & Karsholt, sp. nov., ♀, La Gomera, 14.5 mm. 10.Stomopteryx variabilis Falck & Karsholt, sp. nov., ♂, El Hierro, 13 mm. 11.Stomopteryx variabilis Falck & Karsholt, sp. nov., ♀, El Hierro, 13 mm. 12.Stomopteryx variabilis Falck & Karsholt, sp. nov., ♀, La Palma, 13 mm. 13.Aproaerema polychromella (Rebel, 1902), ♂, Tenerife, 10 mm. 14.Aproaerema polychromella (Rebel, 1902), ♂, Gran Canaria, 8.5 mm. 15.Aproaerema genistae Walsingham, 1908, ♂, Gran Canaria, 8 mm. 16.Aproaerema genistae Walsingham, 1908, ♂, Tenerife, 9.5 mm.
Holotype ♀: SPAIN, TENERIFE; Aguamansa, 1300 m, 1-VIII-1979, leg. P. Stadel Nielsen, genitalia slide 4160OK (ZMUC).
Paratypes: SPAIN, GRAN CANARIA, Barranco de la Virgen, Moya, 400 m, 5 ♂, 20-VII-1984, leg. P. Olsen, B. Skule & P. Stadel Nielsen; Mogán, Barranco de Arguineguín, 1 ♂, 1 ♀, 21-VII-1-VIII-1995, leg. K. Larsen (all ZMUC); Barranco de Guayadeque, 1 ♂, 17-IV-2000, leg. M. Baéz (MB); Ayagaure, Alto Stausse, 1000 m, 1 ♀, 6-IX-2012, leg. A. Werno (AW); Pie de la Cuesta, 500 m, 15 ♂, 13 ♀, 11- 24-VI-2018, leg. P. Falck, genitalia slide 3937PF, 3942PF, 3947PF, 3949PF, 3951PF, 4008PF, 4035PF (PF), same data but, 1 ♀, 17-30-IX-2018, P. Falck (PF, MNCN); Barranquillo Andrés, 700 m, 1 ♂, 2 ♀, 11-24-VI-2018, leg. P. Falck, genitalia slide 4004PF, 4005PF (PF); Los Tilos de Moya, 500 m, 1 ♂, 17- 30-IX-2018, leg. P. Falck (PF); Carreteria, 455 m, 9 ♂, 2 ♀, 8-20-VIII-2020, leg. P. Falck, genitalia slide 4050PF (PF); Barranco de Azuaje, 270 m, 4 ♂, 5 ♀, 8-20-VIII-2020, leg. P. Falck, DNA samples Lepid Phyl 0840PF/CILEP839-21, 0841PF/CILEP840-21, 0843PF/CILEP842-21 (PF); Barranco Moya, 80 m, 1 ♂, 3 ♀, 8-20-VIII-2020, leg. P. Falck (PF). TENERIFE; Aguamansa, 1300 m, 3 ♂, 5 ♀, 29- VII-1979, leg. P. Stadel Nielsen; Las Cãnadas, 2100 m, 4 ♂, 23-VII-1984, leg. P. Olsen, B. Skule & P. Stadel Nielsen; Bco. de Badajoz, 1♂ 11-VII-1994, leg. M. Baéz (MB); Bco. Harques, 2 ♂, 20-VII1995, leg. M. Baéz (MB); Güímar, Barranco de Badajoz, 700 m, 1 ♀, 1-VII-2003, leg. O. Czadek (ZMUC); 3 km N San Andrés, 2 ♀, 10-IX-2014, leg. A. Werno (AW); Anaga, Roque Negro, 615 m, 1 ♂, 7-9-VIII-2018, leg. K. Larsen (ZMUC); Teide, La Crucita, 2190 m, 1 ♀, 8-VIII-2018, leg. K. Larsen (ZMUC); Aguamansa, 1050 m, 20 ♂, 10 ♀, 13-26-VIII-2019, leg. P. Falck, genitalia slides, 4038PF, 4051PF, 4053PF, DNA samples Lepid Phyl 0844PF/CILEP843-21, 0845PF/CILEP844-21, 0845PF/CILEP844-21 (PF, MNCN); Arona, 670 m, 23 ♂, 9 ♀, 21-V-3-VI-2019, leg. P. Falck (PF), same data but, 1 ♂, 1 ♀, 3-16-VIII-2021, leg. P. Falck, genitalia slide 3933PF (PF); Las Manchas, 1050 m, 2 ♂, 21-V-3-VI-2019, leg. P. Falck (PF), same data but, 1 ♂, 1 ♀, 3-16-VIII-2021, leg. P. Falck, genitalia slide 3932PF (PF); Güímar, 500 m, 2 ♂, 5 ♀, 3-16-VIII-2021, leg. P. Falck (PF); El Caletón, 160 m, 2 ♂, 3 ♀, 1-13-VI-2022, leg. P. Falck, genitalia slide 3943PF (PF). LA GOMERA; El Cedro, 1000 m, 1 ♂, 1 ♀, 24-25-VII-1984, leg. P. Olsen, B. Skule & P. Stael Nielsen; El Cedro, 870 m, 3 ♂, 1 ♀, 17- 20-VIII-2018, K. Larsen; 2 km S Hermigua, 520 m, 2 ♀, 18-19-VIII-2018, K. Larsen (all ZMUC); Hermigua, 250 m, 19 ♂, 11 ♀, 9-12-VIII-2021, leg. P. Falck, genitalia slide 3940PF, 3948PF, 4037PF, 4049PF, DNA samples Lepid Phyl 0912PF/CILEP911-21, 0913PF/CILEP912-21, 0914PF/CILEP913- 21 (PF); LA PALMA; Barranco de las Nieves, 250 m, 7 ♂, 13-16-VIII-2018, leg. K. Larsen (ZMUC); Tigalate, 670 m, 1 ♂, 9 ♀, 29-VII-11-VIII-2023, leg. P. Falck, genitalia slide 3945PF, DNA sample Lepid Phyl 1396PF/CILEP1395-23 (PF); Barranco de la Madera, 500 m, 3 ♂, 1 ♀, 29-VII-11-VIII2023, leg. P. Falck, genitalia slide 3935PF, 4011PF, DNA sample Lepid Phyl 1395PF/CILEP1394-23 (PF); Barranco del Rio, 450 m, 3 ♂, 12 ♀, 29-VII-11-VIII-2023, leg. P. Falck, genitalia slide 3939PF, 3952PF, 4010PF (PF, MNCN); Las Toscas, 150 m, 1 ♂, 3 ♀, 29-VII-11-VIII-2023, leg. P. Falck, genitalia slide 4039PF (all PF); EL HIERRO; Frontera, El Sitio, 340 m, 4 ♂, 10-12-VIII-2018, leg. K. Larsen (ZMUC); Erese, 700 m, 14 ♂, 16 ♀, 22-VII-3-VIII-2022, leg. P. Falck, genitalia slide 3938PF, 3946PF, 4036PF, 4048PF (PF, MNCN); Sabinosa, 100 m, 1 ♂, 3 ♀, 22-VII-3-VIII-2022, leg. P. Falck (PF); Tacorón, 170 m, 2 ♂, 8 ♀, 22-VII-3-VIII-2022, leg. P. Falck, genitalia slide 3934PF, 4059PF, DNA sample Lepid Phyl 1228PF/CILEP1227-23 (PF); Cruz de los Reyes, 1360 m, 1 ♂, 1 ♀, 22-VII-3- VIII-2022, leg. P. Falck (PF); Jinama, 1250 m, 2 ♂, 3 ♀, 22-VII-3-VIII-2022, leg. P. Falck, DNA sample Lepid Phyl 1229PF/CILEP1228-23 (PF); Frontera, 280 m, 1 ♂, 22-VII-3-VIII-2022, leg. P. Falck, genitalia slide 3941PF (PF).
Diagnosis: The species is characterized by its high variability. However, all forms are characterized by the yellowish-brown colour along the dorsum and the meadian longitudinal black streak and they resemble no other known Stomopteryx species. In the male genitalia the relatively long pointed lateral projection of the phallus is characteristic. In the female genitalia the triangular signa are characteristic.
Description Adult (Figures 7-12): Wingspan 10-16 mm. Labial palp slender, strongly upturned; segment 2 cream white, with outer surface basally dark brown; segment 3 slightly longer than segment 2, white, lower surface with a black streak and black tip. Antenna blackish; scape black. Head, neck and thorax yellowish white. Tegula yellowish white, basally dark brown. Forewing yellowish brown, grey along costa and mottled with grey and brown scales in apical part; from the base to the distal end of cell a median, broad, black streak; fringe grey with some black-tipped scales, sometimes forming an outer fringe-line. Hindwing grey; fringe grey.
Variation: S.variabilis exhibits considerable geographical variation. The above description applies to specimens from Tenerife and La Palma. Specimens from El Hierro and Gran Canaria have the forewings dark brown, yellowish brown along the dorsum and an indistinct yellowish white postmedian fascia, and the black median streak is narrower and more irregular. The black streak is shorter in specimens from the Gran Canaria. Specimens from La Gomera are very similar to specimens from El Hierro, but the costal half of the forewing is greyish.
Male genitalia (Figures 45, 45a, 45b): Uncus triangular, covered with long strong setae reaching beyond middle of tegumen. Gnathos broadly rounded. Tegumen elongate, elliptical. Valva narrow, evenly rounded, before middle slightly broadened, apex rounded. Vinculum subtriangular with two flaps densely covered with setae covering the phallus. Phallus almost straight, posterior half tapering, distally rounded, lateral projection half the length of phallus, slightly bent, apex pointed.
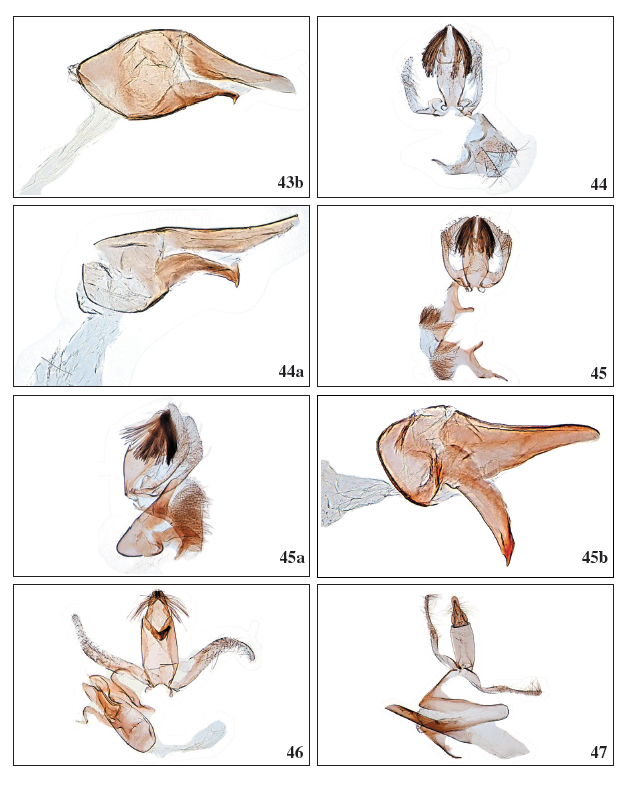
Figures 43b-47.
43b. Stomopteryx cuestaella Falck & Karsholt, sp. nov., ♂, GP4020PF. 44.Stomopteryx minimella Falck & Karsholt, sp. nov., ♂, Gran Canaria, GP4055PF. 44a. Stomopteryx minimella Falck & Karsholt, sp. nov., ♂, phallus, GP4055PF. 45.Stomopteryx variabilis Falck & Karsholt, sp. nov., ♂, Gran Canaria, GP3942PF. 45a.Stomopteryx variabilis Falck & Karsholt, sp. nov., ♂, lateral view, Tenerife, GP4038PF. 45b.Stomopteryx variabilis Falck & Karsholt, sp. nov., ♂, phallus, Tenerife, GP3943PF. 46. Aproaerema genistae Walsingham, 1908, ♂, Tenerife, GP3980PF. 47. Aproaerema mercedella Walsingham, 1908, ♂, Tenerife, GP3963PF.
Female genitalia (Figures 58, 58a): Papillae anales rounded. Posterior apophysis slender, longer than anterior apophysis. Antrum small, rounded and membranous. Ductus bursae membranous, about 1.5 times longer than corpus bursae. Corpus bursae oval with two rounded signa, in lateral view triangular. The signa may vary in size.
DNA barcodes (Figure 66): We obtained full length DNA barcodes (658 bp) from ten specimens and DNA barcode fragments of 621 bp, 622bp 632 bp and 584bp from four specimens. The barcodes fall within three Barcode Index Numbers (BIN) BOLD: AEI4402 (four specimens from Tenerife), BOLD: AEI0944 (three specimens from Gran Canaria) and BOLD: AEN4981 (seven specimens from La Gomera (3), La Palma (2) and El Hierro (2)). The minimum p-distance between the BIN´s is 1.86% (AEN4981 vs. AEI4402), 3.09% (AEI0944 vs. AEI4981) and 3.49% (AEN4981 vs. AEI4402). The maximum intraspecific p-distance is very high 4.28%. The minimum p-distance to nearest neighbour, an unnamed Gelechiidae species from South Africa, is 5.68% .
Biology: Early stages and hostplant are unknown. Adults have been collected at light from the middle of April to the end of September, at altitudes from 150 m to 2190 m.
Distribution: Known from several locations on the islands of Gran Canaria, Tenerife, La Gomera, La Palma and El Hierro. Endemic to the Canary Islands, Spain.
Etymology: The species is named after the variable adults.
Remarks: Despite the high intraspecific divergence in COI and the very high variation in adult appearance between the island populations, we were not able to find morphological differences in the genitalia. Further, the DNA barcodes suggests that only one species is involved. The maximum intraspecific p-distance in BIN BOLD: AEN4981 is only 1.28%, which includes very different looking specimens e.g., between populations from El Hierro and La Palma. Single specimens from Tenerife are similar to specimens from both La Gomera and Gran Canaria. There is also no correlation in size of the signa between the different populations.
The species is figured by Klimesch (1984: 181) as Stomopteryx sp.
Aproaerema Durrant, 1897
Aproaerema Durrant, 1897, Ent. Month. Mag., 33, 221
In its current concept this genus includes more than 90 described species, distributed in all continents except Antartica, but with most species in Europe and South Africa. The larvae of most species feed on Fabaceae.
A. mercedellaWalsingham, 1908 and the three closely related species described as new below form a separate group within the genus Aproaerema, differing especially in the uncus-gnathos complex: the rounded uncus is without pecten, and the gnathos is subtriangular. Other parts of the genitalia are similar to those of other Aproaerema species. Such differences may indicate that these species could belong in a separate genus. However, we refrain from introducing a new genus until there is a better overview of the genera of Anacampsini. This group of species seems to be endemic to the Canary Islands. According to Huemer et al. (2020, Suppl. material 2, NJ tree 10) A. mercedella is nested within the genus Aproaerema.
Aproaerema polychromella (Rebel, 1902) (Figures 13, 14)
Anacampsis polychromellaRebel, 1902. Dt. ent. Z. Iris, 15, 109
Material examined: SPAIN, GRAN CANARIA, Pie de la Cuesta, 500 m, 1 ♂, 17-30-IX-2018, leg. P. Falck, DNA sample Lepid Phyl 0076PF/CILEP76-18. LA GOMERA, Hermigua, 170 m, 1 ♀, 1-8-I-2023, leg. P. Falck, DNA sample Lepid Phyl 1548PF/CILEP1547-24 (PF).
Diagnosis: A rather small species (wingspan 9-11 mm), with basally brownish shining forewings becoming blackish in apical half and characterized by a broad white, apically irregular post-median fascia. The genitalia are similar to those ofA. genistae.
Biology: The biology is imperfectly known, but the larva probably feeds on Fabaceae. (Klimesch, 1984: 162) found adults amongst Lotus sessilifolius DC. Adults are recorded during almost all months of the year, mostly in late autumn, at lower altitudes.
Distribution in the Canary Islands (Spain): First record by Klimesch (1984, p. 162) from Gran Canaria, Tenerife, La Gomera and La Palma. Also, on Fuerteventura (Báez, 1998, p. 188). New island record. El Hierro, Sabinosa, 100 m, 22-VII-3-VIII-2022, leg. P. Falck (PF).
General distribution: A sub-tropical and tropical species occurring in the Mediterranean countries, northwards to SW France; Africa, Near East and India. In recent years mostly as singletons in several areas in NW Europe, probably due to migration (Gregersen & Karsholt, 2022, p. 90).
Remarks: It is surprising that the early stages of this common and widespread species are imperfectly known.
A. polychromella is a rather variable species through its distribution area. The population in the Canary Islands varies in the colour basally of the forewing from dark brown to shining golden, and the post-median fascia varies in width (Figures 13, 14).
Aproaerema genistae Walsingham, 1908 (Figures 15-18, 46, 59)
![17.Aproaerema genistaeWalsingham, 1908, ♀,
Tenerife, 10 mm. 18.Aproaerema genistae Walsingham, 1908, ♀, La Palma, 10 mm.
19. Aproaerema thaumalea Walsingham, 1905, ♂, Tenerife, 9.5 mm. 20.Aproaerema
thaumalea Walsingham, 1905, ♀, Tenerife, 9.5 mm. 21.Aproaerema anthyllidella (Hübner, [1813]), ♀, Gran
Canaria, 12 mm. 22.Aproaerema anthyllidella (Hübner, [1813]), ♀, Tenerife, 9.5
mm. 23.Aproaerema anthyllidella (Hübner, [1813]), ♀, El Hierro, 7 mm. 24.Aproaerema anthyllidella (Hübner, [1813]), ♀, La Palma, 9.5 mm.](../0300-5267-shilap-53-209-5-gf9.png)
Figures 17-24.
17.Aproaerema genistaeWalsingham, 1908, ♀, Tenerife, 10 mm. 18.Aproaerema genistae Walsingham, 1908, ♀, La Palma, 10 mm. 19. Aproaerema thaumalea Walsingham, 1905, ♂, Tenerife, 9.5 mm. 20.Aproaerema thaumalea Walsingham, 1905, ♀, Tenerife, 9.5 mm. 21.Aproaerema anthyllidella (Hübner, [1813]), ♀, Gran Canaria, 12 mm. 22.Aproaerema anthyllidella (Hübner, [1813]), ♀, Tenerife, 9.5 mm. 23.Aproaerema anthyllidella (Hübner, [1813]), ♀, El Hierro, 7 mm. 24.Aproaerema anthyllidella (Hübner, [1813]), ♀, La Palma, 9.5 mm.
Aproaerema genistae Walsingham, 1908.Proc. zool. Soc. Lond., 1907, 933, pl. 51, fig. 8.
Aproaerema adenocarpella Rebel, 1927 in Hering. Zool. Jb. (Syst.) 53, 425
Material examined: SPAIN, GRAN CANARIA, Pinos de Galdar, 1200 m, 1 ♂, 1 ♀, 22-VII-1984, leg. P. Olsen. P. Stadel Nielsen & B. Skule, DNA sample TLMF Lep.25523, genitalia slide 1708 Hendriksen (ZMUC); Pie de la Cuesta, 500 m, 1 ♀, 11-24-VI-2019, DNA sample Lepid Phyl 0069PF/CILEP69-19, same data but 2 ♀, 17-30-IX-2019, leg. P. Falck, DNA samples Lepid Phyl 0071PF/CILEP71-19, 0072PF/CILEP72-19; Ayacata, 1400 m, 1 ♂, 3 ♀, 17-30-IX-2019, leg. P. Falck, genitalia slide GP3982PF, GP3984PF, GP3986PF, GP4030PF. TENERIFE, Las Manchas, 1050 m, 2 ♂, 5 ♀, 17-30-IX-2019, leg. P. Falck, DNA samples Lepid Phyl 0067PF/CILEP67-19, 0068PF/CILEP68-19, genitalia slide GP3980PF, GP3987PF, GP3989PF, GP4039PF, GP4070PF; Arona, 600 m, 1 ♀, 1-20-III2017, leg. P. Falck, genitalia slide GP4075PF; Barranquillo Andrés, 700 m, 1 ♀, 11-24-VI-2019, leg. P. Falck, DNA sample Lepid Phyl 0070PF/CILEP70-19. LA PALMA, Tigalate, 670 m, 1 ♂, 6 ♀, 29-VII-11- VIII-2023, leg. P. Falck, DNA sample Lepid Phyl 1430PF/CILEP1429-24, genitalia slide GP3981PF, GP3985PF, GP4042PF, GP4044PF, GP4045PF, GP4067PF (all PF).
Diagnosis: A rather small species (wingspan 7-11 mm) having black forewings with a more or less light brownish basal half, a slender whitish, oblique post-median fascia, and apical part of wing with grey, black-tipped scales. The genitalia (Figures 46, 59) are similar to those of A. polychromella, but distinctly different from those of A. anthyllidella.
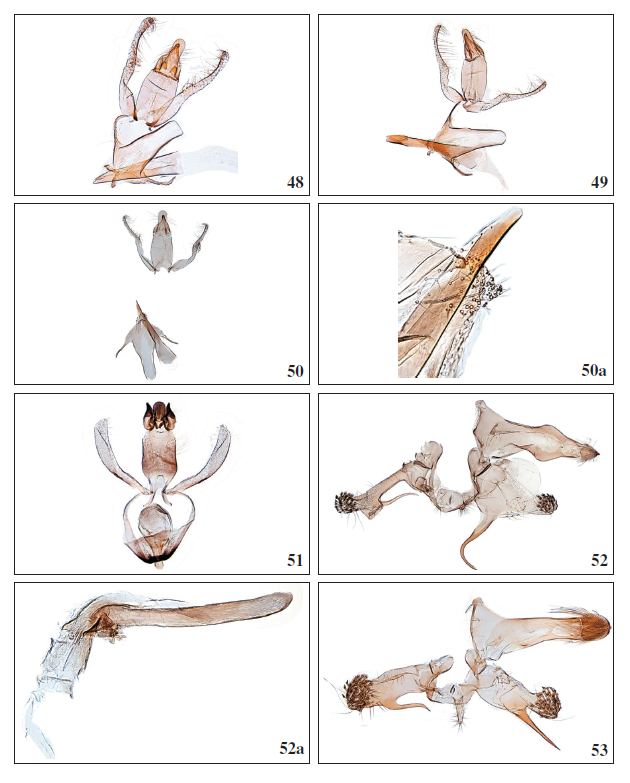
Figures 48-53.
48.Aproaerema ayacatella Falck & Karsholt, sp. nov., ♂, Gran Canaria, GP3974PF. 49.Aproaerema pseudomercedella Falck & Karsholt, sp. nov., ♂, El Hierro, GP3964PF. 50.Aproaerema angustumella Falck & Karsholt, sp. nov., ♂, Tenerife, GP4002PF. 50a. Aproaerema angustumella Falck & Karsholt, sp. nov., ♂, apex of phallus with cornuti, La Palma, GP4052PF. 51.Mesophleps ochracella (Turati, 1926), ♂, Lanzarote, GP3997PF. 52.Anarsia lineatella Zeller, 1839, ♂, Gran Canaria, GP3958PF. 52a. Anarsia lineatella Zeller, 1839, ♂, phallus, GP3958PF. 53. Anarsia acaciae Walsingham, 1896, ♂, Gran Canaria, GP3956PF.
DNA barcode (Figure 66): We obtained full length DNA barcodes (658 bp) from five specimens and DNA barcode fragments of 624 bp, 639bp from two specimens. The barcodes fall within two Barcode Index Numbers (BIN) BOLD: ADL6169 (six specimens from Gran Canaria (4) and Tenerife (2) and BOLD: AEI0944 (one specimen from La Palma). The minimum p-distance between the BINs is 3.38%. The maximum intraspecific p-distance is very high 3.84%. The minimum p-distance to nearest neighbour, Aproaerema polychromella, is 5.64%.
Biology: The larva feeds during May among shoots of Genista canariensis L. (Walsingham, 1908: 934) and Adenocarpus foliolosus DC. (Hering 1927).
Distribution in the Canary Islands (Spain): First record by Walsingham (1908, p. 933) from Tenerife. Additionaly from Lanzarote (Báez, 1998, p. 188), Gran Canaria (Hering, 1927, p. 426) and La Palma (Klimesch, 1984: 163). New island record: La Gomera, Arure, 820 m, 1 ♀, 9-16-III-2024, leg. P. Falck (PF).
General Distribution: Endemic to the Canary Islands (Spain).
Remarks: Aproaerema adenocarpella was synonymized with A. genistae by Klimesch (1984, p. 163).
A. genistae varies in the colour basally of the forewing from almost dark brown to reddish brown, and the post-median fascia varies in width (Figures 15-18). There is no correlation between the different forms and the populations. Despite the divergence in the barcodes between the populations from Gran Canaria/Tenerife and La Palma, we found no morphological differences either in the adults or in the genitalia.
Aproaerema thaumaleaWalsingham, 1905 (Figures 19, 20)
Aproaerema thaumaleaWalsingham, 1905, Ent. mon. Mag., 41, 41
Material examined: SPAIN, FUERTEVENTURA, Lajares, 50 m, 1 ♂, 1-27-XI-2017, leg. P. Falck, DNA sample Lepid Phyl 0077PF/CILEP77-19, same data but, 1 ♂, 6-26-I-2020, leg. P. Falck, genitalia slide GP3990PF. TENERIFE, El Médano, 20 m, 1 ♂, 1 ♀, 1-20-III-2017, leg. P. Falck genitalia slides GP3991PF, GP3993PF (all PF).
Diagnosis: A very characteristic species. The pattern of the forewings resembles those of A. polychromella, but differs by the large, triangular white spot from base to 1/3 of costa. The genitalia are similar to those of A. polychromella.
DNA barcode (Figure 66): Barcode index number (BIN) BOLD: ADO5854.
Biology: The larva feeds on Lotus sessilifolius DC., forming sand-galleries beneath the trailing shoots (Walsingham, 1908, p. 934).
Distribution in the Canary Islands (Spain): First record by Walsingham (1908, p. 934) from Tenerife. Also, on La Gomera (Báez, 1998, p. 188). New island records. Fuerteventura, Jandia, Barranco de Esquinzo, 20-IV-3-V.2000, leg. R. Pass (WS) and Lanzarote, Caleta de Famara, 21-X-10- XI-2019, leg. P. Falck (PF).
General distribution: Canary Islands (Spain), Morocco, Algeria, Libya.
Aproaerema anthyllidella (Hübner, [1813]) (Figures 21-24)
Tinea anthyllidella Hübner, 1813, Samml. eur. Schmett., 8, pl. 48, fig. 330
Gelechia elachistella Stainton, 1859, Ann. Mag. nat. Hist. (3)3, 213, stat. nov
Gelechia psoralella Millière, 1865, Iconogr. Descr. Chenilles Lépid. inédits 2, 83, 99, pl. 61, figs 1-6
Anacampsis infestellaRebel, 1896, Annln naturhist. Mus. Wien, 11, 128
Material examined: SPAIN, FUERTEVENTURA, Corralejo, 10 m, 1 ♂, 7-27-XI-2017, leg. P. Falck, genitalia slide GP3979PF. GRAN CANARIA, Degollada de Tasartico, 150 m, 1 ♂, 28-VII-1995, leg. K. Larsen, genitalia slide ZM4079PF (ZMUC); Pie de la Cuesta, 500 m, 1 ♀, 4-23-III-2019, leg. P. Falck, genitalia slide GP3988PF. TENERIFE, Buenavista del Norte, 1 ♂, 18-IV-1998, leg. K. Larsen, genitalia slide ZM4078PF (ZMUC); El Médano, 10 m, 1 ♀, leg. P. Falck, DNA sample Lepid Phyl 0075PF/CILEP75-18; LA PALMA, Barranco de la Madera, 500 m, 1 ♀, leg. P. Falck, DNA sample Lepid Phyl 1558PF/CILEP1557-24, genitalia slide GP4043PF; Tigalate, 670 m, 1 ♂, 29-VII-11-VIII-2023, leg. P. Falck, genitalia slide GP3983PF; Los Llanos, 150 m, 1 ♂, 9-30-III-2023, leg. P. Falck, genitalia slide GP3978PF. EL HIERRO, Sabinosa, 100 m, 1 ♀, 22-VII-3-VIII-2022, leg. P. Falck, DNA sample Lepid Phyl 1561PF/CILEP1560-24, genitalia slide GP3992PF (all PF).
Diagnosis: A rather small species of variable wingspan (7-13 mm), having black forewings with more or less distinct costal and tornal spots and a small but characteristic whitish spot in the fold. The very characteristic genitalia are figured by, e.g., Gregersen & Karsholt (2022).
Variation (Figures 21-24): A. anthyllidella is a variable species, both in size, colour and wing markings. Females are generally stronger marked than males, with distinct white or yellowish white costal and tornal spots which in some specimens can be fused to a transverse fascia. Some specimens, especially males, are almost black.
DNA barcodes (Figure 66): We obtained full length DNA barcodes (658 bp) from three specimens. The barcodes fall within Barcode Index Number (BIN) BOLD: AAD2266. The maximum intraspecific p-distance is 4.41%. The minimum p-distance to nearest neighbour BOLD: AAD2267, also Aproaerema anthyllidella, is 2.17%.
Biology: The larva is known to feed on a number of different Fabaceae. In the Canary Islands it has been recorded from Lotus glaucus Aiton and Bituminaria bituminosa (L.) C.H. Stirt. (= Psoralea bituminosa), feeding between spun together shoots and leaves which are mined (Walsingham, 1908, p. 932; Klimesch 1984, p. 160-164), and from Medicago sativa L. (label data). Adults are recorded during almost all months of the year, at altitudes from sea level to 1050 m.
Distribution in the Canary Islands (Spain): First recorded from Tenerife by Rebel (1896, p. 127). Additionally, from Fuerteventura (Arenberger, 1999, p. 13), Gran Canaria (Walsingham, 1908, p. 933), La Gomera (Klimesch, 1984, p. 160), La Palma (Rebel, 1938, p. 89) and El Hierro (Báez & Martín, 2001, p. 238). New island record: Lanzarote, Puerto del Carmen, 1-5-II-1994, leg. J. P. Baungaard (ZMUC). A widespread and common species.
General Distribution: Widely distributed throughout the Palaearctic region; North America.
Remarks: Gelechia elachistella was described from the small island of Ilhéu Chão in the Madeira archipelago. Similar to most other specimens of A. anthyllidella from Madeira it is characterized by having more distinct whitish costal and tornal spots when compared with most female specimens from continental Europe. Similar female forms were described by Rebel (1896) as Anacampsis infestella from Tenerife (lectotype figured by Ungureanu & Murariu, 2024), and by Millière (1865) as Gelechia psoralella from southern France.
Walsingham (1908, pp. 932-933) treated specimens from the Canary Islands as Aproaerema psoralella and specimens from Madeira as A. elachistella, separating the latter from A. anthyllidella on the lighter white colour of the head and labial palpi.
Klimesch (1984, pp. 160-161, 164) listed two species in this complex from the Canary Islands: Aproaerema anthyllidella elachistella and Iwaruna psoralella. He figured the male genitalia of both, in dorso-ventral and lateral view, respectively, and the female genitalia of the latter. He stated that he could not see any difference between those of A. anthyllidellaelachistella, and specimens of A. anthyllidella from continental Europe, but treated A. elachistella as a subspecies of A. anthyllidella.
Gelechia psoralella is currently considered as a synonym of Aproaerema anthyllidella (Leraut, 1997, p. 125; Huemer & Karsholt, 2020, p. 73). It is unclear why Klimesch (1984) placed A. psoralella in the genus Iwaruna Gozmány, 1957.
We have not been able to find differences in the genitalia between specimens with more or less distinct costal and tornal spots or other forms of A. anthyllidella from the Canary Islands, Madeira and continental Europe.
The DNA barcode of A. anthyllidella is referred in BOLD to three BINs: AAD2266, AAD2267 and ADR9287, the latter from only one specimen from Spain (Huemer et al. 2020, suppl. material 2). AAD2266 and 2267 cover many specimens from all over Europe, with specimens from Madeira belonging to AAD2266 (P. Huemer, in litt.). As we are not able to separate A. anthyllidella elachistella from the nominotypical subspecies, neither on the genitalia nor in the DNA barcode, and because the alleged differences in size, colour and wing markings fall within the variation found in continental Europe we find no support for treating the populations of A. anthyllidella in the Macaronesian Islands as separate species or subspecies.
Aproaerema mercedella Walsingham, 1908 (Figures 25, 26, 47, 60)

Figures 25-32.
25.Aproaerema mercedellaWalsingham, 1908, ♂, Tenerife, 11 mm. 26.Aproaerema mercedella Walsingham, 1908, ♀, Tenerife, 11 mm. 27.Aproaerema ayacatella Falck & Karsholt, sp. nov., ♂, Gran Canaria, 10.5 mm. 28. Aproaerema ayacatella Falck & Karsholt, sp. nov., ♀, Gran Canaria, 10.5 mm. 29.Aproaerema pseudomercedella Falck & Karsholt, sp. nov., ♂, La Gomera, 10.5 mm. 30.Aproaerema pseudomercedella Falck & Karsholt, sp. nov., ♀, El Hierro, 12 mm. 31. Aproaerema angustumella Falck & Karsholt, sp. nov., ♂, Tenerife, 7.5 mm. 32.Aproaerema angustumella Falck & Karsholt, sp. nov., ♂, Tenerife, 8.5 mm.
Aproaerema mercedellaWalsingham, 1908, Proc. zool. Soc. Lond., 1907, 934
Material examined: SPAIN, TENERIFE, Aguamansa, 1050 m, 9 ♂, 10 ♀, 13-26-VIII-2019, leg. P. Falck, genitalia slide 3963PF, GP4068PF, DNA sample Lepid Phyl 0731PF/CILEP730-20 (PF); Las Mercedes, 750 m, 8 ♂, 9 ♀, 13-26-VIII-2019, leg. P. Falck, genitalia slide 3972PF, 3963PF, DNA sample Lepid Phyl 0730PF/CILEP729-20 (PF).
Diagnosis: A. mercedella resembles A. ayacatella sp. nov. and A. pseudomercedella sp. nov. It is not possible to distinguish the species without examination of the genitalia or by barcoding. In the male genitalia the relatively shorter phallus and the spinule covered vesica are characteristic. It differs from A. ayacatella and A. pseudomercedella by the shorter phallus and by the spinule covered vesica. In the female genitalia the relatively broad ductus bursae and the oval, distinctively granulated corpus bursae are characteristic. It differs from A. ayacatella by the straight relatively broad ductus bursae (twisted in ayacatella) and the granulated corpus bursae. It differs from A. pseudomercedella by the longer and broader ductus bursae and the granulated corpus bursae.
Description. Adult (Figures 25, 26): Wingspan 10-11.5 mm. Labial palp slender, upturned, segment 2 dark brown posteriorly and medially yellowish white, segment 3 slightly shorter than segment 2, yellowish white, apex blackish brown. Antenna dark grey with yellowish rings. Head, neck and thorax yellowish to greyish brown. Tegula dark brown, yellowish at base. Forewing blackish brown; basally an indistinct yellowish brown spot between costa and dorsum; from costa near the base to the middle a rhomboid yellowish mark; at the end of cell a distinct black spot, edged white; apically at costa and from near tornus two outwardly, distinct, yellowish lines, almost forming a postmedian fascia. Fringe yellowish with two parallel, grey fringe-lines. Hindwing grey, with grey fringe. Abdomen grey, with yellowish anal tufts.
Variation: There is often an admixture of rust-brown spots, especially apically and sometimes forming a rust-brown streak from the costal mark to beyond the postmedian fascia.
Male genitalia (Figure 47): Tegumen oblong, lateral margins convex, anterior margin almost without emargination; uncus slender, apex rounded; gnathos subtriangular, shorter than uncus, lateral arms well developed, apex slender with a small hook; valva simple, narrow, digitate, setose, longer than tegumen process; vinculum subtriangular, posteriorly flatly rounded, anteriorly with lateral arms; juxta small, rounded, covered with small spines; phallus straight, tapering towards apex, slightly longer than vinculum; vesica covered with spinules.
Female genitalia (Figure 60): Papillae anales elongate, distally pointed; posterior apophysis long, 2.5 times longer than anterior apophysis; segment VIII cylindrical, as long as anterior apophysis, medially membranous; antrum membranous; ductus bursa relatively broad slightly widening just before colliculum, anterior third evenly widening towards corpus bursae; corpus bursae oval, distinctively granulated.
DNA barcodes (Figure 66): We obtained full length DNA barcodes (658 bp) from two specimens. The barcodes fall within Barcode Index Number (BIN) BOLD: AEG7879. The maximum intraspecific p-distance is 1.12%. The minimum p-distance to nearest neighbour, A. angustumella, is 5.44%.
Biology: One specimen reared from moss-grown dead bark of Prunus lusitanica L. (as Laurocerasus lusitanica L.) from 7-III-1904 and emerged 24-VIII same year (Eaton) (Walsingham, 1908, p. 935). It is unclear if the larva was still feeding, or if it had just entered the bark to pupate. Adults are attracted to light and fly actively in the late afternoon in laurisilva forests during August.
Distribution: Known only from a few locations on the north-eastern part of Tenerife (Spain). Probably endemic to Tenerife.
Aproaerema ayacatella Falck & Karsholt, sp. nov. (Figures 27, 28,48, 61)
Holotype ♀: SPAIN, GRAN CANARIA, Ayacata, 1400 m, 17-30-IX-2018, leg. P. Falck, genitalia slide 3965PF (ZMUC).
Paratypes: SPAIN, GRAN CANARIA, Ayacata, 1400 m, 2 ♀, 17-30-IX-2018, leg. P. Falck, genitalia slide 3971PF, 3973PF (PF); Pie de la Cuesta, 500 m, 1 ♂, 21-VIII-4-IX-2020, leg. P. Falck, genitalia slide 3967PF (PF); Los Tilos de Moya, 500 m, 1 ♀, 17-30-IX-2018, leg. P. Falck, genitalia slide 3975PF, DNA sample Lepid Phyl 0733PF/CILEP732-20 (PF); Barranco de Guayadeque, 700 m, 1 ♂, 21-VIII-4-IX-2020, leg. P. Falck, genitalia slide 3974PF, DNA sample Lepid Phyl 0732PF/CILEP431- 20 (PF, MNCN).
Diagnosis: A. ayacatella is similar to A. pseudomercedella and A. mercedella q.v. In the male genitalia the almost rectangular vinculum, the relatively longer phallus and the vesica without or only with very few spinules are characteristic. It is not possible to distinguish A. ayacatella and A. pseudomercedella by the male genitalia. In the female genitalia the broad and twisted ductus bursae and the small rounded, weakly granulated corpus bursae are characteristic. It is distinguished from A. mercedella, q.v., and from A. pseudomercedella by the broad and twisted ductus bursae.
Description Adult (Figures 27, 28): Wingspan 9-10 mm. Labial palp slender, upturned, segment 2 dark brown posteriorly white, segment 3 slightly shorter than segment 2, white, anteriorly black ringed, apex blackish brown. Antenna dark grey with yellowish rings. Head, neck and thorax greyish brown. Tegula dark brown, yellowish at base. Forewing blackish brown; basally an indistinct yellowish brown spot between costa and dorsum; from costa near the base to the middle a yellowish spot, continuing as an indistinct median streak to beyond the postmedian fascia; at the end of cell a distinct black spot, edged white; apically at costa and from near tornus two outwardly, distinct, yellowish lines, almost forming a postmedian fascia. Fringe yellowish with two parallel grey fringe-lines. Hindwing grey, with grey fringe. Abdomen grey, with yellowish anal tufts.
Male genitalia (Figure 48): Tegumen oblong, lateral margins convex, anterior margin almost without emargination; uncus slender, apex rounded; gnathos subtriangular, shorter than uncus, lateral arms well developed, apex slender with a small hook; valva simple, narrow, digitate, setose, longer than tegumen process; vinculum subrectangular, posteriorly flatly rounded, anteriorly with lateral arms; juxta small, rounded, covered with small spines; phallus straight, tapering towards apex, longer than vinculum; vesica without or with very few spinules.
Female genitalia (Figure 61): Papillae anales elongate, distally pointed; posterior apophysis long, twice as long as anterior apophysis; segment VIII cylindrical, slightly shorter than anterior apophysis, medially membranous; antrum membranous; ductus bursae broad, distinctively widening towards colliculum, just before corpus bursae twisted; corpus bursae small, rounded, weakly granulated.
DNA barcodes (Figure 66): We obtained full length DNA barcodes (658 bp) from two specimens. The barcodes fall within Barcode Index Number (BIN) BOLD: AEG7878. The maximum intraspecific p-distance is high 1.93%. The minimum p-distance to nearest neighbour, A. angustumella, is 7.14%.
Biology: Early stages unknown. The specimens were attracted to light during August and September at altitudes between 500 and 1400 m.
Distribution in the Canary Islands (Spain): Known from a few locations in the central parts of Gran Canaria. Probably endemic to Gran Canaria.
Etymology: The species is named after the small town Ayacata situated near the type-locality.
Aproaerema pseudomercedella Falck & Karsholt, sp. nov. (Figures 29, 30, 49, 62)
Holotype ♀: SPAIN, LA GOMERA, El Cedro, 1000 m, 25-VII-1984, leg. P. Olsen, B. Skule & P. Stadel Nielsen, DNA sample TLMF Lep.25524 (ZMUC).
Paratypes: SPAIN, LA GOMERA, El Cedro, 870 m, 3 ♂, 1 ♀, 25-VII-1984, leg. P. Olsen, B. Skule & P. Stadel Nielsen, genitalia slide 5064OK, same data but 1 ♂, 1 ♀, 17-20-VIII-2018, K. Larsen (all ZMUC), same data but 1 ♂, 2 ♀, 9-12-VIII-2021, leg. P. Falck, genitalia slide 3966PF, 3970PF, 3977PF, DNA samples Lepid Phyl 1433PF/CILEP1432-24, 1434PF/CILEP1433-24 (PF). EL HIERRO, Cruz de los Reyes, 1250 m, 4 ♂, 8 ♀, 22-VII-3-VIII-2022, leg. P. Falck, genitalia slide 3964PF, 3969PF, 3976PF, GP4071PF, DNA samples Lepid Phyl 1435PF/CILEP1434-24, 1436PF/CILEP1435-24; Jinama, 1250 m, 3 ♂, 4 ♀, 22-VII-3-VIII-2022, leg. P. Falck; Frontera, 280 m, 1 ♂, 22-VII-3-VIII-2022, leg. P. Falck (PF, MNCN).
Diagnosis: A. pseudomercedella is similar to A. ayacatella and A. mercedella q.v. In the male genitalia the relatively long phallus and the vesica without or with very few spinules are characteristic. In the female genitalia the short narrow ductus bursae and the very long, narrow, weakly granulated corpus bursae are characteristic and that distinguishes it from A. ayacatella and A. mercedella.
Description Adult (Figures 29, 30): Wingspan 10-12,5 mm. Labial palp slender, upturned, segment 2 dark brown posteriorly and medially yellowish white, segment 3 slightly shorter than segment 2, yellowish white, apical half blackish brown. Antenna dark grey with yellowish rings. Head, neck and thorax yellowish. Tegula dark brown, yellowish at base. Forewing blackish brown; basally an indistinct yellowish brown spot between costa and dorsum; from costa near the base to the middle a yellowish, irregular-shaped spot often fused with the basal spot to near dorsum, continuing as an indistinct median streak to beyond the postmedian fascia; at the end of cell a distinct black spot, edged white; apically at costa and from near tornus two outwardly, distinct, yellowish lines, almost forming a postmedian fascia. Fringe yellowish apically, grey basally, forming two parallel grey fringe-lines. Hindwing grey, with grey fringe. Abdomen grey, with yellowish anal tufts.
Variation: Specimens from La Gomera are on average slightly smaller, have a darker colouration of the forewings and segment 3 of the labial palp is blackish brown in apical third.
Male genitalia (Figure 49): Tegumen oblong, lateral margins convex, anterior margin almost without emargination; uncus slender, apex rounded; gnathos subtriangular, shorter than uncus, lateral arms well developed, apex slender with a small hook; valva simple, narrow, digitate, setose, longer than tegumen process; vinculum subtriangular, posteriorly rounded, anteriorly with lateral arms; juxta small, rounded, covered with small spines; phallus straight, tapering towards apex, longer than vinculum; vesica without or with very few spinules.
Female genitalia (Figure 62): Papillae anales elongate, distally pointed; posterior apophysis long, twice as long as anterior apophysis; segment VIII cylindrical, shorter than anterior apophysis, medially membranous; antrum membranous; ductus bursae relatively narrow and short; corpus bursae very long, narrow, anteriorly slightly widening, weakly granulated.
DNA barcodes (Figure 66): We obtained full length DNA barcodes (658 bp) from three specimens and DNA barcode fragments of 595 bp and 608 pb from two specimens. The barcodes fall within three Barcode Index Numbers (BIN) BOLD: ADM9690 (1 specimen from La Gomera), BOLD: AFS8444 (2 specimens from La Gomera) and BOLD AFS8444 (two specimens from El Hierro). The maximum intraspecific p-distance is very high 6.81%. The distance between BINs is 3.85% (ADM9690, La Gomera vs.AFS8229, El Hierro), 5.92% (AFS8444, La Gomera vs. AFS8229, El Hierro) and 6.81% (ADM9690, La Gomera vs. AFS8444, La Gomera). The minimum p-distance to nearest neighbour, Aproaerema angustumella sp. nov., is 6.01%.
Biology: Early stages and hostplant are unknown. Adults have been collected at light in July and August at altitudes from 250 m to 1250 m.
Distribution: Known only from the islands of La Gomera and El Hierro (Spain) and only from few locations. Probably endemic to La Gomera and El Hierro.
Etymology: The species name is derived from combining the Greek word ψευδο (pseudo = false) and mercedella, referring to its similarity to A. mercedella.
Aproaerema angustumella Falck & Karsholt, sp. nov. (Figures 31, 32, 50, 50a, 63)
Holotype ♀: SPAIN, TENERIFE, El Caletón, 35 m, 19-IX-2-X-2023, leg. P. Falck (ZMUC).
Paratypes: SPAIN, TENERIFE, Las Mercedes, 750 m, 1 ♂, 4 ♀, 13-26-VIII-2019, leg. P. Falck, genitalia slide 4002PF, 4003PF, DNA sample Lepid Phyl 0666PF/CILEP665-20 (PF, MNCN); Aguamansa, 1050 m, 1 ♂, 13-26-VIII-2019, leg. P. Falck (PF); El Caletón, 35 m, 2 ♂, 3 ♀, 19-IX-2-X2023, leg. P. Falck, genitalia slide 4012PF, DNA samples Lepid Phyl 1557PF/CILEP1556-24, 1559PF/CILEP1558-24 (PF); La Tierra del Trigo, 500 m, 1 ♂, 1 ♀, 19-IX-2-X-2023, leg. P. Falck (PF), DNA sample Lepid Phyl 1560PF/CILEP1559-24 (PF). LA PALMA, La Galga, 400 m, 1 ♂, 29-VII-11- VIII-2023, leg. P. Falck, genitalia slide 4052PF, DNA sample Lepid Phyl 1394PF/CILEP1393-23 (PF).
Diagnosis: A. angustumella is very distinctive because of its small size and black forewing with two distinct white fasciae. It may resemble females of Scrobipalpa feralella (Zeller, 1872), but in Scrobipalpa segment 2 of the palp is much more rough-scaled. In the male genitalia the posteriorly slender gnathos and the 5-7 very small cornuti are characteristic. In the female genitalia the long posterior apophysis, the relatively narrow ductus bursae and the rounded corpus bursae are characteristic.
Description Adult (Figures 31, 32): Wingspan 6.5-9 mm. Labial palp slender, upturned, segment 2 dark grey, distally white, segment 3 slightly shorter than segment 2, black, anteriorly white. Antenna black with whitish rings. Head and neck dark grey. Thorax and tegula blackish brown. Forewing blackish; basally an indistinct white spot; from costa 1⁄4 an oblique, white fascia almost reaching dorsum, at 2/3 a post-median white fascia from costa to tornus; just below apex a very small white spot. Fringe blackish grey. Hindwing dark grey. Fringe grey.
Male genitalia (Figures 50, 50a): Tegumen oblong, lateral margins convex, anterior margin almost without emargination; uncus slender, slightly spatulate, apex rounded; gnathos subtriangular, shorter than uncus, lateral arms well developed, apex slender with a small hook; valva simple, narrow, digitate, setose, longer than tegumen process; vinculum subtriangular, posteriorly rounded, anteriorly with lateral arms; juxta small, rounded, covered with small spines; phallus straight, tapering towards apex, slightly longer than vinculum, 5-7 very small cornuti (n=2).
Female genitalia (Figure 63): Papillae anales elongate, distally pointed; posterior apophysis long, twice as long as anterior apophysis; segment VIII cylindrical, same length as anterior apophysis, medially membranous; antrum membranous; ductus bursae relatively narrow, anteriorly slightly broadening; corpus bursae rounded.
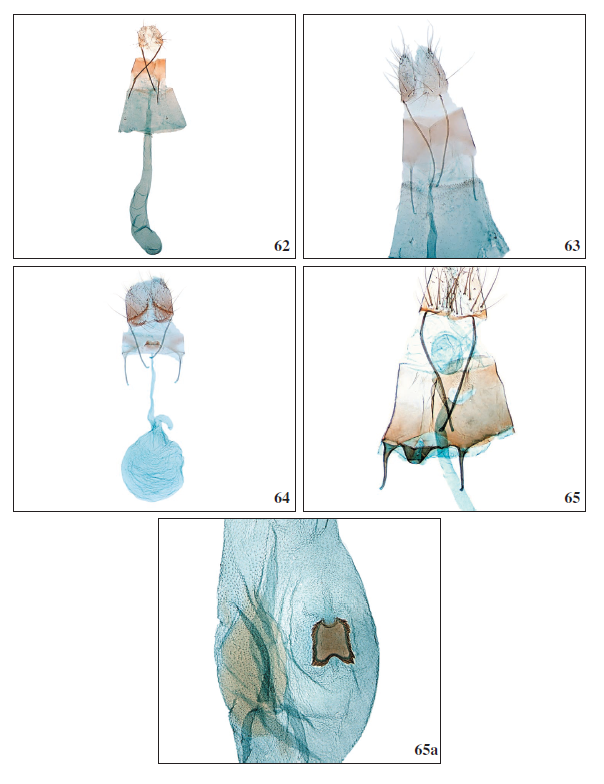
Figures 62-65a.
62. Aproaerema pseudomercedella Falck & Karsholt, sp. nov., ♀, El Hierro, GP3976PF. 63.Aproaerema angustumella Falck & Karsholt, sp. nov., 0, Tenerife, GP4003PF. 64. Mesophleps ochracella (Turati, 1926), ♀, Tenerife, GP3996PF. 65.Anarsia acaciae Walsingham, 1896, ♀, antrum complex, Tenerife, GP3960PF. 65a.Anarsia acaciae Walsingham, 1896, ♀, corpus bursae, GP3960PF.
DNA barcodes (Figure 66): We obtained full length DNA barcodes (658 bp) from four specimens and DNA barcode fragments of 601 bp from one specmen. The barcodes fall within Barcode Index Number (BIN) BOLD: AEE3180 (Tenerife) and BOLD: AFM9356 (La Palma). The minimum divergence between BINs is 2.88%. The maximum intraspecific p-distance is high 3.63%. The minimum p-distance to nearest neighbour, A. pseudomercedella, is 4.92%.
Biology: Early stages and hostplant are unknown. Adults have been collected at light from late July to the beginning of October at altitudes from 35 m to 1050 m.
Distribution: Known only from the islands of Tenerife and La Palma (Spain). Endemic to the Canary Islands.
Etymology: The species is named after the Latin word angustum (= the narrow one) referring to the narrow wings.
Remarks: The specimen from La Palma differs slightly in the shape of gnathos from specimens from Tenerife, but we consider it is due to variation.
Mesophleps Hübner, [1825]. Verz. bek. Schmett., 406
A genus of 39 species distributed through the temperate to tropical parts of the old world, with a single species known from the Americas (Li & Sattler, 2012). All five European species occur in the Canary Islands (Spain).
Segment 2 of labial palp with dorsal scale brush, more or less hiding short segment 3, and yellowish forewings with blackish brown markings.
The genitalia of all species are figured by Li & Sattler (2012).
Mesophleps corsicella Herrich-Schäffer, [1856]
Mesophleps corsicella Herrich-Schäffer, 1856. Neue Schmett. Eur., (1), 7, fig. 47
Material examined: SPAIN, GRAN CANARIA, Ayacata, 1400 m, 1 ♀, 17-30-IX-2018, leg. P. Falck, DNA sample Lepid Phyl 1554PF/CILEP1553-24. LA PALMA, Barranco del Rio, 450 m, 1♀, 29-VII-11- VIII-2023, leg. P. Falck, DNA sample Lepid Phyl 1556PF/CILEP1555-24. TENERIFE, 5 km S Aguamansa, 1600 m, 1 ♀, 1-13-VI-2022, leg. P. Falck, DNA sample Lepid Phyl 1555PF/CILEP1554- 24; Ifonche, 1040 m, 1 ♂, 21-V-3-VI-2019, leg. P. Falck, genitalia slide GP4030PF (all PF).
Diagnosis: A medium-sized species (wingspan 9-18 mm) having relatively wide, pale-yellow forewings more or less overlaid with blackish brown. The characteristic labial palps have segment 2 keel-shaped, with upturned brush and segment 3 tiny. Similar to M. silacella, which is warmer yellow and has less blackish brown on the forewings.
DNA barcodes (Figure 66): We obtained full length DNA barcodes (658 bp) from three specimens all from La Palma. The barcodes fall within Barcode Index Number (BIN) BOLD: AFR9724. The intraspecific p-distance is 0%. The minimum p-distance to nearest neighbour, M. corsicella from Greece, is 1.28% with BIN BOLD: AFR9723.
Biology: The larva feeds in seed capsules of Cistus L. and Helianthemum Mill. species (Li & Sattler, 2012, p. 35). It has not been found in the Canary Islands (Spain).
Distribution in the Canary Islands (Spain): First recorded by Klimesch (1984, p. 166) from Tenerife. New island records: Gran Canaria, San Bartolome de Tirajana, 7-IX-2012, leg. A. Werno (AW); La Palma, Barranco del Río, 29-VII-11-VIII-2023, leg. P. Falck (PF).
General distribution: Canary Island (Spain), Southern Europe from Portugal to Croatia and Greece; Morocco, Lebanon (Karsholt et al. 2023, p. 105).
Mesophleps silacella (Hübner, 1796)
Tinea silacella Hübner, 1796. Samml. eur. Schmett., 8, 63, pl. 17, fig. 117
Diagnosis: A medium sized (wingspan 9-18 mm) species having yellow forewings, with violet brownish scales in the terminal area. The characteristic labial palps have segment 2 keel-shaped, with upturned brush and segment 3 tiny.
Biology: The larva feeds in seed capsule of Helianthemum Mill. It has not been found in the Canary Islands (Spain).
Distribution in the Canary Islands (Spain): Lanzarote: Puerto del Carmen, 1-II-1994, leg. J. P. Baungaard (ZMUC); Fuerteventura, Jandia, Cofete, 460 m, 1-V-1987, leg. W. Schmitz (ZMUC). M. silacella is widely distributed on Lanzarote and Fuerteventura. New to the Canary Islands.
General distribution: Canary Islands (Spain); widely distributed in Central and South Europe to Russia, more disjunctive in North Europe; North Africa, Turkey (Gregersen & Karsholt, 2022, p. 109).
Mesophleps oxycedrella (Millière, 1871)
Gelechia oxycedrella Millière, 1871. Icon. Desc. Chenilles Lépid., 3, 177, 193, pl. 118, figs 1-6
Diagnosis: The yellowish forewings with 4-5 large blackish brown spots make this species unmistakably. The species is variable in size (7-12 mm).
Biology: In the Canary Islands the larva feeds in berries/cones of Juniperus canariensis Guyot (= J. turbinata ssp. canariensis Guyot), and overwinters therein, before pupating on the ground (Klimesch, 1984, p. 166;Guido & Roques, 1996).
Distribution in the Canary Islands (Spain): First recorded by Klimesch (1984, p. 166) from La Gomera. Subsequently from Gran Canaria and La Palma (Báez & Martín, 2001, p. 238) and from Tenerife and El Hierro (Li & Sattler, 2012, p. 36).
General distribution: Canary Islands (Spain), western part of southern Europe and North Africa; Croatia.
Mesophleps trinotella Herrich-Schäffer, [1856]
Mesophleps trinotella Herrich-Schäffer, 1856. Neue Schmett. Eur., (1), 6, fig. 46
Diagnosis: A rather small species (wingspan 8-14 mm). Segment 2 of labial palp with subtriangular scale tuft, about twice length of segment 3; forewing slender, ochre, costal and dorsal margin and apex blackish brown. May be confused with yellowish specimens of the more broad-winged M. ochracella, which has longer segment 3 of the labial palp.
Biology: The larva feeds in seeds of Erysimum L. and Moricandia DC. (Cruciferae). It has not been found in the Canary Islands (Spain).
Distribution in the Canary Islands (Spain): First recorded by Arenberger (1999, p. 13) from Fuerteventura. New island records: Gran Canaria, Fataga, 21-VIII-4-IX-2020, leg. P. Falck (PF); Lanzarote, Urb. Famara, 55 m, 2-8-XI-2018, leg. C. Hviid & B. Skule (ZMUC).
General distribution. Canary Islands (Spain), southern Europe, Croatia, Czech Republic, Hungary, Cyprus, Turkey, North Africa.
Mesophleps ochracella (Turati, 1926) (Figures 33-36, 51, 64)

Figures 33-40.
33. Mesophleps ochracella (Turati, 1926), ♂, Lanzarote, 12.5 mm. 34.Mesophleps ochracella (Turati, 1926), ♀, Lanzarote, 13 mm. 35. Mesophleps ochracella (Turati, 1926), ♀, Fuerteventura, 12 mm. 36.Mesophleps ochracella (Turati, 1926), ♂, El Hierro, 12 mm. 37.Nothris congressariella ssp. tenerifensis Falck & Karsholt, ssp. nov., ♂, Tenerife, 18 mm. 38.Nothris congressariella ssp. tenerifensis Falck & Karsholt, ssp. nov., ♀, Tenerife, 18.5 mm. 39.Anarsia acaciae Walsingham, 1896, ♂, Gran Canaria, 13 mm. 40.Anarsia acaciae Walsingham, 1896, ♀, Fuerteventura, 13 mm.
Nothris ochracella Turati, 1926, Atti Soc. ital. Sci. nat., 65, 69, fig. 32
Material examined: SPAIN, FUERTEVENTURA, Caldereta, 120 m, 1 ♂, 1 ♀, 27-II-19-III-2018, leg. P. Falck, DNA sample Lepid Phyl 0066PF/CILEP66-18, genitalia slide GP4000PF; Betancuria, 400 m, 1♀, 7-27-XI-2017, leg. P. Falck, DNA sample Lepid Phyl 0065PF/CILEP65-18. LANZAROTE, Caleta de Famara, 20 m, 1 ♂, 1♀, 1-13-II-2022, leg. P. Falck, DNA sample Lepid Phyl 1549PF/CILEP1548-24, genitalia slide GP3999PF; El Golfo, 95 m, 1 ♀, 1-13-II-2022, leg. P. Falck, DNA sample Lepid Phyl 1550PF/CILEP1549-24; Femes, 340 m, 1 ♂, 1-13-II-2022, leg. P. Falck, genitalia slide GP3997PF. TENERIFE, Arona, 600 m, 1 ♂, 2 ♀, 3-16-VIII-2021, leg. P. Falck, DNA sample Lepid Phyl 1553PF/CILEP1552-24, genitalia slide GP3994PF, GP3996PF. EL HIERRO, Erese, 700 m, 1 ♂, 2 ♀, 22- VII-3-VIII-2022, leg. P. Falck, DNA sample Lepid Phyl 1551PF/CILEP1550-24, genitalia slide GP3998PF, GP4001PF; Tacorón, 170 m, 2 ♀, 22-VII-3-VIII-2022, leg. P. Falck, DNA sample Lepid Phyl 1552PF/CILEP1551-24, genitalia slide GP3995PF (all PF).
Diagnosis: A medium-sized species (wingspan 9-16 mm). Segment 2 of labial palp with subtriangular scale tuft, segment 3 almost as long as segment 2; forewing variable, from light yellowish brown to almost black, often with an orange streak before termen (Figure 33-36).
DNA barcodes (Figure 66): We obtained full length DNA barcodes (658 bp) from seven specimens from El Hierro (2), Tenerife (1), Fuerteventura (2) and Lanzarote (2). The barcodes fall within Barcode Index Number (BIN) BOLD: AAV4323. The maximum intraspecific p-distance is 0.48% (n = 41). The minimum p-distance to nearest neighbour (a Pyraustinae) is 8.33%.
Biology: Hostplant unknown.
Distribution in the Canary Islands (Spain): LANZAROTE, 0.8 km S Coril, 1.4 km N Tias, 240 m, 2- 8-XI-2018, leg. C. Hviid & B. Skule (ZMUC). FUERTEVENTURA, Corralejo, 14-20-XII-1996, leg. K. Larsen (ZMUC). LA PALMA, El Paso, 400 m, 16-II-2004, leg. H. van der Wolf (RMNH). EL HIERRO, Erese, 22-VII-2-VIII-2022, leg. P. Falck (PF). M. ochracella is common on Lanzarote, Fuerteventura and El Hierro more locally on Tenerife. New to the Canary Islands.
General distribution: Canary Islands (Spain), Mediterranean countries from Portugal and France to Cyprus.
Chelariini
Nothris Hübner, [1825], Verz. bek. Schmett., (23-27), 411
Nothris congressariella (Bruand, 1858) (Figure 37, 38)
Ypsolopha congressariella Bruand, 1858. Ann. Soc. ent. Fr., (3) 6, 471, pl. 11, fig. 7, 7a
Nothris declaratella Staudinger, 1859. Stett. ent. Ztg., 20(7-9), 238
Remarks: This species is in the Canary Islands (Spain) restricted to the mountains of Tenerife. The population there differs in colour and forewing markings from the nominotypical subspecies occurring from the southernmost parts of the British Isles across Europe to North Africa, Turkey and Iran. It is described below as a new subspecies.
Nothris congressariella tenerifensis Falck & Karsholt, ssp. nov. (Figure 37, 38)
Holotype ♀: SPAIN, TENERIFE, Las Cañadas del Teide, 2200 m, e. l. 16-VII-1965, Scrophularia glabrata Aiton. L., leg. J. Klimesch (ZMUC).
Paratypes: SPAIN, TENERIFE, Las Cañadas del Teide, 2200 m, 1 ♀ e. l. 26-IX-1966, Scrophularia glabrata, leg. J. Klimesch (ZMUC); Las Cañadas, 2000 m, 1 ♀, 30-VII-1979, leg. P. Stadel Nielsen (ZMUC); Las Cañadas, Liano de Ucanca, 2200 m, 1 ♀, 1 ♂, e. l. 7-1-1998, leg. K. Larsen (ZMUC); Villaflor, Las Lajas, 1800 m, 1 ♀, 1 ♂, 24-IV-1998, leg. K. Larsen (ZMUC); 5 km N San Andrés, 2 ♀, 2 ♂, 25-IV-1998, leg. K. Larsen (ZMUC); 8 km N Vilaflor, 1700 m, 23 ♂, 19 ♀, 21-V-3-VI-2019, leg. P. Falck, DNA samples Lepid Phyl 0336PF/CILEP335-19, Lepid Phyl 0337PF/CILEP336-19, genitalia slide GP4028PF; 5 km S Aguamansa, 1400 m, 4 ♂, 1 ♀, 21-V-3-VI-2019, leg. P. Falck, genitalia slide GP4027PF (PF, MNCN).
Diagnosis: A rather large gelechiid (wingspan 16-21 mm) with brown forewings with two black spots in the middle of the wing and sometimes a black patch near base. It differs from the nominotypical subspecies by its darker brown forewings, by having less (or no) whitish at the basal half of costa, and by being almost without black on the veins in the apical part of the forewing. Segment 2 of the labial palp with a large projecting tuft, laterally compressed with scales projecting forward and downwards separates it from similar looking species in the Canary Islands (Spain).
The male and female genitalia are similar to those of the nominotypical subspecies. They are figured by Klimesch (1984, p. 179) and Karsholt & Šumpich (2015, pp. 480, 494-495).
DNA barcodes (Figure 66): We obtained full length DNA barcode (658 bp) from one specimens and DNA barcode fragments of 602 bp from one specimen. The barcodes fall within Barcode Index Number (BIN) BOLD: AAE5563. The maximum intraspecific p-distance is 1.44% (n = 11). The minimum divergence between populations from Europe and The Canary Islands is 1.02%. The minimum p-distance to nearest neighbour, Nothris verbascella ([Denis & Schiffermüller], 1775), is 4.52%.
Biology: The larva feeds on Scrophularia glabrata, at first mining the leaves and later among leaves spun together (Klimesch, 1984, pp. 160, 180, fig. 64). N. congressariella tenerifensis is commonly found in the pine-forest zone (700-2400 m) as a larva among spun leaves of its hostplant, occasionally adults are attracted to light.
Distribution: First recorded by Klimesch (1984, p. 159). Only known from the mountains of Tenerife, at altitudes between 1400 and 2200 m.
Etymology. The subspecies is named after the island of Tenerife.
Remarks: Klimesch (1984, p. 169) noted that the Canary Islands population may deserve status as a subspecies. However, he refrained from naming it. We have examined material of N. congressariella from its total distribution area in order to evaluate the distinctiveness of the population occurring in Tenerife. See also our discussion about subspecies in the discussion chapter at the end of this paper.
Anarsia Zeller, 1839, Isis von Oken, 1839, 190
A large genus which in its current concept includes more than 100 described species distributed throughout the Old World. Males have segment 3 of the labial palp reduced, and often asymmetric genitalia.
Anarsia lineatella Zeller, 1839 (Figures 52, 52a)
Anarsia lineatella Zeller, 1839. Isis von Oken, 1839, 190
Material examined: SPAIN, GRAN CANARIA, Barranco de Guayadeque, 460 m, 2 ♂, 1 ♀, 4-23-III2019, leg. P. Falck, DNA samples Lepid Phyl 1455PF/CILEP1454-24, Lepid Phyl 1456PF/CILEP1455-24, genitalia slide GP3959PF, GP3962PF; Ayacata, 1400 m, 1 ♂, 1♀, 4-23-III2019, leg. P. Falck, genitalia slide GP3954PF, GP3955PF (all PF).
Diagnosis: Wingspan 11-15 mm. Similar to A. acaciae Walsingham, 1896 (see that species). The genitalia are figured by, e.g., Karsholt & Gregersen (2017).
DNA barcodes (Figure 66): We obtained DNA barcode fragments of 621bp and 603pb from two specimens. The barcodes fall within Barcode Index Number (BIN) BOLD: AAD7849. The maximum intraspecific p-distance is 0.48% (n = 21). The minimum p-distance to nearest neighbour, A. innoxiella Gregersen & Karsholt, 2017 is 1.75%.
Biology: The larvae feed in fruits of different Rosaceae, especially Prunus L. to which they may be harmful. To our knowledge it has not been found in the Canary Islands.
Distribution in the Canary Islands (Spain): A. lineatella is common in several locations on the island of Gran Canaria. New to the Canary Islands. Perhaps recently introduced.
General distribution: Central and southern Europe and North Africa, eastwards through the Middle East and Turkey to Central Asia and China; introduced to North America.
Anarsia acaciae Walsingham, 1896 (Figures 39, 40, 53, 53a, 65, 65a)
Anarsia acaciae Walsingham, 1896. Proc. zool. Soc. Lond., 1896, 278
Material examined: SPAIN, FUERTEVENTURA, Corralejo, 10 m, 1 ♀, 7-27-XI-2017, leg. P. Falck, genitalia slide GP3957PF; Caldereta, 120 m, 7-27-XI-2017, leg. P. Falck, genitalia slide GP3953PF. LANZAROTE, Mojón Blanco, Orzola, 1 ♂, 1 ♀, 20 m, 21-X-10-XI-2019, leg. P. Falck, DNA sample Lepid Phyl 1454PF/CILEP1453-24, genitalia slide GP3961PF; Femes, 340 m, 1 ♀, 1-13-II-2022, leg. P. Falck, DNA sample Lepid Phyl 1452PF/CILEP1451-24. GRAN CANARIA, El Sao, 110 m, 3 ♂, 1 ♀, 4- 23-III-2019, leg. P. Falck, DNA samples Lepid Phyl 1451PF/CILEP1450-24, Lepid Phyl 1453PF/CILEP1452-24, genitalia slide GP3956PF, GP4031PF (all PF).
Diagnosis: Wingspan 13-16 mm. Colour and wing markings resemble A. lineatella but differ in the lighter basal part of costa. Males are easily recognised by a characteristic black hair pencil from the base of costa in the hindwings (figure 39). Such a hair pencil is absent in A. lineatella. The male genitalia (figures 53, 53a) are less asymmetric than those of A. lineatella (figure 52, 52a). The female genitalia (figures 65, 65a) differ from those of A. lineatella in having longer anterior apophyses.
DNA barcodes (Figure 66): We obtained DNA barcode fragments of 624bp, 621bp, 602bp and 589pb from four specimens. The barcodes fall within Barcode Index Number (BIN) BOLD: ADA2395. The maximum intraspecific p-distance is 1.36% (n = 24). The minimum p-distance to nearest neighbour, Anarsia sp., is 3.01%.
Biology: The larva feeds on Acacia Mill. and related Fabaceae. In the Canary Islands it has been found on Vachellia farnesiana (L.) Wight. & Arn. (Klimesch, 1984, p. 165, as Acacia farnesiana).
Distribution in the Canary Islands (Spain): First recorded by Klimesch (1984, p. 165) from La Gomera. New island records: Fuerteventura, Corralejo, 27-II-19-III-2018, leg. P. Falck (PF); Lanzarote, Mojón Blanco, Orzola, 21-X-10-XI-2019, leg. P. Falck (PF); Gran Canaria, Maspalomas, 6- XI-1991, leg. P. Grotenfelt (MZH); Tenerife, Arona, 1-20-III-2017, leg. P. Falck (PF).
General distribution: Canary Islands (Spain), North Africa, Middle East, Yemen, East Africa.
Discussion
When we started this series of papers dealing with Lepidoptera of the Canary Islands, we stated that the Lepidoptera of the islands are considered to be well-known (Falck et al. 2019, p. 34). This is not the case, especially for the Microlepidoptera. The most recent checklist of Canary Island Lepidoptera (Vives Moreno, 2014) lists 11 species of Anacampsinae. Here we record 23 species. We still do not have the exact number of species occurring in these islands for the remaining subfamilies of Gelechiidae which we plan to publish in three forthcoming papers, but they include also many species which are either new for the islands or new to science.
Among the Anacampsinae of the Canary Islands the tribe Anacampsini is the largest with 20 species, whereas the Chelariini includes only three species. The species of Anacampsini are placed in three genera with seven species of Stomopteryx, eight species of Aproaerema and five species of Mesophleps. Whereas all species of Mesophleps occur also in continental Europe, the two other genera have diversified in the Canary Islands with respectively five and four endemic species. In particular the Aproaerema mercedella group is specialized, and their closest relatives are currently unknown.
In this paper we describe two new subspecies and synonymise one. Whereas the category of subspecies is widely used for Papilionoidea and Macroheterocera it is little used in the Gelechiidae. This is partly because many Gelechiidae are rather uniformly, dull coloured with more or less indistinct markings, but probably also because taxonomists of this family have been busy in describing new species and thus paid less attention to geographical variation. It may therefore be relevant to deal briefly with the term subspecies.
Geographic variation is often termed as a subspecies. This category is below species level and covers populations which differ from the main (nominotypical) population of a species. Whereas there is overall agreement on what constitutes a species within Lepidoptera this is not the case for subspecies. That is much more up to individual researchers’ preferences and decisions. Often subspecies are populations with different colour or wing markings, but with similar genitalia. Most authors would agree that a subspecies must be geographically separated from the main population of the species (= the nominotypical subspecies), but not all insist that there should be no overlap in distribution or appearance of the adult. It may also influence the opinion if the population in question has a different bionomy (e.g. another hostplant) and also the size of the adults may be considered.
One may consider subspecies as a stage in the evolutionary process where a species is in a process of splitting into two or more species due, e.g., to geographical separation, changes in climatic conditions or adaptations to new niches. In such cases we find it relevant to name subspecies, thereby making both the scientific community and local authorities aware of the presence of populations which need taxonomic or conservation attention.
We give status as subspecies to populations which differ from the nominotypical subspecies both in the DNA barcode and in the colour and markings of the forewing, but where we could not find morphological differences in the genitalia. This is not the case with Aproaerema anthyllidellaelachistella (Klimesch, 1984). Here specimens with the characters of the forewings (strongly marked females) are also found among populations in continental Europe, there are only slight/unclear differences in the DNA barcode, and we could not find differences in the genitalia and so treat elachistella as a junior synonym of A. anthyllidella.
Acknowledgments
We are grateful to the following for providing help during the preparation of the manuscript of this paper: Marcos Báez (La Laguna, Tenerife, Spain); Dr. Lauri Kaila (Finnish Museum of Natural History, Helsinki, Finland); Willibald Schmitz (Bergisch Gladbach, Germany); Andreas Werno (Nunkirchen, Germany); Dr. Hugo van der Wolf (Neunen, The Netherlands) for loan of specimens, and to Carsten Hviid, Knud Larsen, Bjarne Skule and Per Stadel Nielsen (all Denmark) for presenting specimens, which were used in this paper, to the collection of ZMUC; to Dr. Oleksiy Bidzilya (Staatliches Museum für Naturkunde Stuttgart, Germany) and Dr. Peter Huemer (Tiroler Landesmuseum, Innsbruck, Austria) for information; to Stella D. Beavan (Zeal Monachorum, UK) and Robert J. (Bob) Heckford (Plymouth, UK) who read the final manuscript carefully and improved both the English language and the content; to Javier Gastón (Bilbao, Spain) for the photographic review and adjustments; and finally Dr. Antonio Vives (Madrid, Spain) for translating the abstract into Spanish, for editing our manuscript, and for his help with obtaining permission to collect Lepidoptera in the Canary Islands for the Scientific Project of SHILAP.
References
Arenberger, E. (1999). Microlepidoptera von Fuerteventura (Kanarische Inseln) (Insecta: Lepidoptera). SHILAP Revista de lepidopterología, 27(105), 11-18.
Báez, M., & Martín, E. (2001). Orden Lepidoptera. Pp. 236-249. InIn M. Arechavaleta, S. Rodríguez, N. Zurita & A. García (eds). Lista de especies silvestres de Canarias (hongos, plantas y animales terrestres). Gobierno de Canarias.
Báez, M., & Martín, E. (2004). Orden Lepidoptera. In I. Izquierdo, J. L. Martin, N. Zurita & M. Arechavaleta (eds). Lista de especies silvestres de Canarias (hongos, plantas y animales terrestres)(2nd edn., pp. 247-260). Gobierno de Canarias. https://chrome-extension://efaidnbmnnnibpcajpcglclefindmkaj/http://bba.bioucm.es/cont/docs/456.pdf
Báez, M., & Oromi, P. (2010). Planipennes, Trichoptera, Lepidoptera, Diptera, Hymenoptera. Pp. 302-328. In M. Arechavaleta, S. Rodríguez, N. Zurita & A. García (eds). Lista de especies silvestres de Canarias. Hongos, plantas y animales terrestres. 2009 (pp. 302-328). Gobierno de Canarias. https://chrome-extension://efaidnbmnnnibpcajpcglclefindmkaj/https://islandlab.uac.pt/fotos/publicacoes/publicacoes_Lista_Especies_Silv estres_Canarias2010.pdf
Falck, P., & Karsholt, O. (2023). The Symmocinae and Holcopogoninae in the Canary Islands and Madeira, with descriptions of 13 new species (Lepidoptera: Autostichidae). SHILAP Revista de lepidopterología, 51(202), 269-314. https://doi.org/10.57065/shilap.462
Falck, P., Karsholt, O., & Slamka, F. (2019). New data on Pyraloidea from the Canary Islands, Spain (Lepidoptera: Pyraloidea). SHILAP Revista de lepidopterología, 47(185), 33-48. https://doi.org/10.57065/shilap.725
Guido, M., & Roques, A. (1996). Impact of the phytophagous insect and mite complex associated with cones of Junipers (Juniperus phoenicea L. and J. cedrus Webb and Berth.) in the Canary Islands. Ecologia mediterranea, 22, 1-10. https://doi.org/10.3406/ecmed.1996.1797
Hering, M. (1927). Die Minenfauna der Canarischen Inseln. Zoologische Jahrbücher, Abteilung für Systematik, Ökologie und Geographie der Tiere, 53, 405-486.
Huemer, P., & Karsholt, O. (2010). Gelechiidae II (Gelechiinae: Gnorimoschemini). In P. Huemer, O. Karsholt & M. Nuss (eds). Microlepidoptera of Europe (Vol. 6). https://doi.org/10.1163/9789004260986
Huemer, P., & Karsholt, O. (2020). Commented checklist of European Gelechiidae (Lepidoptera). ZooKeys, 921, 65140. https://doi.org/10.3897/zookeys.921.49197 PMid:32256151 PMCid:PMC7109147
Huemer, P., Karsholt, O., Aarvik, L., Berggren, K., Bidzilya, O., Junnilainen, J., Landry, J.-F., Mutanen, M., Nupponen, K., Segerer, A., Šumpich, J., Wieser, C., Wiesmair, B., & Hebert, P. D. N. (2020). DNA barcode library for European Gelechiidae (Lepidoptera) suggests greatly underestimated species diversity. ZooKeys, 921, 141-157. https://doi.org/10.3897/zookeys.921.49199 PMid:32256152 PMCid:PMC7109146
Janse, A. J. T. (1949-63). Gelechiadae. The Moths of South Africa, (Vol. 5, 1949-54; Vol. 6, 1958-63). Pretoria.
Karsholt, O. (2004-2024). Gelechiidae. In O. Karsholt & E. J. van Nieukerken (eds): Lepidoptera. Fauna Europaea [last update of Gelechiidae: version 2.4 January 2011]. https://fauna-eu.org/
Karsholt, O., Mutanen, M., Lee, S., & Kaila, L. (2013). A molecular analysis of the Gelechiidae (Lepidoptera, Gelechioidea) with an interpretative grouping of its taxa. Systematic Entomology, 38, 334-348. https://doi.org/10.1111/syen.12006
Karsholt, O., & Šumpich, J. (2015). A review of the genus Nothris Hübner, 1825, with description of new species (Lepidoptera: Gelechiidae). Zootaxa, 4059, 471-498. https://doi.org/10.11646/zootaxa.4059.3.3
Karsholt, O., & Šumpich, J. (2018). A new Stomopteryx Heinemann, 1870 from southern Spain (Lepidoptera: Gelechiidae). SHILAP Revista de lepidopterología, 46(182), 235-240. https://doi.org/10.57065/shilap.821
Karsholt, O., Baldizzone, G., & Gomboc, S. (2023). Overview of the Gelechiidae moths (Lepidoptera) of Krk Island, Croatia: diversity, distribution and ecological insights. Gortania, Botanica, Zoologia, 45, 95-124. https://www.researchgate.net/publication/377635209https://www.dropbox.com/scl/fi/3rl3payqbxm961khdusna/Karsholt-Baldizzone-Gomboc-2023-Gelechiidae-ofKrk.pdf?rlkey=lqb4emq5asvmvh34hc00ewww7&dl=0
Klimesch, J. (1984). Beiträge zur Kenntnis der Microlepidopteren-Fauna des Kanarischen Archipels. 6. Beitrag: Gelechiidae. Vieraea, 13(1983), 145-182. https://bibdigital.rjb.csic.es/viewer/16261/?offset=#page=147&viewer=picture&o=bookmark&n=0&q=
Leraut, P. (1997). Liste systématique et synonymique des Lépidoptères de France, Belgique at Corse. Alexanor (Supplement).
Li, H., & Sattler, K. (2012). A taxonomic revision of the genus Mesophleps Hübner, 1825 (Lepidoptera: Gelechiidae). Zootaxa, 3373, 1-82. https://doi.org/10.11646/zootaxa.3373.1.1
Pitkin, L. M. (1986). A technique for the preparation of complex male genitalia in Microlepidoptera. Entomologist’s Gazette, 37, 173-179.
Rebel, H. (1892). Beitrag zur Microlepidopterenfauna des canarischen Archipels. Annalen des Naturhistorischen Museums Wien, 7, 241-284, pl. XVII. https://www.biodiversitylibrary.org/item/134318#page/5/mode/1up
Rebel, H. (1896). Dritter Beitrag zur Lepidopterenfauna der Canaren. Annalen des Naturhistorischen Museums in Wien, 11, 102-147, pl. III.
Rebel, H. (1938). Achter Beitrag zur Lepidopterenfauna der Kanaren. Annalen des Naturhistorischen Museums in Wien, 49, (43)-(68).
Sohn, J.-C., Regier, J. C., Mitter, C., Adamski, D., Landry, J.-F., Heikkila, M., Park, K.-T., Harrison, T., Mitter, K., Zwick, A., Kawahara, A. Y., Cho, S., Cummings M. P., & Schmitz, P. (2016). Phylogeny and feeding trait evolution of the mega-diverse Gelechioidea (Lepidoptera: Obtectomera): New insight from 19 nuclear genes. Systematic Entomology, 41, 112-132. https://doi.org/10.1111/syen.12143
Ungureanu, V., & Murariu, D. (2024). Anacampsis infestella (Rebel, 1896). https://clasate.cimec.ro/Detaliu_en.asp?tit=anacampsis-infestella-Anacampsis-infestella-Rebel-1896&k=29F5F68B07FE49A187705CD5DE6F9CB6
Vives Moreno, A. (2014). Catálogo sistemático y sinonímico de los Lepidoptera de la Península Ibérica, de Ceuta, de Melilla y de las islas Azores, Baleares, Canarias, Madeira y Salvajes (Insecta: Lepidoptera). Improitalia.
Walsingham, L. (1908). Microlepidoptera of Tenerife. Proceedings of the Zoological Society of London, 1907, 911- 1034, pls 51-53. https://www.biodiversitylibrary.org/item/98530#page/585/mode/1up
Wang, Q.-Y., & Li, H.-H. (2016). Phylogeny of the superfamily Gelechioidea (Lepidoptera: Obtectomera), with an exploratory application on geometric morphometrics. Zoologica Scripta, 49, 307-328. https://doi.org/10.1111/zsc.12407
World Flora Online (2024). https://www.worldfloraonline.org/.
Author notes
a Autor para correspondencia: per.falck@live.dk
