ARTÍCULO
COMPOSITION OF A BAT ASSEMBLAGE (MAMMALIA: CHIROPTERA) IN THE NATURAL RESERVE SALTO MORATO, EAST COAST OF PARANÁ, SOUTH BRAZIL
Composição da assembleia de morcegos (Mammalia: Chiroptera) na Reserva Natural Salto Morato, costa oeste do Paraná, sul do Brasil.
COMPOSITION OF A BAT ASSEMBLAGE (MAMMALIA: CHIROPTERA) IN THE NATURAL RESERVE SALTO MORATO, EAST COAST OF PARANÁ, SOUTH BRAZIL
Mastozoología Neotropical, vol. 27, núm. 1, pp. 53-60, 2020
Sociedad Argentina para el Estudio de los Mamíferos
Recepción: 23 Septiembre 2019
Aprobación: 20 Febrero 2020
Abstract: Brazilian fauna is characterized by megadiverse groups. Within those, there are bats that are directly responsible for important ecosystem services, such as pollination, dispersion and pest control. Despite of their importance, there are several geographical gaps in the Brazilian bat fauna knowledge. Thus, the present study aimed to analyze the composition of bat assemblage in a Conservation Unit in the Atlantic Forest biome, East Coast of Paraná state, southern Brazil. Samplings were carried out during six nights, in average, per month between 2013 and 2014. Captures were performed by 18 mist nets installed at different heights in the forest. Reaching sampling sufficiency, we found high species richness (n =25 spp.), which imply the important role of Conservation Units to maintain bat diversity. The high number of captures (n =1201) can be explained by sampling in different forest height, which may indicate the occurrence of spatial variation in habitat use. Moreover, the current results can assist the management and administration of protected areas, as well as the species conservation in situ.
Keywords: abundance patterns, Atlantic Forest, conservation, diversity, inventory, southern Brazil.
Resumo: A fauna brasileira é caracterizada por grupos megadiversos. Dentro desses grupos estão os morcegos, os quais são responsáveis por importantes serviços ecossistêmicos, como polinização, dispersão e controle de pragas. Apesar de sua importância, existem várias lacunas geográficas no conhecimento da fauna de morcegos do Brasil. Assim, o presente estudo teve como objetivo analisar a composição da assembleia de morcegos em uma Unidade de Conservação no bioma Mata Atlântica, no litoral leste do Paraná, Região Sul do Brasil. As amostragens foram realizadas entre 2013 e 2014, durante em media por seis noites mensais. As capturas foram realizadas por 18 redes de neblina instaladas em diferentes alturas na floresta. Atingindo a suficiência amostral, encontramos alta riqueza de espécies (n=25 spp.), o que implica o importante papel das Unidades de Conservação na manutenção da diversidade de morcegos. O grande número de capturas (n =1201) pode ser explicado pela amostragem em diferentes alturas na floresta, o que pode indicar ocorrência de variação espacial no uso do habitat. Além disso, os resultados atuais podem auxiliar no gerenciamento e administração de áreas protegidas, bem como na conservação de espécies in situ.
Palavras-chave: biodiversidade, conservação, inventário, Bosque Atlantico, padrões de abundância, sul do Brasil.
INTRODUCTION
Brazil is a megadiverse country in terms of its faunal groups, among which are the bats. Currently, there are 182 bat species occurring within Brazilian boundaries (Nogueira et al. 2018), which represents about 13,15% of the global bat fauna (Taylor & Tuttle 2019). Beyond the high richness, several species were described in Brazil lately (Miranda et al. 2007; Nogueira et al. 2018; Dias et al. 2013; Feijó et al. 2015; Moratelli & Dias 2015), expanding the knowledge on Brazilian bat fauna.
Among the Brazilian biomes, Amazon owns the highest specific richness – 146 spp., followed by Atlantic Forest – 118 spp., Cerrado – 101 spp., Caatinga – 77 spp., Pantanal – 60 spp., and Pampa– 24 spp. (Paglia et al. 2012; Varzinczak et al. 2015; Carvalho et al. 2017). The Atlantic Forest is the one that represents greater diversity of bats (Marinho- Filho 1996), with greater knowledge about its richness and species distribution (Bergallo et al. 2003; Bernard et al. 2011; Muylaert et al. 2017). Despite scientific knowledge increases in the Atlantic Forest, great rates of habitat loss and high levels of fragmen tation reduced the Biome to only 13% of its original coverage (Scarano & Ceotto 2015).
The maintenance of the remaining fraction of the Atlantic Forest is partly due to the implantation of Conservation Units (CU). In South America, Atlantic Forest’s protected areas increased in the last decades (Galindo & Câmara 2003; Tabarelli et al. 2005). On the other hand, the range of effectively protected territory within CUs is still small, about 2% of the total range of this Biome and, among the forest remnants 24% are protected within conservation areas (Tabarelli et al. 2005).
In Brazil, among the CUs that cover Atlantic Forest fragments, the Reserva Particular do Patrimônio Natural Salto Morato (RSM) – freely translated to “Private Reserve of the Natural Patrimony Salto Morato”, with other annex CUs in Paraná and São Paulo –, constitute the largest continuous remnant of this Biome. Specifically for bats, the East Coast of Paraná was considered as with low priority for inventories, region where the RSM is located (Miretzki 2003). Meanwhile, recent studies in this region described the occurrence of new species for the State (Scultori et al. 2009a;b;c; Carvalho et al. 2014; 2019), showing that despite the historical research, bat fauna on this portion of Atlantic Forest is still poorly known (Varzinczak et al. 2015).
To fill these knowledge gaps inventories may be carried out, which are considered base studies for conservation actions and ecological macro approaches (Bergallo et al. 2003). Although the inventories provide static perception of biodiversity patterns (Silveira et al. 2010) and for bat communities tend to vary seasonally (e.g. Moya et al. 2008; Mello 2009), the resulting information of these studies are important to manage and conserve protected areas. In this context, the present study had the goal to analyze the composition of bat assemblage in a Conservation Unity in the Atlantic Forest, East Coast of Paraná state, southern Brazil.
MATERIAL AND METHODS
Study area
Private Reserve of the Natural Patrimony Salto Morato is located in the Guaraqueçaba municipality, North Coast of Paraná state (25°09’98"N e 48°17’90"W – Fig. 1). According to the Koeppen classification, the region presents Cfa climate, which is characterized by hot summers, without a well-defined dry season (Alvares et al. 2013).
The RSM has an area of 2,340 ha that is inserted in the Environmental Protection Area of Guaraqueçaba (Straube & Urben-Filho 2005) that covers 282,444 ha (SISBIO 2015) of Atlantic Forest (IBGE 2012). Due to its colonization history, the RSM has different vegetation succession stages. We carried out samplings in a secondary vegetation, with canopy of 12 to 25 meters high of Dense Submontane Forest.
Sampling protocol
Samplings were carried out between September 2013 and August 2014, during 61 capture nights (an average of six nights monthly). Capture nights were not consecutive in cases of heavy rain, so not all months had six capture nights. The capture of bats was performed with mist nets installed at different heights, which aimed to sample the different strata (understory, sub-canopy and canopy) used by bats. Thus, for each night, 18 nets were opened – three nets of 12 x 2.5m; nine nets of 9 x 2.5 m and six nets of 6 x 2.5 m – all Avinet brand with mesh of 36 mm. Nets were installed inside and in forests edges, on trails and over a water body, opened for six hours after the twilight and checked every 20 minutes. The total sampling effort was 139,995 m2.h (Straube & Bianconi 2002).
The bats captured were allocated in individual cotton bags and taken to the field base, where they were identified.
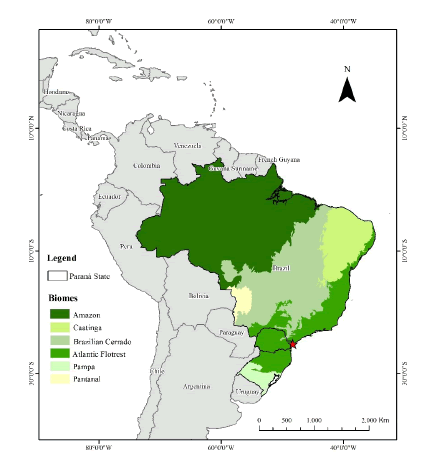
Fig. 1
Location Map Salto Morato Nature Reserve (F), eastern coast of the state of Paraná, southern Brazil.
There, we did biometrics and tagged each specimen with numbered metal rings. After that, the specimens were released where they were caught. For all captured species, up to 10 individuals were collected as testimonial material, fixed in humid via and forwarded to the scientific collection of Laboratório de Zoologia e Ecologia de Vertebrados (LABZEV) of the Universidade do Extremo Sul Catarinense (Appendix 1). All procedures followed the American Society of Mammalogists protocols for the use of mammals in research (Sikes et al. 2016). The taxonomic identification followed Barquez et al. (1999), Gardner (2007) and Díaz et al. (2016).
The sample sufficiency was calculated by the species accumulation curve using the rarefaction method, considering each night as a sampling unit. In addition, Chao1 and Bootstrap estimators were the inventory complementary analysis. We used Chao1 since this estimator allows us to assess the richness expected in the sampling area taking into consideration the number of species represented in one or two sample units (giving more weight to rare species). On the other hand, we used Bootstrap because it weights all species equally, not considering their frequency or abundance (all species are given equal weight). Since our species abundance are almost equally divided between rare and abundant species, we believe it is efficient to estimate richness in these two different ways, so similar results from both estimators may reach more confident results.
The analysis were performed in the software EstimateS 9.1 (Colwell 2013) considering 999 randomizations.
Recapture were not considered in sample sufficiency, although rates were calculated for each species.
RESULTS
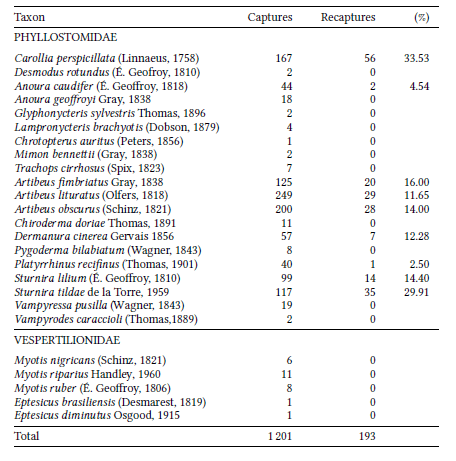
We obtained 1 201 captures of 1 008 individuals, with a recapture rate of 16.06% from 25 species from two families and 18 genera (Table 1). Phyllostomidae was the dominant family in terms of capture number (98% of the sample) and richness (20 species) when compared to Vespertilionidae – 2% of the sample and five species.
Artibeus lituratus (n =249), Artibeus fimbriatus (n =200) and Carollia perspicillata (n =167) were the most frequent species (Table 1), which together correspond to 51.29% of the sample. A total of 193 recapture events were recorded involving 10 species (Table 1), of which Carollia perspicillata (33,53%), Sturnira tildae (29,91%) and Artibeus fimbriatus (16%) have the highest recapture rates (Table 1).
The accumulation species curve shows an asymp tote trend (Fig. 2). The estimators Chao1 and Bootstrap indicate the occurrence of 26.1 and 26.6 species respectively, what suggests that 95.7% and 93.9% of the total expected species for RSM area were sampled.
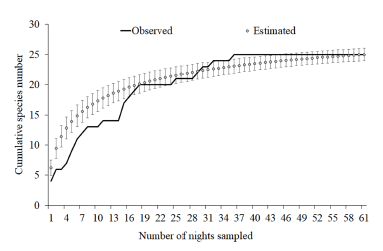
Fig. 2
Cumulative bat species sampled and the species richness estimated by rarefaction method, based on the standardized samples sampled at Reserva Particular do Patrimônio Natural Salto Morato (RNSM), located in Guaraqueçaba, northern coast of Paraná state.
DISCUSSION
We recorded a high species richness at RSM (n = 25 spp.), reinforcing that the Coastal region of Paraná is an important area for bat conservation in the Atlantic Forest (Varzinczak et al. 2015). Considering that there are 70 species in the entire State and 71 in Brazil’s South Region, RSM shelters 35.7% and 35.2% of the bat fauna for these regions, respectively (Passos et al. 2010; Carvalho et al. 2014; 2017; Portella et al. 2017; Carvalho et al. 2019). Also, for Vampyrodes caraccioli and Lasiurus egregius this is the unique location with a confirmed record in the State (Carvalho et al. 2014; 2019). For Glyphonycteris sylvestris, the records at RSM represent its second confirmed record in Paraná state, also captured at the Parque Nacional do Iguaçú (Sekiama et al. 2001). Furthermore, Chiroderma doriae, Chrotopterus auritus and Mimon bennettii are included as priority species for conservation actions in Paraná (Miranda et al. 2009). These characteristics show the important role that RSM plays to conserve bats in the Atlantic Forest, mainly in its southern portion.
Our data about the species accumulation curves, with the richness estimators, suggest that the composition of the bat assemblage in RSM was enough sampled (95.7% and 93.9%). This scenario is probably given to two attributes: (1) sampling at different heights, that provides detailing of the bat fauna composition (Bernard 2001; Carvalho et al. 2013; Gregorin et al. 2017); and (2) the high number of captures (n =1 008 captures, more 193 recaptures). It is estimated that, at least for phyllostomids in Atlantic Forest of southeast Brazil, 1 000 captures are required to consider an area as successfully sampled (Bergallo et al. 2003). This number was overcame in the RSM (Number of Phyllostomidae captures: 1174 individuals, see Table 1). However, even with sampling sufficiency for that family, species as Artibeus planirostris (Spix, 1823), Tonatia bidens (Spix, 1823), Myotis levis (I. Geoffroy, 1824), Molossus molossus (Pallas, 1766), Molossus rufus (É. Geoffroy, 1805), Thyroptera tricolor Spix, 1823 and Lasiurus egregius (Peters, 1970) were not recorded on the present study even been recorded in RSM yet (Kaku-Oliveira 2010; Passos et al. 2010; Carvalho et al. 2019). Even enough sufficiently sampled inventories, as ours, tend to not record all the occurring species in a given area.
The abundance pattern of bat species at RSM is similar to what was observed in other Atlantic Forest assemblages (Esbérard 2003), and in most of the Neotropical environments (Estrada & Coates- Estrada 2002; Bernard & Fenton 2002; Bordignon & França 2009): Few species are abundant, and the vast majority is rare. Among the species with greater number of captures, Artibeus lituratus, Carollia perspicillata, Artibeus fimbriatus and Sturnira lilium are among the most abundant in Southern Brazil (Sekiama et al. 2001; Bianconi et al. 2004; Reis et al. 2006; Carvalho et al. 2013), including the Paraná state (Kaku-Oliveira 2010; Rubio et al. 2014). This characteristic is associated to the ecological tolerance of these species, which can adapt to modified environments (Reis et al. 2006).
The recapture rate was high when compared to other studies developed in the Atlantic Forest (Esbérard et al. (2014) recaptured an average of 9 and Bôlla et al. (2017) an average of 6 bats per year). For two species (Carollia perspicillata and Sturnira tildae) recapture rates were over 29%, which could indicate changes in the habitat use patterns caused by anthropic impacts as lack of food, increasing in individuals displacement and diminishing foraging sites fidelity (Pedro et al. 2001) or even a natural fluctuation, as suggested by Aguirre et al. (2003). Nevertheless, this hypothesis seems not been justified since RSM is a Conservation Unit inserted in a large continuous of Atlantic Forest. Other possibility is that this attribute is related to biological and ecological characteristics of species, as space use. Carollia perspicillata potentially presents large foraging territory (Heithaus et al. 1975), but the effectively used area can be very small (Fleming et al. 1972; Cloutier & Thomas 1992), justifying its high recapture rate. We could not find data on this attribute for Sturnira tildae.
Comprehending how bats explore the environment is fundamental to evaluate the dynamics of the assemblages in natural and fragmented areas. The temporal availability of food resources may also influence the recapture rate. Species such as Carollia perspicillata, Sturnira tildae and Sturnira lilium, that base their diets in resources with small temporal variation in their availability (e.g. Marinho- Filho 1991; Mello et al. 2004; 2008; Barboza-Marquez & Aguirre 1999), tend to show greater fidelity to the foraging areas. Species that use resources that vary less temporally tend to show smaller foraging areas, what would elevate the recapture rates. Those species that use ephemeral resources, which require moving through a larger are to reach energy demand (Passos et al. 2010; Bianconi et al. 2006; Mello 2009) tend to be less loyal to foraging sites (Egert-Berg et al 2018). The habitat use is a complex characteristic, that is related to several factors (biotics, abiotics and ecological).
In the opposite, species as Artibeus fimbriatus, Artibeus lituratus and Artibeus obscurus, that use the resources with high temporal variation (Passos et al. 2010; Passos & Graciolli 2004), tend to show lower fidelity to foraging locations, mainly the males (Lewis 1995). In addition, A. lituratus is known by its long-distance displacements (see Bianconi et al. 2006; for displacements recorded with banding method): 19.4 km (Carvalho et al. 2017) and 113 km (Arnone et al. 2016), both in the Atlantic Forest.
Due to the high anthropogenic influence on Atlantic Forest environments, studies developed in conserved areas, such as RSM, are of great impor tance. The data collected at these areas enable conservationists and stakeholders to evaluate the impact of anthropic actions over the biodiversity of bats and other species related. Moreover, because it is a Conservation Unit, the data resulting from the studies about assemblages’ composition can assist the management and administration of other protected areas too, as well as species conservation actions in situ.
Acknowledgments
The legal licenses to capture, mark and colect bats were granted by SISBIO under the number 36103-3. We thank Fundação Boticário de Proteção à Natureza for the financial and logistical support for the for the research (Partnership term RNSM_089_2018); FAPESC for financial support to LABZEV (Edital Jovens Pesquisadores, Termo de Outorga N°: 2017TR1706); CAPES for the doctoral scholarships granted to BFLL, VM and Master Scholarship granted to LSB and CNPq to the Master Scholarship granted to DASB.
REFERENCES
Aguirre, L. F., X. Velez-Liendo, A. Muñoz, & A. Selaya. 2003. Patrones de distribución y zoogeografía de los murciélagos de Bolivia. Revista Boliviana de Ecología y Conservación Ambiental 14:3-17.
Alvares, A. C. , J. L. Stape, P. C. Sentelhas, J. L. M. Gonçalvez, & G. Sparovek. 2013. Köppen’s climate classification map for Brazil. Meteorologische Zeitschrift, 22(1): 711-728. https://doi.org/10.1127/0941-2948/2013/0507
Arnone, I. S. , E. Trajano, A. Pulchério-Leite, & F. C. Passos. 2016. Long-distance movement by a great fruit- eating bat, Artibeus lituratus (Olfers, 1818), in southeastern Brazil (Chiroptera, Phyllostomidae): evidence for migration in Neotropical bats? Biota Neotropica 16(1): e0026. https://doi.org/10.1590/1676-0611-bn-2015-0026
Barboza-Marquez, K. & L. F. Aguirre. 2010. Patrones reproductivos del murciélago frugívoro de cola corta (Carollia perspicillata) relacionados con la fenología de Piper en un bosque montano de Bolivia. Revista Boliviana de Ecología y conservación Ambiental 27: 43-52.
Barquez, R. M., M. A. Mares, & J. K. Braun. 1999. The Bats of Argentina. Special Publications Museum of Texas Tech University 42: 1-275.
Bergallo, H. G. , C. E. L. Esberard, M. A. R. Mello, V. Lins, R. Mangolin,G. G. S. Melo, & M. Baptista. 2003. Bat Species Richness in Atlantic Forest: What Is the Minimum Sampling Effort? Biotropica 35(2): 278-288. https://doi.org/10.1111/j.1744-7429.2003.tb00286.x
Bernard, E. 2001. Vertical stratification of bat communities in primary forests of Central Amazon, Brazil. Journal of Tropical Ecology 17(1): 115-126. https://doi.org/10.1017/s0266467401001079
Bernard, E. , L. M. S. Aguiar, & R. B. Machado. 2011. Discovering the Brazilian bat fauna: a task for two centuries? Mammal Review 41(1): 23-39. https://doi.org/10.1111/j.1365-2907.2010.00164.x
Bernard E., & M. B. Fenton. 2002. Species diversity of bats (Mammalia: Chiroptera) in forest fragments, primary forests, and savannas in central Amazonia, Brazil. Canadian Journal of Zoology 80: 1124-1140. https://doi.org/10.1139/z02-094
Bianconi, G. V. , S. B. Mikich, & W. A. Pedro. 2004. Diversidade de morcegos (Mammalia, Chiroptera) em remanescentes florestais do município de Fênix, noroeste do Paraná, Brasil. Revista Brasileira de Zoologia 21(4): 943-954. https://doi.org/10.1590/s0101-81752004000400032
Bianconi, G. V. , S. B. Mikich, & W. A. Pedro. ( 2006) Movements of bats (Mammalia, Chiroptera) in Atlantic Forest remnants in southern Brazil. Revista Brasileira de Zoologia 23: 1199-1206. https://doi.org/10.1590/s0101-81752006000400030
Bôlla, D. A. S. et al. ( 2017) Assembleia de morcegos (Mammalia: Chiroptera) em ambiente de Restinga alterada no sul do Brasil. Neotropical Biology and Conservation 12(2): 135-142.
Bordignon, M. O. & A. O. França. 2009. Riqueza, diversidade e variação atitudinal em uma comunidade de morcegos filostomídeos (Mammalia: Chiroptera) no Centro-Oeste do Brasil. Chiroptera Neotropical (15): 425-433. https://doi.org/10.1590/s0101-81752006000400004
Carvalho, F. , M. E. Fabián, & J. O. Menegheti. 2013. Vertical structure of in assemblage of bats (Mammalia: Chiroptera) in a fragment of Atlantic Forest in Southern Brazil. Zoologia 30(5): 491-498. https://doi.org/10.1590/s1984-46702013000500004
Carvalho, F. , V. Mottin, J. M. D. Miranda, & F. C. Passos. 2014. First record of Vampyrodes caraccioli (Thomas, 1889) (Chiroptera: Phyllostomidae) for the state of Paraná, and range extension to southern region of Brazil. Check List 10: 1189-1194. https://doi.org/10.15560/10.5.10.5.1189
Carvalho, F. , D. A. S. Bolla, F. M. Patel, J. M. D. Miranda, S. L. Althoff, &J. J. Zocche. 2017. Ampliação de distribuição de Eumops patagonicus (Chiroptera: Molossidae) e primeiro registro em ambiente de restinga na costa leste do brasil. Mastozoología Neotropical 24(2): 443–450.
Carvalho, F. , D. A. S. Bôlla, K. P. Supi, L. S. Biz, B. F. L. Luciano, & J. J. Zocche. 2019. First record of Lasiurus egregius (Peters, 1870) (Chiroptera, Vespertilionidae) in Paraná state, southern Brazil. Check List 15(6): 1099-1105. https://doi.org/10.15560/15.6.1099
Carvalho, F. , D. A. S. Bolla, J. M. D. Miranda, & J. J. Zocche. 2017. Deslocamentos de morcegos frugívoros (Chiroptera: Phyllostomidae), entre diferentes fitofisionomias da Mata Atlântica, no Sul do Brasil. Revista Brasileira de Biociências 15(2):78-82. https://doi.org/10.1590/s0101-81752002000100023
Cloutier, D. , & D. W. Thomas. 1992. Carollia perspicillata. Mammalian Species 417:1-9.
Colwell, R. K. 2013. EstimateS: Statistical estimation of species richness and shared species from sample. Version 9. 1 http://viceroy.colorado.edu/estimates/. Accessed on:2019-03-27.
Delciellos, A. C. , A. Motta, D. Dias, B. Almeida, & O. Rocha- Barbosa. 2018. Bats of the Serra da Bocaina National Park, southeastern Brazil: an updated species list and a distribution extension for Trinycteris nicefori (Sanborn, 1949). Biota Neotropica. 18(4): e20180537. https://doi.org/10.1590/1676-0611-bn-2018-0537
Dias, D. , C. E. L. Esbérard, & R. Moratelli. 2013. A new species of Lonchophylla (Chiroptera, Phyllostomidae) from the Atlantic Forest of southeastern Brazil, with comments on L. bokermanni. Zootaxa 3722(3): 347-360. https://doi.org/10.11646/zootaxa.3722.3.4
Díaz, M. M. , S. Solari, L. F. Aguirre, L. M. S. Aguiar, & R. M. Bar Ez. (eds. ). 2016. Clave de identificación de los murciélagos de Sudamérica. 1 st edition. Programa de Conservación de los Murciélagos de Argentina, Argentina. https://doi.org/10.35537/10915/67189
Egert-Berg K, et Al. 2018. Resource ephemerality drives social foraging in bats. Current Biology 28:3667-3673. https://doi.org/10.1016/j.cub.2018.09.064
Esbérard, C. E. L. 2003. Diversidade de morcegos em área de Mata Atlântica regenerada no sudeste do Brasil. Revista Brasileira de Zoociências 5(2):189-204.
Esbérard, C. E. L. , J. L. Luz, L. M. Costa, & H. G. Bergallo. 2014. Bats (Mammalia, Chiroptera) of an urban park in the metropolitan area of Rio de Janeiro, southeastern Brazil. Iheringia, Série Zoologia 104(1): 59-69. https://doi.org/10.1590/1678-4766201410415969
Esbérard, C. E. L. , M. S. M. Godoy, L. Renovato, & W. D. Carvalho. 2017. Novel long-distance movements by Neotropical bats (Mammalia: Phyllostomidae) evidenced by recaptures in southeastern Brazil. Studies on Neotropical Fauna and Enriroment 52(1):1-6. https://doi.org/10.1080/01650521.2016.1273751
Estrada, A. , & R. Coates-Estrada. 2002. Bats in continuous forest, forest fragments and in an agricultural mosaic habitat-island at Los Tuxtlas, Mexico. Biological Conservation 103(2): 237–245. https://doi.org/10.1016/s0006-3207(01)00135-5
Feijó, A. , P. A. Rocha, & S. L. Althoff. 2015. New species of Histiotus (Chiroptera: Vespertilionidae) from northeastern Brazil. Zootaxa 4048: 412-427. https://doi.org/10.11646/zootaxa.4048.3.4
Fenton, M. B. ,&N. M. Simmons. (eds. ). 2014. Bats: A World of Science and Mystery. Névraumont Publishing Company. New York.
Fleming, T. H. , E. T. Hooper , & D. E. Wilson. 1972. Three central American bat communities: Structure, reproductive cycles and movement patterns. Ecology 53(4): 555-569. https://doi.org/10.2307/1934771
Galindo, C. L. , & I. G. Câmara. 2003. (Eds. ) The Atlantic Forest of South America: biodiversity status, threats, and outlook. 1 st Edition. Fundação SOS Mata Atlântica. São Paulo.
Gardner, A. L. 2007. Order Chiroptera Blumenbach, 1779. Mammals of South America: Marsupials, Xenarthrans, Shrews, and Bats. (A. L. Gardner Ed. ). The University of Chicago Press. Chicago and London. https://doi.org/10.7208/chicago/9780226282428.001.0001
Giannini, N. P. 1999. Selection of diets and elevation by sympatri. species of Sturnira in an Andean rainforest. Journal of Mammalogy 80:1186-1195. https://doi.org/10.2307/1383169
Gregorin, R. et al. 2017. Vertical stratification in bat assemblages of the Atlantic Forest of south-eastern Brazil. Journal of Tropical Ecology 33(5):299-308. https://doi.org/10.1017/s026646741700027x
Heithaus, E. R. , T. H. Fleming, & P. A. Opler. 1975. Foraging patterns and resource utilization in seven species of bats in a seasonal Tropical Forest. Ecology 56(4): 841-854. https://doi.org/10.2307/1936295
IBGE. Instituto Brasileiro de Geografia e Estatística. (ed.). 2012. Manual Técnico da Vegetação 2 st. Edition. Brasileira. IBGE. Rio de Janeiro. https://doi.org/10.17143/ciaed/xxiilciaed.2017.00322
Kalko, E. 1998. Organization and diversity of tropical bat communities through space and time. Zoology 101: 281-297.
Kaku-Oliveira, N. Y. 2010. Estrutura de comunidade, reprodução e dinâmica populacional de morcegos (Mammalia, Chiroptera) na Reserva Natural do Salto Morato, Guaraqueçaba. Masters dissertation. Federal University of Paraná, Curitiba, Brazil.
Lewis, S.E. 1995. Roost Fidelity of bats: A Review. Journal of Mammalogy 76:482-496. https://doi.org/10.2307/1382357
Luz, J. L. , L. M. Costa, E. C. Lourenço, & C. E. L. Esbérard. 2011. Morcegos (Mammalia: Chiroptera) da Reserva Rio das Pedras, Rio de Janeiro, Sudeste do Brasil. Biota Neotropica 11(1): 1-7. https://doi.org/10.1590/s1676-06032011000100009
Marinho-Filho, J. S. 1996. Distribution of bat diversity in the southern and southeastern Brazilian Atlantic Forest. Chiroptera Neotropical 2(2): 51-54.
Marinho-Filho, J. S. 1991. The coexistence of two frugivorous bats and the phenology of their food plants in Brazil. Journal of Tropical Ecology 7(1): 59-67. https://doi.org/10.1017/s0266467400005083
Mello, M. A. R. , G. M. Schittini, P. Selig, & H. G. Bergallo. 2004. Seasonal variation in the diet of the Carollia perspicillata (Chiroptera: Phyllostomidae) in a Atlantic Forest area in south-eastern Brazil. Mammalia 68(1): 49-55. https://doi.org/10.1515/mamm.2004.006
Mello, M. A. R. , E. K. V. Kalko, & W. R. Silva. 2008. Movement. of the bat Sturnira lilium and its role as a seed disperser of Solanaceae in the Brazilian Atlantic Forest. Journal of Tropical Ecology 24: 225-228. https://doi.org/10.1017/s026646740800480x
Mello, M. A. R. 2009. Temporal variation in the organization of a Neotropical assemblage of leaf-nosed bats (Chiroptera: Phyllostomidae). Acta Oecologica 35: 280-286. https://doi.org/10.1016/j.actao.2008.11.008
Miranda, J. M. D. , M. F. M. Azevedo-Barros, & F. C. Passos. 2007. First Record of Histiotus leophotis Thomas (Chiroptera, Vespertilionidae) from Brazil. Revista Brasileira de Zoologia 24(4): 1188-1191. https://doi.org/10.1590/s0101-81752007000400040
Miranda, J. M. D. , I. P. Bernardi, & F. C. Passos. 2009. Plano de conservação para morcegos. Planos de conservação para espécies de mamíferos ameaçados. (Instituto Ambiental do Paraná eds. ) Instituto Ambiental do Paraná. Curitiba. https://doi.org/10.18068/iisbrns2015.geoap236
Miretzki, M. 2003. Morcegos do Estado do Paraná, Brasil (Mammalia, Chiroptera): riqueza de espécies, distribuição e síntese do conhecimento atual. Papéis Avulsos de Zoologia 43(6): 101-138. https://doi.org/10.1590/s0031-10492003000600001
Moratelli, R. , & D. Dias. 2015. A new species of nectar-feeding bat, genus Lonchophylla, from the Caatinga of Brazil (Chiroptera, Phyllostomidae). ZooKeys 514: 73-91. https://doi.org/10.3897/zookeys.514.10013.figure2
Moya, M. I. , F. Montaũo-Centellas, L. F. Aguirre, J. Tordoya, J. Martínez, & M. I. Galarza. 2008. Variación temporal de la quiropterofauna en un bosque de yungas en Bolivia. Mastozoología Neotropical 15(2): 349-357.
Muylaert, R. D. L. et al. 2017. ATLANTIC BATS: a data set of bat communities from the Atlantic Forests of South America. Ecology 98(12): 3227.
Nogueira, M. R. et al. 2018. Updated checklist of Brazilian bats: version 2018. 1. Comitê da Lista de Morcegos do Brasil. Sociedade Brasileira para o Estudo de Quirópteros (SBEQ) http://www.sbeq.net/updatelist.
Nogueira, M. R. , I. P. Lima, A. L. Peracchi, & N. B. Simmons. 2012. New Genus and Species of Nectar-Feeding Bat from the Atlantic Forest of Southeastern Brazil (Chiroptera: Phyllostomidae: Glossophaginae). American Museum Novitates 3747: 1-30. https://doi.org/10.1206/3747.2
Paglia, A. P. et al. 2012. Lista Anotada dos Mamíferos do Brasil. Arlington: Occasional Papers in Conservation Biology, 6: 1-82.
Passos, F. C. , W. R. Silva, W. A. Pedro, & M. R. Bonin. 2003. Frugívoria em morcegos (Mammalia, Chiroptera) no Parque Estadual Intervales, sudeste do Brasil. Revista Brasileira de Zoologia 20(3): 511-517. https://doi.org/10.1590/s0101-81752003000300024
Passos, F. C. , & G. Graciolli. 2004. Observações da dieta de Artibeus lituratus (Olfers) (Chiroptera, Phyllostomidae) em duas áreas do sul do Brasil. Revista Brasileira de Zoologia 21(3):487- 489. https://doi.org/10.1590/s0101-81752004000300010
Passos, F. C. , J. M. D. Miranda, I. P. Bernardi, N. Y. Kaku- Oliveira, & L. C. Munster. 2010. Morcegos da Região Sul do Brasil: análise comparativa da riqueza de espécies, novos registros e atualizações nomenclaturais (Mammalia, Chiroptera). Iheringia 100(1): 25-34. https://doi.org/10.1590/s0073-47212010000100004
Pedro,W. A. , F. C. Passos, & B. K. Lim. 2001. Morcegos (Chiroptera, Mammalia) da Estação Ecológica dos Caetetus, estado de São Paulo. Chiroptera Neotropical, 7(1-2): 136-140. https://doi.org/10.1590/s0101-81752006000100025
Portella, T. P. , N. Y. Kaku-Oliveira, J. S. Barros, & G. C. Sessegolo. 2017. First record of the Vulnerable bat Furipterus horrens (Cuvier, 1828) (Chiroptera: Furipteridae) in the state of Paraná, southern Brazil. Check List 13(4): 127-134. https://doi.org/10.15560/13.4.127
Reis, N. R. , A. L. Peracchi, I. P. Lima, & W. A. Pedro. 2006. Riqueza de espécies de morcegos (Mammalia, Chiroptera) em dois diferentes habitats, na região centro-sul do Paraná, sul do Brasil. Revista Brasileira de Zoologia 23(3): 813-816. https://doi.org/10.1590/s0101-81752006000300028
Rubio, M. B. G. , L. H. Varzinczak, I. P. Bernardi, F. C. Passos, & J. M. D. Miranda. 2014. Bats from two sites of the Paraná State coastal area, southern Brazil. Chiroptera Neotropical 20(1): 1255-1263.
Scarano, F. R. , & P. Ceotto. 2015. Brazilian Atlantic forest: impact, vulnerability, and adaptation to climate change. Biodiversity and Conservation 24:2319-2331. https://doi.org/10.1007/s10531-015-0972-y
Scultori, C., D. Dias, & A. L. Peracchi. 2009a. Mammalia, Chiroptera, Phyllostomidae, Lampronycteris brachyotis (Dobson, 1879): first record in the state of Paraná, southern Brazil. Check List 5(4): 872-875. https://doi.org/10.15560/5.4.872
Scultori, C., D. Dias, & A. L. Peracchi. 2009b. Mammalia, Chiroptera, Phyllostomidae, Artibeus cinereus: first record in the state of Paraná, Southern Brazil. Check List 5(2): 325-329. https://doi.org/10.15560/5.2.325
Scultori, C., D. Dias, & A. L. Peracchi. 2009c. Mammalia, Chiroptera, Phyllostomidae, Platyrrhinus recifinus: first record in the state of Paraná, southern Brazil. Check List 5(2): 238-242. https://doi.org/10.15560/5.2.238
Sekiama, L. M. , N. R. Reis, A. L. Peracchi, & J. V. Rocha. 2001. Morcegos do Parque Nacional do Iguaçu, Paraná (Mammalia, Chiroptera). Revista Brasileira de Zoologia 18(3): 749-754. https://doi.org/10.1590/s0101-81752001000300011
Sikes, R. S. et al. 2016. Guidelines of the American Society of Mammalogists for the use of wild mammals in research and education. Journal of Mammalogy 97(3): 663-688. https://doi.org/10.1093/jmammal/gyw078
Silveira, L. F. et al. 2010. Para que servem os inventários de fauna? Estudos Avançados, 24:173-207. https://doi.org/10.1590/s0103-40142010000100015
SISBIO. 2015. Área de Proteção Ambiental de Guaraqueçaba. http://www.icmbio.gov.br/portal/biodiversidade/unidades-deconservacao. Accessed on: 2019-02-12.
Straube, F. C. , & G. V. Bianconi. 2002. Sobre a grandeza e a unidade utilizada para estimar esforço de captura com utilização de redes-de-neblina. Chiroptera Neotropical 8(1-2): 150-153.
Straube, F. C. , & A. Urben-Filho. 2005. Avifauna da Reserva Natural Salto Morato (Guaraqueçaba, Paraná). Atualidades Ornitológicas 124:12-33.
Tabarelli, M. , L. P. Pinto, J. M. C. Silva, M. M. Hirota, & L. C. Bedê. 2005. Desafios e oportunidades para a conservação da biodiversidade na Mata Atlântica brasileira. Megadiversidade 1(1): 132-138.
Taylor, M., & M. Tuttle. (eds.). 2019. Bats: An illustrated guide all species. Smithsonian Books. Washington, D. C.
Tavares, V. C. , F. A. Perini, & J. A. Lombardi. 2007. The bat communities (Chiroptera) of the Parque Estadual do Rio Doce, a continuous remnant of Atlantic Forest in outheastern Brazil. Lundiana 8:35-47.
Varzinczak, L. H. , I. P. Bernardi, & F. C. Passos. 2015. Is the knowledge of bat distribution in the Atlantic Rainforest sufficient? Comments about new findings and a case study in the Paraná State coastal area, Brazil. Mammalia 80(3): 263-269. https://doi.org/10.1515/mammalia-2014-0130
APPENDIX 1

Información adicional
Editor asociado: R. Moratelli.