SECCIÓN ESPECIAL - ARTÍCULO
ALERT RESPONSE OF GUANACOS IN THE BOLIVIAN GRAN CHACO IS AFFECTED BY A CHANGING ENVIRONMENT DRIVEN BY WOODY PLANT ENCROACHMENT
La respuesta de alerta de los guanacos en el Gran Chaco boliviano se ve afectada por un entorno cambiante impulsado por la invasión de plantas leñosas
ALERT RESPONSE OF GUANACOS IN THE BOLIVIAN GRAN CHACO IS AFFECTED BY A CHANGING ENVIRONMENT DRIVEN BY WOODY PLANT ENCROACHMENT
Mastozoología Neotropical, vol. 28, núm. 1, p. 555, 2021
Sociedad Argentina para el Estudio de los Mamíferos
Recepción: 14 Octubre 2020
Aprobación: 02 Febrero 2021
Abstract: In the Bolivian Gran Chaco, woody plant encroachment (WPE) is gradually replacing open habitats such as grassland and savannah. This change in vertical structuring of native open vegetation may create hunting cover for predators. Therefore, animal species naturally dependent on open habitats, enabling effective visual scanning of their environment, may need to adapt to a denser vegetation structure to survive. Hence, we expected a negative consequence of increasing WPE intensity in the grasslands of the Bolivian Gran Chaco, on the population of guanacos (Lama guanicoe) at the species’ northerly distribution limit. We analysed guanaco alert responses under different conditions, whether the animals: 1) modified their response according to habitat structure; 2) varied their escape response according to their group size and composition; and 3) responded differently to observers (these approaching either on foot or horseback). The habitat variable (open versus dense) was the strongest predictor of an alert response, irrespective of whether guanacos were alone or in groups. Contrary to our expectation, guanacos were more alert when in open habitat. In addition, we found no evidence for the effect of habitat type on group size. Finally, there was a significant association between observation method and guanaco alert response. We suggest that the current combination of relatively high densities of predators such as puma (Puma concolor), and jaguar (Panthera onca) together with WPE-driven replacement of natural, open habitat preferred by Chacoan guanacos, could diminish the potential recovery and survival of this population of guanacos, currently threatened in Bolivia.
Keywords: Camelidae, change in vegetation structure, conservation, Gran Chaco.
Resumen: En el Gran Chaco Boliviano, la invasión de plantas leñosas (WPE) está reemplazando gradualmente hábitats abiertos como pastizales y sabanas. Este cambio en la estructura vertical de la vegetación abierta nativa puede crear una cobertura de caza para los depredadores. Por lo tanto, las especies animales que dependen naturalmente de hábitats abiertos, lo que permite un escaneo visual efectivo de su entorno, pueden necesitar adaptarse a una estructura de vegetación más densa para sobrevivir. Por lo mencionado anteriormente, esperábamos una consecuencia negativa del aumento de la intensidad del WPE en los pastizales del Gran Chaco boliviano, sobre la población de guanacos (Lama guanicoe) en el límite de distribución norte de la especie. Analizamos las respuestas de alerta del guanaco bajo diferentes condiciones, ya sea que los animales: 1) modificaran su respuesta de acuerdo con la estructura del hábitat; 2) variaran su respuesta de escape de acuerdo con el tamaño y la composición de su grupo; y 3) respondieran de manera diferente a los observadores (que se acercaban a pie o a caballo). La variable de hábitat (abierto versus denso) fue el predictor más fuerte de una respuesta de alerta, independientemente de si los guanacos estaban solos o en grupos. Contrariamente a nuestras expectativas, los guanacos estaban más alerta cuando estaban en hábitat abierto. Además, no encontramos evidencia del efecto del tipo de hábitat en el tamaño del grupo. Finalmente, hubo una asociación significativa entre el método de observación y la respuesta de alerta del guanaco. Sugerimos que la combinación actual de densidades relativamente altas de depredadores como el puma (Puma concolor) y el jaguar (Panthera onca), junto con el reemplazo impulsado por el WPE del hábitat natural abierto preferido por los guanacos del Chaco, podría disminuir la recuperación potencial y supervivencia de esta población de guanacos, actualmente amenazada en Bolivia.
Palabras clave: Cambio en la estructura de la vegetación, Camélidos, Conservación, Gran Chaco.
INTRODUCTION
In the Bolivian Gran Chaco, woody plant encroachment (WPE) is causing a structural change in grassland and savannah communities and consequently affecting animal species dependant on the quantity and quality of those habitats (Archer et al. 2017). Consequently, as WPE advances and the native grasslands give way to early and later stages of encroachment, animal species naturally adapted to an open habitat may require rapid adaptation to newer and denser environments or suffer habitat displacement (Grant et al. 2004; Sirami et al. 2009; Sauer & Link 2011; Sirami & Monadjem 2012; Archer et al. 2017). Despite its ecological relevance, and the long-term declines in the abundance and distribution of many grassland species (Skowno & Bond 2003; Coppedge 2004; Sirami & Monadjem 2012), very few studies have tackled the response effects of WPE-driven habitat transformation on animal species behaviour.
In general, habitat selection by herbivores should be influenced by the structure and composition of the vegetation, especially as a more vertical habitat structuring reduces visibility and hence capacity of said herbivores to detect a predator (Lima 1987). Therefore, if the vegetation structure is integral to the provision of effective visual scanning enabling, in turn, substantial rates of escape from predators (Elgar 1989; Laundre et al. 2001; Beauchamp 2003, 2008; Li & Jiang 2008; Riginos & Grace 2008), one expects that WPE will lower those probabilities for animals that inhabit affected areas (Mannan & Steidl 2013; Archer et al. 2017). Indeed, antipredator responses amongst ungulates vary greatly depending on several factors, among those are habitat structure and composition (Creel et al. 2014). Examples of the latter are altering their level of vigilance or patterns of aggregation (Prins & Iason 1989; Hunter & Skinner 1998; Caro 2005; Creel & Winnie 2005; Taraborelli et al. 2014) under high and low levels of predation risk, such as in the case of a large ungulate in South America, the guanaco Lama guanicoe (Marino 2010). In general, guanacos like other ungulates that depends on their visibility to avoid predators are expected to favour open habitats with low vegetation (Sarno et al. 2008). Furthermore, even in Tierra del Fuego Island, at the southernmost tip of the species distribution (Muñoz & Simonetti 2013) where guanacos use the forest comparatively more extensively than in other areas of its distribution, guanacos are mainly grazers and only become browsers due to competition with domestic sheep (Raedeke 1980).
We studied a relict and almost extinct population of guanacos, in the north-eastern fringe of the species range in the Gran Chaco (Cuéllar & Núñez 2009). Whilst the Chacoan population of guanacos persists in an area characterized by a mosaic of vegetation cover with various extents of WPE (Navarro & Fuentes 1999; Pinto & Cuéllar Soto 2017), their distribution is analogous to that of two top guanaco predators, the jaguar (Panthera onca), and puma (Puma concolor) (Maffei et al. 2004; Kelly et al. 2008).
We tested whether guanacos: 1) modify their vigilance behaviour in relation to vegetation structure and 2) vary their escape response according to group size and composition; larger groups being expected to exhibit a lowered tendency for flight than lone individuals or groups with offspring, and 3) responded differently to observers (these approaching either on foot or horseback). We analysed the response of guanacos to human presence as an antipredator response (Taraborelli et al. 2014). Our assumption was that both habitat structure and group size would influence alert response of guanacos living in groups in this changing environment.
MATERIALS AND METHODS
Study area
The study area extends from 19°45’ to 20°30’ S and from 62°00’ to 63°00’ W, on the fluvial megafans of the Río Grande and the Río Parapetí in the Bolivian Gran Chaco (May et al. 2008), in the extreme south of Santa Cruz department, Bolivia. The study area includes the southwest corner of the Kaa-Iya National Park and part of the Indigenous Isoseño Communal Land (see Cuéllar Soto et al. 2020; for a detailed description of the study site). The climate is predominantly semi-arid (Peel et al. 2007), with annual rainfall ranging from 200 to 350 mm (Taber et al. 1997).
Data collection
In a time period of five years (2004-2009), we conducted 693 transect surveys (between 06:00 and 18:00 h) within a survey area of 791km2, of which 454 yielded successful observations. Mean transect length was 10.5 km (range: 7-14km), and transects were travelled on foot or on horseback. In addition, we followed Navarro’s classification to deter- mine of stages of WPE in the study area (Navarro & Fuentes 1999; Navarro 2002). We assigned two habitat categories according to vegetation structure: 1) open grassland and scrub constitutes low vegetation cover [heigh around 0.40 m and dominated mainly by Lippia sp. (Verbenaceae), Pappophorum krapovickasii (Poaceae), and Aristida mendocina (Poaceae)]; and 2) shrubland or woody vegetation [heigh around 4 m, dominated by Pithecellobium chacoense (Fabaceae)].
Once the guanacos were encountered we recorded the structure and size of the groups, presence/absence of offspring (chulengos) and their response from the first moment of visual contact. Their responses were classified into two main categories: 1) continuing activity or passive response; and 2) alert response (standing in a vigilant position, alarm call and evasion either by walking or running).
Data analysis
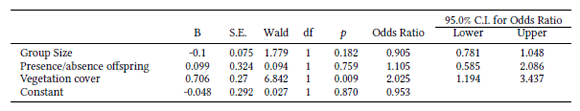
We analysed data gathered on foot and on horseback separately, as we initially detected an observer effect on guanaco response. We performed a logistic regression with three independent predictors: group size, habitat coverage and presence/absence of offspring.
For the statistical analysis, we considered group size as a continuous variable, and we categorised guanaco groups into two categories according to the presence or absence of offspring.
We tested for a relationship between type of alert response (dependent variable) and group size, presence or absence of offspring, and habitat cover (predictor independent variables).
Then we performed logistic regressions to assess the influence of a few factors on the likelihood that guanacos would show an alert response to our presence as an indication of predator avoidance behaviour. We included interaction terms to test, for example, if the effect of group size on the response depended on habitat structure. The statistical analyses were performed with SPSS software (version 18.0; Chicago: SPSS Inc. 2009).
RESULTS
Guanacos appeared to react differently to observers approaching on foot to those approaching on horseback. There was a significant association between observation method and guanaco vigilance response (χ2=44.60, df=1, p=0.00). An alert response was significantly more likely when the approach was on foot than on horseback.
Vigilance response vs. social and habitat variables
The model made significant predictions of an alert response (χ2= 8.73, df=3, p < 0.033). It explained between 3.2% (Cox & Snell R Square) and 4.3% (Nagelkerke R Square) of the variance in guanaco response, and correctly classified 58.1% of cases. Only vegetation structure made a statistically significant contribution to the model (Table 1). The odds ratio for habitat cover was 2.02, indicating that the alert response of guanacos encountered in relatively lower vegetation coverage was about twice as likely as that for high vegetation cover. The odds ratio of 0.90 for the predictor “group size” was <1, indicating that for every additional individual in the group, individual guanacos were 0.9 times less likely to show an alert response, controlling for other factors in the model. There was no evidence that the effect of habitat depended on group size: the interaction term was not statistically significant (χ2=0.01, df=1, p=0. 92). Group size and habitat were not confounded predictors: mean group sizes in both open and closed habitat are similar (Mean(low)=3.10, SE=0.15; Mean(high)= 3.02, SE=0.21). The model made no significant predictions of an alert response on a set of 164 observations made on horseback (χ2= 3.95, df=3, p=0.266).
Social structure
From the accumulated observations we identified three forms of guanaco social organisation: adults only, family groups (with one or two offspring), and solitary individuals (male or female). The mean group size was 2.8 (SD±1.8). Solitary adults andsolitary juveniles (female or male) were observedmore often than larger groups (Fig. 1).
DISCUSSION
In our study, we found vegetation cover to be the strongest predictor of an alert response which, in turn, was not affected by whether guanacos were alone or in groups. Contrary to our expectation, guanacos were twice as responsive in what we classified as open areas, these being relatively scarce and forming a patchwork surrounded by areas of dense cover.
Cuéllar Soto et al. (2020) have previously reported that, for guanacos, forage areas of preference were limited to patches of open vegetation. Perhaps guanacos in our study area have greater perceptions of risk in open habitats, because there were surrounded by thick vegetation. Therefore, represent a high risk for guanacos due to potential ambush from predators (Bank et al. 2002, 2003). Consequently, guanacos may consider these open areas to be more dangerous than denser areas, which could be perceived as counter- intuitive due to heightened anxiety when nearer cover, potential ambush sites, in proper grasslands. Furthermore, if poor visibility, associated with high vegetation cover, increases risk of predation then this should drive adaptations for increased vigilance regardless of group size. This latter point contrasts with the general ecological concept that group size is an important factor determining vigilance patterns in mammalian herbivores (Elgar 1989; Lima & Dill 1990; Childress & Lung 2003; Lung & Childress 2006; Marino & Baldi 2008). However, additional, and specific information need to be collected in order to confirm whether the current small groups and solitary individuals have a similar response (Taraborelli et al. 2019).
During the course of our investigation, we encountered solitary individuals, both males and females, more frequently than groups (Fig. 1). Given the polygynous nature typical to guanacos (Franklin 1983), and the importance of territoriality and site fidelity (Young & Franklin 2004), lone males are unlikely to secure mating opportunities with females. Although they are sometimes seen briefly with solitary territorial males after the end of the mating period, females are thought to be receptive only to territorial males within family groups (Franklin 1982, 1983). However, additional data is required to confirm mating success, birth rates, juvenile mortality rates, and emigration or dispersal rates related to the lack of open areas from WPE.
Establishing a guanaco alertness rating gives us a proxy for how anxious they may feel or how they perceive risk in a changing landscape. Regardless of the method used, guanacos invariably moved away within two minutes of being sighted. This contrasts with observations in open habitats of Argentina, where guanacos do not flee after having detected observers (Marino & Baldi 2008; Taraborelli et al. 2014) or become rapidly habituated if harassment ceases as reported in a guanaco population inhabiting Argentinean Patagonian arid lands (Marino & Johnson 2012). However, the response from Chacoan guanacos could be just different given their unique current woody environment, or natural history as mentioned by Stankowich (2008). Our method of searching for guanacos in open environments dif- fered greatly from the standard transect methods used elsewhere, such as Patagonian steppe (Novaro et al. 2000; Franklin & Grigione 2005; Marino & Baldi 2008; Schroeder et al. 2013), Andean foothills (Bank et al. 2003), or Puna plateaux (Lucherini 1996), or the Monte desert (Acebes et al. 2010). Overall visibility and access, in all these latter cases, were very much greater compared to our study site where we observed that within 5m of relatively open areas, there could be several intermediate stages of WPE, each offering various amounts of cover. Therefore, the dominant category, in our observations (a low vegetation mix of open grass and scrubland) is likely to have been considered as high vegetation cover, by guanacos, in other distinct areas of their distribution range.
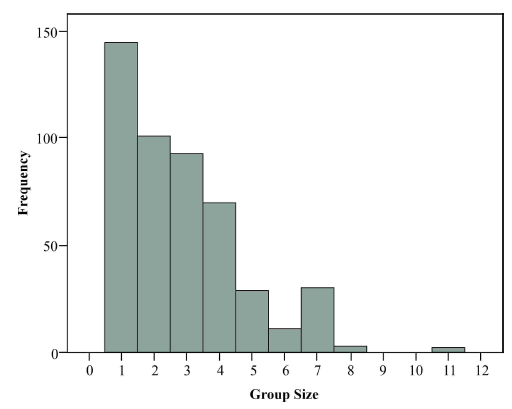
Fig. 1
Guanaco group sizes (including offspring) observed in the study population (n=484).
Although hunting had stopped in the study area by 2001, guanacos responded differently to human observers depending on whether they were approached on foot or on horseback. Apparently, guanacos were still very much wary of human presence, as they are reported to be elsewhere when subjected to strong hunting pressure (Donadio & Buskirk 2006). We conclude that this difference in response was based on the history of harassment as reported for other species by Stankowich (2008) by sport hunters rather than the presence of domestic ungulates which are becoming more frequently in the guanaco distribution range. Aside from people, the guanaco’s only other important predator, outside the Gran Chaco region, is the puma (Wilson 1984; Bank & Franklin 1998; Novaro et al. 2000; Bank et al. 2003; Marino & Baldi 2008). Although we have no specific data to show that the response behaviour of guanacos was linked to predation, we found evidence that both jaguars and pumas occurred within the same study area, and that, similar to guanacos, were diurnally active (Cuéllar Soto & Noss 2014).
The combination of facts that, in the last few decades, the Chacoan population of guanacos has been severely reduced to a small number and its potential primary habitat transformed due to WPE (Cuéllar Soto et al. 2017) deserves attention and action for rigorous protection and conservation ef- forts. Furthermore, specific parallel studies need to be conducted to test whether management of WPE may improve visibility and benefit guanacos in their detection of predators given that, even within relatively open environments, guanacos concentrated in areas with the sparsest vegetation cover.
Acknowledgments
U.F.J Pardiñas kindly reviewed the Spanish abstract and D. Voglino helped with the edition of the FIgure. A. J Noss, and L. James contributed in different phases of the project. We are grateful to several institutions for providingfunds, The Wildlife Conservation Society, The Gordonand Betty Moore Foundation within the Amazon-Andes Conservation Program (AACP), The Shared EarthFoundation, Whitley Fund for nature, and Kaa-IyaFoundation. We also thank the para-biologists and parkrangers from the Kaa-Iya national park. Carlos Galliari and U.F.J Pardiñas kindly reviewed the Spanish abstract and D. Voglino helped with the edition of the figure. A. J Noss, and L. James contributed in different phases of the project.
REFERENCES
Acebes, P., J. Traba, J. E. Malo, R. Ovejero, & C. E. Borghi. 2010. Density and habitat use at different spatial scales of a guanaco population (Lama guanicoe) in the Monte desert of Argentina. Mammalia 74:57–62. https://doi.org/10.1515/MAMM.2009.071
Archer, S. R., E. M. Andersen, K. I. Predick, S. Schwinning, R.J. Steidl, & S. R. Woods. 2017. Woody Plant Encroachment:Causes and Consequences. Pp 25–83 in Rangeland Systems (D.Briske, ed.). Springer Series on Environmental Management.Springer, Cham https://doi.org/10.1007/978-3-319-46709-22
Bank, M. S., & W. Franklin. 1998. Mountain lion (Puma concolor patagonica) feeding observations and attacks on guanacos (Lama guanicoe). Mammalia 62:599–605.
Bank, M. S., R. J. Sarno, N. K. Campbell, & W. L. Franklin. 2002. Predation of guanacos (Lama guanicoe) by southernmost mountain lions (Puma concolor) during a historically severe winter in Torres del Paine National Park, Chile. Journal of Zoology 258:215–222.
Bank, M. S., R. J. Sarno, & W. L. Franklin. 2003. Spatial distribution of guanaco mating sites in southern Chile: conservation implications. Biological Conservation 112:427–434.
Beauchamp, G. 2003. Group-size effects on vigilance: a search for mechanisms. Behavioural Processes 63:111–121.
Beauchamp, G. 2008. What is the magnitude of the group-size effect on vigilance? Behavioral Ecology 19:1361–1368.
Caro, T. 2005. Antipredator defenses in birds and mammals. University of Chicago Press, Chicago.
Childress, M. J., & M. A. Lung. 2003. Predation risk, gender and the group size effect: does elk vigilance depend upon the behaviour of conspecifics? Animal Behaviour 66:389–398.
Coppedge, B. 2004. Predicting juniper encroachment and CRP effects on avian community dynamics in southern mixed-grass prairie, USA. Biological Conservation 115:431–441.
Creel, S., & J. A. Winnie. 2005. Responses of elk herd size to fine-scale spatial and temporal variation in the risk of predation by wolves. Animal Behaviour 69:1181–1189. https://doi.org/10.1016/j.anbehav.2004.07.022
Creel, S., P. Schuette, & D. Christianson. 2014. Effects of predation risk on group size, vigilance, and foraging behavior in an African ungulate community, Behavioral Ecology 25: 773–784. https://doi.org/10.1093/beheco/aru050
Cuéllar Soto, E., P. J. Johnson, D. W. Macdonald, G. A. Barrett, & J. Segundo. 2020. Woody plant encroachment drives habitat loss for a relict population of a large mammalian herbivore in South America. Therya 11:484–494. https://doi.org/10.12933/therya-20-1071ISSN2007-3364
Cuéllar Soto, E., P. J. Johnson, & D. W. Macdonald. 2017. The diets of cattle and guanaco in the relict Chacoan savannahs of Bolivia. Ecología en Bolivia 52:77–87.
Cuéllar Soto, E., & A. J. Noss. 2014. Diversidad de mamíferos y participación local en la conservación en el Gran Chaco Boliviano. Therya 5:39–60.
Cuéllar, E., & A. Núñez. 2009. Lama guanicoe (Müller, 1776): Artiodactyla-Camelidae. Libro Rojo de la fauna silvestre de vertebrados de Bolivia. (L. F. Aguirre, R. Aguayo, J. Balderrama, Cortéz & T. Tarifa, eds.). Ministerio de Medio Ambiente y Agua de Bolivia, La Paz.
Donadio, E., & S.W. Buskirk. 2006. Flight behavior in guanacos and vicunas in areas with and without poaching in western Argentina. Biological Conservation 127: 139–145. https://doi.org/10.1016/j.biocon.2005.08.004
Elgar, M. A. 1989. Predator vigilance and group size in mammals and birds- a critical review of the empirical evidence. Biological Reviews of the Cambridge Philosophical Society 64:13–33.
Elgar, M. A., P. J. Burren, & M. Posen. 1984. Vigilance and perception of flock size in foraging house sparrows (Passer domesticus). Behaviour 90:215–223. https://doi.org/10.1163/156853984x00146
Franklin, W. L. 1982. Biology, ecology, and relationship to man of the South American camelids. Mammalian biology in South America (M. A. Mares & H. H. Genoways, eds.). Pymatuning Laboratory of Ecology and University of Pittsburgh Press., Pittsburgh.
Franklin, W. L. 1983. Contrasting socioecologies of South America’s wild camelids: the vicuña and the guanaco. Advances in the study of mammalian behavior: 1. (J. F. Eisenberg & D. G. Kleiman, eds.). Special publication American Society of Mammalogists.
Franklin, W. L., & M. M. Grigione. 2005. The enigma of guanacos in the Falkland Islands: the legacy of John Hamilton. Journal of Biogeography 32:661–675. https://doi.org/10.1111/j.1365-2699.2004.01220.x
Grant, T., E. Madden, & G. Berkey. 2004. Tree and shrub invasion in northern mixed-grass prairie: Implications for breeding grassland birds. Wildlife Society Bulletin 32:807–818.
Hunter, L. T. B., & J. D. Skinner. 1998. Vigilance behaviour in African ungulates: the role of predation pressure. Behaviour 135:195–211. https://doi.org/10.1163/156853998793066320
Kelly, M. J., A. J. Noss, M. S. Di Bitetti, L. Maffei, R. L. Arispe, A. Paviolo, C. D. De Angelo, & Y. E. Di Blanco. 2008. Estimating puma densities from camera trapping across three study sites: Bolivia, Argentina, and Belize. Journal of Mammalogy 89:408–418. https://doi.org/10.1644/06-mamm-a-424r.1
Laundre, J. W., L. Hernandez, & K. B. Altendorf. 2001. Wolves, elk, and bison: reestablishing the "landscape of fear" in Yellowstone National Park, USA. Canadian Journal of Zoology- Revue Canadienne De Zoologie 79:1401–1409.
Li, Z. Q., & Z. A. Jiang. 2008. Group size effect on vigilance: Evidence from Tibetan gazelle in Upper Buha River, Qinghai-Tibet Plateau. Behavioural Processes 78: 25–28.
Lima, S. L. 1987. Vigilance while feeding and its relation to the risk of predation. Journal of Theoretical Biology 124:303–316.
Lima, S. L., & L. M. Dill. 1990. Behavioral decisions made under the risk of predation - a review and prospectus. Canadian Journal of Zoology-Revue Canadienne de Zoologie 68:619–640.
Lucherini, M. 1996. Group size, spatial segregation and activity of wild sympatric vicuñas Vicugna vicugna and guanacos Lama guanicoe. Small ruminant research 20:193–198.
Lung, M. A., & M. B. Childress. 2006. The influence of conspecifics and predation risk on the vigilance of elk (Cervus elaphus) in Yellowstone National Park. Behavioral Ecology 18:12–20. https://doi.org/10.1093/beheco/arl066
Maffei, L., E. Cuéllar, & A. Noss. 2004. One thousand jaguars Panthera onca in Bolivia’s Chaco? Camera trapping in the Kaa- Iya National Park. Journal of Zoology 262:295–304. https://doi.org/10.1017/s0952836903004655
Mannan, R. W., & R. J. Steidl. 2013. Habitat. Wildlife management and conservation: Contemporary principles and practices (P. Krausman & J. Cain, eds.). Baltimore, MD: John Hopkins University Press.
Marino, A., & A. Johnson. 2012. Behavioural response of free-ranging guanacos (Lama guanicoe) to land-use change: habituation to motorised vehicles in a recently created reserve. Wildlife Research 39:503–511.
Marino, A. 2010. Costs and benefits of sociality differ between female guanacos living in contrasting ecological conditions. Ethology 116:1–12. https://doi.org/10.1111/j.1439-0310.2010.01812.x
Marino, A., & R. Baldi. 2008. Vigilance patterns of territorial guanacos (Lama guanicoe): The role of reproductive interests and predation risk. Ethology 114:413–423. https://doi.org/10.1111/j.1439-0310.2008.014851.x
May, J. H., R. Zech, & H. Veit. 2008. Late Quaternary paleosol-sediment-sequences and landscape evolution along the Andean piedmont, Bolivian Chaco. Geomorphology 98:34–54.
Muñoz, A. E., & J. A. Simonetti. 2013. Diet of guanaco in sheep-free rangeland in Tierra del Fuego, Chile. Ciencia e Investigación Agraria 40:185–191.
Navarro, G. 2002. Ecología estructural y dinámica de áreas quemadas en el PN-ANMI Kaa-Iya del Gran Chaco. Ecología del fuego en el Parque Nacional y Área Natural de Manejo Integrado Kaa-Iya del Gran Chaco (J. Guerrero, ed.). Proyecto Kaa-Iya, Santa Cruz, Bolivia.
Navarro, G., & A. Fuentes. 1999. Geobotánica y sistemas ecológicos de paisaje en el Gran Chaco de Bolivia. Revista Boliviana de Ecología y Conservación Ambiental 5:25–50.
Novaro, A.J., M. C. Funes, & R. S. Walker. 2000. Ecological extinction of native prey of a carnivore assemblage in Argentine Patagonia. Biological Conservation 92:25–33. https://doi.org/10.1016/s0006-3207(99)00065-8
Peel, M.C., B. L. Finlayson, & T. A. Mcmahon. 2007. Updated world map of the Koppen-Geiger climate classification. Hydrology and Earth System Sciences 11:1633–1644.
Pinto, C., & E. Cuéllar Soto. 2017. Multi-temporal analysis of a vegetation mosaic form by a serial stage of the Chacoan forest on well-drained soils, Kaa-Iya del Gran Chaco National Park. Ecología en Bolivia 52:7–20. ISSN 1605-2528.
Prins, H. H. T., & G. R. Iason. 1989. Dangerous lions and nonchalant buffalo. Behaviour 108:262–296
Raedeke, K. 1980. Food habits of the guanaco Lama guanicoe of Tierra del Fuego, Chile. Turrialba 30:177–181.
Riginos, C., & J. B. Grace. 2008. Savanna tree density, herbivores, and the herbaceous community: Bottom-up vs. top-down effects. Ecology 89:2228–2238.
Sarno, R. J., M. M. Grigione, & L. D. Arvidson. 2008. Lack of response of an open-habitat ungulate to the presence of predator urine. Revista Chilena De Historia Natural 81:179–183.
Sauer, J. R., & W. A. Link. 2011. Analysis of the North American breeding bird survey using hierarchical models. The Auk 128:87–98
Schroeder, N. M. et al. 2013. Including species interactions in resource selection of guanacos and livestock in Northern Patagonia. Journal of Zoology 281:213–225.
Sirami, C., & A. Monadjem. 2012. Changes in bird communities in Swaziland savannas between 1998 and 2008 owing to shrub encroachment. Diversity and Distributions 18:390–400.
Sirami, C., C. Seymour, G. Midgley, & P. Barnard. 2009. The impact of shrub encroachment on savanna bird diversity from local to regional scale. Diversity and Distributions 15:948–957.
Stankowich, T. 2008. Ungulate flight responses to human disturbance: A review and meta-analysis. Biological Conservation 141:2159–2173. https://doi.org10.1007/s13364-014-0186-910.1016/j.biocon.2008.06.026
Skowno, A., & W. Bond. 2003. Bird community composition in an actively managed savanna reserve, importance of vegetation structure and vegetation composition. Biodiversity and Conservation 12:2279–2294.
Skowno, A., & W. Bond. 2003. Bird community composition in an actively managed savanna reserve, importance of vegetation structure and vegetation composition. Biodiversity and Conservation 12:2279–2294.
Taber, A., G. Navarro, & M. A. Arribas. 1997. A new park in the Bolivian Gran Chaco - An advance in tropical dry forest conservation and community-based management. Oryx 31:189– 198.
Taraborelli, P., R. Ovejero, M. E. Mosca Torres, N. Schroeder, P. Moreno, P. Gregorio, E. Marcotti, A. Marozzi, & P. Carmanchahi. 2014. Different factors that modify anti-predator behaviour in guanacos (Lama guanicoe). Acta Theriologica 59:529–539. https://doi.org10.1007/s13364-014-0186-9
Taraborelli, P., P. Moreno, & M. E. Mosca Torres. 2019. Are there different vigilance strategies between types of social units in Lama guanicoe? Behavioural Processes 167:103914. ISSN 0376-6357, https://doi.org/10.1016/j.beproc.2019.103914.
Wilson, P. 1984. Puma predation on guanacos in Torres del Paine National Park, Chile. Mammalia 48:515–522.
Young, J. K., & W. L. Franklin. 2004. Territorial fidelity of male Guanacos in the Patagonia of southern Chile. Journal of Mammalogy 85:72–78. https://doi.org/10.1644/1545-1542(2004)085<0072:tfomgi>2.0.co;2
Información adicional
Editor asociado: C. A. Galliari
Cite as: Cuéllar Soto, E. & J. Segundo. 2021. Alert response of guanacos in the Bolivian Gran Chaco is affected by a changing environment driven by woody plant encroachment. Mastozoología Neotropical, 28(1):e0555. https://doi.org/10.31687/saremMN.21.28.1.0.27