ARTÍCULO
CRANIAL MORPHOLOGICAL VARIABILITY OF A SMALL NEOTROPICAL CAT REVEALED BY GEOMETRIC MORPHOMETRICS
VARIABILIDADE MORFOLÓGICA CRANIANA DE UM PEQUENO FELINO NEOTROPICAL, REVELADA PELA MORFOMETRIA GEOMÉTRICA
CRANIAL MORPHOLOGICAL VARIABILITY OF A SMALL NEOTROPICAL CAT REVEALED BY GEOMETRIC MORPHOMETRICS
Mastozoología Neotropical, vol. 28, núm. 2, p. 472, 2021
Sociedad Argentina para el Estudio de los Mamíferos
Recepción: 07 Junio 2020
Aprobación: 05 Marzo 2021
Abstract: The jaguarundi (Herpailurus yagouaroundi) has one of the most extensive latitudinal ranges among felids of the Western Hemisphere. Its wide geographic distribution and range of habitats may result in patterns of cranial morphological variation. Thus, we investigated the hypothesis of the existence of morphological ecotypes adapted to specific environments used by the species. The crania of 54 museum specimens from three different ecoregions in Brazil were digitized in ventral, dorsal, and lateral views. No sexual dimorphism was detected. Our hypothesis was supported by the fact that the specimens from the Amazon were significantly larger than those from the Atlantic Forest and Uruguayan savanna. Cranial shape variation between sexes, as well as among ecoregions, was mostly explained by the effect of size. Correlations between geographical distance and cranial shape were not significant. There was a significant correlation between cranium size and latitude, in a pattern that is the reverse of Bergmann’s rule, with larger specimens in lower latitudes. The environmental variables positively correlated with cranium size indicated that larger cats occurred in regions with greater temperature and precipitation. Resource availability might be the cause of the observed variation in cranium size, since jaguarundis probably show different prey size preferences along the species distribution range. However, more ecological data for most ecoregions are needed to test the “resource rule” and to fully understand the patterns and causes of cranial variation in this cat.
Keywords: Amazon, Felidae, geometric morphometrics, Herpailurus yagouaroundi, intraspecific variation.
Resumo: O jaguarundi (Herpailurus yagouaroundi) possui uma das faixas latitudinais mais extensas entre os felídeos do Hemisfério Ocidental. Sua ampla distribuição geográfica e variedade de habitats podem resultar em padrões de variação morfológica do crânio. Assim, investigamos a hipótese da existência de ecótipos morfológicos adaptados aos ambientes específicos utilizados pela espécie. Os crânios de 54 espécimes de museus de três diferentes ecorregiões no Brasil foram digitalizados nas vistas ventral, dorsal e lateral. Não foi detectado dimorfismo sexual no tamanho. Nossa hipótese foi corroborada, pois os espécimes da Amazônia são significativamente maiores do que os da Mata Atlântica e da Savana Uruguaia. A variação da forma do crânio entre os sexos, bem como entre ecorregiões, está correlacionada com a variação de tamanho. Correlações entre distância geográfica e forma do crânio não foram significativas. Houve correlação significativa entre tamanho e latitude, em um padrão inverso da regra de Bergmann, com espécimes maiores observados nas menores latitudes. As variáveis ambientais correlacionadas positivamente com o tamanho indicaram felinos maiores ocorrendo em regiões com maior temperatura e precipitação. A disponibilidade de recursos pode ser a causa da variação de tamanho observada, uma vez que aparentemente o jaguarundi preda presas de tamanhos diferentes ao longo de sua distribuição. Porém, são necessários mais dados ecológicos das demais ecorregiões, para testar a "regra do recurso" e elucidar os padrões e causas da variação do crânio deste felino.
Palavras-chave: Amazônia, Felidae, Herpailurus yagouaroundi, morfometria geométrica, variação intraespecífica.
INTRODUCTION
The jaguarundi, Herpailurus yagouaroundi É. Geoffroy Saint-Hilaire 1803, is the most distinct of all Neotropical cats, with short legs, a small head, rounded ears, and unmarked pelage (Hunter 2015). This small cat has one of the most extensive latitudinal ranges, second only to its closest relative, the puma, Puma concolor Linnaeus 1771, among felids of the Western Hemisphere (Johnson et al. 2006). This species occurs in the eastern and western low- lands of northern Mexico, throughout Central and South America to southeastern Brazil and central Argentina (Oliveira 1998; Caso et al. 2015).
The jaguarundi occupies a great variety of habitats from dry to wet forests, savanna woodlands, wet subalpine scrub savannas, swamplands, semi-arid scrub, chaparral, and dense grasslands (Hunter 2015). Within Brazil, the species occurs in all habitat types, from tropical and subtropical moist broadleaf forests (the Amazon basin in the north and the Atlantic Forest throughout the eastern seaboard), including the Cerrado in the central region, the Caatinga in the northeastern, the flooded grasslands in the western region, and towards the Uruguayan savanna in the southern region of the country; however, it is not particularly abundant anywhere (Oliveira et al. 2010; Almeida et al. 2013). This cat feeds mainly on small vertebrates, with mammals constituting nearly 60% of its prey items, followed by birds and reptiles (Tófoli et al. 2009; Giordano 2016). Despite being considered a generalist predator that occupies a broad spectrum of Neotropical habitats, this species has been the subject of little empirical research (Giordano 2016).
Current knowledge regarding the ecology and behavior of the jaguarundi is based mostly on diet data from subtropical moist broadleaf forests (Wang 2002; Tófoli et al. 2009; Rocha-Mendes et al. 2010; Bianchi et al. 2011; Rinaldi et al. 2015). In addition, ecological modelling of the two principal coat colors revealed that the dark/grey pelage is associated with wet, dense forests, whereas the red coat color is mainly associated with dry, open habitats (Silva et al. 2016). This spatial pattern does not seem to reflect a genetic structure but is caused by spatially heterogeneous, natural selection instead.
Knowledge of the variations in other morphological characteristics of jaguarundi is even scarcer. Particularly, the cranial morphology of the species has only been explored in family wide studies focused on evolutionary trends (Slater & Valkenburgh 2009; Morales & Giannini 2010; Sicuro 2011), functional morphology (Kiltie 1984; Sicuro & Oliveira 2011), and ontogeny (Segura et al. 2013). The jaguarundi is the smallest species of the Puma lineage, with an elongated braincase, an intermediate postorbital width and a relatively narrow masseteric scar at the zygomatic arch. Despite their phylogenetic proximity, only a few features could be considered conspicuous in all members of this lineage (as are the distinguished elongated angular processes) reinforcing the idea that its constituent species possess markedly derived skulls (Sicuro & Oliveira 2011).
The jaguarundi’s low and narrow skull is characterized by a short facial area, inconspicuous zygomatic arches, and an enlarged and convex braincase (Oliveira 1998). During ontogeny, its “adult” cranial shape is reached while the animal is still growing in size, and before sexual maturity (Segura et al. 2013). Additionally, this species overlaps extensively in skull size with most of the small Neotropical felids (Morales & Giannini 2010), and is considered functionally identical to the margay, Leopardus wiedii Schinz 1821, based on the relative maximum gape and jaw length (Kiltie 1984).
Some authors have proposed that there is a constant tension between divergent natural selection and unifying gene flow in the jaguarundi, making it an attractive model for in-depth evolutionary analyses (Silva et al. 2016). Patterns of morphological variation in the cranium of carnivorans with large distribution areas have been described as the result of adaptations to the range of environmental conditions (Gay & Best 1996), availability of resources (McNab 2010; Schiaffini 2016), or even to reduce competition with ecologically similar species occurring in sympatry (Bubadué 2016). Consequently, the wide geographic distribution of jaguarundi and its diverse range of habitats could give rise to different morphological ecotypes adapted to their respective environments (Turesson 1922).
We hypothesize that there should be a differentiation in cranium size or shape between populations from different ecoregions. We also wondered whether selected abiotic variables influenced cranial morphological variation. Thus, the goal of this study was to investigate cranium size and shape variation in jaguarundi. This will hopefully provide a basis for understanding how selective pressures, environmental heterogeneity, and geographic distance can modulate the evolution of the cranial morphology of this small carnivoran.
MATERIALS AND METHODS
Sample
The crania of 54 adult jaguarundi specimens (20 males, 13 females, and 21 unsexed) were photographed in ventral (n = 54), dorsal (n = 49), and lateral views (n = 53) with a Sony DSC-H9 digital camera placed at a fixed distance of 24 cm, using a support platform and 1.3 × zoom. Millimeter graph paper was used as a photographic background for subsequent scale referencing. The photographs were compiled using tpsUtil 1.64 (Rohlf 2013), and landmarks were digitized on ventral (17), dorsal (17), and lateral (11) views using tpsDig 2.26 (Rohlf 2015). The landmarks can be defined as type I (ventral: 4; dorsal: 1, 4; lateral: 3, 5, 6), type II (ventral: 1, 2, 3, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17; dorsal: 2, 3, 9, 10, 13, 14, 17; lateral: 4, 7, 8) and type III (dorsal: 5, 6, 7, 8, 11, 12, 15, 16; lateral: 1, 2, 9, 10, 11) (Fig. 1), according to Bookstein (1991). To avoid errors associated with modifications in the cranium during growth of the animals, the study included only adult individuals, which were classified as having complete permanent dentition (Segura et al. 2013).
For morphometric analysis, only specimens with known locality were used (Table 1). In the case of those for which the corresponding geographical coordinates were missing, the collection locality was georeferenced using Google Earth (Google 2018). The location records of the specimens were classified based on the ecoregions proposed by Olson et al. (2001), with some adaptations due to our small sample size, using the QGIS 2.18.25 software (Qgis Development Team 2018). The Amazon forest comprises a plethora of ecoregions, but all belong to humid forests. Therefore, those specimens from the moist forest ecoregions of Madeira-Tapajós, Uatuma-Trombetas, Xingu-Tocantins-Araguaia, Tapajós-Xingu, Tocantins/Pindare, and Marajó Várzea were grouped and labeled as “Amazon”. The specimens sampled in Alto Paraná Atlantic forest, Araucaria moist forest, Serra do Mar coastal forest, and Bahia Interior forest were clustered as “Atlantic Forest” since these ecoregions are all associated with forested formations (IBGE 1993).
Thus, for the analyses the specimens were grouped into three major Brazilian ecoregions (Fig. 2): Amazon (n = 16; 7 males, 5 females, and 4 unsexed), Atlantic Forest (n = 25; 7 males, 6 females, and 12 unsexed), and Uruguayan savanna (n = 13; 6 males, 2 females, and 5 unsexed). A list of the collections of the Brazilian institutions visited and the specimens analyzed is provided in Table 1. Each specimen was also assigned a value for the latitude of the locality where it was recorded, plus 19 bioclimatic variables (Table S1) taken from the WorldClim version 2.1 database (www.worldclim.org, Fick & Hijmans 2017) at a spatial resolution of 30s (∼ 1 km2).
Data analysis
First, landmark coordinates were superimposed using generalized Procrustes analysis (GPA), which removes effects not related to shape, such as position, scale, and orientation (Dryden & Mardia 1998). Although GPA scales all configurations to the same size, it does not remove the size-related component of shape variation. For this reason, it is necessary to test whether there is allometry by examining the correlation of cranium size and shape using multivariate regression (Monteiro 1999; Klingenberg 2016). Sexual dimorphism in cranium size (i.e., centroid size) was evaluated by Student’s t-test. Sexual dimorphism in cranial shape (i.e., Procrustes coordinates), as well as the interaction between the factors “sex” and “ecoregion” for both size and shape, was analyzed using Procrustes ANOVA (Adams & Otárola-Castillo 2013).
Size differences among the distinct ecoregions were tested using an ANOVA on log-transformed centroid size values, followed by Tukey’s tests for multiple comparisons. The hypothesis of the existence of differences in cranial shape among the ecoregions was tested using Procrustes ANOVA. The variation in shape was explored using principal component analysis (PCA), a dimensionality reduction technique. In the PCA plots, minimum-convex polygons were generated automatically by joining individuals by sex and ecoregion. We performed a linear discriminant analysis on the subset of principal components (PCs), which explained 99% of shape variance to calculate the correct classification percentages for each group (i.e., ecoregion). We used the leave-one-out procedure for cross-validation (Baylac & Friess 2005).
The relationship between cranium size and latitude was analyzed using multivariate regression. The association between size and the 19 bioclimatic variables was performed with a single ordinary least squares (OLS) regression analysis, conducted separately for each bioclimatic variable on centroid size (Table S1). Then, to avoid multicollinearity (Graham 2003), the correlation coefficients (r) between the predictor variables (i.e., bioclimatic variables) were calculated. For those with r >0.7, we applied a stepwise algorithm to retain the variables that provided the most suitable explanation. Finally, subsequent multiple regression analyses were performed to evaluate the contribution of each predictor variable to cranium size in the presence of the others. To avoid Type I errors related to spatial structure in the data, we tested the presence of spatial autocorrelation by applying Moran’s I index (Diniz-Filho et al. 2003) in the residuals of the OLS.
It is possible to outline a correspondence between genotype and phenotype, thus enabling the examination of the isolation-by-distance model. This is a commonly used model in genetic studies, which predicts that genetic divergence increases with geographical distance (Wright 1943). The localities of the specimens were used to generate a geographical distance matrix with Geographic Distance Matrix Generator v1.2.3 (Ersts 2018). A Mahalanobis distance matrix was generated with the Procrustes distance of the consensus shape of the specimens. These matrices were correlated through an RV coefficient (Heo & Gabriel 1997), to calculate the relationship between the morphological variation and the geographic distance of the specimens.
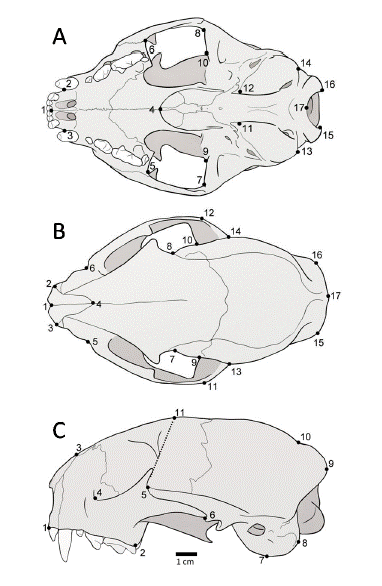
Fig. 1
Landmarks digitized in the crania of Herpailurus yagouaroundi. Ventral view (A): midpoint of incisive series (1); anteriormost point of canines (2–3); posteriormost point of interpalatine suture (4); anterior point (5–6), external edge (7–8) and internal edge (9–10) of temporal fossa; anterior (11–12) and posterior (13–14) extremes of tympanic bulla; internal apex of occipital condyle (15–16); inferiormost point in the foramen magnum (17). Dorsal view (B): anteriormost point of the sutura internasalis (1); tip of nasal process (2–3); midpoint of sutura frontonasalis (4); infraorbital foramen (5–6); narrowest point between frontals (7–8); anterior point of squamous (9–10); most external posterior point of zygomatic arch (11–12); posterior point of squamous (13–14); lateralmost point of occipital (15–16); inion (17). Lateral view (C): anteriormost (1) and posteriormost (2) points of dentary row; anteriormost point of sutura frontonasalis (3); lacrimal foramen (4); dorsal (5) and ventral (6) ends of the squamous-jugal suture along the zygomatic arch; ventralmost point of tympanic bulla (7); tip of paracondylar process (8); posteriormost point of occipital (9); intersection of temporal line and sagittal crest (10); projection of the orbital processes onto the frontal midline (11).
Statistical analyses were conducted in R v.3.2.2 (R Development Core Team 2018) using the packages MASS (Venables & Ripley 2013), ape (Paradis et al. 2004), stats (R Development Core Team 2018), ade4 (Dray & Dufour 2007), letsR (Vilela & Villalobos 2015), and geomorph (Adams & Otárola-Castillo 2013).
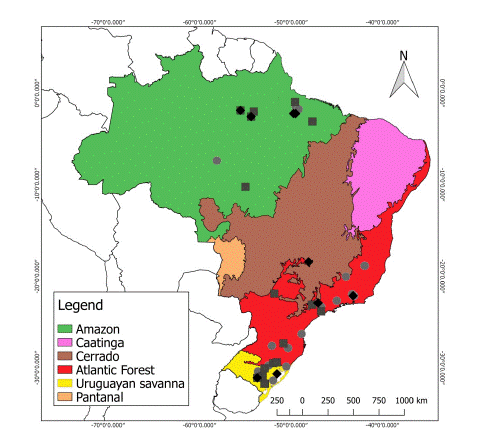
Fig. 2
Geographic distribution of Herpailurus yagouaroundi specimens analyzed (squares: males, diamonds: females, circles: unsexed), indicating their ecoregions of origin.
RESULTS
Sexual dimorphism
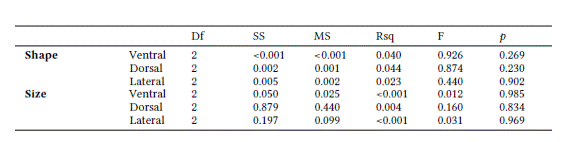
No sexual dimorphism in size was observed in any cranial view (ventral: t = -1.741, df = 22.2, p = 0.095; dorsal: t = -1.793, df = 23.821, p = 0.085; and lateral: t = -1.986, df = 24.225, p = 0.058). The Tukey’s test also showed no sexual dimorphism in the cranium by ecoregion: Amazon (ventral: p = 0.338; dorsal: p = 0.258; lateral; p = 0.378), Atlantic Forest (ventral: p = 0.190; dorsal: p = 0.516; lateral; p = 0.274), and Uruguayan savanna (ventral: p = 0.503; dorsal: p = 0.581; lateral; p = 0.751).
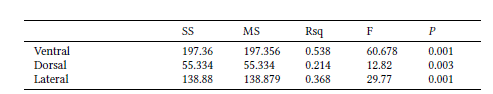
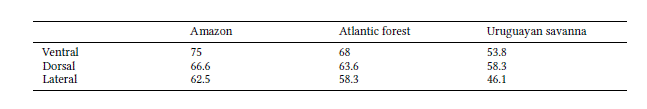
The effect of size was significant, with different percentages of shape variation predicted by size for the ventral (31.7%; p = 0.001), dorsal (33.9%; p = 0.001), and lateral (20.55%; p = 0.001) views. The Procrustes ANOVA indicated no significant sexual dimorphism in shape (ventral: R2 = 0.067, p = 0.054; lateral: R2 = 0.054, p = 0.08), except for dorsal view (R2 = 0.10, p = 0.019). By plotting the first two PCs, the PCA displayed relative overlap between male and female cranial shapes in all views (Fig. 3). The first nine PCs were associated with 95% of the total variance in the ventral view, while the first seven explained 95% of the variation in the dorsal view, and the first 10 were associated with 94.7% of the variation in the lateral view.
The Procrustes ANOVA indicated no significant interaction (p >0.05) between the factors “sex” and “ecoregion” for either cranium size or shape in any of the views (Table 2). Thus, males, females, and the unsexed specimens were pooled together for all subsequent comparisons among ecoregions.
Cranium size
The ANOVA showed significant differences in cranium size among specimens from different ecoregions for the three views (p <0.001). Subsequent Tukey’s tests revealed that the specimens from Amazon were significantly different from those from the other ecoregions sampled. As the results were similar for all cranial views, we opted to present only the boxplot for the ventral view (Fig. 4).
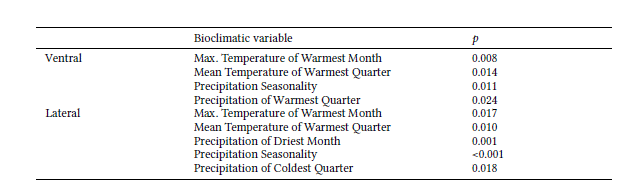
There was a significant association between size and latitude in the three cranial views (Table 3). Of the bioclimatic variables evaluated, the maximum temperature of the warmest month, mean temperature of the warmest quarter, precipitation seasonality, and precipitation of the warmest quarter explained 65.3% of the cranium size variation in the ventral view. These first three variables, together with precipitation of the driest month and precipitation of the coldest quarter, were the most important for 62.9% of the size variance in the lateral view. There was no correlation between the independent variables and cranium size for the dorsal view (Table 4). No Moran’s I coefficients were significant, meaning that no spatial patterns were apparent in the cranium size data (Table S2).
Cranial shape
The multivariate regression of shape on size revealed a significant size effect on the three cranial views. The amount of shape predicted by size was 20.7% for ventral (p = 0.001), 27.8% for dorsal (p = 0.001), and 12.6% for lateral (p = 0.001) views. Significant shape differences among ecoregions were found in the Procrustes ANOVA (ventral: R2 = 0.185, p = 0.001, dorsal: R2 = 0.158, p = 0.002, and lateral: R2 = 0.089, p = 0.001).
By plotting PC1 with PC2, PCA did not cluster separate groups among ecoregions. The only relative differentiation is seen along the first axis for ventral view, where the overlap between Amazonian specimens and the other ecoregions is minimal. The first 10 PCs cumulatively explained 95.2% of the total variance in ventral view, while eight PCs explained 95.2% in dorsal view, and the first 11 explained 94.5% in lateral view (Fig. 5). The percentages of correct classifications for the cranial views are shown in Table 5.
The correlations between the geographical distance and the Procrustes distances of the consensus shape were conducted separately for the sexes. There were no significant correlations for ventral (males: p = 0.472; females: p = 0.370), dorsal (males: p = 0.256; females: p = 0.388), and lateral views (males: p = 0.315; females: p = 0.185).
DISCUSSION
Sexual dimorphism
Our results did not indicate significant sexual dimorphism in size in the jaguarundi, which agrees with previous studies by Sicuro & Oliveira (2011) and Morales & Giannini (2010). Despite the trend common to most felid species that males tend to be 5% to 10% larger than females (Wilson & Mittermeier 2009), these authors stated that the jaguarundi might not follow this dimorphic pattern. However, Segura et al. (2013) found significant sexual dimorphism in cranium size in this species. Without statistical analysis of the data, Oliveira (1998) presented values of the species’ mean total body length and weight with the males and females from Brazil averaging 109.9 cm and 5.6 kg and 98.9 cm and 4.5 kg, respectively. Our measurements of total cranial length (Table 1) also showed that the average values for male specimens were larger than those for females (Amazon: males = 127.84 mm, females = 120.99 mm; Atlantic Forest:males = 111.45 mm, females = 104.26 mm; Uruguayan savanna: males = 113.46 mm, females = 104.77 mm),but our analyses still did not find significant sexual dimorphism in cranium size (i.e., centroid size) inthe sampled ecoregions.
According to Segura et al. (2013), both sexes exhibit the same ontogenetic trajectory, but female jaguarundi reaches the adult size at an earlier age than males. Despite the relative overlap between sexes we found in all views displayed in PCA, most male specimens were placed toward positive values. The mean shape along PC1 (Fig. 3) follows the morphofunctional pattern in Neotropical felids, where males have a narrower postorbital constriction and a longer temporal fossa, which is reflected in a stronger bite force through the action of the muscle temporalis (Morales & Giannini 2010). Even though the differences in size between sexes were non-significant, cranial size and shape were statistically correlated (Klingenberg 2016) and allometry accounted for moderate proportions (20.5% – 33.9%) of the sexual shape variation. Thus, what we see here might reinforce the evidence for allometric changes of cranial shape as reported for carnivorans in general (Ercoli 2017), where allometric and sexual dimorphic differences usually affect muscular insertion areas.
Cranium size
Among ecoregions, a clear size difference was found, with the specimens from the Amazon being significantly larger than specimens from the Atlantic Forest and Uruguayan savanna ecoregions. This difference is the opposite of the size patterns reported for other felid species that occur in the same regions, such as the jaguar Panthera onca Linnaeus, 1758 and the puma. Unlike our results previously described for the jaguarundi, these species tend to be smaller in dense tropical forests, with larger jaguars found in more open environments, such as Cerrado and Pantanal, and larger pumas found in drier areas and temperate regions (Iriarte et al. 1990; Gay & Best 1996; Gambarini et al. 2016).
Size differentiation might occur as a mechanism for sympatric species to reduce niche overlap (Kiltie 1984). However, according to Morales & Giannini (2010), the jaguarundi overlaps extensively in skull size and morphology with most small Neotropical cats. Thus, they hypothesized that the species segregated along another niche dimension (temporal segregation), considering its daily activity pattern as the key ecological character for the jaguarundi to reduce competition and allow species coexistence (Di Bitetti et al. 2010; Dias et al. 2019). In this context, we believe that the cause for the significant differences in cranium size found among the jaguarundis of the studied ecoregions might not be primarily related to mechanisms to reduce niche overlap with similar and closely related species.
According to Gay & Best (1996), the pattern of variation in size of pumas, the jaguarundi’s closest relative, is correlated with latitude, consistent with Bergmann’s rule, according to which endothermic species tend to be larger in higher latitudes and cooler climates (Bergmann 1847). Our results also showed a correlation of cranium size with latitude for jaguarundis, however, the pattern of variation is the reverse of Bergmann’s rule because the larger specimens are from the ecoregion at the lower sampled latitudes, that is, closest to the equator, between 10° and 0°S. Measures of total cranial length (Table 1) reinforced this pattern, with the Amazonian jaguarundi reaching the highest average of 124.78 mm, while cranial length from Uruguayan savanna and Atlantic Forest specimens averaged 111.36 mm and 109.49 mm, respectively. We have compiled some few body mass values, as follows: Amazon, one male (7.6 kg, MNHN 24901); Uruguayan savanna, one male (5.25 kg, MCNU 4125); and Atlantic Forest, one male (5.25 kg, UFSC 3531), and two females (4 kg, UFSC 3792; 3 kg, MNHN 79548).
Although nearly 50% of the mammalian carnivores already studied follow Bergmann’s rule (Meiri et al. 2004), its validity has been criticized (McNab 1999).
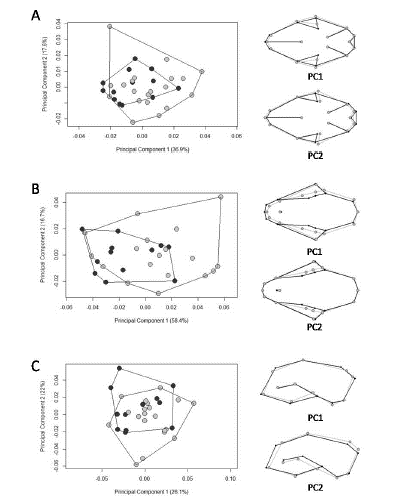
Fig. 3
Scatterplots of the two first principal components (PCs) of cranial shape variation in Herpailurus yagouaroundi specimens from Brazil, in ventral (A), dorsal (B) and lateral (C) views. Black wireframes represent positive PC scores and grey wireframes negative PC scores. Males are represented by light grey circles and females by dark grey circles.
Another phenomenon has been proposed by McNab (2010), called the “resource rule”, which establishes that the common element in the geographic variation in body size of mammals is the variation in the abundance and size of resources, and the factors that influence their availability or quality. Although the jaguarundi inhabits most of the Neotropics, its food habits are poorly understood in most of its range, especially in the Amazonian region. The only published data from this region was based on a very small sample lacking estimates of prey availability (Tirelli et al. 2018). This study revealed armadillo genus Dasypus Linnaeus 1758 and agouti Dasyprocta azarae Lichtenstein 1823 as prey species, both taxa weighing over 1 kg. Preys of this weight are consumed in low amounts in other ecoregions of Brazil (Trovati et al. 2008; Silva-Pereira et al. 2011; Migliorini et al. 2018). Within the Brazilian portion of the Uruguayan savanna, the jaguarundi seems to exhibit specialized food habits, with a diet comprising mainly guinea pigs genus Cavia Pallas 1766 (Migliorini et al. 2018), weighing less than 1 kg. Thus, our hypothesis is that jaguarundis have larger crania in the Amazon ecoregion than in other environments because they have specialized in hunting larger prey. Additionally, even if jaguarundis preying on armadillo and agouti in the Amazon proved to be an artifact caused by the small sample size of that study, some authors have pointed out that Sigmodontinae rodent lineages tend to increase in size in these forested regions (Rodríguez et al. 2008; Maestri et al. 2016).
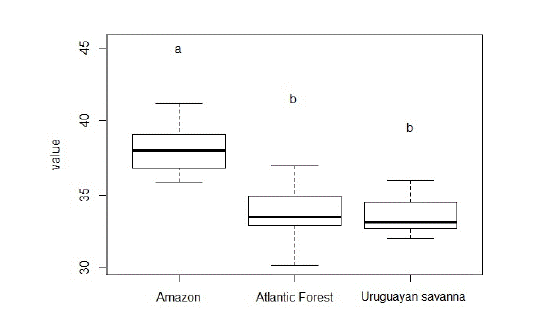
Fig. 4
Boxplot for log-centroid size values of the ventral view of Herpailurus yagouaroundi crania among ecoregions in Brazil. The bold black lines represent median values, the boxes the first interquartile, and the bars the second interquartile. Letters above the plots represent the two significant different groups suggested by Tukey’s test.
The bioclimatic variables positively correlated with the jaguarundi’s cranium size indicate that larger cats occur in regions with higher temperatures during the warmest period and with the highest precipitation. This result could be related to environmental factors, such as habitat and climate stability. Similar trends have been reported for some New World monkeys, such as capuchins Cebus Erxleben 1777, Sapajus Linnaeus 1758, and howlers Alouatta Lacépède 1799 (Meloro et al. 2014), and marsupials e.g., wooly opossums: Caluromys J. A. Allen 1900 (Magnus et al. 2017). Since food resources varies seasonally, with high availability of fruit during the rainy seasons in most tropical regions, these mammals seem to respond to habitat productivity, becoming larger in regions with less climate seasonality and high and constant rate of rainfall, such as in the Amazon forest. It is important to note that abiotic variables associated with size variation, such as temperature and precipitation, are correlated when we consider large ecoregions such as Amazon versus the Atlantic Forest. The Amazonian region has warmer temperatures and more precipitation in comparison with the Atlantic Forest or Uruguayan Savanna.
Cranial shape
Shape differentiation among ecoregions was statistically significant, although the jaguarundis from the Amazon, Atlantic Forest, and Uruguayan savanna overlapped on the PCA plots (Fig. 5). The highest percentage of correct classification for skull shape (75%) was for the Amazonian specimens in ventral view, which was also the only grouping relatively differentiated on PCA, which represented specimens with a wider interior edge of the temporal fossa and slightly narrower and longer crania (Fig. 5A). We found that the variation in shape among ecoregions was significantly associated with variation in size. The co-evolution between size and shape is a known trend in Felidae skull morphology (Morales & Giannini 2010; Sicuro 2011), given that felids exhibit a remarkable interspecific homogeneity that is functionally associated with a uniform bite (Christiansen 2008).
Selection to maintain high bite forces to body mass implies elongation and elevation of the posterior part of the crania in larger species (Christiansen 2008), as we observed in the cranial shape variation in the jaguarundi specimens. According to Sicuro & Oliveira (2011), some craniomandibular shape changes in felids may result from casuistic fixation of phenotypic fluctuations rather than specific selections. Despite this, skull-jaw performance is always preserved, as required by the selective pressures related to their fit to hypercarnivory (Sicuro 2011). Moreover, general felid skull size is correlated with bite force and prey size (Sicuro & Oliveira 2011). Thus, our data reinforce the “resource rule” hypothesis (McNab 2010), as the evolutionary allometric trend in larger jaguarundi from the Amazon might reflect the consumption of large prey in that ecoregion.
The non-significant correlation between the geographical distance and the Procrustes distance of the consensus shape indicates that jaguarundi cranial shape variation does not follow a pattern of isolation-by-distance. This seems to be consistent with the cat’s genetic variability reported by Ruiz-García et al. (2018), who proposed a single subspecies for Central and South America. However, a study by Pires (2012) in which mitochondrial DNA and microsatellite markers were used, suggested that isolation-by-distance may have played a role in limiting gene flow between populations from the south and north of the Amazon River. Although our sample included four specimens from north of the river, we did not find any significant correlation between geographical distance and morphology. As also noted by Pires (2012), this could be the result of gene flow between these broad regions until the recent past.
CONCLUSIONS
The aim of this study was to explore the patterns of morphological variation in the cranium of jaguarundi. The hypothesis of the existence of ecotypes adapted to their respective environments was supported, since we found significantly larger specimens in the Amazon ecoregion relative to the samples from the Atlantic Forest and Uruguayan savanna and significant differences in shape among these ecoregions. We hypothesize that the larger cranium size of Amazonian specimens is related to a possible specialization to feeding on prey over 1 kg. Unfortunately, information on the carnivoran guild composition in the Amazon region is still limited. More data concerning the ecology of this species is still needed to assess the effect of prey size and availability on the cranium size of this small predator. Cranial shape variation between ecoregions was mostly explained by the effect of size, and no correlation with geographical distance was detected. Further studies could benefit from sampling additional ecoregions, to fully understand the patterns of cranial variation in this cat and its possible causes.
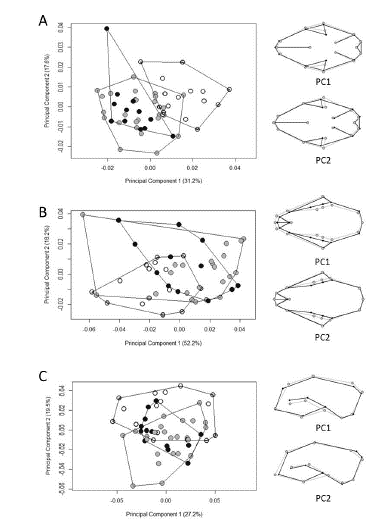
Fig. 5
Scatterplots of the two first axes of the principal components (PCs) of cranial shape variation in Herpailurus yagouaroundi specimens from Brazil, in ventral (A), dorsal (B) and lateral (C) views. Specimens from the Amazon are represented as white circles, from the Atlantic Forest as grey circles and from the Uruguayan savanna as black circles. Black wireframes represent positive PC scores and grey wireframes negative scores.
Acknowledgments
We thank all the curators of the following institutions for their kindness in providing the necessary information and for allowing access to the collections: Dr. Tatiane Trigo (FZB/RS), Dr. Carla Fontana (MCT-PUCRS), Dr. Alexandre Uarth Christoff (MCNU), Dr. Maurício Graipel (LAMAq- UFSC), Juliana Barros (MZUSP), Karina Rebelo de Almeida (ZUEC-MAM), Dr. João Alves de Oliveira (MNHN), Dr. Fernando Perini (CCT-UFMG), MSc. Claudia Guimarães Costa (MCN-M) and Dr. Alexandra Bezerra (MPEG). We are also grateful to the postgraduate scholarship granted by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) – finance Code 001, as well as the support by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) - grant proc. No. 309219/2018-3 for the first and second authors, respectively. Special thanks to Felipe Bortolotto Peters for his continued contribution in providing material for the scientific collection and for the research carried out at LABIMAVE. The authors are also thankful for the assistance with the statistical analysis of Tiago Gomes dos Santos. Additional thanks to Mauro Anderson da Silva Bossi for contributions throughout the development of this study.
REFERENCES
Adams, D. C., & E. Otárola-Castillo. 2013. Geomorph: an R package for the collection and analysis of geometric morphometric shape data. Methods in Ecology and Evolution 4:393-399. https://doi.org/10.1111/2041-210x.12035
Almeida, L. B., D. Queirolo, B. M. Beisiegel, & T. G. Oliveira. 2013. Avaliação do estado de conservação do Gato-mourisco Puma yagouaroundi (É. Geoffroy Saint-Hilaire, 1803) no Brasil. Biodiversidade Brasileira 3:99-106.
Baylac, M., & M. Friess. 2005. Fourier descriptors, Procrustes superimposition and data dimensionality: an example of cranial shape analysis in modern human populations. Modern morphometrics in physical anthropology (D. E. Slice, ed.). Springer-Verlag, Boston. https://doi.org/10.1007/0-387-27614-96
Bergmann, C. 1847. Ueber die Verhältnisse der Wärmeökonomie der Tiere zu ihrer Grösse. Göttinger Studien 3:595-708.
Bianchi, R. D. C., A. F. Rosa, A. Gatti, & S, L. Mendes. 2011. Diet of margay, Leopardus wiedii, and jaguarundi, Puma yagouaroundi (Carnivora: Felidae) in Atlantic rainforest, Brazil. Zoologia 1:127- 132. https://doi.org/10.1590/s1984-46702011000100018
Bookstein, F. L. 1991. Morphometric tools for landmark data. Geometry and biology. Cambridge University Press. New York.
Bubadué, J. M. 2016. Character displacement under influence of Bergmann’s rule in Cerdocyon thous (Mammalia: Canidae). Hystrix 2:1-8.
Caso, A., T. De Oliveira, & S. V. Carvajal. 2015. Herpailurus yagouaroundi. The IUCN Red List of Threatened Species: e.T9948A50653167. https://doi.org/10.2305/iucn.uk.2015- 2.rlts.t9948a50653167.en
Christiansen, P. E. R. 2008. Evolution of skull and mandible shape in cats (Carnivora: Felidae). PLoS ONE, 3:e2807. https://doi.org/10.1371/journal.pone.0002807
Dias, D. M., R. L. Massara, C. B. De Campos, & F. H. G. Rodrigues. 2019. Feline predator–prey relationships in a semi-arid biome in Brazil. Journal of Zoology 307:1-10. https://doi.org/10.1111/jzo.12647
Di Bitetti, M. S., C. D. De Angelo, Y. E. Di Blanco, & A. Paviolo. 2010. Niche partitioning and species coexistence in a Neotropical felid assemblage. Acta Oecologica 36:403-412. https://doi.org/10.1016/j.actao.2010.04.001
Diniz-Filho, J. A. F., L. M. Bini, & B. A. Hawkins. 2003. Spatial autocorrelation and red herrings in geographical ecology. Global Ecology and Biogeography 12:53-64. https://doi.org/10.1046/j.1466-822x.2003.00322.x
Dray, S., & A. B. Dufour. 2007. The ade4 package: implementing the duality diagram for ecologists. Journal of Statistical Software 22:1-20. https://doi.org/10.18637/jss.v022.i04
Dryden, I. L., & K. V. Mardia. 1998. Statistical analysis of shape. 1st edition. John Wiley & Sons, Inc, New York.
Ercoli, M. D. 2017. Morphofunctional analysis of the mastoid region of the extinct South American mustelid †Stipanicicia pettorutii. Earth and Environmental Science Transactions of the Royal Society of Edinburgh 106:337-349. https://doi.org/10.1017/s1755691016000220
Ersts P. J. 2018. Geographic Distance Matrix Generator. Ver. 1.2.3. Importar imagen American Museum of Natural History, Center for Biodiversity and Conservation. https://www.biodiversityinformatics.amnh.org/opensource/gdmg
Fick, S. E, & R. J. Hijmans. 2017. WorldClim 2: new 1km spatial resolution climate surfaces for global land areas. International Journal of Climatology 37:4302-4315. https://doi.org/10.1002/joc.5086
Gambarini, A., L. D. Mota, M. Haberfeld, & R. C. Paula. 2016. Panthera onca: À sombra das florestas. 1st edition. Avis Brasilis, Vinhedo.
Gay, S. W., & T. L. Best. 1996. Relationships between abiotic variables and geographic variation in skulls of pumas (Puma concolor: Mammalia, Felidae) in North and South America. Zoological Journal of the Linnean Society 117:259-282. https://doi.org/10.1111/j.1096-3642.1996.tb02190.x
Giordano, A. J. 2016. Ecology and status of the jaguarundi Puma yagouaroundi: a synthesis of existing knowledge. Mammal Review 46:30-43. https://doi.org/10.1111/mam.12051
Google. 2018. Google Earth website. https://www.earth.google.com/
Graham, M. H. 2003. Confronting multicollinearity in ecological multiple regression. Ecology 84:2809-2815. https://doi.org/10.1890/02-3114
Heo, M., & K. R. Gabriel. 1997. A permutation test of association between configurations by means of RV coefficient. Communication in Stattistics – Simulation and Computation 27:843-856. https://doi.org/10.1080/03610919808813512
Hunter, L. 2015. Wild cats of the world. Bloomsbury Publishing. London.
[IBGE] Instituto Brasileiro de Geografia Estatística. 1993. Mapa de Vegetação do Brasil. Map 1:5,000,000. Rio de Janeiro (Brazil): IBGE. https://doi.org/10.17143/ciaed/xxiilciaed.2017.00322
Iriarte, J. A., W. L. Franklin, W. E. Johnson, & K. H. Redford. 1990. Biogeographic variation of food habits and body size of the America puma. Oecologia 85:185-190. https://doi.org/10.1007/bf00319400
Johnson, W. E. et al. 2006. The late Miocene radiation of modern Felidae: a genetic assessment. Science 311:73-77. https://doi.org/10.1126/science.1122277
Kiltie, R. A. 1984. Size ratios among sympatric neotropical cats. Oecologia 61:411-416. https://doi.org/10.1007/bf00379644
Klingenberg, C. P. 2016. Size, shape, and form: concepts of allometry in geometric morphometrics. Development Genes and Evolution 226:113-137. https://doi.org/10.1007/s00427-016-0539-2
Maestri, R. et al. 2016. Geographical variation of body size in sigmodontine rodents depends on both environment and phylogenetic composition of communities. Journal of Biogeography 43:1192-1202. https://doi.org/10.1111/jbi.12718
Magnus, L. Z., R. F. Machado, & N. Cáceres. 2017. Comparative ecogeographical variation in skull size and shape of two species of woolly opossums (genus Caluromys). Zoologischer Anzeiger 267:139-150. https://doi.org/10.1016/j.jcz.2017.03.003
McNab, B. K. 1999. On the comparative ecological and evolutionary significance of total and mass-specific rates of metabolism. Physiological and Biochemical Zoology 72:642-644. https://doi.org/10.1086/316701
McNab, B. K. 2010. Geographic and temporal correlations of mammalian size reconsidered: a resource rule. Oecologia 164:13-23. https://doi.org/10.1007/s00442-010-1621-5
Meiri, S., T. Dayan, & D. Simberloff. 2004. Carnivores, biases and Bergmann’s rule. Biological Journal of the Linnean Society 81:579-588. https://doi.org/10.1111/j.1095-8312.2004.00310.x
Meloro, C. et al. 2014. In and out the Amazonia: evolutionary ecomorphology in howler and capuchin monkeys. Evolutionary Biology 41:38-51. https://doi.org/10.1007/s11692-013-9244-5
Migliorini, R. P., F. B. Peters, M. O. Favarini, C. B. Kasper. 2018. Trophic ecology of sympatric small cats in the Brazilian Pampa. PLoS ONE 13:e0201257. https://doi.org/10.1371/journal.pone.0201257
Monteiro, L. R.1999. Multivariate regression models and geometric morphometrics: the search for causal factors in the analysis of shape. Systematic Biology 48:192-199. https://doi.org/10.1080/106351599260526
Morales, M. M., & N. P. Giannini. 2010. Morphofunctional patterns in Neotropical felids: species co-existence and historical assembly. Biological Journal of the Linnean Society 100:711-724. https://doi.org/10.1111/j.1095-8312.2010.01461.x
Oliveira, T. G.1998. Herpailurus yagouaroundi. Mammalian Species 578:1-6. https://doi.org/10.2307/3504500
Oliveira, T. G. et al. 2010. Ocelot ecology and its effect on the small-felid guild in the lowland Neotropics. Biology and Conservation of Wild Felids (D. Macdonald & A. Loveridge, Orgs.). Oxford University Press. New York.
Olson, D. M. et al. 2001. Terrestrial Ecoregions of the World: A New Map of Life on Earth. BioScience 51:933-938.
Paradis, E., J. Claude, & K. Strimmer. 2004. APE: Analyses of phylogenetics and Evolution in R language. Bioinformatics 20:289-290. https://doi.org/10.1093/bioinformatics/btg412
Pires, C. B. 2012. Diversidade Genética e Filogeografia de Puma yagouaroundi (Mammalia, Carnivora, Felidae). Dissertação de mestrado. Pontifícia Universidade Católica do Rio Grande do Sul, Porto Alegre, Brazil. https://doi.org/10.5327/z2447-211520161600027
Qgis Development Team, 2018. QGIS Geographic Information System. Open Source Geospatial Foundation Project. http://www.qgis.osgeo.org
R Development Core Team. 2018. R: a language and environment for statistical computing. R Foundation for Statistical Computing. Vienna: Austria. http://www.rproject.org
Rinaldi, A. R., F. H. Rodriguez, A. L. Carvalho, & F. C. Passos. 2015. Feeding of small Neotropical felids (Felidae: Carnivora) and trophic niche overlap in antropized mosaic landscape, South Brazilian. Biotemas 28:155-168. https://doi.org/10.5007/2175-7925.2015v28n4p155
Rocha-Mendes, F., S. B. Mikich, J. Quadros, & W. A. Pedro. 2010. Feeding ecology of carnivores (Mammalia, Carnivora) in Atlantic forest remnants, southern Brazil. Biota Neotropica 10:21-30. https://doi.org/10.1590/s1676-06032010000400001
Rodríguez, M. Á., M. Á. Olalla-Tárraga, & B. A. Hawkins. 2008. Bergmann’s rule and the geography of mammal body size in the Western Hemisphere. Global Ecology and Biogeography 17:274-283. https://doi.org/10.1111/j.1466-8238.2007.00363.x
Rohlf, F. J. 2013. tpsUtil, file utility program. Ver. 1.64. Department of Ecology and Evolution, State University of New York at Stony Brook.
Rohlf, F. J. 2015. tpsDig, digitize landmarks and outlines. Ver. 2.19. Department of Ecology and Evolution, State University of New York at Stony Brook.
Ruiz-García, M., M. Pinedo-Castro, & J. M. Shostell. 2018. Mitogenomics of the jaguarundi (Puma yagouaroundi, Felidae, Carnivora): Disagreement between morphological subspecies and molecular data. Mammalian Biology 93:153-168. https://doi.org/10.1016/j.mambio.2018.09.004
Schiaffini, M. I. 2016. A test of the Resource’s and Bergmann’s rules in a widely distributed small carnivore from southern South America, Conepatus chinga (Molina, 1782) (Carnivora: Mephitidae). Mammalian Biology 81:73-81. https://doi.org/10.1016/j.mambio.2014.11.007
Slater, G. J., & B. Van Valkenburgh. 2009. Allometry and performance: the evolution of skull form and function in felids. Journal of Evolutionary Biology 22:2278-2287. https://doi.org/10.1111/j.1420-9101.2009.01845.x
Segura, V., F. Prevosti, & G. Cassini. 2013. Cranial ontogeny in the Puma lineage, Puma concolor, Herpailurus yagouaroundi, and Acinonyx jubatus (Carnivora: Felidae): a three dimensional geometric morphometric approach. Zoological Journal of Linnean Society 169:235-250. https://doi.org/10.1111/zoj.12047
Sicuro, F. L. 2011. Evolutionary trends on extant cat skull morphology (Carnivora: Felidae): a three dimensional geometrical approach. Biological Journal of Linnean Society 103:176-190. https://doi.org/10.1111/j.1095-8312.2011.01636.x
Sicuro, F. L., & L. F. B. Oliveira. 2011. Skull morphology and functionality of extant Felidae (Mammalia: Carnivora): a phylogenetic and evolutionary perspective. Zoological Journal of Linnean Society 161:414-462. https://doi.org/10.1111/j.1096-3642.2010.00636.x
Silva-Pereira, J. E., R. F. Mororios, D. R. Bilski, & F. C. Passos. 2011. Diets of three sympatric Neotropical small cats: Food niche overlap and interspecies differences in prey consumption. Mammalian Biology 76:308-312. https://doi.org/10.1016/j.mambio.2010.09.001
Silva, L. G. et al. 2016. Biogeography of polymorphic phenotypes: mapping and ecological modelling of coat colour variants in an elusive Neotropical cat, the jaguarundi (Puma yagouaroundi). Journal of Zoology 299:295-303. https://doi.org/10.1111/jzo.12358
Tirelli, F. P., T. R. De Freitas, F. Michalski, A. R. Percequillo, & E. Eizirik.2018. Using reliable predator identification to investigate feeding habits of Neotropical carnivores (Mammalia, Carnivora) in a deforestation frontier of the Brazilian Amazon. Mammalia 83:415-427. https://doi.org/10.1515/mammalia-2018-0106
Tófoli, C. F., F. Rohe, & E. Z. F. Setz. 2009. Jaguarundi (Puma yagouaroundi) (Geoffroy, 1803) (Carnivora, Felidae) food habits in a mosaic of Atlantic Rainforest and eucalypt plantations of southeastern Brazil. Brazilian Journal of Biology 69:871-877. https://doi.org/10.1590/s1519-69842009000400015
Trovati, R. G., C. B. Campos, & B. A. Brito. 2008. Nota sobre convergência e divergência alimentar de canídeos e felídeos (Mamalia: Carnivora) simpátricos no Cerrado brasileiro. Neotropical Biology and Conservation 3:95-100.
Turesson, G. 1922. The species and the variety as ecological units. Hereditas 3:100-113.
Venables, W. N., & B. D. Ripley. 2013. MASS: Modern applied statistics with S-PLUS. 4th edition. Springer Science & Business Media, New York.
Vilela, B., & F. Villalobos. 2015. letsR: a new R package for data handling and analysis in macroecology. Methods in Ecology and Evolution 10:1229-1234. https://doi.org/10.1111/2041-210x.12401
Wang, E. 2002. Diets of ocelots (Leopardus pardalis), margays (L. wiedii), and oncillas (L. tigrinus) in the Atlantic rainforest in southeast Brazil. Studies on Neotropical Fauna and Environment 37:207-212. https://doi.org/10.1076/snfe.37.3.207.8564
Wilson, D. & Mittermeier, R. 2009. Handbook of the Mammals of the World: Carnivores. Vol. 1. Lynx Editions. Barcelona.
Wright, S. 1943. Isolation by distance. Genetics. 28:114-138.
Información adicional
Editor asociado: Aldo Iván Vassallo
Cite as: Migliorini, R. P., R. Fornel, & C. B. Kasper. 2021. Cranial morphological variability of a small neotropical cat revealed by geometric morphometrics. Mastozoología Neotropical, 28(2):e0472. https://doi.org/10.31687/saremMN.21.28.2.0.02.e0472