ARTÍCULO
AERIAL INSECTIVOROUS BATS IN THE BRAZILIAN PANTANAL: DIVERSITY AND ACTIVITY PATTERNS IN RESPONSE TO HABITAT AND MICROCLIMATE
MORCEGOS INSETÍVOROS AÉREOS NO PANTANAL BRASILEIRO: DIVERSIDADE E PADRÕES DE ATIVIDADE EM RESPOSTA AO HABITAT E AO MICROCLIMA
AERIAL INSECTIVOROUS BATS IN THE BRAZILIAN PANTANAL: DIVERSITY AND ACTIVITY PATTERNS IN RESPONSE TO HABITAT AND MICROCLIMATE
Mastozoología Neotropical, vol. 28, núm. 2, p. 604, 2021
Sociedad Argentina para el Estudio de los Mamíferos
Recepción: 30 Junio 2021
Aprobación: 14 Octubre 2021
Abstract: In tropical environments, landscape heterogeneity is a key factor determining species richness gradients, while climatic conditions are commonly related to the occurrence and activity patterns of most animal species. We used acoustic monitoring data to test the influence of microclimate variables and habitat on the activity pattern of aerial insectivorous bats in three distinct environments in northern Pantanal, Brazil – Cambarazal, Murundu field and Riparian area. We hypothesized that foraging and feeding activities should be positively related to humidity and temperature; and species composition should differ between habitats. In 30 sampling nights, we obtained 6049 records and identified 16 species and three species complexes distributed in five families. There were no significant differences in species richness between the three habitats, but the riparian area, the most heterogeneous environment sampled, presented the highest levels of foraging and feeding activities. Species composition varied with the sampled habitat, demonstrating that different species use and perceive distinct landscapes in different ways. Only the foraging activity of open-space bats responded to the interaction between relative humidity and different habitat types during the Pantanal’s dry season. Our results, obtained exclusively by acoustic sampling, show that many aerial insectivore species and activity patterns are subsampled in studies done exclusively with mist-nets.
Keywords: Acoustic monitoring, aerial insectivorous bats, Chiroptera, microclimate, Pantanal.
Resumo:
Em ambientes tropicais, a heterogeneidade da paisagem é um fator chave na determinação de gradientes de riqueza de espécies, enquanto as condições climáticas são comumente relacionadas aos padrões de ocorrência e atividade da maioria das espécies animais. Utilizamos dados de monitoramento acústico para testar a influência das variáveis do microclima e do habitat no padrão de atividade de morcegos insetívoros aéreos em três ambientes distintos no norte do Pantanal, Brasil - Cambarazal, campo de Murundu e área ripícola. Nossa hipótese é que as atividades de navegação e forrageamento devem estar positivamente relacionadas à umidade e temperatura; e a composição das espécies deve diferir entre os habitats. Em 30 noites de amostragem, obtivemos 6049 registros e identificamos 16 espécies e três complexos de espécies distribuídas em cinco famílias. Não houve diferenças significativas na riqueza de espécies entre os três habitats, mas a área ripária, o ambiente mais heterogêneo amostrado, apresentou os maiores níveis de atividades de navegação e forrageamento. A composição das espécies variou com o habitat amostrado, demonstrando que diferentes espécies usam e percebem paisagens distintas de maneiras diferentes. Apenas a atividade de navegação dos morcegos de espaço-aberto respondeu à interação entre humidade relativa do ar e os diferentes tipos de habitat durante a estação seca do Pantanal. Nossos resultados, obtidos exclusivamente por amostragem acústica, mostram que muitas espécies de insetívoros aéreos e padrões de atividade são subamostrados em estudos feitos exclusivamente com redes de neblina.
Palavras-chave: Chiroptera, microclima, monitoramento acústico, morcegos insetívoros aéreos, Pantanal.
INTRODUCTION
Heterogeneous and complex environments tend to present higher levels of diversity (Tews et al. 2004). Indeed, environmental heterogeneity is a key factor determining species richness as higher availability of niche space, roosts, and feeding resources, as well as increased opportunities for isolation and divergent adaptation are thought to enhance species coexistence, persistence, and diversification (Stein et al. 2014). Particularly in tropical environments, the community structure of several animal groups tends to reflect aspects of spatial heterogeneity (Tews et al. 2004). For example, in Neotropical rainforests, bats of the Phyllostomidae vary in the way they use the vertical strata of the vegetation (Tews et al. 2004; Kalko et al. 2008; Ramos Pereira et al. 2010), with different wing shapes optimized for foraging in more open or more cluttered environments of the understory or canopy. Insectivorous bats are no exception to these patterns (Griffin 1971; O’donnell 2000; Adams et al. 2009; Adams 2012; Torquetti et al. 2017; Dias-Silva et al. 2018). Particularly in tropical environments, this guild is known to show a clear pattern of vertical stratification, with those species best adapted to cluttered spaces able to explore the understory, several others exploring the free spaces immediately below the canopy, and open-space foragers exploring mostly the space above the canopy (Marques et al. 2016). While using uncluttered environments, they may depend on some landscape elements provided by vegetation patches not only to forage, but also to roost or to use as commuting routes (Toffoli 2016). So, vegetation patches both with vertical and horizontal structure may be crucial for the maintenance of populations of aerial insectivores (Manning et al. 2006).
Climatic conditions are also related to the occurrence and activity patterns of most animal species because they are responsible for determining the temporal and spatial availability of different resources, particularly food, but often also shelters (Bozinovic et al. 2011; Liu et al. 2020). In environments with well-marked seasons, the level of activity of aerial insectivorous bats varies in response to fluctuations in weather conditions and their own energy requirements throughout the year (Hayes 1997; Barros et al. 2014; Reher et al. 2019). Temperature influences the activity patterns of aerial insectivorous bats directly or indirectly: directly by affecting bats’ physiological and metabolic mechanisms (McCain 2007) or indirectly through its influence on the density of insects available at a given space and time (Schowalter 2016). Relative humidity also affects bats, either in roost selection (Torquetti et al. 2017) or in foraging habitat selection (O’donnell 2000), in addition to influencing the propagation and attenuation of the sounds emitted by echolocating bat species (Snell-Rood 2012; Mutumi et al. 2016; Chaverri & Quirós 2017). The main consequence of atmospheric attenuation in high frequency sounds is the restriction of the effective range of the echolocation of bats, which can be limited to maximum operational ranges of a few meters (Griffin 1971; Lawrence & Simmons 1982).
The Pantanal is the largest alluvial plain on Earth, occupying 150000 km2, approximately 1.8% of the Brazilian territory (Mapbiomas 2020). The area shows high vegetational mosaicism resulting from the joint influence of the neighbouring phytogeographic domains of the Cerrado, Chaco, Amazon, and Atlantic Forest, as well as of the hydrological regime, with marked seasonal alternation between inundation and severe drought (Mittermeier et al. 2003;Nunes Da Cunha & Junk 2004). Such biogeographic scenario, spatial heterogeneity and seasonal environmental variations influence the regional set of bat species that occur in the biome (Fischer et al. 2018). Of the 181 species of bats known to occur in Brazil (Garbino et al. 2020), 65 occur in the Pantanal (Alho et al. 2011; Fischer et al. 2018; Fischer 2019), where bats represent the richest mammalian group, accounting for approximately 40% of the mammal fauna (Nunes Da Cunha & Junk 2004; Tomas et al. 2011). However, most inventories done in the region have used mostly mist-nets or harp traps (Oliveira 2016; Fischer et al. 2018), which ends up underestimating aerial insectivorous bats (Kunz & Parsons 2009; Puechmaille et al. 2012; Hintze et al. 2016; Ramos Pereira et al. 2018; Hintze et al. 2020). In fact, this pattern is not exclusive of the Pantanal— in Brazil and all of the Neotropic, while the majority of bat species are edge-space or open-space aerial insectivores, studies with acoustic monitoring are still scarce (but see Jung & Kalko 2011; Estrada-Villegas et al. 2012; Barataud 2013; Bader et al. 2015; Hintze et al. 2016; López-Baucells et al. 2016; Marques et al. 2016; Silva & Bernard 2017; Arias-Aguilar et al. 2018). Acoustic monitoring is a non-invasive technique that makes it possible to explore different aspects of bat ecology. As some of the acoustic parameters of bat calls are species-specific, their analysis allows the identification of the emitting species and, therefore, can support studies on bat occurrence and distribution, composition, diversity, habitat use, activity patterns and foraging behavior (Vaughan et al. 1997; O’Farrell & Miller 1999; Coleman & Barclay 2013; Ochoa et al. 2000; MacSwiney et al. 2008; Mendes et al. 2017; Militão 2017).
Here, using acoustic monitoring, we aim to investigate the effects of environmental variables—habitat type and microclimate—on the occurrence, activity and diversity of aerial insectivorous bats in the Pantanal biome, during the dry season. To our knowledge this is the first study based exclusively on acoustic sampling of aerial insectivorous bats in the Pantanal.
We hypothesize that bat species composition differs between habitats with different vegetation structure and predict that more heterogeneous habitats show greater diversity of aerial insectivorous bats. Additionally, we hypothesize that microclimate (temperature and relative humidity) affects bat activity: activity should be higher in wetter and warmer nights. As in dry environments or with pronounced seasonality, water availability strongly influences bat and insect activity (McCain 2007; Korine et al. 2016; Schowalter 2016), we expect relative humidity to have an even greater effect on bat activity than temperature.
MATERIALS AND METHODS
Study area
The study was done in the northern Pantanal (Fazenda Santa Lúcia; 16°53’38.25"S; 55°54’24.98"W), Barão de Melgaço, State of Mato Grosso (Fig. 1). Mato Grosso is the third largest state in Brazil, occupying an area of 903206997 km2 (IBGE 2021). Located in the Center-West region of the Brazilian territory, it borders Bolivia and is divided into three main domains: Amazon, Cerrado and Pantanal.
Temperature seasonality and especially precipitation seasonality are quite marked. The total annual precipitation varies between 1000 and 1600 mm, concentrated in the wet season (November to April), while in the dry season (May to October) it is less than 40 mm (Hofmann et al. 2010). During the inundation period (November to April), a large part of the Fazenda Santa Lúcia area gets flooded. The landscape presents the marked spatial heterogeneity, characteristic of the Pantanal plains. Sampling sites were classified into one of the three main physiognomies occurring in the region: i) Cambarazal (12 sampling sites): the most homogeneous sampled area is a forest area composed mainly of the tree species Vochysia divergens Pohl (popularly known as cambará). These trees can exceed 20 m in height and are commonly found in wetlands (Nascimento & Nunes Da Cunha 1989; Arieira & Nunes Da Cunha 2012); ii) Murundu field (14 sampling sites): floodplain formation of earthbound fields, covered with woody vegetation typical of the Cerrado biome known as murundus. During the flood season, they remain dry, serving as shelter for several animals in the area (Marimon et al. 2012); iii) Riparian area (four sampling sites): dense forest areas associated with the São Lourenço river and interspersed with areas of Scheelea phalerata palm locally known as “acurizal” and swamps; this is the most heterogeneous sampled area.
Acoustic monitoring
We conducted acoustic surveys in September and October of 2015, during the Pantanal’s dry season. We sampled 30 sites separated by a minimum distance of 2.5 km. For each site, acoustic monitoring was done for one night during 6 hours after dusk, between 18:00h and 00:00h. Sound recordings were made with two Pettersson D500X bat detectors (sampling rate 256 kHz, 16 bits) (Pettersson Electronic AG, Uppsala, Sweden) configured to record files of 15 seconds. Temperature and humidity sensors were set up next to the bat detector, taking measurements at 1-min intervals during the same period of bat sampling.
Acoustic analyses
Species identification was done through manual analysis of recordings in Raven Pro 1.5 Software (Bioacoustics Research Program 2015) selecting a Hamming window with a 1024 Fast Fourier Transformation (FFT; overlap 93%) to minimize the effect of spectral dispersion. We measured the following parameters: frequency modulation (FM), number of harmonics (nH), peak frequency (PF), duration (D), inter-pulse interval (IPI), frequency of maximum energy (FME), maximum frequency (Fmax), minimum frequency (Fmin) and bandwidth (BW = Fmax-Fmin). We identified bat recordings to the species level, following the acoustic identification key for Brazilian bats (Arias-Aguilar et al. 2018). Ambiguous identification of species or genera due to similarities in signal structure and frequency were organized in potential species complexes.
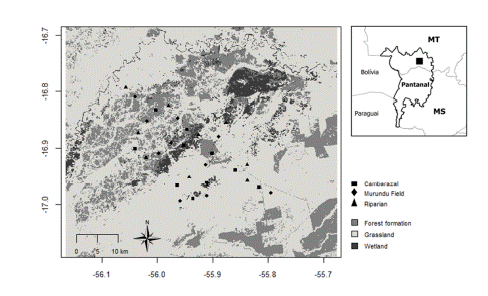
Fig. 1
Map of the study area, Fazenda Santa Lucia, MT, Brazil, in the Pantanal Biome, with the locations of the sampling sites for each habitat type.
We grouped the different species within two guilds according to their eco-morphological characteristics: open-space and edge-space foragers, following Denzinger & Schnitzler (2013). We quantified bat foraging activity by the number of bat passes, which is defined as a sequence of at least three bat echolocation calls during a sound recording and feeding activity by number of terminal phases, per 30-min for each one of the two guilds.
Statistical analyses
Activity patterns were related to the predictor variables: mean temperature (°C) and mean relative humidity (H%). To assess the influence of environmental variables on total foraging and feeding activity for each one of the guilds we used generalized linear models (GLM). As data evidenced overdispersion we opted for a Quasi-Poisson error distribution, considering all possible combinations of the predictor variables.
We selected the best-adjusted models using the second-order Akaike Information Criterion (AICc) and the determination coefficient (R2), according to the guidelines of Burnham & Anderson (2002). We considered as the best-supported models those with 6AICc<2. Although humidity and temperature were slightly correlated (r2 = -0.31), we chose to use the two variables as predictors in the models, as they are expected to have different effects on total foraging and feeding bat activity and for each one of the evaluated foraging guilds.
To compare bat community composition between habitats we performed a Multivariate Permutational Analysis of Variance (PERMANOVA) using distance matrices with 999 permutations (Anderson & Walsh 2013). We estimated species richness using the rarefaction and extrapolation method of Hill numbers (q = 0) for incidence data, with 95% confidence intervals and standard error obtained by the bootstrap method, following the procedures proposed by Chao et al. (2014). All statistical analyzes were done in R, version 3.5.1 (R Core Team 2019), using vegan (Oksanen et al. 2013), AICcmodavg (Mazerolle & Mazerolle 2019), permute (Simpson et al. 2019), lattice (Sarkar et al. 2020) and iNEXT (Hsieh et al. 2019) packages.
RESULTS
Species richness and composition
Overall, we recorded 6049 bat passes and 387 feeding buzzes. We identified 16 species and three complexes belonging to five families: Emballonuridae (Peropteryx macrotis, Rhynchonycteris naso, Saccopteryx bilineata); Molossidae (Molossops temminckii, Molossus currentium, Molossus molossus, Molossus rufus, Promops centralis, Promops nasutus, Tadarida brasiliensis, the complex Cynomops/Eumops/Nyctinomops andthe complex Eumops/Nyctinomops); Mormoopidae (Pteronotus gymnonotus); Noctilionidae (Noctilio albiventris and Noctilio leporinus) and Vespertilionidae (Eptesicus chiriquinus, Eptesicus furinalis, Myotis cf. simus, and the Myotis nigricans/Myotis riparius complex).
The taxa presenting highest activity were Noctilio albiventris (1455 bat passes), Eumops/Nyctinomops complex (828 bat passes), the Myotis nigricans/Myotis riparius complex (688 bat passes), Molossops temminckii and Eptesicus furinalis (536 bat passes each); these represented 66.83% of the total bat passes (Table 1).
We detected 14 species and three complexes in each area. Richness estimates were the following: i) Cambarazal – 19.062 ± 3.140 (standard error); ii) Murundu field – 19.786 ± 4.251; and Riparian area –20.375 ± 5.473. There were no significant differences between habitats in terms of species richness estimates (all comparisons between pairs p>0.05). Species composition significantly differed between habitats (Permanova: r2 = 0.11 p <0.01; Fig. 2). The main difference in the composition between the habitats was the occurrence of Promops centralis that was only detected in the Murundu field and Promops nasutus that was absent from the Riparian area. Rhynchonycteris naso occurred only in the Riparian area, and Saccopteryx bilineata was absent from the Murundu field.
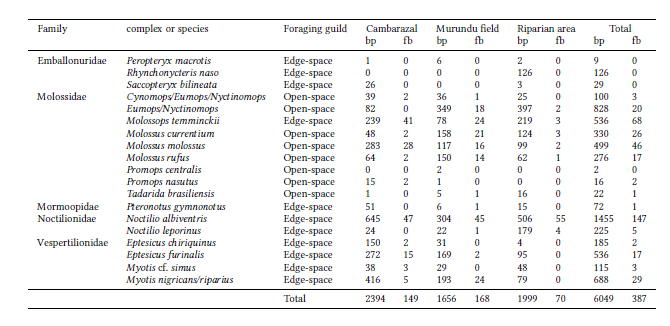
Fig. 2
Species composition grouped by guild in the three habitats surveyed: Cambarazal, Murundu field and Riparian area. The size of the bars represents the proportion of each species in the sampled habitat.
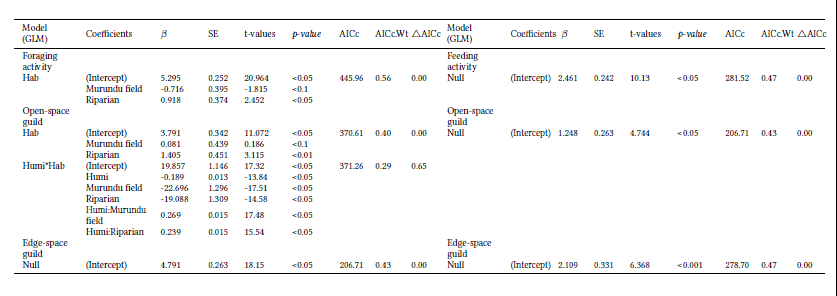
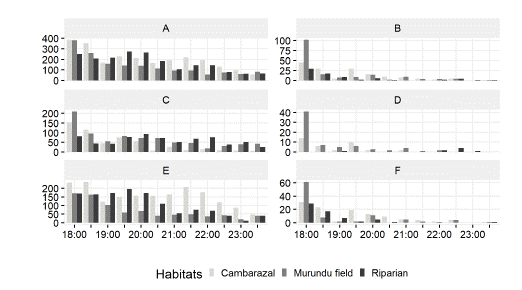
Fig. 3
Absolute number of bat passes in each habitat per 30-min interval 18h and 00h: A) Total bat foraging activity, B) Total bat feeding activity, C) Open-space bats foraging activity, D) Open-space bats feeding activity, E) Edge-space bats foraging and F) Edge-space bats feeding activity.
Effect of habitat and microclimate on bat activity
Activity of edge-space foragers represented 65.73% (3976) of the total bat passes and 70.28% (272) of the total feeding buzzes. Noctilio albiventris, the complex Myotis riparius/Myotis nigricans, Molossops temminckii and Eptesicus furinalis accounted for the greatest number of bat passes (1455; 688; 536 and 536, respectively) and feeding buzzes (147; 29; 69 and 17, respectively). These species accounted for 80.86% and 95.96% of the foraging and feeding activity of this guild. Bats were active throughout the sampling period in the three sampled habitats (Fig. 3).
The open-space guild accounted for 34.27% (2073) of the total number of bat passes and 29.72% (115) of the total number of feeding buzzes. The Eumops/Nyctinomops complex, Molossus molossus, and Molossus currentium presented the highest number of bat passes (828; 499 and 330, respectively) and feeding buzzes (20; 46 and 26, respectively). Altogether they represented 79.93% and 80.00% of the foraging and feeding activity of this guild.
A summary of the best-adjusted (D.AICc<2) models is presented in Table 2 (Tab. S1, Fig. S1). Total foraging activity varied with habitat. The whole of the insectivorous bat assemblage, open-space and edge-space bats foraged significantly more in the Riparian habitat, also, edge-space bats foraged significantly less in the Murundu field (Fig. 4).
The foraging activity of the open-space guild varied with habitat and with the interaction between humidity and habitat types, edge-space foraging activity did not respond to any of the habitat or microclimate variables. Feeding activity did not respond significantly to any of the habitat or microclimate variables, for the whole of aerial insectivores, open-space or edge-space foragers.
The Riparian area, the most heterogeneous of the areas sampled, presented the highest levels of activity, which was also concentrated in the first hours of the night, until 20:30 h. In this habitat the relative humidity varied between 70.0% and 95.1%. The frequency of foraging calls was higher in high relative humidity 91.8 to 97.3 H%. The peak period of foraging activity was similar between Cambarazal and Murundu field (Fig. 5), occurring in the early evening, between sunset and the following 2 hours (18:00h to 20:00h). The relative humidity in these habitats varied between 64.3% and 97.3%, with the peak of foraging activity occurring above 80%. Feeding activity was also similar in the two environments and occurred between 75.3 and 91.7% relative humidity. Feeding buzzes were mostly recorded in the lower range of humidity in this habitat 70 to 75.2%.
DISCUSSION
Species composition and richness
Our data showed that bat species composition varied between Cambarazal, Murundu field and Riparian area, supporting our initial hypothesis that there would be differences in the composition of the bat assemblages between habitats with different vegetation structure. However, our results did not support the hypothesis that more heterogeneous habitats would present greater richness of bats, as richness estimates were similar for the three surveyed habitats. The Pantanal floodplain presents contrasting conditions throughout the year, with floods alternating with several months of hydric stress, resulting in high seasonal productivity and strong environmental heterogeneity (Alho 2008). Thus, different areas of the Pantanal allow the presence of different bat assemblages during the dry and wet seasons. Indeed, the Pantanal bat fauna is composed by species that thrive in the floodplain, in addition to few abundant species occurring in the subregions close to their original geographical distributions out of the Pantanal. Therefore, the assemblage is filtered by the regional connections with the neighbouring phytogeographic domains, a key factor in determining the high richness per area of the Pantanal’s bat fauna (Fischer et al. 2018).
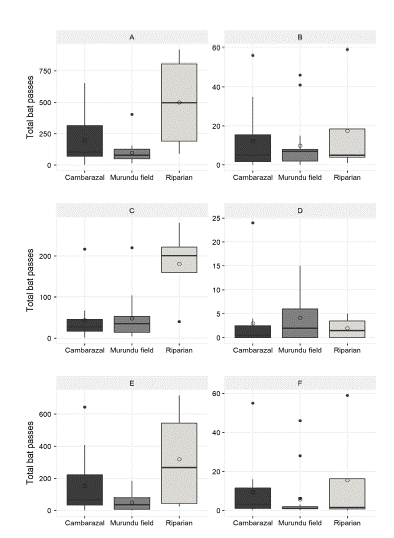
Fig. 4
A) Total foraging bat activity, B) total bat feeding activity, C) total open-space guild foraging activity, D) total open-space guild feeding activity, E) total edge-space guild foraging activity and F) total edge-space guild feeding activity in the three sampled habitats. The y-axis corresponds to the number of bat-passes in 30-min intervals (the box represents the first quartile, the second quartile - median, and the third quartile, whiskers indicate the minimum and maximum disregarding outliers, the open circle represents the mean and black dots represents the outliers).
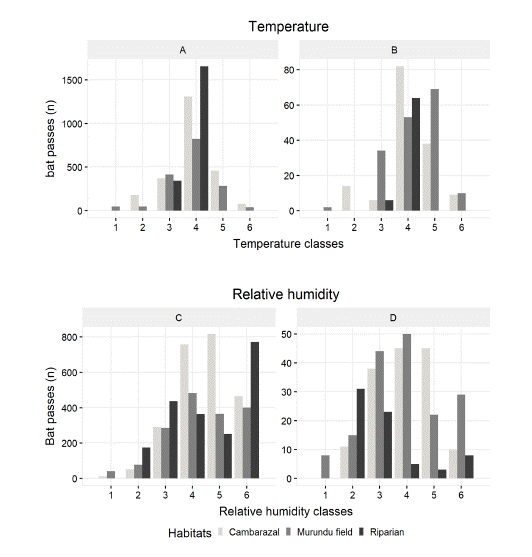
Fig. 5
Total bat activity pattern according to temperature and relative humidity. For temperature (°C), in A) total bat navigation activity and B) total bat feeding activity, the x-axis is divided into six classes (15.7-18.5, 18.6-21.5,21.6-24.4,24.5- 27.4,27.5-30.2,30.3-33.3). For relative humidity (%), in C) total bat foraging activity and D) total bat feeding activity the x-axis is divided into six classes (64.3-69.7,69.8-75.2,75.3-80.7,80.8-86.2,86.3-91.7,91.8-97.2).
The resources provided by the extensive, despite homogeneous, forest formation of the Cambarazal, with potentially greater availability of insects, availability of roosts and protection against winds (Russ et al. 2003), probably favour a diverse bat assemblage, as well as the other sampled areas, although no species has shown a preference for foraging or feeding in this habitat.
The Murundu field is characterized as a large open area with small islands of vegetation, creating connectivity between different environments. Due to their quantity and distribution in the Pantanal landscape, Murundu fields seem to act as important stepping-stones for bats between open areas and physiognomies of denser vegetation. In addition, some species that occur in the Pantanal are highly specialized in foraging and feeding in open spaces. This is the case of Promops centralis, registered exclusively in this habitat; Promops centralis is a fast-flying insectivorous species, which navigates and forages high above the canopy (Kalko et al. 2008), making its capture with mist-nets extremely difficult; on the other hand, the echolocation calls of P. centralis are easily distinguishable from any other species (Hintze et al. 2020).
The Riparian area presents great diversity of tree species and diverse vertical strata, making this habitat extremely heterogeneous. Still, while this habitat was not richer than Cambarazal or the Murundu field, it presented the highest bat activity. Some species, particularly those closely associated with water, were exclusively detected in this habitat; this is the case of Rhynchonycteris naso, which feeds mostly by capturing insects above the water surface (Plumpton & Jones 1992; Nogueira & Pol 1998), and of Noctilio leporinus, which feeds on fishes it captures on waterbeds (Altenbach 1989; Zortea & Aguiar 2001).
Foraging and feeding during the Pantanal’s dry season
Contrarily to our predictions, the general activity of aerial insectivorous bats was neither explained by temperature nor by humidity during the sampling period—the Pantanal’s dry season. However, the interaction between humidity and different habitat types was an important predictor of the activity of open-space foraging bats. The interaction was equally positive for all habitats, with a greater effect on the Murundu field. The activity peak of this guild concentrated above 75% relative humidity. The narrow variation in night air humidity across all habitats perhaps explains the lack of significance of air humidity in the remaining predictive models. In dry regions, low relative humidity leads to increased water loss through evaporation in bats, which must be partially compensated by an increase in the quantity of water drank (Rainho & Palmerim 2011). So, foraging and feeding when the air relative humidity levels are higher, during the dry season, may be strategic for bats that fly high and long-distances, especially in open habitats. Similarly, while it is known that the feeding activity of aerial insectivores tends to increase in warmer nights due to higher arthropod activity (Racey & Swift 1985; Aldridge & Rautenbach 1987; Schowalter 2016), all of our sampled nights were reasonably warm and within narrow temperature ranges, which probably explains the lack of significance of temperature in the models. Regarding the three different sampled habitats, foraging activity was higher in the Riparian area, which represents the most heterogeneous sampled habitat, and also where water is persistently available. In fact, the dry season in the Pantanal can be rather extreme (Alho 2008; Marengo et al. 2015), so habitats that are a reliable and consistent source of water may become key for bats and other animals (Alho & Gonçalves 2005; Mamede & Alho 2006; Monadjem & Reside 2008; Tomas et al. 2011). Previous studies in arid areas have shown the same pattern between bat foraging and feeding activity and proximity to bodies of water (Zukal & Rehák 2006; Barros et al. 2014; Hintze et al. 2016; Marques et al. 2016). In addition, riparian forests have already been highlighted as important linear elements of the landscape for bats, both for activity and in number of species (Russo & Jones 2003; Fukui et al. 2006; Barros et al. 2014). The water bodies tend to provide more food resources for bats, as they function as breeding grounds for various insects, in addition to being used as flying corridors by aerial insectivores (?Brooks 2009; Hagen & Sabo 2011; Marques et al. 2016).
Cambarazal and Murundu field presented similar levels of bat foraging activity, with the Murundu field showing significantly less foraging activity for edge-space foraging bats. Many bat species select habitats with the presence of trees to navigate and feed (Russ et al. 2003; Brooks 2009). Indeed, in Cambarazal, vegetation edges possibly provide various micro- climates, which may allow bats to search for sites with higher or lower ambient temperatures along the night, allowing for an optimization of the regulation of their body temperatures. The combination of these factors may be responsible for the high number of bat passes, especially those belonging to edge-space foragers.
Unlike the other two habitats, where the activity of edge-space foragers was higher, in Murundu field the activity of open-space and edge-space foragers was similar. This may result from the higher availability of open spaces between the vegetation in this habitat, allowing for its increased use by open-space foragers. The ability of bats to fly allows them to move large distances between distinct landscape patches (Bernard & Fenton 2003; Stevens et al. 2007). Natural open environments help maintain a high diversity of bats, as open-space foragers are adapted for faster and less manoeuvrable flight, while edge- space foragers are intermediate in terms of flight speed and manoeuvrability and often use vegetation patches on the edge of open areas (Schnitzler & Kalko 2001; Monadjem & Reside 2008).
While our sampling period was not enough to detect temporal changes in species-specific patterns of activity it is quite likely that during the flood season the activity of species that feed on resources associated to water increases also in the environments where they are less active or absent in the dry season.
The importance of acoustic monitoring
The previous study by Oliveira (2016) in the same study area, solely based on mist-net sampling, registered 27 species in five families (Emballonuridae, Phyllostomidae, Noctilionidae, Molossidae and Vespertilionidae) with 59.26% of the species belonging to the Phyllostomidae. Using solely acoustic monitoring we added nine new species and one family to the study area: Peropteryx macrotis and Saccopteryx bilineata (Emballonuridae), Molossus currentium, Molossus rufus, Promops centralis, Promops nasutus and Tadarida brasiliensis (Molossidae), Eptesicus chiriquinus, (Vespertilionidae), and Pteronotus gymnonotus (Mormoopidae). Besides, at least one additional species from the Cynomops/Eumops/Nyctinomops complex occurs in the area. The use of acoustic monitoring is definitely key to expand our knowledge on the distribution, occurrence and ecology of aerial insectivorous bats in this Neotropical biome.
CONCLUSIONS
Our sampling scheme based on acoustic monitoring revealed that several bat species end up undetected in studies exclusively based on mist-nets. While Cambarazal, Murundu field and Riparian area present similar bat richness, composition varied between these habitats, demonstrating that different species use and perceive distinct landscapes in different ways. The activity of open-space bats responded to the interaction between relative humidity and different habitat types, while the activity edge-space was not influenced by microclimatic conditions. Our results point out the need to preserve the spatial heterogeneity typical of the Pantanal biome as different habitats may support different local bat assemblages, while offering exclusive resources for different bats. Our work also presented some limitations: as a pilot study with acoustic monitoring in the area we were unable to increase the spatial and temporal replicates (particularly by sampling during the wet season), which would allow for a better understanding of the richness, composition, and activity patterns of insectivorous bats in the region of the northern Pantanal, especially considering the marked seasonal changes that this area undergoes throughout the year. Does species composition and activity change in response to the seasonal flooding regime? Are there evident species-specific patterns? What is the effect of the environmental degradation resulting from anthropogenic actions in the area, particularly now that the Pantanal has suffered intense and extensive fires of human origin resulting from the laxation on environmental ruling (Libonati et al. 2020)? These are important questions that we expect to answer in the future.
Acknowledgments
The authors would like to thank the support of the National Museum - UFRJ, particularly Professor Dr. Luiz Flamarion Barbosa de Oliveira and all those that helped during fieldwork. CFC and MJRP were supported by National Council for Scientific and Technological Development (CNPq) scientific initiation and productivity grants, respectively, and AAA was supported by a MSc scholarship provided by the Coordination for the Improvement of Higher Education Personnel (CAPES).
REFERENCES
Adams, M. D., S. L. Bradley, & O. F, Kris. 2009. Vegetation structure influences the vertical stratification of open-and edge-space aerial-foraging bats in harvested forests. Forest Ecology and Management 258(9):2090-2100. https://doi.org/10.1016/j.foreco.2009.08.002
Adams, M. D. 2012. Vertical stratification of insectivorous bats (Microchiroptera) in harvested forests: assessing the role of structural clutter in shaping patterns of flight activity. PhD Thesis. Australia: School of Biological Sciences, University of Wollongong.
Aldridge, H. D. J. N., & I. L. Rautenbach. 1987. Morphology, echolocation and resource partitioning in insectivorous bats. The Journal of Animal Ecology 56(3):763-778. https://doi.org/10.2307/4947
Alho, C. J. R., & H. C. Gonçalves. 2005. Biodiversidade do Pantanal. Ecologia e Conservação. Campo Grande-MS: Editora UNIDERP.
Alho, C. J. R. 2008. Biodiversity of the Pantanal: response to seasonal flooding regime and to environmental degradation. Brazilian Journal of Biology 68(4):957-966. https://doi.org/10.1590/S1519-69842008000500005
Alho, C. J. R., E. Fischer., L. F. Oliveira-Pissini., & C. F. Santos. 2011. Bat-species richness in the Pantanal floodplain and its surrounding uplands. Brazilian Journal of Biology 71(1):311-320. https://doi.org/10.1590/s1519-69842011000200010
Altenbach, J. S. 1989. Prey capture by the fishing bats Noctilio leporinus and Myotis vivesi. Journal of Mammalogy 36:390-399. https://doi.org/10.2307/1381532
Anderson, M. J., & D. C. Walsh. 2013. Permanova, Anosim, and the Mantel test in the face of heterogeneous dispersions: what null hypothesis are you testing? Ecological Monographs 83(4):557- 574. https://doi.org/10.1890/12-2010.1
Arias-Aguilar, A., F. Hintze, L. M. Aguiar, V. Rufray, E. Bernard, & M. J. Ramos Pereira. 2018. Who’s calling? Acoustic identification of Brazilian bats. Mammal Research 63(3):231-253. https://doi.org/10.1007/s13364-018-0367-z
Arieira, J., & C. Nunes Da Cunha. 2012. Estrutura populacional do cambará (Vochysia divergens Pohl, Vochysiaceae), espécie monodominante em floresta inundável no Pantanal Mato-Grossense. Oecologia Australis 16(4):819-831. https://doi.org/10.4257/oeco.2012.1604.07
Bader, E., K. Jung K, E. K. V. Kalko, R. A. Page, R. Rodriguez, & T. Sattler. 2015. Mobility explains the response of aerial insectivorous bats to anthropogenic habitat change in the Neotropics. Biological Conservation 186:97-106. https://doi.org/10.1016/j.biocon.2015.02.028
Barataud, M. et al. 2013. Identification et écologie acoustique des chiroptères de Guyane française. Le Rhinolophe 19(1):103-145.
Barros, M. A., D. Pessoa, & A. M Rui. 2014. Habitat use and seasonal activity of insectivorous bats (Mammalia: Chiroptera) in the grasslands of southern Brazil. Zoologia (Curitiba) 31(2):153-161. https://doi.org/10.1590/S1984-46702014000200006
Bernard, E., & M. B. Fenton. 2003. Bat mobility and roosts in a fragmented landscape in Central Amazonia, Brazil. Biotropica. 35:262–277. https://doi.org/10.1111/j.1744-7429.2003.tb00285.x
Bioacoustics Research Program. 2015. Raven Pro 1.5: Interactive Sound Analysis Software. Ithaca, NY: The Cornell Lab of Ornithology. Available from: http://ravensoundsoftware.com/
Bozinovic, F., P. Calosi, & J. L. Spicer. 2011. Physiological correlates of geographic range in animals. Annual Review of Ecology, Evolution, and Systematics 42:155-179. https://doi.org/10.1146/annurev-ecolsys-102710-145055
Brooks, R. T. 2009. Habitat-associated and temporal patterns of bat activity in a diverse forest landscape of southern New England, USA. Biodiversity and Conservation 18(3):529-545. https://doi.org/10.1007/s10531-008-9518-x
Burnham, K. P., & D. R. Anderson. 2002. Model selection and multimodel inference: a practical information-theoretic approach, 2nd ed. New York: Springer, 2.
Chaverri, G., & O. E. Quirós. 2017. Variation in echolocation call frequencies in two species of free-tailed bats according to temperature and humidity. The Journal of the Acoustical Society of America. 142(1):146-150. https://doi.org/10.1121/1.4992029
Chao, A. et al. 2014. Rarefaction and extrapolation with Hill numbers: a framework for sampling and estimation in species diversity studies. Ecological Monographs 84:45-67. https://doi.org/10.1890/13-0133.1
Coleman, J. L., & R. M. R., Barclay. 2013. Prey availability and foraging activity of grassland bats in relation to urbanization. Journal of Mammalogy 94(5):1111-1122. https://doi.org/10.1644/12-MAMM-A-217.1
Dai Fukui, M. M., S. Nakano, & T. Aoi. 2006. Effect of emergent aquatic insects on bat foraging in a riparian forest. Journal of Animal Ecology 75(6):1252-1258. https://doi.org/10.1111/j.1365-2656.2006.01146.x
Denzinger, A., & H-U. Schnitzler. 2013. Bat guilds, a concept to classify the highly diverse foraging and echolocation behaviors of microchiropteran bats. Frontiers in Physiology 4:164. https://doi.org/10.3389/fphys.2013.00164
Dias-Silva, L., G. T. Duarte, R. Alves, M. J. Ramos Pereira, & A. Paglia. 2018. Feeding and social activity of insectivorous bats in a complex landscape: The importance of gallery forests and karst areas. Mammalian Biology 88:52-63. https://doi.org/10.1016/j.mambio.2017.11.005
Estrada-Villegas, S., J. M. Brian, & E. K. V. Kalko. 2012. Climate, habitat, and species interactions at different scales determine the structure of a Neotropical bat community. Ecology 93(5):1183- 1193. https://doi.org/10.1890/11-0275.1
Fischer, E. et al. 2018. Bats in the dry and wet Pantanal. Hystrix, the Italian Journal of Mammalogy 29(1):11-17. https://doi.org/10.4404/hystrix-00019-2017
Fischer, E. 2019. Secretaria Regional do Pantanal [Internet]. Sociedade Brasileira para o Estudo de Quirópteros (SBEQ). Versão 2019-2. [accessed 2020 Apr 26]. Available from: http://www.sbeq.net/pantanal
Fukui, D., M. Murakami, S. Nakano, & T. Aoi. 2006. Effect of emergent aquatic insects on bat foraging in a riparian forest. Journal of Animal Ecology 75:1252-1258. https://doi.org/10.1111/j.1365-2656.2006.01146.x.
Garbino, G. S. T. et al. 2020. Updated checklist of Brazilian bats. [Internet] Comitê da Lista de Morcegos do Brasil—CLMB. Sociedade Brasileira para o Estudo de Quirópteros (SBEQ). Versão 2020. [accessed 2020 Sep 26]. Available from: https://www.sbeq.net/lista-de-especies
Griffin, D. R. 1971. The importance of atmospheric attenuation for the echolocation of bats (Chiroptera). Animal Behaviour 19(1):55-61. https://doi.org/10.1016/s0003-3472(71)80134-3
Hagen, E. M., & J. L. Sabo. 2011. A landscape perspective on bat foraging ecology along rivers: does channel confinement and insect availability influence the response of bats to aquatic resources in riverine landscapes? Oecologia 166(3):751-760. https://doi.org/10.1007/s00442-011-1913-4
Hayes, J. P. 1997. Temporal variation in activity of bats and the design of echolocation-monitoring studies. Journal of Mammalogy 78(2):514-524. https://doi.org/10.2307/1382902
Hintze, F., E, Barbier, & E. Bernard. 2016. Emballonuridae Gervais, 1855 (Chiroptera) of reserve biologica de Saltinho (Atlantic forest), in Brazil, revealed by echolocation. Check List 12(4):19-25. https://doi.org/10.15560/12.4.1925
Hintze, F. et al. 2020. Molossid unlimited: extraordinary extension of range and unusual vocalization patterns of the bat, Promops centralis. Journal of Mammalogy 101(2):417-432. https://doi.org/10.1093/jmammal/gyz167
Hofmann, G. S., J. L. P. C. Hasenack, L. D. Oliveira, H. Hasenack, J. L. & P. Cordeiro. 2010. O clima na Reserva Particular de Patrimônio Natural SESC Pantanal. Rio de Janeiro: SESC, Departamento Nacional.
Hsieh, T. C., K. H. Ma, & A. Chao. 2019. iNEXT- package: Interpolation and extrapolation for species diversity
IBGE - Instituto Brasileiro de Geografia e Estatística. 2021. [Internet]. CONHEÇA CIDADES E ESTADOS DO BRASIL. [accessed 2021 Apr 20]. Available from: https://cidades.ibge.gov.br/brasil/mt/panorama.
Jung, K., & E. K. V. Kalko. 2011. Adaptability and vulnerability of high flying Neotropical aerial insectivorous bats to urbanization. Diversity and Distributions 17(2):262-274. https://doi.org/10.1111/j.1472-4642.2010.00738.x
Kalko, E. K. V., S. Estrada Villegas, M. Schmidt, M. Wegmann, & C. F. J. Meyer. 2008. Flying high - Assessing the use of the aerosphere by bats. Integrative and Comparative Biology 48(1):60-73. https://doi.org/10.1093/icb/icn030
Korine, C., R. Adams, D. Russo, M. Fisher-Phelps, & D. Jacobs. 2016. Bats and water: anthropogenic alterations threaten global bat populations. Bats in the Anthropocene: Conservation of Bats in a Changing World (C. C. Voigt & T. Kingston, eds.). Springer Open, Cham, Switzerland. https://doi.org/10.1007/978-3-319-25220-98
Kunz, T. H., & S. Parsons. 2009. Ecological and behavioral methods for the study of bats. 2nd ed. Johns Hopkins University Press, Baltimore, Maryland. 600:1-29. https://doi.org/10.1644/jmammal/92-2-475
Lawrence, B. D, & J. A. Simmons. 1982. Measurements of atmospheric attenuation at ultrasonic frequencies and the significance for echolocation by bats. The Journal of the Acoustical Society of America 71(3):585-90. https://doi.org/10.1121/1.387529
Libonati, R., C. C. Da Camara, L. F. Peres, L. A. S. De Carvalho, & L. C. Garcia. 2020. Rescue Brazil’s burning Pantanal wetlands. Nature 588:217-219. https://doi.org/10.1038/d41586-020-03464-1
Liu, H., Q. Ye, & J. J. Wiens. 2020. Climatic-niche evolution follows similar rules in plants and animals. Nature Ecology and Evolution 4(5):753-763. https://doi.org/10.1038/s41559-020-1158-x
López-Baucells, A., R. Rocha, P. E D. Bobrowiec, E. Bernard, J. Palmeirim, & C. Meyer. 2016. Field Guide to Amazonian Bats. Editora INPA, Manaus.
MacSwiney, G M. C., F. M. Clarke, & P. A. Racey. 2008. What you see is not what you get: the role of ultrasonic detectors in increasing inventory completeness in Neotropical bat assemblages. Journal of Applied Ecology 45(5):1364-1371. https://doi.org/10.1111/j.1365-2664.2008.01531.x
Mamede, S. B., & C. J. R. Alho. 2006. Response of wild mammals to seasonal shrinking-and-expansion of habitats due to flooding regime of the Pantanal, Brazil. Brazilian Journal of Biology 66(4):991-998. https://doi.org/10.1590/S1519-69842006000600006
Manning, A. D., J. Fischer, & D. B. Lindenmaye. 2006. Scattered trees are keystone structures–implications for conservation. Biological Conservation 132(3):311-321. https://doi.org/10.1016/j.biocon.2006.04.023
Mapbiomas. 2020. Collection of Brazilian Land Cover & Use Map Series [Internet]. Version 4.1 (1985-2018). [accessed 2020 Apr 26]. Available from: https://mapbiomas.org/
Marengo, J. A., G. S. Oliveira, & L. M. Alves. 2015. Climate Change Scenarios in the Pantanal. Dynamics of the Pantanal Wetland in South America (I. Bergier & M. Assine, eds.). The Handbook of Environmental Chemistry, vol 37. Springer, Cham. https://doi.org/10.1007/6982015357
Marimon, B. S. et al. 2012. Florística dos campos de murundus do Pantanal do Araguaia, Mato Grosso, Brasil. Acta Botanica Brasilica 26(1):181-196. https://doi.org/10.1590/S0102-33062012000100018
Marques, J. T., M. J. Ramos Pereira, & J. M. Palmeirim. 2016. Patterns in the use of rainforest vertical space by Neotropical aerial insectivorous bats: all the action is up in the canopy. Ecography 39(5):476-486. https://doi.org/10.1111/ecog.01453
Mazerolle, M. J., & M. M. J Mazerolle. 2019. Package ‘AICcmodavg’. R package, version 2.2-2.
McCain, C. M. 2007. Could temperature and water availability drive elevational species richness patterns? A global case study for bats. Global Ecology and Biogeography 16:1-13. https://doi.org/10.1111/j.1466-822x.2006.00263.x
Mendes, E. S., C. Fonseca, S. F. Marques, D. Maia, & M. J. Ramos Pereira. 2017. Bat richness and activity in heterogeneous landscapes: guild-specific and scale-dependent? Landscape Ecology 32(2):295-311. https://doi.org/10.1007/s10980-016-0444-0
Militão, E. S. G. 2017. Padrão de atividade temporal de morcegos insetívoros no Cerrado. Masters Dissertation. Brasília (DF): Universidade de Brasília. https://doi.org/10.14393/19834071.2016.31993
Mittermeier, R. A., et al. 2003. Wilderness: Earth’s last wild places. Conservation International, Washington.
Monadjem, A., & A. Reside. 2008. The influence of riparian vegetation on the distribution and abundance of bats in an African savanna. Acta Chiropterologica 10:339-348. https://doi.org/10.3161/150811008X414917
Mutumi, G. L., D. S. Jacob, & H. Winker. 2016. Sensory drive mediated by climatic gradients partially explains divergence in acoustic signals in two horseshoe bat species, Rhinolophus swinnyi and Rhinolophus simulator. PLoS ONE 11(1):e0148053. https://doi.org/10.1371/journal.pone.0148053
Nascimento, M. T., & C. Nunes Da Cunha. 1989. Estrutura e composição florística de um cambarazal no Pantanal de Poconé-MT. Acta Botanica Brasilica 3(1):3-23. https://doi.org/10.1590/S0102-33061989000100001
Nogueira, M. R., & A. Pol. 1998. Observações sobre os hábitos de Rhynchonycteris naso (Wied-Neuwied, 1820) e Noctilio albiventris (Desmarest, 1818) (Mammalia, Chiroptera). Revista Brasileira de Biologia, São Carlos 58(3):473-480.
Nunes Da Cunha, C., & W. J. Junk. 2004. Year-to-year changes in water level drive the invasion of Vochysia divergens in Pantanal grasslands. Applied Vegetation Science 7:103-110. https://doi.org/10.1111/j.1654-109X.2004.tb00600.x
Nunes Da Cunha, C., & W. J. Junk. 2011. A preliminary classification of habitats of the Pantanal of Mato Grosso and Mato Grosso do Sul, and its relation to national and international wetland classification systems. The Pantanal: Ecology, biodiversity and sustainable management of a large neotropical seasonal wetland (W. J. Junk, C. J. da Silva, C. Nunes da Cunha & K. M. Wantzen, eds.). Sofia: Pensoft.
Ochoa, J. G., M. J. O’Farrell, & B. W. Miller. 2000. Contribution of acoustic methods to the study of insectivorous bat diversity in protected areas from northern Venezuela. Acta Chiropterologica 2(2):171-183.
O’donnell, C. F. 2000. Influence of season, habitat, temperature, and invertebrate availability on nocturnal activity of the New Zealand long-tailed bat (Chalinolobus tuberculatus). New Zealand Journal of Zoology 27:207-221. https://doi.org/10.1080/03014223.2000.9518228
O’Farrell, M. J., & B. W. Miller. 1999. Use of Vocal Signatures for the Inventory of Free-flying Neotropical Bats 1. Biotropica 31(3):507-516. https://doi.org/10.1111/j.1744-7429.1999.tb00394.x
Oksanen, J. et al. 2013. Package ‘vegan’. Community Ecology Package 2(9):1-295.
Oliveira, M. B. D. 2016. Implicações do mosaico da paisagem na estrutura e composição de espécies de quirópteros no norte do Pantanal. Masters Dissertation. Rio de Janeiro (RJ): Universidade Federal Rural do Rio de Janeiro
Puechmaille, S. J., W. M. Hizem, B. Allegrini, & A. Abiadh. 2012. Bat fauna of Tunisia: review of records and new records, morphometrics and echolocation data. Vespertilio 16:211-239.
Plumpton, D. L., & J. K. Jones. 1992. Rhynchonycteris naso. Mammalian Species 413:1-5. https://doi.org/10.2307/3504230
R Core Team. 2019. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. Available from: https://www.R-project.org/
Racey, P. A., & S. M. Swift. 1985. Feeding ecology of Pipistrellus pipistrellus (Chiroptera: Vespertilionidae) during pregnancy and lactation. I. Foraging behaviour. The Journal of Animal Ecology 54:205-215. https://doi.org/10.2307/4631
Rainho, A., & J. M. Palmerim. 2011. The Importance of Distance to Resources in the Spatial Modelling of Bat Foraging Habitat. PLoS ONE 6(4):e19227. https://doi.org/10.1371/journal.pone.0019227
Ramos Pereira, M. J., J. T. Marques, J. M. Palmerim. 2010. Vertical stratification of bat assemblages in flooded and unflooded Amazonian forests. Current Zoology 56(4):469-478. https://doi.org/10.1093/czoolo/56.4.469
Ramos Pereira, M. J., C. Fonseca, & L. M. Aguiar. 2018. Loss of multiple dimensions of bat diversity under land-use intensification in the Brazilian Cerrado. Hystrix, the Italian Journal of Mammalogy 29(1):25-32. https://doi.org/10.4404/hystrix-00020-2017
Reher, S., H. Rabarison, M. Schoroth, & K. Dausmann. 2019. Seasonal movements of insectivorous bat species in southwestern Madagascar. Malagasy Nature (13):117–124.
Russ, J. M., M. Briffa, & W. I. Montgomery. 2003. Seasonal patterns in activity and habitat use by bats (Pipistrellus spp. and Nyctalus leisleri) in Northern Ireland, determined using a driven transect. Journal of Zoology 259(3):289-299. https://doi.org/10.1017/S0952836902003254
Russo, D., & G. Jones. 2003. Use of foraging habitats by bats in a Mediterranean area determined by acoustic surveys: Conservation implications. Ecography 26:197-209. https://doi.org/10.1034/j.1600-0587.2003.03422.x
Sarkar, D., M. D. Sarkar, & S. Kernsmooth. 2020. Package ‘lattice’. R package.
Schnitzler, H. U., & E. K. Kalko. 2001. Echolocation by insect-eating bats: we define four distinct functional groups of bats and find differences in signal structure that correlate with the typical echolocation tasks faced by each group. Bioscience 51(7):557-569.
Schowalter, T. 2016. Insect ecology – An ecosystem approach (4th ed.). Cambridge, MA: Academic Press.
Silva, C. R., & E. Bernard. 2017. Bioacoustics as an important complementary tool in bat inventories in the Caatinga drylands of Brazil. Acta Chiropterologica 19(2):409-418. https://doi.org/10.3161/15081109ACC2017.19.2.017
Simpson, G. L., R. C. Team, D. M. Bates, J. Oksanen, & M. G. L. Simpson. 2019. Package ‘permute’. R package.
Snell-Rood, E. C. 2012. The effect of climate on acoustic signals: does atmospheric sound absorption matter for bird song and bat echolocation? The Journal of the Acoustical Society of America 131(2):1650-1658. https://doi.org/10.1121/1.3672695
Stein, A., K. Gerstner, & H. Kreft. 2014. Environmental heterogeneity as a universal driver of species richness across taxa, biomes and spatial scales. Ecology Letters 17:866-880. https://doi.org/10.1111/ele.12277
Stevens, R. D., C. López-González & S. J. Presley. 2007. Geographical ecology of Paraguayan bats: spatial integration and metacommunity structure of interacting assemblages. Journal of Animal Ecology 76(6):1086-1093. https://doi.org/10.1111/j.1365-2656.2007.01288.x
Tews, J. et al. 2004. Animal species diversity driven by habitat heterogeneity/diversity: The importance of keystone structures. Journal of Biogeography 31(1):79-92. https://doi.org/10.1046/j.0305-0270.2003.00994.x
Toffoli, R. 2016. The importance of linear landscape elements for bats in a farmland area: The influence of height on activity. Journal of Landscape Ecology 9(1):49-62. https://doi.org/10.1515/jlecol-2016-0004
Tomas, W. M., N. C. Cáceres, A. P. Nunes, E. Fischer, G. Mourão, & Z. Campos. 2011. Mammals in the Pantanal wetland, Brazil. The Pantanal: ecology, biodiversity and sustainable management of a large Neotropical seasonal wetland (W. J. Junk, C. J. Silva, C. N. Cunha & K. M. Wantzen, eds.). Pensoft Publishers, Moscow.
Torquetti, C. G., M. X. Silva, & S. A. Talamoni. 2017. Differences between caves with and without bats in a Brazilian karst habitat. Zoologia (Curitiba) 34:1-7. https://doi.org/10.3897/zoologia.34.e13732
Vaughan, N., G. Jones, & S. Harris. 1997. Habitat use by bats (Chiroptera) assessed by means of a broad-band acoustic method. Journal of Applied Ecology 34:716-730. https://doi.org/10.2307/2404918
Zortea, M., & L. Aguiar. 2001. Foraging behavior of the fishing bat, Noctilio leporinus (Noctilionidae). Chiroptera Neotropical 7(1-2):140-142.
Zukal, J., & Z. Rehák. 2006. Flight activity and habitat preference of bats in a karstic area, as revealed by bat detectors. Folia Zoologica-Praha 55(3):273-281.
Información adicional
Editor asociado: Dra. Andrea Previtali
Cite as: da Costa, C. F., A. Arias-Aguilar, & M. J. Ramos Pereira. 2021. Aerial insectivorous bats in the brazilian pantanal: diversity and activity patterns in response to habitat and microclimate. Mastozoología Neotropical, 28(2):e0604. https://doi.org/10.31687/saremMN.21.28.2.0.09.e0604