ARTÍCULO
Evaluation of mycelium composite materials produced by five Patagonian fungal species
Evaluation of mycelium composite materials produced by five Patagonian fungal species
Maderas. Ciencia y tecnología, vol. 24, 35, 2022
Universidad del Bío-Bío
Received: 17 April 2021
Accepted: 09 May 2022
Abstract: Mycelium composites consist of particulate lignocellulosic materials, e.g., sawdust from the timber industry structured as a solid matrix resulting from the mycelial growth. Many protocols have been proposed based on different strains and substrates. However, the influence of intrinsic elements, such as the structure of the hyphal system on the main parameters required by the industry still needs to be researched. The main goal of this work is to assess the performance of five Patagonian lignocellulolytic fungal species for producing mycelium composites. Strains of these species were studied in order to assess the relation between basidiome hyphal structure and the hyphal structure of mycelium-based materials. Comparisons of the hardness in the Janka scale were performed with commercial expanded polystyrene. Composites resulting from the growth of Pleurotus ostreatus, Nothophellinus andinopatagonicus and Funalia trogii successfully formed composites, showing a lower quality than Ganoderma austral. Ryvardenia cretacea in turn completely failed to colonize the substrate. The material resulting from the growth of Ganoderma australe on pine sawdust (a substrate chosen based on its local availability) is proposed as a good substitute with improved resistance.
Keywords: Fungal agglomerates, Ganoderma australe, hyphal system, patagonia strains, sawdust substrate.
Introduction
The accumulation of plastic waste as a result of human activities has redirected the attention of scientists towards the substitution of non-biodegradable materials with environmentally friendly options. Lignocellulosic wastes produced by the timber industry are a good source of raw materials for many bioprocesses. Bio-agglomerates are solid matrices resulting from the growth of fungal mycelium on lignocellulosic waste matter (Koutinas et al. 2004). In Patagonia, large amounts of lignocellulosic residues from the timber industry do not receive any additional treatment and are burnt or treated as domestic waste and, besides the environmental issues, they also cause operational difficulties due to the lack of efficient disposal options (Loguercio et al. 2008). A very interesting feature of mycelium-based materials is the possibility of revalorization and recycling of biological waste. Fungal agglomerates or mycelium composite materials are fully biobased and biodegradable, and they can be discarded at the end of their life cycles with little to no cost or environmental damage. Moreover, these composites can be composted in different ways (Meng et al. 2017).
The use of biomass derived from agro-industrial activities, including forestry and paper manufacture, as well as from the food industry, is especially relevant to the global economy (Sanchez 2009). Mycelium composite production is an interesting procedure for the revalorization of industrial waste and replace non-biodegradable materials like plastic or expanded polystyrene (Holt et al. 2012, Jones et al. 2017, Girometta et al. 2018).
The applications and environmental advantages of mycelium composites have attracted increasing interest (Jones et al. 2020). Mycelium-based materials (Bayer et al. 2008) have been tested for their technical properties (Holt et al. 2012), such as performance as alternative packaging materials, thermal and acoustic insulation, and, particularly, design materials, like furniture and decorative objects (Pelletier et al. 2013, Haneef et al. 2017, Camere and Karana 2018). Mechanical parameters of the mycelium composites still need more research (Bruscato et al. 2019). However, few studies address how mycelial microstructure determines properties of the final composite (Lelivelt 2015, Haneef et al. 2017, Jones et al. 2017).
Within the substrate, the fungal mycelium creates a three-dimensional matrix that binds the wood matter into a solid low-weight material, comparable to expanded polystyrene (Lelivelt 2015). The composition of the hyphal system of each fungal species varies depending on the binding capability of its hyphae: from straight to single-branched hyphae (generative hyphae), through thick-walled to solid straight elements (skeletal hyphae) with a low number of branches (arboriform or skeleto-binding hyphae) to profusely branching elements with tortuous and contorted branches (binding hyphae). A monomitic hyphal system only presents generative hyphae, a dimitic hyphal system presents generative hyphae and skeletal or skeleto-binding hyphae; whereas a trimitic hyphal system presents generative, skeletal and binding hyphae. Those different configurations of the hyphal systems are reflected in physical characteristics, e.g., hardness, consistency, or flexibility of basidiomes.
Members of Basidiomycota seem to be the most useful organisms to produce optimal mycelial matrices (Lelivelt 2015). To date, 36 fungal species have been used or are mentioned in patents for mycelial-material applications (Elsacker et al. 2020). Their mycelia work as a network of biopolymers whose mechanical properties depend on the characteristics of the individual behavior of hyphae, elastic properties, orientation, and connectivity within the network (Islam et al. 2017). Inherent biological characteristics also influence mycelial density; an especially good example being the mono-, di- and trimitic hyphal networks of the Basidiomycota (Jones et al. 2017). The three main hyphal types are generative, binding (also known as ligative), and skeletal. The number of different hyphal types present in a species is described as the “mitic” system. Wood-degrading fungi have the ability to degrade cell wall components using extracellular enzymes (Martínez et al. 2005). Since fungi are absorbotrophic organisms, lignin, cellulose, and hemicellulose degradation occurs extracellularly through two types of enzyme systems: a hydrolytic system, which involves hydrolases for the degradation of polysaccharides (cellulose, hemicelluloses and pectin); and a unique degradative-oxidative and extracellular system, which degrades lignin and oxidizes the phenolic units (Sanchez 2009). Depending on their ability to decay lignin or cellulose, wood fungi are characterized respectively as white or brown rot (Schmidt 2006). These particularly active degradative systems confer these organisms the unique capability of growing and obtaining energy from the complex chemical network that constitutes wood.
The main goal of this work is to evaluate the potential of five fungal species from the Patagonian forests with different hyphal systems to produce a mycelial composite using wood industry waste.
Materials and methods
We selected strains of five species from the Patagonian Andean forests with different hyphal structures, i.e. Pleurotus ostreatus (Jacq.) P. Kumm. (monomitic, white rot, strain CIEFAPcc 619), Funalia trogii (Berk.) Bondartsev & Singer, (trimitic, white rot, CIEFAPcc 606) Nothophellinus andinopatagonicus (J.E. Wright & J.R. Deschamps) (di- to trimitic, white rot, CIEFAPcc 627), Rajchenb. & Pildain, Ganoderma australe (Fr.) Pat. (dimitic, white rot, strain CIEFAPcc 657) and Ryvardenia cretacea (Lloyd) Rajchenb. (monomitic, brown rot, Strain CIEFAPcc 182). All strains were incubated in malt extract agar 2% (MEA) medium in Petri dishes in the dark at 24 ºC. After the inoculation in the center of the plate, radial growth was measured every 3 days until the mycelium completely covered the plate. The growth rate was determined using triplicate cultures, adjusting to a linear model and was expressed in cm per day (Zervakis et al. 2001). Hyphal structures were described following Nobles (1965) and Pegler (1996), and compared with reference literature (Rajchenberg 2006).
Mycelium composite fabrication conditions
We developed mycelium composite test discs (five replicates for each strain) of 10 cm day x 1,5 cm in height growing the five species on poplar sawdust using glass Petri dishes of 9 cm diam x 1 cm height as molds. Poplar sawdust with 80 % water content (w/w) was autoclaved 1 h at 121 ºC. After cooling, the sawdust was inoculated with 30% (wet weight) spawn of each species consisting of mycelium-colonized oat grains, which were previously boiled in water for 1 h and autoclaved for 20 min at 121 °C. The inoculated sawdust was placed into the molds, and incubated in the dark at 24 ºC for 21 days or until all the substrate was covered by mycelium. All samples were dried at 80 °C until reaching constant weight.
Morphological analyses of test discs
The final test discs were visually inspected. To observe the hyphal structure, 1g of dry mycelium composite was extracted and disrupted using hot concentrated NaOH as suggested by Decock et al. (2013) with the modifications proposed by Gomez-Montoya et al. (2017) with Congo Red for light microscope observation.
Mechanical properties of test discs
Hardness tests
Hardness was measured following the Janka method according to Murace et al. (2010) measuring the maximum load applied to embed an 11,28 mm steel ball halfway through its diameter into the material, at a speed of 6 mm/min. Five sample replicates were tested, measuring the load required for linear penetration. Measure triplicates were performed on each sample. As a reference, hardness was measured also on expanded polystyrene in order to evaluate the performance of the novel materials in replacing this plastic. A preliminary assay using commercial wood substitutes, such as particle board, indicated that the resistances of these materials are out of range and cannot be replaced by our mycelium agglomerates.
Material improvement treatment
The strain producing the best performing composite was selected to optimize composite features by modifying fabricating conditions. We tested the addition of different amounts of CaCO3 (0, 2,5 or 5,0 %) as a supplement to stabilize pH in the composite mix (Stamets and Chilton 1983). Other parameters and culture processing were performed as in the previously explained experiments. The measured parameters in the final test discs were density, dry weight loss, hardness, water absorbance, and flammability. Density was calculated from the weight after drying and the volume of each specimen. Dry weight loss was calculated as the difference between dry weight of the substrate and dry weight of the composite.
Water absorbance
Water absorbance was measured as the weight gain of the test disks after placing them in containers filled with distilled water. Within 2 and 24 hours samples were removed from the water and weighed. In order to test flammability, dry samples were exposed to a flame to estimate the required time of fire exposure for ignition.
Enzymatic assay
To extract extracellular enzymes, 2 g of solid samples were taken from each mold and stirred in 10 ml distilled water at 20 °C for 20 minutes, then centrifuged for 15 minutes at 5000 rpm and filtered.
Endoglucanases enzymatic activity was determined with a reaction mixture containing 0,4 ml of carboxymethylcellulose (0,5 % in 100 mM sodium acetate buffer, pH 4,8) and 50 μl of supernatant. Released reducing sugars were determined by the Somogyi-Nelson method (Nelson 1944, Somogyi 1952). The absorbance reading was performed at 540 nm (Wood and Bhat 1988).
Laccase enzymatic activity was determined using 0,5 mM ABTS, (2,2'-azinobis (3-ethylbenzthiazoline-6-sulfonate)) in 0,1 M Na acetate buffer, pH 3,6 as substrate. The reaction started by adding 20 µl of enzyme in 2,5 ml of substrate incubated at 30 °C. The reading was made at 420 nm (ε420 = 36/mM cm), taking the initial and a final absorbance at 10 seconds (Kuhar et al. 2015).
Manganese peroxidase (MnP) was determined with phenol red as substrate at a concentration of 0,01 % in 0,1 M sodium succinate buffer, pH 4,5 and 0,2 mM H2O2. The reaction was stopped by adding 40 μl of NaOH 5 N. The reaction product was read at 610 nm (ε610= 22 mM-1 cm-1) (Glenn and Gold 1985).
Statistical analysis
All experiments were performed in five replicates. Mean values and standard error of the means were determined. Data from material optimization and enzymatic analysis were subjected to one way ANOVA. Differences detected by ANOVA were analyzed using Tukey's test. Data were examined for normality (modified Shapiro-Wilks test, α = 0,05) and for homoscedasticity (Levene's test, α=0,05). Non-parametric analyses were performed when certain data sets did not comply with the homoscedasticity assumption, using the non-parametric test of Kruskal Wallis (α = 0,05). All analyses were performed using the Infostat software (Di Rienzo et al. 2016). Figures were created using the GraphPad Prism 7.0 (GraphPad Software Inc., La Jolla, CA).
Results and Discussion
Strain characterization
The strains of G. australe, P. ostreatus and F. trogii presented similar average growth rates on MEA, with values of 0,47 cm/day, 0,44 cm/day, and 0,45 cm/day respectively (Figure 1). Ryvardenia cretacea showed a lower growth rate (0,24 cm/day), whereas N. andinopatagonicus showed the lowest rate (0,07 cm/day) (Figure 1). The five selected strains presented different hyphal structures, all of which were present in MEA culture. Data corresponding to basidiomes were obtained from literature (Table1).
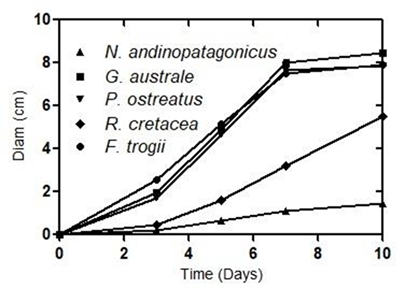
Figure 1:
Average growth curves of all strains on MEA in Petri dishes. Standard error smaller than 5% in all cases.
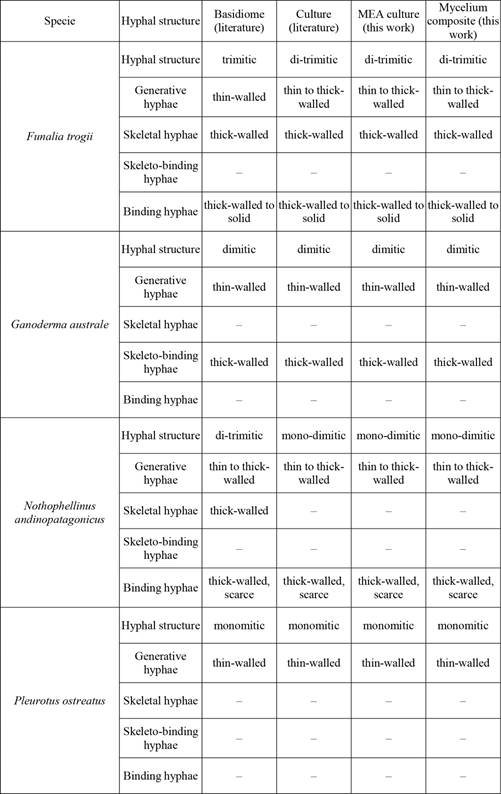
Bibliographic data were summarized from Asef (2012), Ibañez (1995), Menolli-Junior et al. (2010), Urcelay and Robledo (2009), Petre and Tănase (2013), Rajchenberg and Greslebin (1995), Rajchenberg (2006), Rajchenberg et al. (2015) and Wright and Deschamps (1972).
Morphological characteristics of the mycelium composite test discs
The strains colonized the substrates within 20 to 35 days, with the exception of Ryvardenia cretacea, which was unable to colonize the solid substrate even after 50 days of incubation. Pleurotus ostreatus test discs contained only abundant clamped generative hyphae with 2-5 μm diam. regular branches (Figure 2a, Figure 2b). Nothophellinus andinopatagonicus discs contained straight to branched generative hyphae with simple septa and thin to slightly thickened walls and a wide (Figure 2c and Figure 2d) hyphae (2-5 μm dia.). The same variation was observed in our agar cultures. Ganoderma australe (Figure 2e and Figure 2f) showed abundant generative hyphae (2-5 µm dia.), with clamp connections and thin walls. Long unbranched skeletal hyphae with thickened walls and no septa were also abundant. Branching binding hyphae, (2-4 µm dia) were also observed. Furthermore, curly helicoid branches were observed emerging from skeletal elements, which were not reported by Wright and Deschamps (1972). Funalia trogii discs contained clamped generative hyphae (Figure 2g and Figure 2h), abundant skeletal and binding hyphae of thickened walls and a profuse branching pattern. These features are summarized in Table 1.
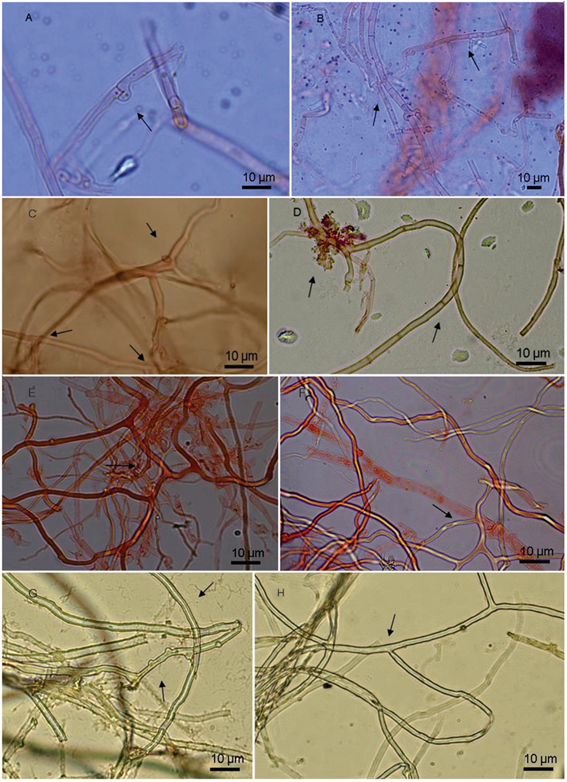
Figure 2:
Hyphal structure exhibited by the composite test discs.
A-B) Postreatus thin-walled hyaline clamped generative hyphae; C-D) N. andinopatagonicus, C) generative hyphae with short and digitiform lateral branches, D) thick-walled simple septate generative hypahe; E-F) G. australe E) thick-walled brownish skeleto-binding hyphae, F) wide hyaline thin-to-slightly-thick-walled clamped generative hyphae; G - H) F. trogii G) thick-walled hyaline skeletal hyphae, with thickened walls and multiple branching.
Mechanical qualitative properties of the mycelium composite test discs
Pleurotus ostreatus fully colonized the molds after 20 days. The test discs exhibited a good covering of the substrate with a moderate adhesion to the mold. Mycelial covering was white and cottony, moderately hard, and somewhat friable. Nothophellinus andinopatagonicus completely colonized after 35 days. Test discs were firstly covered by a white cottony mycelium, which later turned brown. They showed a notable hardness and a low friability to the touch and a rough surface. Full covering of the substrates took 20 days for Ganoderma australe, producing a notably hard composite, which was easy to extract from the molds. A complete covering of the substrate was observed as well as the formation of a dense, highly smooth mycelium in the upper part of the plate. The mycelium was first whitish and became brownish after complete colonization. It developed an elastic and firm layer, resulting in friability matrices. Funalia trogii reached full covering of the substrate after 20 days of incubation, and basidiome primordia started to form towards the edges of the plate. The first colonizing mycelium was white, without changes in color, and more dense near the inoculum. The test discs showed medium hardness and friability and they were rough on their surface.
Hardness test
Ganoderma australe showed the highest resistance to penetration. Enduring an average maximum load of 38,69 kg, the matrix conserved its structure after the measurement. The average maximum load values endured by N. andinopatagonicus, F. trogii and P. ostreatus were 23,13 kg, 7,46 kg and 4,27 kg respectively. In these three cases, material broke apart after the application of the load. A sample of expanded polystyrene was also subjected to the measurement, resisting an average load value of 5,27 kg, higher than P. ostreatus but considerably below the charges needed to damage the samples produced by G. australe. In addition, density of the bioagglomerates formed by each strain was evaluated, obtaining values higher than the expanded polystyrene reference. All values are represented in Table 2 and Figure 3. No direct relation between the maximum load and the density of the material was evidenced by our assays. Pleurotus ostreatus showed overall damage after maximum load and severe structural disorganization. Nothophellinus andinopatagonicus suffered several cracks and fissures, as did F. trogii. Ganoderma australe maintained its structure and only presented occasional cracks due to the effort.
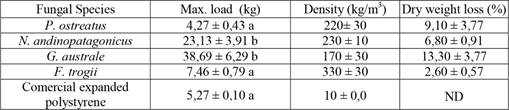
Data are mean values ± SE. Different letters refer to significant differences (p value < 0,05) among strains for each treatment.
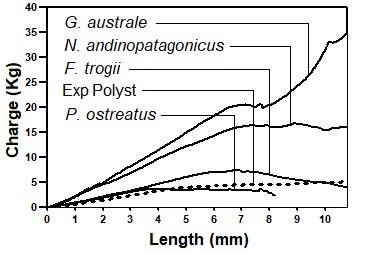
Figure 3:
Hardness test graph of each strain, using the Janka method. Pleurotus ostreatus bio-agglomerate broke apart before finishing the end of the test. Each line represents the average load developed during penetration.
Calcium addition
CaCO₃ did not improve hardness (Table 3). On the contrary, a diminished response to forcible penetration was evidenced during the test. Density of the material was notably increased by this addition, a parameter that is not desired for a packaging material. No significant (p value > 0,05) improvement in the avoidance of water absorption could be verified in these treatments. Density values were around 200 kg/m3, showing a minimum of 0,18 g/cm3 in the treatment without CaCO₃ and reaching the maximum of 240 kg/m3 in the treatment with 5 % CaCO₃. The highest value of water absorbance (water absorption w/w over dry weight) in 2 hours was 56,03 % in the 5 % CaCO₃ treatment and the minimum was 48,68 % in the treatment without CaCO₃. The same parameter measured after 24 hours increased up to 65 % in the treatment without CaCO₃ with the lower value of 62 % in the 2,5 % CaCO₃ treatment. The heaviest load value obtained was 22,72 kg in the treatment without CaCO₃ and the minimum value was 7,07 kg in the 5 % CaCO₃ treatment. Analysis of flammability showed that the bioagglomerate is flammable under direct fire. The flame exposure time needed for ignition was in all cases shorter than 15 seconds but also very variable and this is aligned with the results shown by Jones et al. (2017b). The time elapsed from ignition to extinction of the flame was approximately 4,5 min for all materials assayed. Cellulase and laccase activities showed no correlation (direct or inverse) with the formed materials (compare Table 3 with Figure 4). Although these measures can be used to test the ability of a strain to grow on a given substrate, our assays comparing the same strain under different calcium concentrations do not support their value as a predictor of the physical properties of the agglomerate. Although more data are needed to reach any conclusions, MnP activity values show inverse correlation with the hardness. This is probably an artifact due to the long known enhanced stability of this enzyme in the presence of Ca (Sutherland et al. 1997).
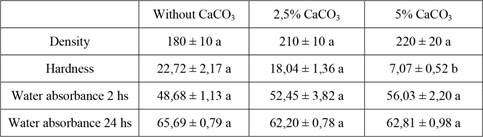
Data are mean values ± SE. Different letters refer to significant differences (p value < 0,05) among strains for each treatment.
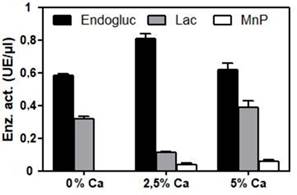
Figure 4:
Endoglucanase, manganese peroxidase and laccase enzyme activities in different Ca++ concentrations. Data are mean values ± SE.
Understanding the biological bases of the material resistance is necessary for the large-scale development of this novel technology. This work attempts to better understand how the mycelial traits influence the resulting material. Although no available studies directly relate hyphal structure with mechanical properties (like mitic system or hyphal growth performance), evidence suggests a strong relationship between these variables (Haneef et al. 2017, Lelivelt 2015, Girometta et al. 2018, Jones et al. 2018). Pleurotus ostreatus, Ganoderma australe, Funalia trogii, and Nothophellinus andinopatagonicus strains produce mycelium composites of compact structure, agglutinating all the substrate and improving the resistance, while Ryvardenia cretacea did not achieve complete colonization during the assays.
Ganoderma australe composites showed the highest material performance of all the mycelium composites. Its test discs present a dimitic hyphal system structure with profuse branched skeleto-binding hyphae forming an intricate and intertwined network. This structure is correlated with the configuration in agarized media and the basidiome (Wright and Deschamps 1972, Rajchenberg and Greslebin 1995, Levin 1998). Nothophellinus andinopatagonicus presented a similar pattern to G. austral. Funalia trogii, a species with a trimitic hyphal system, failed to form a continuous agglomerate and showed limited colonization in the center of the mold. This was probably due to the limitation of oxygen supply during the incubation period or the chemical characteristics of the substrate (Appels et al. 2019, Elsacker et al. 2019). Pleurotus ostreatus and Ryvardenia cretacea strains exhibit monomitic systems in their basidiomes (Lechner et al. 2002, Rajchenberg 2006) as well as in agarized culture (Rajchenberg 1996, Lechner et al. 2002). Ryvardenia cretacea was not able to grow in lignocellulosic substrate under the studied conditions. This seems to indicate that the basidiome consistency (hard and chalky in Ryvardenia and soft in Pleurotus) is not a good predictor of the quality of the resulting material. Accordingly, we suggest that not only is the hyphal system relevant to the constitution of the mycelium composite, but it is probably also necessary a metabolic component of each species and its extracellular matrix. Ryvardenia cretacea is a brown rot species, unlike the other strains assayed (Rajchenberg 2006). To our knowledge, no brown rot species are known to produce tenacious mycelia, but we did not find a rationale for this apparent correlation.
Ganoderma species present a dimitic hyphal system with skeleto-binding hyphae (Costa Rezende et al. 2017, Costa Rezende et al. 2020), and several species have been tested to produce mycelium-based materials with positive or negative results (Holt et al. 2012, Haneef et al. 2017, Vallas and Courard 2017, Xing et al. 2018).
In the last few years, some interesting works reporting mechanical properties on mycelium-based materials have been published. However, the lack of an appropriate framework unifying criteria on the parameters that need to be quantified and the methodologies is still an obstacle for accurate comparisons among different results. Elsacker et al. (2021) reports promising results regarding the flexural strength of materials based on Ganoderma resinaceum and Trametes versicolor. Data on uniaxial compression tests have been provided and discussed by Islam et al. (2018), showing also that the studied materials are resistant enough for industrial applications. However, standardized parameters are needed before accurate comparative data can be tabulated and used for strain and condition selection by the industry.
Conclusions
This work is the first to characterize the growth of Ganoderma australe. The intricate network of the thick-walled profusely branched skeleto-binding hyphae gives the composite a high resistance to cut; thus, generating a tenacious layer in agarized medium and a rigid structure in lignocellulosic substrate. The tension generated by the mycelial network is such that it does not only agglomerate the substrate, but it also shrinks the matrix, separating it from the mold walls. This decrease in the volume of the substrate is observed mainly in advanced maturation stages of the material (especially after 12 days).
Hyphal structures in the agarized culture are equivalent to the ones exhibited by the same species in the agglomerate material (excepting Ryvardenia cretacea, which failed to completely colonize the substrate). However, the differences observed between P. ostreatus and the results reported by Bruscato et al. (2019) on P. albidus, suggest that even close species can show different performances. Although the hardening effect of calcium salts on the substrates is well known by commercial mushroom producers, this hardness seems not to be reflected in the resistance to penetration measured in the Janka scale. On the contrary, an increased friability is evidenced by the chalk-like consistency of the analyzed materials. For this reason, we conclude that under the analyzed conditions, this kind of amendment does not contribute to the quality of the studied agglomerates.
We emphasize the importance of testing the hardness and other physical properties of novel biological materials, since this information is of paramount importance to find possible technological applications for them. The range of resistance to penetration assessed here suggests that composites based on Pleurotus ostreatus or Funalia trogii are acceptable biodegradable alternatives to expanded polystyrene, while Ganoderma australe is more adequate for applications requiring a higher resistance.
Acknowledgments
We thank Alejandro Jovanovski from the Wood Technology Area of the CIEFAP. We acknowledge the financial support provided by FONCYT (Grant PICT 2018 - 3781 to FK).
References:
Appels, F.V.; Camere, S.; Montalti, M.; Karana, E.; Jansen, K.M.; Dijksterhuis, J.; Wösten, H.A. 2019. Fabrication factors influencing mechanical, moisture-and water-related properties of mycelium-based composites. Material Desing161: 64-71. https://doi.org/10.1016/j.matdes.2018.11.027
Asef, M.R. 2012. Intersterility groups of Pleurotus ostreatus complex in Iran. Mycology 3(2): 147-152.
Bayer, E.; McIntyre, G.; Swersey, B.L. 2008. Method for producing grown materials and products made thereby. U.S. Patent Application US 2008/0145577 A1.
Bruscato, C.; Malvessi, E.; Brandalise, R.N.; Camassola, M. 2019. High performance of macrofungi in the production of mycelium-based biofoams using sawdust-Sustainable technology for waste reduction. Journal Cleaner Production 234: 225-232. https://doi.org/10.1016/j.jclepro.2019.06.150
Camere, S.; Karana, E. 2018. Fabricating materials from living organisms: An emerging design practice. Journal Cleaner Production 186: 570-584. https://doi.org/10.1016/j.jclepro.2018.03.081
Costa Rezende, D.H.C.; Robledo, G.L.; Góes Neto, A.; Reck, M.A.; Crespo, E.M.; Drechsler Dos Santos, E.R. 2017. Morphological reassessment and molecular phylogenetic analyses of Amauroderma s. lat. Raised new perspectives in the generic classification of the Ganodermataceae family. Persoonia 39 (2017 December): 254-269. https://doi.org/10.3767/persoonia.2017.39.10
Costa-Rezende, D.H.; Robledo, G.L.; Drechsler-Santos, E.R.; Glen, M.; Gates, G.; de Madrignac Bonzi, B.; Popoff, O.;Crespo, E.; Góes- Neto, A. 2020. Taxonomy and phylogeny of polypores with ganodermatoid basidiospores (Ganodermataceae). Mycol Progress 19: 725-741. https://doi.org/10.1007/s11557-020-01589-1
Decock, C.; Amalfi, M.; Robledo, G.L.; Castillo, G. 2013. Phylloporia ouraguensis, an undescribed species on Myrtaceae from French Guiana. Cryptogam Mycol 34: 15-27. https://doi.org/10.7872/crym.v34.iss1.2013.15
Di Rienzo, J.; Casanoves, F.; Balzarini, M.; Gonzalez, L.; Tablada, M.; Robledo, C. 2016. InfoStat versión 2016. Grupo InfoStat, FCA, Universidad Nacional de Córdoba: Argentina.
Elsacker, E.; Vandelook, S.; Brancart, J.; Peeters, E.; De Laet, L. 2019. Mechanical, physical, and chemical characterisation of mycelium-based composites with different types of lignocellulosic substrates. PLoS One 14(7): e0213954.
Elsacker, E.; Vandelook, S.; Van Wylick, A. 2020. A comprehensive framework for the production of mycelium-based lignocellulosic composites. Science Total Environment. https://doi.org/10.1016/j.scitotenv.2020.138431
Elsacker, E.; Søndergaard, A.; Van Wylick, A.; Peeters, E.; De Laet, L. 2021. Growing living and multifunctional mycelium composites for large-scale formwork applications using robotic abrasive wire-cutting. Construction and Building Materials283. e122732. https://doi.org/10.1016/j.conbuildmat.2021.122732
Girometta, C.; Picco, A.M.; Baiguera, R.M.; Dondi, D.; Babbini, S.; Cartabia, M.; Pellegrini, M.; Savino, E. 2018. Physical-Mechanical and Thermodynamic Properties of Mycelium-Based Biocomposites: A Review. Sustainability 11(1): 1-22. https://doi.org/10.3390/su11010281
Glenn, J.K.; Gold, M.H. 1985. Purification and characterisation of an extracellular Mn(II)-dependent peroxidase from the lignin degrading basidiomycete Phanerochaete chrysosporium. Archives of Biochemistry and Biophysics 242: 329-341. https://doi.org/10.1016/0003-9861(85)90217-6
Gomez-Montoya, N.; Dreschler-Santos, E.R.; Ferreira-Lopes, V.; Tomsovsky, M.; Urcelay, C.; Robledo, G. 2017. New insights on Trametopsis tomšovský (Polyporales Gäum) based on phylogenetic evidence and morphological analyses of neotropical species. Phytotaxa 311(2): 155-167. http://hdl.handle.net/11336/37394
Haneef, M.; Ceseracciu, L.; Canale, C.; Bayer, I.S.; Heredia-Guerrero, J.A.; Athanassiou, A. 2017. Advanced materials from fungal mycelium: fabrication and tuning of physical properties. Scientific Reports 7. e41292. https://doi.org/10.1038/srep41292
Holt, G.A.; Mcintyre, G.; Flagg, D.; Bayer, E.; Wanjura, J.D.; Pelletier, M.G. 2012. Fungal mycelium and cotton plant materials in the manufacture of biodegradable molded packaging material: Evaluation study of select blends of cotton byproducts. J Biobased Mater Bioenergy 6(4): 431-439. https://doi.org/10.1166/jbmb.2012.124
Islam, M.R.; Tudryn, G.; Bucinell, R.; Schadler, L.; Picu, R.C. 2018. Mechanical behavior of mycelium-based particulate composites. Journal Material Science53(24): 16371-16382. https://doi.org/10.1007/s10853-018-2797-z
Ibañez, C.G. 1995. Contribución al estudio de hongos xilófagos en la provincia de Misiones, Argentina. (Basidiomycetes, Aphyllophorales) I. Ganodermataceae e Hymenochaetaceae. Boletín de la Sociedad Argentina de Botánica 30(3-4): 213-230.
Islam, M.R.; Tudryn, G.; Bucinell, R.; Schadler, L.; Picu, R.C. 2017. Morphology and mechanics of fungal mycelium. Scientific Reports 7(1). e13070. https://doi.org/10.1038/s41598-017-13295-2
Jones, M.; Huynh, T.; Dekiwadia, C.; Daver, F.; John, S. 2017. Mycelium composites: A review of engineering characteristics and growth kinetics. Journal of Bionanoscience 11(4): 241-257. https://doi.org/10.1166/jbns.2017.1440
Jones, M.; Bhat, T.; Wang, C.H.; Moinuddin, K.; John, S. 2017b. Thermal degradation and fire reaction properties of mycelium composites. In Proceedings of the 21st International Conference on Composite Materials, Xi’an, China. pp.20-25.
Jones, M.; Huynh, T.; John, S. 2018. Inherent species characteristic influence and growth performance assessment for mycelium composite applications. Advanced Materials Letters9(1): 71-80. https://aml.iaamonline.org/article_15116_d4d4ba530ebff9f5b3fdabe8c9a7c6df.pdf
Jones, M.; Mautner, A.; Luenco, S.; Bismarck, A.; John, S. 2020. Engineered mycelium composite construction materials from fungal biorefineries: A critical review. Materials & Design 187. e108397. https://doi.org/10.1016/j.matdes.2019.108397
Koutinas, A.A.; Wang, R.; Webb, C. 2004. Restructuring Upstream Bioprocessing: Technological and Economical Aspects for Production of a Generic Microbial Feedstock from Wheat. Biotechnology and Bioengineering 1-15. https://doi.org/10.1002/bit.10888
Kuhar, F.; Castiglia, V.; Levin, L. 2015. Enhancement of laccase production and malachite green decolorization by co-culturing Ganoderma lucidum and Trametes versicolor in solid-state fermentation. International Biodeterioration & Biodegradation 104: 238-243. https://doi.org/10.1016/j.ibiod.2015.06.017
Lechner, B.E.; Petersen, R.; Rajchenberg, M.; Albertó, E. 2002. Presence of Pleurotus ostreatus in Patagonia, Argentina. Revista Iberoamericana de Micología 19(2): 111-114.
Lelivelt, R.J.J. 2015. The mechanical possibilities of mycelium materials. Thesis Master. Eindhoven University of Technology. Eindhoven, Netherlands.
Levin, L. 1998. Biodegradación de materiales lignocelulósicos por Trametes trogii (Aphyllophorales, Basidiomycetes). Tesis Doctoral. Facultad de Ciencias Exactas y Naturales, Universidad de Buenos Aires: Buenos Aires.
Loguercio, G.A.; Jovanovsky, A.; Molina, J.C.; Pantaenius, P. 2008. Residuos de biomasa de forestaciones y aserraderos de la región Andina de las Provincias de Chubut y Neuquén. Evaluación preliminar de oferta. Publicación Técnica. CIEFAP 34. CIEFAP - JICA: Esquel, Argentina.
Martínez, A.T.; Speranza, M.; Ruiz-Dueñas, F.J.; Ferreira, P.; Camarero, S.; Guillén, F.; Gutiérrez, A. 2005. Biodegradation of lignocellulosics: microbial, chemical, and enzymatic aspects of the fungal attack of lignin. International Microbiology8(3): 195-204. https://scielo.isciii.es/pdf/im/v8n3/07%20Martinez.pdf
Meng, L.; Li, W.; Zhang, S.; Wu, C.; Lv, L. 2017. Feasibility of co-composting of sewage sludge, spent mushroom substrate and wheat straw. Bioresource Technology 226: 39-45. https://doi.org/10.1016/j.biortech.2016.11.054
Menolli- Junior, N.; Asai, T.; Capelari, M.; Paccola-Meirelles, L.D. 2010. Morphological and molecular identification of four brazilian commercial isolates of Pleurotus spp. and cultivation on corncob. Brazilian Archives of Biology and Technology 53(2): 397-408. https://doi.org/10.1590/S1516-89132010000200019
Murace, M.; Spavento, E.; Keil, G.; Saparrat, M. 2010. Pudrición castaña: efectos sobre las propiedades de resistencia mecánica de la madera. Quebracho - Revista de Ciencias Forestales 18(1-2): 37-46. https://www.redalyc.org/pdf/481/48118695004.pdf
Nelson, N. 1944. A photometric adaptation of the Somogyi method for the determination of glucose. Journal of Biological Chemistry 153(2): 375-380.
Nobles, M.K. 1965. Identification of cultures of wood-inhabiting Hymenomycetes. Canadian Journal of Botany 43: 1097-1139.
Pelletier, M.G.; Holt, G.A.; Wanjura, J.D.; Bayer, E.; McIntyre, G. 2013. An evaluation study of mycelium based acoustic absorbers grown on agricultural by-product substrates. Industrial Crops and Products 51: 480-485. https://doi.org/10.1016/j.indcrop.2013.09.008
Pegler, D.N. 1996. Hyphal analysis of basidiomes. Mycological Research 100(2): 129-142.
Petre, C.V.; Tănase, C. 2013. Description of the culture characteristics of some lignicolous basidiomycetes species grown on three synthetic media. Journal of Plant Development 20: 105-114. https://azkurs.org/pars_docs/refs/27/26641/26641.pdf#page=106
Rajchenberg, M. 1996. Los hongos pudridores de Nothofagus pumilio (lenga): identificación de los cultivos puros. Bosque 17: 87-100.
Rajchenberg, M. 2006. Los políporos (Basidiomycetes) de los bosques Andino Patagónicos de Argentina. Bibliotheca Mycologica 201. https://www.schweizerbart.de/publications/detail/isbn/3443591035
Rajchenberg, M.; Greslebin, A. 1995. Cultural characters, compatibility tests and taxonomic remarks of selected polypores of the Patagonian Andes forests of Argentina. Mycotaxon 56: 325-346.
Rajchenberg, M.; Pildain, M.B.; Bianchinotti, M.V.; Barroetavena, C. 2015. The phylogenetic position of poroid Hymenochaetaceae (Hymenochaetales, Basidiomycota) from Patagonia, Argentina. Mycologia 107(4): 754-767.
Sanchez, C. 2009. Lignocellulosic residues: biodegradation and bioconversion by fungi. Biotechnology Advances 27(2): 185-194. https://doi.org/10.1016/j.biotechadv.2008.11.001
Schmidt, O. 2006. Wood and tree fungi. Biology, damage, protection, and use. Springer: Berlin, Heidelberg, New York, 334p.
Somogyi, M. 1952. Notes on sugar determination. Journal of Biological Chemistry 195: 19-23.
Stamets, P.; Chilton, J.S. 1983. The mushroom cultivator: a practical guide to growing mushrooms at home. Agarikon Press: Olympia Washington. p.415.
Sutherland, G.R.; Zapanta, L.S.; Tien, M.; Aust, S.D. 1997. Role of calcium in maintaining the heme environment of manganese peroxidase. Biochemistry 36(12): 3654-3662. https://doi.org/10.1021/bi962195m
Vallas, T.; Courard, L. 2017. Using nature in architecture: Building a living house with mycelium and trees. Frontiers of Architectural Research 6(3): 318-328. https://doi.org/10.1016/j.foar.2017.05.003
Urcelay, C.; Robledo, G. 2009. Positive relationship between wood size and basidiocarp production of polypore fungi in Alnus acuminata forest. Fungal Ecology 2(3): 135-139.
Wood, T.M.; Bhat, K.M. 1988. Methods for measuring cellulase activities. Methods Enzymol 160: 87-112.
Wright, J.E.; Deschamps, J.R. 1972. Basidiomicetos xilófilos de los bosques andinopatagónicos. RIA. Revista de investigaciones agropecuarias Serie 5 (9): 111-196.
Xing, Y.; Brewer, M.; El-Gharabawy, H.; Griffith, G.; Jones, P. 2018. Growing and testing mycelium bricks as building insulation materials. IOP Conference Series: Earth and Environmental Science 121(2). e022032
Zervakis, G.; Philippoussis, A.; Ioannidou, S.; Diamantopoulou, P. 2001. Mycelium growth kinetics and optimal temperature conditions for the cultivation of edible mushroom species on lignocellulosic substrates.Folia Microbiologica46(3). e231.
Author notes
♠Corresponding author: fkuhar@gmail.com
