Received: 18 July 2017
Revised document received: 03 November 2017
Accepted: 03 November 2017
Published: 02 December 2017
Funding
Funding source: Cuban Ministry of Public Health
Contract number: project No. 1502301
Award recipient: This study was supported by the by the Cuban Ministry of Public Health (project No. 1502301).
Abstract: Context: Ciprofloxacin is the most commonly used fluoroquinolone and is prescribed as the antibiotic of choice in the treatment of several microbial infections. Some clinical reports have suggested that ciprofloxacin may induce QT-interval prolongation and Torsades de Pointes arrhythmias. This drug is a weak inhibitor of a rapid component of the cardiac delayed rectifier potassium current IKr, but there are few electrophysiological data available to assess whether ciprofloxacin has the potency to provoke QT prolongation and subsequent Torsades de Pointes arrhythmias. Aims: To evaluate the effect of ciprofloxacin on the contractile and electrical activity of isolated rat hearts. Methods: The Langendorff technique was performed in rat hearts, and the effects of ciprofloxacin (0.001 - 100 μM) were measured on the cardiac force of contraction and on the RR, QRS and QTc intervals. The arrhythmogenic potential and the ventricular fibrillation threshold were evaluated with ciprofloxacin. Results: Ciprofloxacin decreased the force of contraction of all hearts studied, in a concentration-dependent manner. The estimated IC50 for the inotropic negative effect was 0.15 ± 0.04 μM. Ciprofloxacin significantly prolonged the QRS complex, QTc and RR interval. Significant arrhythmic effects with ciprofloxacin were shown and the ventricular fibrillation threshold was decreased. Conclusions: These results suggest that ciprofloxacin exerted effects on cardiac Na+, K+ and Ca2+ channels. The actions of ciprofloxacin require further studies at the cellular level. These conclusions may account for clinical data that have been reported previously.
Keywords: antibiotic, arrhythmia, ciprofloxacin, ECG, fluoroquinolones, QT prolongation.
Resumen: Contexto: La ciprofloxacina es la fluoroquinolona más comúnmente empleada como antibiótico de elección en el tratamiento de diversas infecciones microbianas. Algunos reportes clínicos han sugerido que la ciprofloxacina puede prolongar el intervalo QT y causar arritmias del tipo Torsión de Puntas. Este fármaco es un débil bloqueador del componente rápido de la corriente de potasio cardiaca rectificadora retardada IKr, pero hay pocos estudios electrofisiológicos que permitan explicar su potencia de acción para provocar la prolongación del intervalo QT y las consecuentes arritmias del tipo Torsión de Puntas. Objetivos: Evaluar los efectos de ciprofloxacina sobre la actividad contráctil y eléctrica de corazones aislados de rata. Métodos: Se llevó a cabo la técnica de Langendorff en los corazones de rata y se midieron los efectos de ciprofloxacina (0.001 – 100 μM) sobre la fuerza de contracción cardiaca y sobre los intervalos RR, QRS y QTc. Se evaluó el potencial arritmogénico y la variación del umbral de fibrilación ventricular con ciprofloxacina. Resultados: La ciprofloxacina disminuyó la fuerza de contracción de todos los corazones estudiados de manera dependiente de la concentración. El valor de IC50 estimado para el efecto inotrópico negativo de ciprofloxacina fue de 0.15 ± 0.04 μM. El complejo QRS y los intervalos RR y QTc fueron prolongados significativamente con ciprofloxacina. Se observaron eventos arrítmicos con este compuesto y disminuyó significativamente el umbral de fibrilación ventricular. Conclusiones: Estos resultados sugieren que la ciprofloxacina ejerce acciones sobre canales cardiacos de Na+, K+ y Ca2+, lo cual requiere la realización de otros estudios a nivel celular. Estas conclusiones pueden explicar los hallazgos clínicos reportados previamente.
Palabras clave: antibiótico, arritmia, ciprofloxacina, ECG, fluoroquinolonas, QT prolongado.
INTRODUCTION
Ciprofloxacin, like other fluoroquinolones, is widely used for the treatment of various types of bacterial infections. Prolongation of the QT interval is an adverse effect associated with the use of fluoroquinolones (Owens, 2004; Briasoulis et al., 2011). Drug-induced QT prolongation increases the risk for Torsade de Pointes (TdP), a potentially fatal ventricular arrhythmia (Roden, 2004; 2016). The proarrhythmic side effect of these antimicrobial agents is receiving more and more attention since the withdrawal of grepafloxacin and sparfloxacin from the market due to adverse cardiac events (Dupon et al., 1996; Jaillon et al., 1996; Owens, 2004).
Fluoroquinolones prolong the QT interval by blocking voltage-gated potassium channels, especially the rapid component of the delayed rectifier potassium current IKr, expressed by HERG (the human ether-a-go-go-related gene) (Bischoff et al., 2000; Anderson et al., 2001; Kang et al., 2001; Owens, 2004). The IC50 of ciprofloxacin for hERG K+ currents has been reported to be 966 μM in Chinese hamster ovary cells (Kang et al., 2001). On the other hand, Lacroix et al. (2003), reported that the maximum blockade of IKr was 47.6 ± 1.9% at 335 μM of ciprofloxacin in human embryonic kidney (HEK) 293 cells. Fluoroquinolones are thus, weak inhibitors of IKr compared with other drugs with specific inhibitory mechanisms (Owens, 2004, Sanguinetti and Tristani-Firouzi, 2006; Owens and Nolin, 2006) and the association between prolongation of the QT interval and TdP is complex and depends on concurrent risk factors; eg, genetic predisposition, female sex, age, underlying heart disease, and co-medication with drugs that are known to increase the risk of TdP (Abo-Salem et al., 2014; Roden, 2016).
Ciprofloxacin prolonged action potential duration in a concentration-dependent manner in canine isolated cardiac Purkinje fibers (Patmore et al., 2000). In Langendorff-perfused rabbit hearts ciprofloxacin increased the QT interval and monophasic action potentials duration (Milberg et al., 2007). Likewise, in halothane-anesthetized guinea pig, ciprofloxacin, at 10 mg/kg, prolonged PR, QRS and QT intervals and the duration of the monophasic action potentials (Matsuo et al., 2013).
Some clinical reports have suggested that ciprofloxacin may induce QT-interval prolongation or TdP arrhythmias in the presence or absence of co-administered drugs (Noel et al., 2003; Prabhakar and Krahn, 2004; Letsas et al., 2006; Keivanidou et al., 2009; Haring and Bauer, 2012). Also, ciprofloxacin inhibits CYP1A2 and CYP3A4, known CYP-mediated drug interactions that potentially leading to a decreased clearance of several proarrhythmic agents. These interactions pose an underestimated risk in the application of ciprofloxacin (Haring and Bauer, 2012).
There are few electrophysiological data available to assess whether ciprofloxacin has the potency to provoke QT prolongation and subsequent TdP. Besides, information is limited regarding the contractile actions of ciprofloxacin in the heart. The present study assessed the effects of ciprofloxacin on the electrical and contractile activity of rat hearts. To better analyze the proarrhythmic potential of ciprofloxacin, the study further evaluated its arrhythmogenic effects and how it affects the ventricular fibrillation threshold.
MATERIAL AND METHODS
Animals
Male adult (7-8 weeks) Wistar rats were obtained from the National Center for Laboratory Animal Reproduction (CENPALAB; La Habana). Prior to the experiment, animals were adapted for seven days to laboratory conditions (controlled temperature 25 ± 2°C, relative humidity 60 ± 10%, and 12 h light/dark cycles). Tap water and standard diet for rodents supplied by CENPALAB were freely provided. All procedures were also conducted according to the European Commission guidelines for the use and care of laboratory animals and approved by the Ethical Committee for Research of the Center (No. 02-2015, folio 3, book 01, 2015). The minimum number of animals (n = 6) required to obtain consistent data were employed.
Isolated hearts
As previously reported (Galán et al., 1998), under pentobarbital anesthesia rat hearts were removed and placed in cold Tyrode (see below). Rat hearts were carefully dissected, mounted on a Langendorff column and perfused at constant flow (10 mL/min) with a Tyrode solution of the following composition (mmol/L): 140 NaCl, 2.5 KCl, MgCl2, 2 CaCl2, 10 Tris-hydroxymethylaminomethane, 10 Glucose (pH = 7.4, gassed with O2; T = 35°C). A bipolar platinum recording electrode was placed on the ventricular epicardium to record the surface electrocardiogram (ECG). Another bipolar platinum electrode was placed near the atrioventricular ring and was connected to an electronic stimulator.
To record the force of contraction (FC), the cardiac apex was fixed to a force-displacement transducer with a surgical 6-0 silk thread. ECG and FC values were recorded at the spontaneous heart rate and at a fixed stimulus rate (200-ms RR interval).
The incidence of arrhythmia was analyzed in accordance with the Lambeth Conventions (II) as ventricular premature beats, ventricular tachycardia, and other types of arrhythmias (bigeminy and salvo) (Curtis et al., 2013).
The ventricular fibrillation threshold (VFT) was determined using a stimulation program that consisted of twenty 2-ms duration current pulses at 2-ms interval. Current intensity (mA) was measured with the stimulus-isolating unit. VFT was the current intensity at which at least five spontaneous arrhythmic complexes were observed after the end of the stimulus train.
Ciprofloxacin and chemicals
Ciprofloxacin hydrochloride monohydrate (molecular weight = 385.82) was a gift of BioCubaFarma, and it was diluted in the bathing solution on the day of the experiment. All other chemicals were from Sigma Aldrich.
Statistical analysis
Results are expressed as means and standard errors of means. Statistical significance was evaluated using Student’s t-test. Differences were considered statistically significant at p<0.05.
RESULTS AND DISCUSSION
The present study examined the effects of ciprofloxacin on rat cardiac contraction. Fig. 1A shows that ciprofloxacin exerted a negative inotropic effect in concentration-dependent manner. The maximum inhibitory effect of ciprofloxacin (100 μM) was 87.1 ± 4.8% of the control force amplitude. Concentration-effect relationships yielded an IC50 value of 0.15 ± 0.04 μM. The negative inotropic effect of ciprofloxacin has not been reported before, this action indicated the existence of an inhibitory contractile mechanism that could be via their action on any of the mechanisms that lead to a decreasing the cellular calcium entry during the excitation-contraction coupling such as Na+ and Ca2+ channels, the Na-Ca exchanger, the ryanodine receptor or the Ca-ATPase (Bers, 2001). Thus, further studies will be needed to clarify its mechanisms.
Ciprofloxacin, in a concentration-dependent manner, affected the spontaneous electrical activity of isolated rat hearts, it prolonged the RR, QRS and corrected QT (QTc = QT/√RR), in a statistically significant manner (Fig. 1B-D). Ciprofloxacin decreased the heart rate with significant changes in RR interval since 0.1 μM, at lower concentrations (0.001 and 0.01 μM), ciprofloxacin showed a tendency to increase the RR interval but without statistical significance; the pre-drug control RR was 241.2 ± 21.0 ms, it was increased to 1687 ± 57.8 ms at the highest concentration (100 μM). QRS interval was not significantly affected by ciprofloxacin at concentrations from 0.001 to 1 µM; however significant changes were detected in QRS duration since 10, 30 and 100 μM of ciprofloxacin (from 9.4 ± 0.8 ms in control to 13.1 ± 1.1 ms, 16.4 ± 4.1 and 17.1 ± 3.5 ms, respectively). QTc was also prolonged by ciprofloxacin, but with statistical significance at concentrations as low as 0.1 μM; the pre-drug control QTc was 105.6 ± 6.9 ms.
These results are in agreement with those of Matsuo et al. (2013) who reported that ciprofloxacin, at 10 mg/kg, in halothane-anesthetized guinea pig, decreased the heart rate and prolonged the QT interval and the duration of the monophasic action potential of the ventricle. They also reported that the PR interval and QRS were also increased by ciprofloxacin at 10 mg/kg, suggesting that the drug inhibited cardiac K+ channels as well as Na+ and Ca2+ channels in vivo. In Langendorff-perfused rabbit hearts, ciprofloxacin led to a significant increase in QT interval and monophasic action potentials duration and exhibited reverse-use dependence (Milberg et al., 2007). Abdelrady et al. (2017), measuring QT, QTc, QRS and ST segment changes, highlighted that administration of ciprofloxacin (150 or 300 mg/kg) to healthy rats or rats with myocardial infarction, increased all these values compared to their control group.
In contrast, ciprofloxacin, orally administered to healthy male dogs at the dosage of 25 mg/kg weight twice daily for 14 days, produced no significant changes in heart rate, QT, QTc (Bazett's formula) and QTc (Fridericia's formula) (Omobowale et al., 2016).
Another finding of the present study resulted in significant arrhythmic effects with ciprofloxacin in isolated hearts from rats. Of six hearts studied, five developed arrhythmias. Ventricular premature beats, bigeminies, and ventricular salvos occurred with ciprofloxacin since 0.1 μM, but no episodes of TdP were shown (Fig. 2).
The ventricular fibrillation threshold (VFT) was significantly decreased by ciprofloxacin since 0.1 μM. At 0.1 μM and 1 μM of ciprofloxacin, the VFT was decreased in 58.3 ± 8.3% and 56.1 ± 10.6%, respectively, compared with the control condition. These data provide direct evidence for the potential role of ciprofloxacin on the susceptibility to arrhythmias of the isolated rat heart, as assessed by the fall in the VFT. However, in this study, the decrease in the VFT and the proarrhythmic effects were independent of ciprofloxacin concentration.
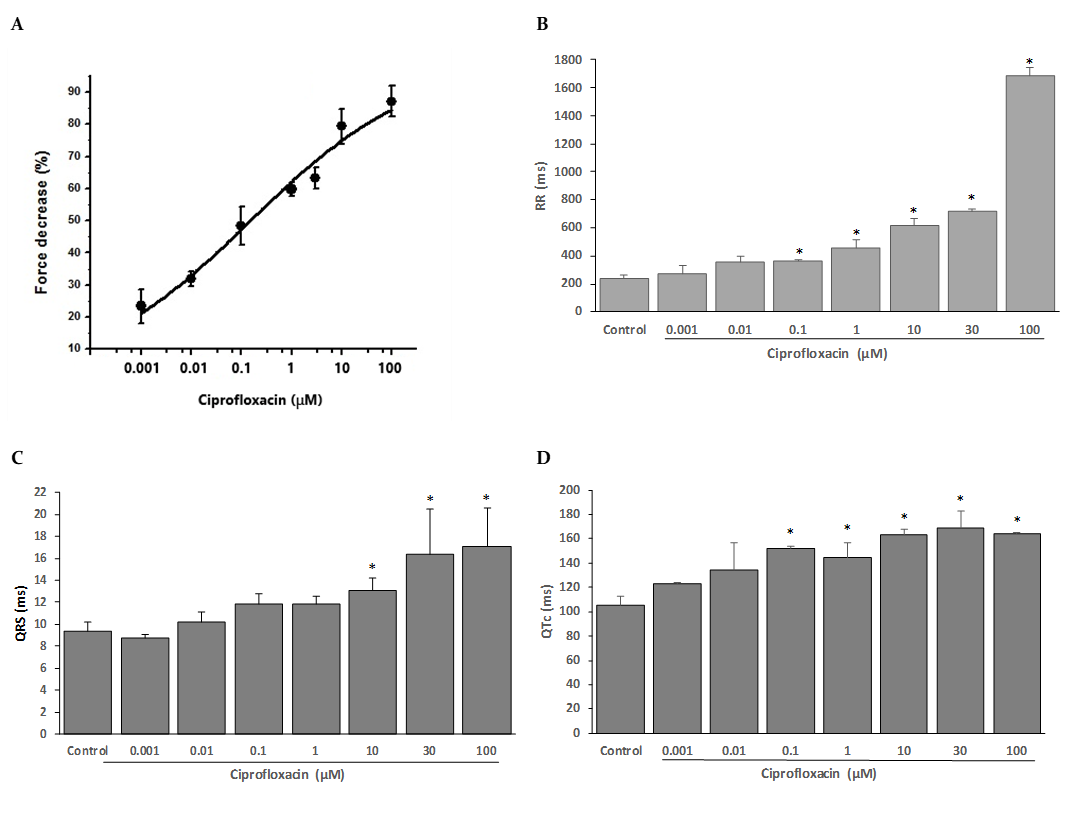
Figure 1
Effects of ciprofloxacin on the contractile and electrical activity of isolated, Langendorff-perfused rat hearts.
A: Concentration-response curves for the inhibition of force of contraction by ciprofloxacin. Experimental data (n = 6 for each point) were fitted to a Hill function. B-D: Effect of ciprofloxacin on the RR (B), QRS (C) and QTc (D) interval of the ECG recorded in isolated rat hearts. The data represent mean ± SEM for n=6.*p<0.05 compared to control values.
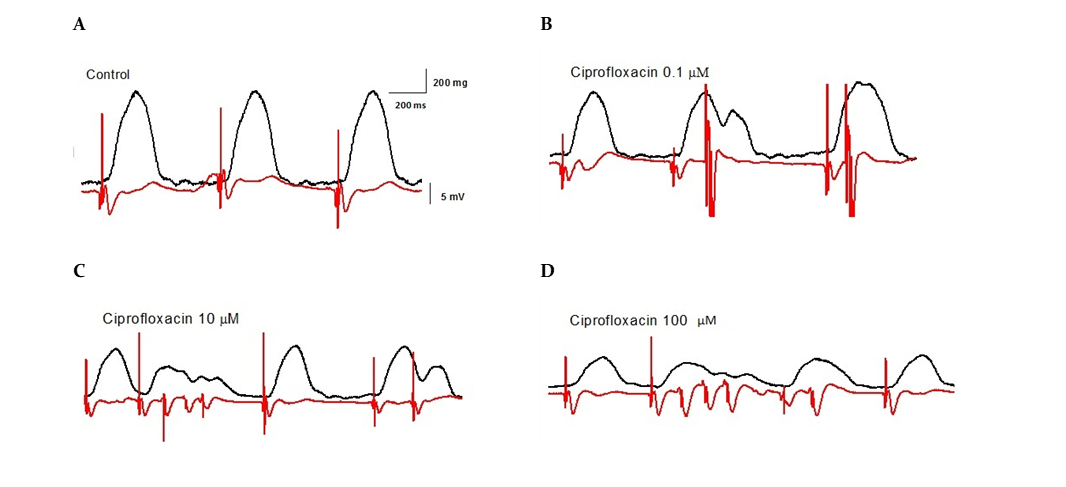
Figure 2
Examples of the arrhythmic effects of ciprofloxacin at different concentrations in isolated, Langendorff-perfused rat hearts on the force of contraction (black upper trace) and ECG (red lower trace).
Trace A is a control recording, trace B indicates the induction of bigeminy by 0.1 μM of ciprofloxacin. Application of 10 μM of ciprofloxacin-induced ventricular premature beats and ventricular salvos (trace C). Ventricular premature beats and ventricular salvos were recorded in the presence of 100 μM of ciprofloxacin (trace D).
Additional pro-arrhythmic mechanisms have been described for ciprofloxacin, such as triangulation of the action potential and dispersion of repolarisation. In the presence of low potassium concentration, four of 10 ciprofloxacin-treated Langendorff-perfused rabbit hearts showed arrhythmic recordings with Early Afterdepolarizations and TdP (40%) (Milberg et al., 2007).
All quinolones demonstrated reverse use dependence as a typical marker of IKr-blocking drugs that is regarded as a risk factor for TdP. It contributes importantly to proarrhythmia associated with these drugs because of a marked increase in dispersion of repolarization. This increase in dispersion of repolarization by all fluoroquinolones is typical for this class of drugs. It results in electrical heterogeneity, representing the appropriate substrate that most likely underlies circus movement re-entry in TdP (Milberg et al., 2007).
Another aspect to keep in mind is that the individual response to a repolarization prolonging drug that might reduce the repolarization reserve plays an important role in the development of proarrhythmia. The concept of repolarization reserve was introduced by Dan Roden (1998), as a patient-specific response to a repolarization prolonging drug underlying acquired QT syndrome. It suggests that risk factors and genetic defects set the stage for this abnormal response (Roden, 2016). When multiple subclinical lesions influence the repolarization process because of clinical risk factors such as hypokalemia or bradycardia, superimposition of IKr-blocking drugs can result in TdP. In this context, administration of fluoroquinolones is of particular importance in patients with predisposing risk factors reflecting decreased repolarization reserve (Prabhakar and Krahn, 2004) and may act as an amplifier that increases the probability of TdP (Owens, 2004; Owens and Nolin, 2006; Abo-Salem et al., 2014).
Ciprofloxacin remains the safest fluoroquinolone to date regarding TdP risk. A few cases of TdP have been reported to occur in association with ciprofloxacin administration (Frothingham, 2001; Prabhakar and Krahn, 2004; Flanagan et al., 2006; Haring and Bauer, 2012; Itoh et al., 2016). This may be related to the fact that some drugs block the hERG potassium channel and prolong QT with a minimal TdP risk like ranolazine (Antzelevitch et al., 2004) or amiodarone (Wu et al., 2008), likely because of additional block of inward currents, such as the late sodium current or the L-type calcium current (Antzelevitch and Shimizu, 2002; Sanguinetti and Tristani-Firouzi, 2006; Roden, 2016). Preclinical studies have suggested that late sodium or calcium current block can shorten hERG potassium channel block-induced action potential and QT prolongation and prevent TdP (Chezalviel-Guilbert et al., 1995; Shimizu and Antzelevitch, 1997; Martin et al., 2004; Belardinelli et al., 2013).
CONCLUSIONS
The present findings clearly highlight the negative inotropic action of ciprofloxacin in a concentration-dependent manner, and the prolonging RR, QTc, and QRS actions. These results suggest that ciprofloxacin exerted a multi-ion channel–blocking action in the heart, and it that requires further studies. These conclusions may account for clinical data that have been reported previously.
Acknowledgements
This work was supported by the Cuban Ministry of Public Health (Project No. 1502301).
REFERENCES
Abdelrady AM, Zaitone SA, Farag NE, Fawzy MS, Moustafa YM (2017) Cardiotoxic effect of levofloxacin and ciprofloxacin in rats with/without acute myocardial infarction: Impact on cardiac rhythm and cardiac expression of Kv4.3, Kv1.2 and Nav1.5 channels. Biomed Pharmacother 92: 196–206.
Abo-Salem E, Fowler JC, Attari M, Craig D. Cox, Perez-Verdia A, Panikkath R, Nugent K (2014) Antibiotic-induced cardiac arrhythmias. Cardiovasc Ther 32: 19–25.
Anderson ME, Mazur A, Yang T, Roden DM (2001) Potassium current antagonist properties and proarrhythmic consequences of quinolone antibiotics. J Pharmacol Exp Ther 296: 806–810.
Antzelevitch C, Belardinelli L, Zygmunt AC, Burashnikov A, Di Diego JM, Fish JM, Cordeiro JM, Thomas G (2004) Electrophysiological effects of ranolazine, a novel antianginal agent with antiarrhythmic properties. Circulation 110: 904–910.
Antzelevitch C, Shimizu W (2002) Cellular mechanisms underlying the long QT syndrome. Curr Opin Cardiol 17: 43–51.
Belardinelli L, Liu G, Smith-Maxwell C, Wang WQ, El-Bizri N, Hirakawa R, Karpinski S, Li CH, Hu L, Li XJ, Crumb W, Wu L, Koltun D, Zablocki J, Yao L, Dhalla AK, Rajamani S, Shryock JC (2013) A novel, potent, and selective inhibitor of cardiac late sodium current suppresses experimental arrhythmias. J Pharmacol Exp Ther 344: 23–32.
Bers DM (2001) Excitation-contraction Coupling and Cardiac Contractile Force. Second edition. Dordrecht, The Netherlands: Kluwer Academic Press.
Bischoff U, Schmidt C, Netzer R, Pongs O (2000) Effects of fluoroquinolones on HERG currents. Eur J Pharmacol 406: 341–343.
Briasoulis A, Agarwal V, Pierce WJ (2011) QT prolongation and torsade de pointes induced by fluoroquinolones: Infrequent side effects from commonly used medications. Cardiology 120: 103–110.
Chezalviel-Guilbert F, Davy JM, Poirier JM, Weissenburger J (1995) Mexiletine antagonizes effects of sotalol on QT interval duration and its proarrhythmic effects in a canine model of torsade de pointes. J Am Coll Cardiol 26: 787–792.
Curtis MJ, Hancox JC, Farkas A, Wainwright CL, Stables CL, Saint DA, Clements-Jewery H, Lambiase PD, Billman GE, Janse MJ, Pugsley MK, Ng GA, Roden DM, Camm AJ, Walker MJ (2013) The Lambeth Conventions (II): Guidelines for the study of animal and human ventricular and supraventricular arrhythmias. Pharmacol Ther 139: 213–248.
Dupont H, Timsit JF, Souweine B, Gachot B, Wolff M, Regnier B (1996) Torsades de pointe probably related to sparfloxacin. Eur J Clin Microbiol Infect Dis 15: 350–351.
Flanagan MC, Mitchell ES, Haigney MCP (2006) Ciprofloxacin-induced torsade de pointes. Intern J Cardiol 113: 239–241.
Frothingham R (2001) Rates of torsades de pointes associated with ciprofloxacin, ofloxacin, levofloxacin, gatifloxacin, and moxifloxacin. Pharmacotherapy 21: 1468–1472.
Galán L, Talavera K, Vassort G, Alvarez JL (1998) Characteristics of Ca2+ channel blockade by oxodipine and elgodipine in rat cardiomyocytes. Eur J Pharmacol 357: 93–105.
Haring B, Bauer W (2012) Ciprofloxacin and the risk for cardiac arrythmias: culpric delicti or watching bystander? Acta Cardiol 67(3): 351–354.
Itoh H, Crotti L, Aiba T, Spazzolini C, Denjoy I, Fressart V, Hayashi K, Nakajima T, Ohno S, Makiyama T, Wu J, Hasegawa K, Mastantuono E, Dagradi F, Pedrazzini M, Yamagishi M, Berthet M, Murakami Y, Shimizu W, Guicheney P, Schwartz PJ, Horie M (2016) The genetics underlying acquired long QT syndrome: impact for genetic screening. Eur Heart J 37(18): 1456–1464.
Jaillon P, Morganroth J, Brumpt I, Talbot G (1996) Overview of electrocardiographic and cardiovascular safety data for sparfloxacin. J Antimicrob Chemother 37 (Suppl. A): 161–167.
Kang G, Wang L, Chen XL, Triggle DJ, Rampe D (2001) Interactions of a series of fluoroquinolone antibacterial drugs with the human cardiac K+ channel HERG. Mol Pharmacol 59: 122–126.
Keivanidou A, Arnaoutoglou C, Krommydas A, Papanikolaou G, Tsiptses K, Chrisopoulos C, Kirpizidis C (2009) Ciprofloxacin induced acquired long QT syndrome in a patient under class III antiarrhythmic therapy. Cardiol J 16: 172–174.
Lacroix P, Crumb WJ, Durando L, Ciottoli GB (2003) Prulifloxacin: in vitro (HERG current) and in vivo (conscious dog) assessment of cardiac risk. Eur J Pharmacol 477: 69–72.
Letsas KP, Sideris A, Kounas SP, Efremidis M, Korantzopoulos P, Kardaras F (2006) Drug-induced QT interval prolongation after ciprofloxacin administration in a patient receiving olanzapine. Int J Cardiol 109: 273–274.
Martin RL, McDermott JS, Salmen HJ, Palmatier J, Cox BF, Gintant GA (2004) The utility of hERG and repolarization assays in evaluating delayed cardiac repolarization: influence of multi-channe block. J Cardiovasc Pharmacol 43: 369–379.
Matsuo K, Fujiwara K, Omuro N, Kimura I, Kobayashi K, Yoshio T, Takahara A (2013) Effects of the fluoroquinolone antibacterial drug ciprofloxacin on ventricular repolarization in the halothane-anesthetized guinea pig. J Pharmacol Sci 122: 205–212.
Milberg P, Hilker E, Ramtin S, Cakir Y, Stypmann J, Engelen MA, Mönnig G, Osada N, Breithardt G, Haverkamp W, Eckardt L (2007) Proarrhythmia as a class effect of quinolones: Increased dispersion of repolarization and triangulation of action potential predict torsades de pointes. J Cardiovasc Electrophysiol 18(6): 647–654.
Noel GJ, Natarajan J, Chien S, Hunt TL, Goodman DB, Abels R (2003) Effects of three fluoroquinolones on QT interval in healthy adults after single doses. Clin Pharmacol Ther 73: 292–303.
Omobowale TO, Oridupa OA, Abiola JO, Orotusin FT, Fabiyi BO, Akangbou PJ (2016) Comparative effects of ciprofloxacin and levofloxacin on heart rate and QT/QTc measurements in dogs. Pharm Chem J 3(3): 26–30.
Owens RC Jr (2004) QT Prolongation with antimicrobial agents understanding the significance. Drugs 64(10): 1091–1124.
Owens RC Jr, Nolin TD (2006) Antimicrobial-associated QT interval prolongation: pointes of interest. Clin Infect Dis 43: 1603–1611.
Patmore L, Fraser S, Mair D, Templeton A (2000) Effects of sparfloxacin, grepafloxacin, moxifloxacin, and ciprofloxacin on cardiac action potential duration. Eur J Pharmacol 406: 449–452.
Prabhakar M, Krahn AD (2004) Ciprofloxacin-induced acquired long QT syndrome. Heart Rhythm 1: 624–626.
Roden DM (1998) Taking the “idio” out of “idiosyncratic”: Predicting torsades de pointes. Pacing Clin Electrophysiol 21: 1029–1034.
Roden DM (2004) Drug-induced prolongation of the QT interval. N Engl J Med 350: 1013–1022.
Roden DM (2016) Predicting drug-induced QT prolongation and torsades de pointes. J Physiol 594(9): 2459–2468.
Sanguinetti MC, Tristani-Firouzi M (2006) hERG potassium channels and cardiac arrhythmia. Nature 440: 463–469.
Shimizu W, Antzelevitch C (1997) Sodium channel block with mexiletine is effective in reducing dispersion of repolarization and preventing torsade de pointes in LQT2 and LQT3 models of the long-QT syndrome. Circulation 96: 2038–2047.
Wu L, Rajamani S, Shryock JC, Li H, Ruskin J, Antzelevitch C, Belardinelli L (2008) Augmentation of late sodium current unmasks the proarrhythmic effects of amiodarone. Cardiovasc Res 77: 481–488.
Additional information
CONFLICT OF INTEREST: The authors declare no conflict of interest.
Citation Format: Galán-Martínez L, Calderín-Pulido
AD, Fleites-Vázquez A, Álvarez JL (2018) Ciprofloxacin, an antibiotic with cardiac
actions on isolated rat hearts. J Pharm Pharmacogn
Res 6(2): 65–71.
Alternative link
http://jppres.com/jppres/pdf/vol6/jppres17.292_6.2.65.pdf (pdf)
http://jppres.com/jppres/cardiac-actions-of-ciprofloxacin/ (html)
