Received: 18 August 2017
Revised document received: 15 January 2018
Accepted: 21 January January 2018
Abstract: Context: According to a report by the World Health Organization (WHO), there were 1.4 million deaths worldwide in 2015 from tuberculosis (TB), with 3.9% being new cases and 21% being previously treated cases of multidrug-resistant tuberculosis (MDR-TB). Aims: To review the literature concerning the costing analysis situation of MDR-TB treatment. Methods: The study was conducted as a systematic review, with a modified checklist being used as the vital instrument. A search was performed of three databases (PubMed, Cochrane, and Scopus) using the terms (cost OR economic, socioeconomic, expenditure, burden, fee, charge, budget impact) AND (resistance OR multidrug resistance, MDR) AND (tuberculosis OR TB, Mycobacterium tuberculosis) in order to identify relevant articles published from 2006 to the present. Results: A total of 1238 abstracts were identified, and 12 papers were ultimately included in the study. The quantity of the published articles was found to increase during in the period 2008 to 2016. Almost all the studies were based on patients’ and healthcare systems’ perceptions. The main data sources used were medical establishments and the reports of various relevant organizations. Primary data were used twice as much as secondary data. All the costing types, including direct costs and indirect costs, were mentioned, albeit not with the same frequency. Conclusions: Africa owns one third of the articles included. Further, it was found that MDR-TB should be treated using ambulatory care rather than hospital-based models. Future research studies should focus on Asia, where drug resistance has proved to be a challenging issue.
Keywords: cost, literature review, multi-drug resistant tuberculosis, systematic review, tuberculosis.
Resumen: Contexto: El informe mundial sobre la tuberculosis, presentado por la Organización Mundial de la Salud (OMS) en 2016, muestra que en 2015 hubo 1,4 millones de muertes por tuberculosis, 3,9% de nuevos casos y 21% de casos tratados con tuberculosis multirresistente. Objetivos: Revisar la literatura científica concerniente al análisis del costo del tratamiento de la tuberculosis multirresistente a fármacos. Métodos: Este estudio se realizó como una revisión sistemática utilizando una lista de comprobación modificada como un instrumento vital. Una búsqueda en tres bases de datos (PubMed, Cochrane y Scopus) con los términos de (cost OR economic, socioeconomic, expenditure, burden, fee, charge, budget impact) AND (resistance OR multidrug resistance, MDR) AND (tuberculosis OR TB, Mycobacterium tuberculosis) se utilizó para abordar una serie de artículos publicados desde 2006 hasta la actualidad. Resultados: Se identificaron un total de 1.238 resúmenes y se incluyeron 12 artículos en este estudio. La cantidad de artículos publicados aumentó en el período 2008-2016. La mayoría de los estudios se basaron en la percepción del paciente y del sistema de salud. La fuente de datos utilizada fue principalmente del establecimiento médico y los informes de las organizaciones. Los datos primarios se utilizaron dos veces más que los datos secundarios. Todos los tipos de costos, costos directos e indirectos, fueron mencionados, pero no en la misma frecuencia. Conclusiones: África posee un tercio de los artículos incluidos. La TB-MDR debe ser tratada con atención ambulatoria en lugar de modelos basados en hospitales. Las investigaciones futuras deberían centrarse en Asia, donde la resistencia a los medicamentos ha sido un tema desafiable.
Palabras clave: costo, revisión de literatura, revisión sistemática, tuberculosis, tuberculosis multirresistente.
INTRODUCTION
Tuberculosis (TB) is a potentially fatal infectious disease that commonly affects the lungs (pulmonary TB), although it can also affect other sites, for example, the bones, joints, skin, meninges, and kidneys (extra pulmonary TB) (WHO, 2016). It is caused by the bacillus Mycobacterium tuberculosis (WHO, 2016). The disease spreads via the respiratory tract, which means that a droplet of fluid containing the tubercle bacilli is released into the air whenever a TB patient exhales, coughs, or sneezes and, further, that it could be taken into the nasal passages and lungs of a susceptible person nearby through the air (WHO, 2016).
According to the World Health Organization’s (WHO’s) Global Tuberculous Report 2016, there were 1.4 million deaths worldwide from TB in 2015, with an additional 0.4 million deaths being seen among people who tested positive for the human immunodeficiency virus (HIV), and 10.4 million new TB cases (including among 1.2 million HIV-positive people, 5.9 million men, 3.5 million women, and 1 million children). Despite being characterized by a lower contagion risk than other infectious diseases, 5–15% of people infected with M. tuberculosis will develop TB during their lifetime. This proportion is much higher among people who are co-infected with HIV (WHO, 2016).
Multidrug-resistant TB (MDR-TB) is defined as TB that is resistant to at least isoniazid and rifampicin, which are the two most commonly used and effective first-line anti-TB drugs. Extensively drug-resistant TB (XDR-TB) is defined as MDR-TB plus resistance to at least one fluoroquinolone and a second-line injectable agent (WHO, 2016). There are tests available for TB that is resistant to first- and second-line anti-TB drugs, including Xpert MTB/RIF, which simultaneously tests for TB and resistance to rifampicin; rapid line probe assays (LPAs), which test for resistance to rifampicin and isoniazid (referred to as first-line LPAs); a rapid LPA that tests for resistance to fluoroquinolones and injectable anti-TB drugs (referred to as a second-line LPA); and sequencing technologies. First-line LPAs were first recommended by the WHO in 2008, while the second-line LPA was first recommended in May 2016. At the same time, in 2016 the WHO issued guidance that people with TB that proves resistant to rifampicin, with or without resistance to other drugs, should be treated with an MDR-TB treatment regimen (WHO, 2016).
In 2015, the WHO estimated that 3.9% (95% confidence interval [CI]: 2.7–5.1%) of new cases and 21% (95% CI: 15–28%) of previously treated cases worldwide had MDR/RR-TB. There were an estimated 480,000 new cases of MDR-TB as well as an additional 100,000 people with rifampicin-resistant TB who were also newly eligible for MDR-TB treatment. Some 51% (30–70%) of patients with MDR-TB exhibit resistance to a fluoroquinolone or a second-line injectable agent, or both. The countries with the largest numbers of MDR/RR-TB cases (accounting for 45% of the global total) are China, India, and the Russian Federation. Based on data collected from 40 countries with a high TB or MDR-TB burden (or both), there is a slight trend for cases of MDR-TB to increase as a proportion of all TB cases in such countries, with the burden of MDR-TB either increasing faster or decreasing more slowly than the overall TB burden in each country (WHO, 2016).
According to the WHO report, progress in TB prevention, diagnosis, and treatment requires adequate funding sustained over many years. In 126 low- and middle-income countries, which accounted for 97% of the total global number of TB cases reported in 2015, funding for TB prevention, diagnosis, and treatment reached 6.6 billion United States (US) dollars ($) in 2016, with the majority of the cost ($4.4 billion, 67%) stemming from the diagnosis and treatment of drug-susceptible TB, although the amount associated with multidrug-resistant TB (MDR-TB) is estimated to double from $1.7 billion in 2016 to $3.6 billion by 2020 (WHO, 2016).
Data from costing analysis studies conducted prior to 2012 have indicated that the average cost per patient for MDR-TB treatment in the Philippines (Tupasi et al., 2006), Peru (Suarez et al., 2002), Estonia, and Tomsk (WHO, 2009) was $3613, $2423, $10,880, and $14,657, respectively, while the best estimates of the cost per disability-adjusted life year (DALY) averted were $143, $163, $598, and $745, respectively. In 2016, the WHO reported that the cost per patient treated is usually within the range of $100–1000 for drug-susceptible TB and $2000–20,000 for MDR-TB (WHO, 2016).
Cost analysis data represent part of the evidence needed to inform decisions regarding whether and how to scale up MDR-TB interventions. In addition, data concerning cost are important for budgeting and financing the expansion of services, as well as for the analysis of affordability. Nevertheless, costing analyses have tended to focus on TB diagnosis and treatment without a concentration on MDR-TB. According to an initial literature search, only one systematic review of the cost of treatment for MDR-TB was conducted in 2012 (Fitzpatrick and Floyd, 2012), which is why an up-to-date review is needed to assess the quality of studies concerning the economic burden of MDR-TB. Therefore, this study performed an updated literature review of the costing analysis situation with regard to MDR-TB treatment.
MATERIAL AND METHODS
Study design
This study was conducted as a systematic review of articles that analyzed the cost of MDR-TB treatment in various countries. After searching the literature, the authors filtered all the results in order to choose several articles that met the pre-identified selection criteria. When eligibility testing had been completed, the data analysis was performed in two stages, namely quantity and quality appreciation, based on two separate checklists, respectively.
Literature search
A search strategy was designed in order to identify articles published from 2006 to the present. All the search results from the three utilized databases, namely PubMed, Cochrane, and Science Direct, were combined and any duplicates were discarded. The keywords (cost OR economic, socioeconomic, expenditure, burden, fee, charge, budget impact) AND (resistance OR multidrug resistance, MDR) AND (tuberculosis OR TB, Mycobacterium tuberculosis) were used to identify eligible studies. To limit the occurrence of undesirable articles, these keywords and MeSH terms were searched in the “Title/Abstract” category.
Selection criteria
The included studies were required to meet the following inclusion criteria: (a) “The article must consider MDR-TB”; (b) “The outcome of the article presented the direct cost, non-direct cost, or economic burden of MDR-TB”; and (c) “The study was published in English and related to humans”. In addition, if the issues were not categorized in the health sector, not related to humans, or applied prior to 2006, then the studies would be rejected. Any studies that could be categorized as conference abstracts, posters, oral communications, or textbooks were also excluded during the screening phase.
Data assessment
All the included studies were appraised in three stages in order to collect the necessary data. The first stage involved summing up the general information concerning each study, including the year of publication, country, the period of time in which the research was conducted, and the unit of currency used to calculate the cost. It was important to check whether these factors affected the assessment results. In the second stage, every article was appraised with regard to quantity. There are numerous costing checklists available, with aspects concerning both costing health services and costing analysis. In this study, the authors created a checklist based on that summarized in Yazbeck (2001). The checklist was edited to ensure it was suitable for achieving the objective of this study. The checklist featured 16 items that were divided into two parts, namely the methodology and the results of the articles. In the results part, the authors used statistics to determine which independent variables were associated significantly or non-significantly with the cost pf MRD-TB. Moreover, the total cost per episode was determined in order to provide an overview of the economic burden of the treatment of MDR-TB. The assessment was carried out independently by the three authors. Differences in the assessment outcomes were remedied by a discussion involving all the authors, who then arrived at a collective conclusion.
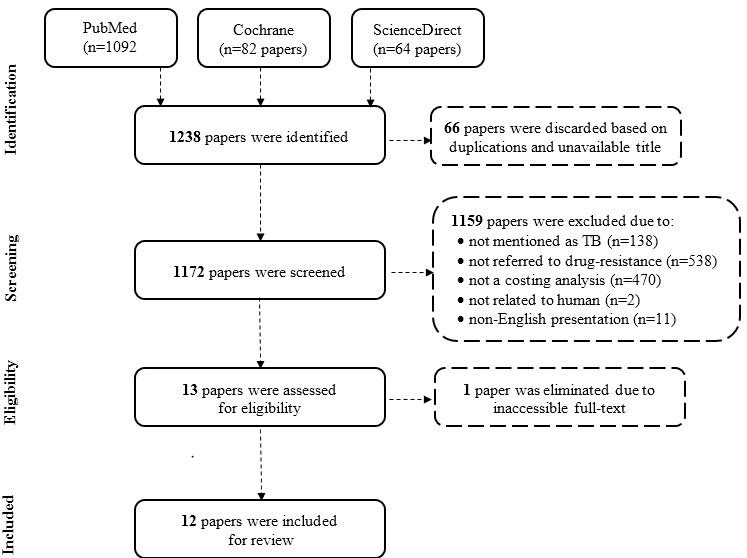
Figure 1
Flow diagram of the systematic review.
RESULTS AND DISCUSSION
A total of 1238 papers were identified through the search performed in December 2016 of three databases, namely PubMed (n=1082), Cochrane (n=82), and Science Direct (n=64). When considering duplicates, 66 titles were excluded. By means of abstract scanning, those titles that were not published in English (n=11), not related to humans (n=2), tuberculosis infection (n=138), drug resistance (n=538), or cost analysis (n=470), and unavailable as a full text (n=2) were also eliminated. Ultimately, 12 papers were included in the assessment. The screening process is illustrated in Fig. 1.
General characteristics
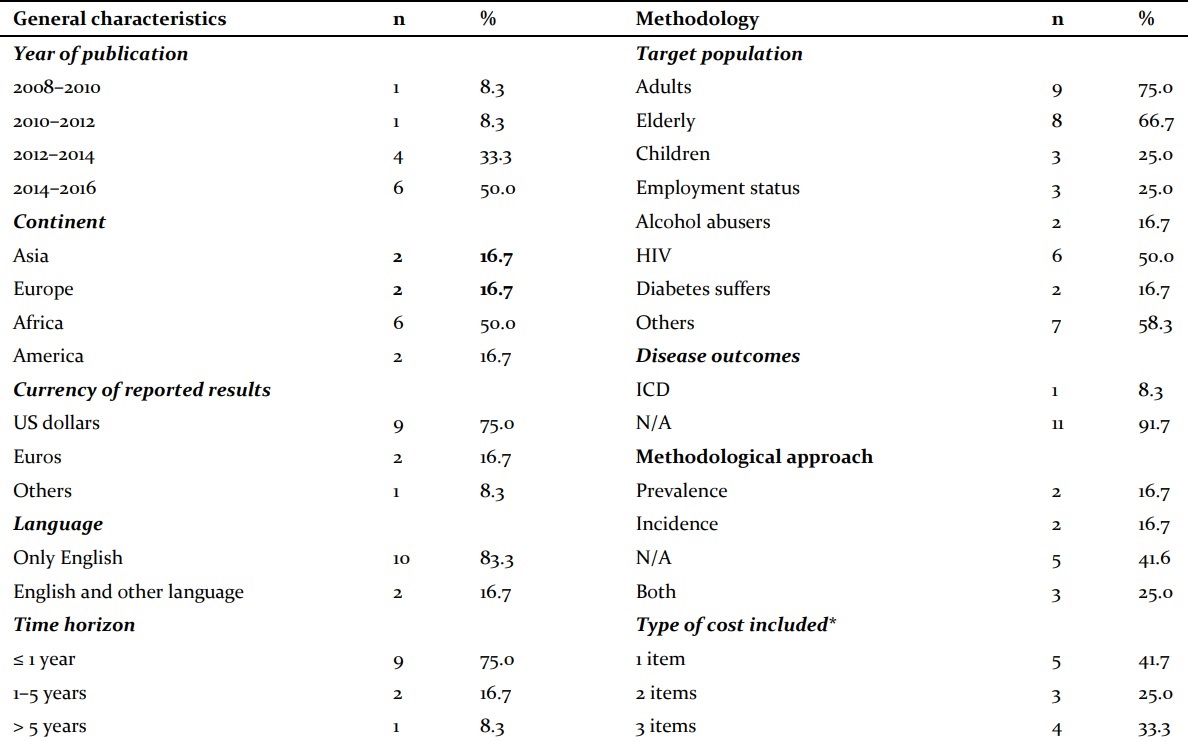
Quite clearly, the number of published articles increased during the period 2008–2016. There were two articles (Mohammadi et al., 2008; Rouzier et al., 2010) published from 2008 to 2012 and four articles published between 2013 and 2014 ( Floyd et al., 2012; Pantoja et al., 2013; Pooran et al., 2013; Schnippel et al., 2013;), but over the next two years, there were six articles (Diel et al., 2014; Marks et al., 2014; Cox et al., 2015; du Toit et al., 2015; Ramma et al., 2015; Musa et al., 2016) published. An increase in the number of published articles indicates an increase in interest in researching MDR-TB. As shown in Table 1, the time horizon of the studies was commonly less than one year (n=9, 75%), although three studies had a time horizon of more than one year (Mohammadi et al., 2008; Floyd et al., 2012; Marks et al., 2014). Six research studies were carried out in Africa (50.0%), while the continents of Asia, Europe, and the Americas shared the six remaining studies (17% for each). Among the 12 studies, five were conducted in South Africa. It is noteworthy that one study was conducted in 36 countries (Pantoja et al., 2013). The main currency used was the US dollar (n=9, 75%), although two articles used Euros (€) and one used Oman dollars (OMR). Due to the applied exclusion criteria, English was the official language of all the articles, although two articles (17%) used French as an added language (Mohammadi et al., 2008; Rouzier et al., 2010).
Methodology
Table 1 shows that studies based on patients’ and healthcare systems’ perceptions were common, accounting for 66% of all studies, followed by the views of society and payers, which accounted for 25% and 17%, respectively. The studies were found to be less reliant on the views of providers (8.3%).
General characteristics, methodology, and results of the included studies (n = 12).
General characteristics, methodology, and results of the included studies (n = 12).
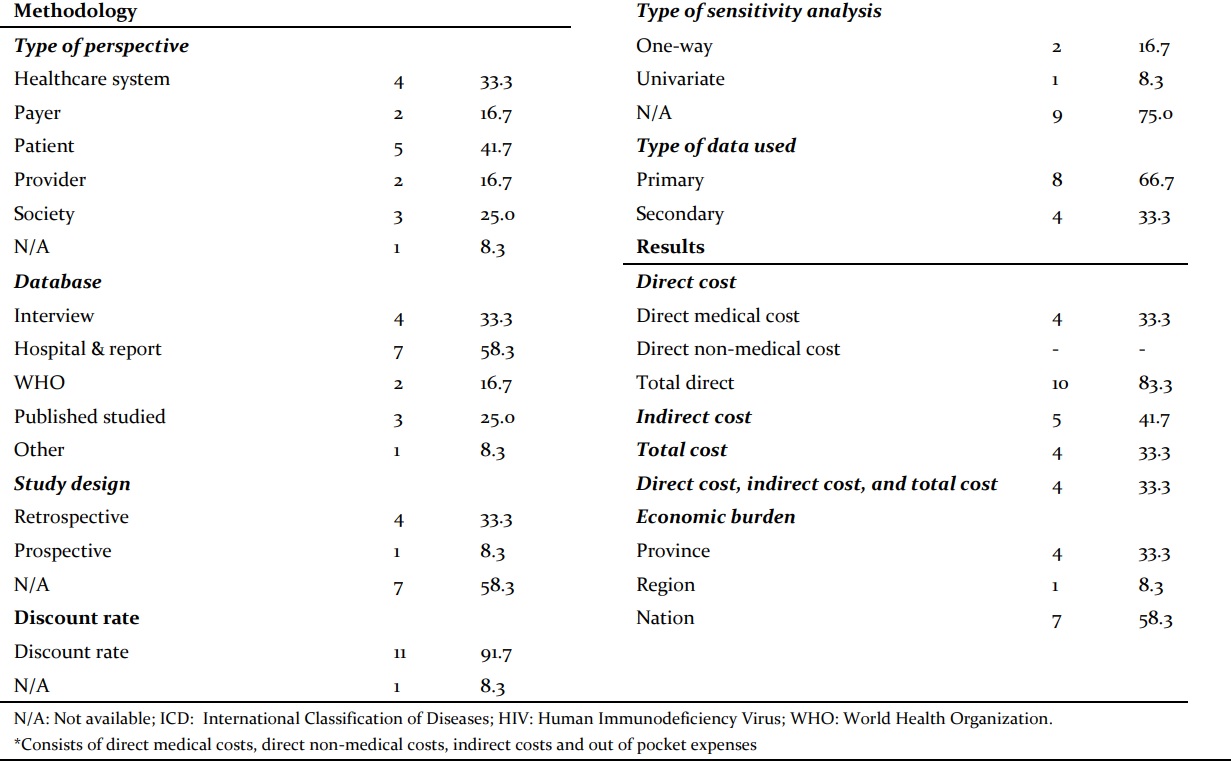
The main data sources used were medical establishments and the reports of relevant organizations (n=7, 58.3%). Interviews were also commonly used to collect research data (n=4, 33.3%). Reliable data sources (published studies and the WHO) accounted for 41.7% (n=5) of the total number of articles. One article used online data provided by pages (Marks et al., 2014). The majority of articles used a combination of data sources, including interviews with staff, interviews with patients, clinical records, and national sources. There were twice as many studies using primary data (n=8, 66.7%) than secondary data (n=4, 33.3%), since most studies used reports from health authorities and interviews. The studies were more commonly designed with a retrospective approach (n=4, 33.3%) than a prospective approach (n=1, 8.3%). The remaining seven studies did not follow either of these approaches, accounting for 58.3% of the total number of studies.
The majority of the target populations in the studies were adults (n=9, 75%) and the elderly (n=8, 66.7%). People infected with HIV were a significant focus of half the total number of studies (n=6, 50%). Among the other participant groups, alcohol abusers, diabetes patients, children, unemployed people, pregnant women, and those with pulmonary symptoms, as well as subgroups based on education level, were also mentioned in small proportions (<25%). In the 12 selected studies, only one study conducted in Germany (Diel et al., 2014) used the International Classification of Diseases (ICD) code to filter data (8.3%), while the remaining 11 articles (91.7%) did not mention it. The methodological approaches used in the studies were equal in terms of the prevalence- and incidence-based approaches (n=4, 33.3%). There were three studies that used both methods (Rouzier et al., 2010; Floyd et al., 2012; Pooran et al., 2013), while five studies did not explain their methodological approaches (41.7%).
The discount rate was a well-considered factor in the majority of studies. Nine studies were conducted during a period of less than one year, so the discount rate was 0%, while the others (n=2) had a longer study period and used the discount rate to convert the treatment cost from the study year into the current year. Only one article (du Toit et al., 2015) failed to mention the discount rate throughout the whole text. The notion of a sensitivity analysis was an underexplored area that was not available in nine of the 12 articles (75%), although two articles used a one-way analysis (Pantoja et al., 2013; Musa et al., 2016) and one article used a univariate analysis (Pooran et al., 2013).
Costing analysis results of the articles
In Table 1, it could be seen that ten articles calculated the total direct cost of MDR-TB, although only four of them considered the direct medical costs, while no articles computed the direct non-medical costs. Five articles (41.7%) estimated the indirect cost, while four (33.3%) calculated the total cost. Four articles used all three components of the treatment cost, namely the direct cost, indirect cost, and total cost, to assess the economic burden. The investigated studies were most commonly national (n=7, 58.3%), followed by provincial studies (n=4, 33.3%) and one regional study (n=1, 8.3%).
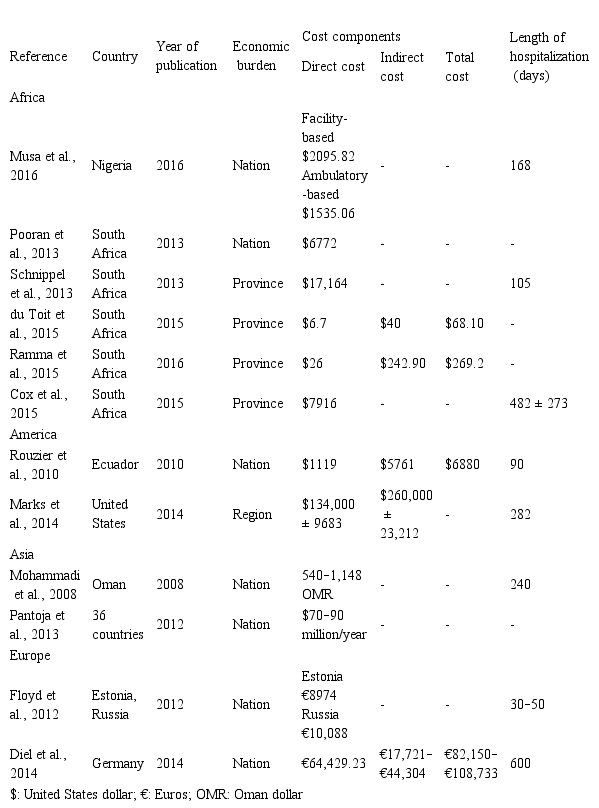
In terms of the cost types, the direct medical cost was included in all 12 studies, while none of them estimated the direct non-medical cost. A minority of studies (n=5, 41.7%) mentioned the indirect cost, with all those studies save for that by Marks et al. (2014) providing a figure for the total cost. Details concerning the results of the cost analyses performed in the 12 studies are summarized in Table 2. The cost data were measured in currency units per patient. Only Pantoja et al. (2013) presented the cost in US dollars per year, since that article featured the largest scope (36 countries) and summed up the worldwide figure rather than the national or provincial figures provided in the remaining studies. The USA paid the highest cost at $134,000 ± 9683 and $260,000 ± 23,212 for the direct and indirect costs, respectively (Marks et al., 2014), while the results found by du Toit et al. (2015) were the lowest, with figures of $6.7 and $40, respectively. In addition, more than half the studies included information about the length of sick days. As seen in Table 2, this number ranged from 30 days (Floyd et al., 2012) to eight months (Mohammadi et al., 2008).
Details of the costing analyses of the included studies (n = 12).
DISCUSSION
The results of the present study demonstrate that a remarkable number of prior studies (n=6, 50%) were conducted in Africa. It is noteworthy that all six of these African studies were published within the last four years, showing an upward trend in research concerning MDR-TB in Africa. The interdependence between poverty and TB has been mentioned in several studies (Ramma et al., 2015, Aia et al., 2016). Not only does poverty increase the risk of contracting TB, but TB also aggravates poverty (Aia et al., 2016). South Africa, which is one of the world’s 22 high TB burden countries with the second highest number of notified RR-TB and MDR-TB cases, was the setting for five out of the six publications. The scope of the studies is diverse; however, there are similarities in results. First, the cost per patient of MDR-TB is much greater than that of drug-sensitive TB. According to Pooran et al. (2013), these costs were $6772 and $257, respectively. Likewise, Schnippel et al. (2013) concluded that the cost of MDR-TB is 40 times higher than the cost of successful drug-susceptible treatment. Second, the majority of the cost stemmed from hospitalization, including general hospital expenses as well as building and staff costs. Schnippel et al. (2013) estimated that the inpatient cost of MDR-TB is over $16,300, which accounts for 95% of the mean total cost per patient. Third, the more serious the patient’s condition, the higher the required cost. Ramma et al. (2015) showed that the mean monthly patient costs associated with the diagnosis and treatment of RR/MDR-TB during the intensive phase and continuation phase are $235 versus $188.
The present study has several limitations that must be acknowledged with the regard to interpreting of the findings. This review was impeded by a number of assumptions and uncertainties. The assessment of cost was restricted to published evidence. Gray or unpublished literature is unlikely to have undergone peer review, and its methodological quality cannot be guaranteed. The number of databases searched was more modest than in other reviews, and a language constraint was also applied, so that there might be a number of publications that were ignored. As the selection and assessment criteria were established based on the authors’ subjective points of view, the quality of the included studies may not be appraised appropriately. Moreover, during the statistical analysis as well as the presentation of the text, errors could have arisen.
However, this review does have a number of strengths. A systematic review was explicitly and transparently conducted. When compared to the last published review concerning MDR-TB, which was performed in 2012, this review is up-to-date, including as it does ten papers published within the last five years. The 12 included studies scored well on the related checklist. Although the authors worked independently, they combined efforts to standardize the results. This study recommends that future investigations should focus on Asia, where drug resistance has proved to be a serious social challenge. It would also be valuable for future cost analyses to have an unequivocal appearance with a clear perspective as well as an explicit study design and calculation method.
CONCLUSIONS
This study was designed as a systematic review using a self-combined checklist to provide an overview of the quality of prior cost analyses concerning MDR-TB. At present, a considerable number of studies have examined the cost of the diagnosis and treatment of MDR-TB. The majority of them were conducted in Africa, where TB as well as MDR-TB result in a high burden. It is recommended that MDR-TB should be treated using ambulatory care rather than hospital-based models. Further studies conducted in Asia are needed due to the lack of information concerning the TB situation in that continent.
Acknowledgements
The authors wish to acknowledge the pharmacists who voluntarily participated in this study. They also wish to express their gratitude to all the lecturers in the Faculty of Pharmacy, University of Pharmacy and Medicine at Ho Chi Minh City, for their support.
REFERENCES
Aia P, Kal M, Lavu E, John LN, Johnson K, Coulter C, Ershova J, Tosas O, Zignol M, Ahmadova S, Islam T (2016) The burden of drug-resistant tuberculosis in Papua New Guinea: Results of a large population-based survey. PLoS One 11(3): e0149806.
Cox H, Ramma L, Wilkinson L, Azevedo V, Sinanovic E (2015) Cost per patient of treatment for rifampicin-resistant tuberculosis in a community-based programme in Khayelitsha, South Africa. Trop Med Int Health 20(10): 1337–1345.
Diel R, Nienhaus A, Lampenius N, Rusch-Gerdes S, Richter E (2014) Cost of multi drug resistance tuberculosis in Germany. Respir Med 108(11): 1677–1687.
du Toit E, Squire SB, Dunbar R, Machekano R, Madan J, Beyers N, Naidoo P (2015) Comparing multidrug-resistant tuberculosis patient costs under molecular diagnostic algorithms in South Africa. Int J Tuberc Lung Dis 19(8): 960–968.
Fitzpatrick C, Floyd K (2012) A systematic review of the cost and cost effectiveness of treatment for multidrug-resistant tuberculosis. Pharmacoeconomics 30(1): 63–80.
Floyd K, Hutubessy R, Kliiman K, Centis R, Khurieva N, Jakobowiak W, Danilovits M, Peremitin G, Keshavjee S, Migliori GB (2012) Cost and cost-effectiveness of multidrug-resistant tuberculosis treatment in Estonia and Russia. Eur Respir J 40(1): 133–142.
Marks SM, Flood J, Seaworth B, Hirsch-Moverman Y, Armstrong L, Mase S, Salcedo K, Oh P, Graviss EA, Colson PW, Armitige L, Revuelta M, Sheeran K (2014) Treatment practices, outcomes, and costs of multidrug-resistant and extensively drug-resistant tuberculosis, United States, 2005-2007. Emerg Infect Dis 20(5): 812–821.
Mohammadi A, Nassor ZS, Behlim T, Mohammadi E, Govindarajan R, Al Maniri A, Smego RA Jr. (2008) Epidemiological and cost analysis of multidrug-resistant tuberculosis in Oman. East Mediterr Health J 14(6): 1240–1245.
Musa BM, John D, Habib AG, Kuznik A (2016) Cost-optimization in the treatment of multidrug resistant tuberculosis in Nigeria. Trop Med Int Health 21(2): 176–182.
Pantoja A, Fitzpatrick C, Vassall A, Weyer K, Floyd K (2013) Xpert MTB/RIF for diagnosis of tuberculosis and drug-resistant tuberculosis: A cost and affordability analysis. Eur Respir J 42(3): 708–720.
Pooran A, Pieterson E, Davids M, Theron G, Dheda K (2013) What is the cost of diagnosis and management of drug resistant tuberculosis in South Africa? PLoS One 8(1): e54587.
Ramma L, Cox H, Wilkinson L, Foster N, Cunnama L, Vassall A, Sinanovic E (2015) Patients' costs associated with seeking and accessing treatment for drug-resistant tuberculosis in South Africa. Int J Tuberc Lung Dis 19(12): 1513–1519.
Rouzier VA, Oxlade O, Verduga R, Gresely L, Menzies D (2010) Patient and family costs associated with tuberculosis, including multidrug-resistant tuberculosis, in Ecuador. Int J Tuberc Lung Dis 14(10): 1316–1322.
Schnippel K, Rosen S, Shearer K, Martinson N, Long L, Sanne I, Variava E (2013) Costs of inpatient treatment for multi-drug-resistant tuberculosis in South Africa. Trop Med Int Health 18(1): 109–116.
Suarez PG, Floyd K, Portocarrero J, Alarcón E, Rapiti E, Ramos G, Bonilla C, Sabogal I, Aranda I, Dye C, Raviglione M, Espinal MA (2002) Feasibility and cost-effectiveness of standardised second-line drug treatment for chronic tuberculosis patients: A national cohort study in Peru. Lancet 359(9322): 1980–1989.
Tupasi TE, Gupta R, Quelapio MI, Orillaza RB, Mira NR, Mangubat NV, Belen V, Arnisto N, Macalintal L, Arabit M, Lagahid JY, Espinal M, Floyd K (2006) Feasibility and cost-effectiveness of treating multidrug-resistant tuberculosis: A cohort study in the Philippines. PLoS Medicine 3(9): e352.
WHO - World Health Organization (2016) Global tuberculosis report. Available from: http://www.who.int/tb/publications/global_report/en/ [Consulted June 12, 2017].
WHO - World Health Organization (2009) The economics of managing multidrug-resistant tuberculosis in countries of the former Soviet Union. Geneva: WHO.
Yazbeck A (2001) Making Costing Fun! PowerPoint presentation at the session entitled “How Much Does it Cost?” in the WBI’s Adjusting to Change Learning Program Core Course on Population, Reproductive Health and Health Sector Reform. World Bank, Washington, USA.
Additional information
CONFLICT OF INTEREST: The authors declare no conflict of interest.
Citation Format: Tran QV, Le PH, Ngo NHY, Vo NX, Vo TQ (2018) An updated literature review concerning the treatment cost of
multidrug-resistant tuberculosis. J
Pharm Pharmacogn Res 6(2): 117–125.
Alternative link
http://jppres.com/jppres/pdf/vol6/jppres17.305_6.2.117.pdf (pdf)
http://jppres.com/jppres/systematic-review-on-treatment-cost-of-multidrug-resistant-tuberculosis/ (html)
