Everything is chemistry: Challenges for a sustainable future

Esta obra está bajo una Licencia Creative Commons Atribución-NoComercial-SinDerivar 4.0 Internacional.
Recepción: 27 Julio 2023
Aprobación: 08 Marzo 2024
DOI: https://doi.org/10.7203/metode.15.27181
Abstract: Copper is one of the key metals required for the digital and energy transition and so its demand will increase in the coming years. However, primary extraction poses increasing environmental problems because of the progressive decline in the mineral concentration of copper deposits (ore grade). In this context, electronic waste is becoming a very promising source of secondary copper. However, this form of copper recovery presents a number of technological and chemical challenges, including the use of renewable energy, separation of plastics from other waste, and increasing process efficiency. Given the thermodynamic limitations of these processes, other non-technological factors are becoming very important in the transition.
Keywords: energy transition, circular economy, recycling, secondary resources, copper.
Industrial civilisation is at a crossroads. After more than two centuries of running on fossil fuels, the urgency of the climate crisis is prompting a shift towards cleaner energy. More and more renewable energy sources (mainly solar panels and wind turbines) are being deployed, while new transmission lines and the introduction of electric vehicles is also simultaneously accelerating (Deetman et al., 2021). Nonetheless, these technologies are not without environmental and social impacts. There are several potential problems in areas in which these technologies are installed, including occupation of fertile land, impact on fauna, and opposition from local populations, generally because of a lack of adequate planning. However, the footprint of energy transition technologies goes beyond this; it begins with the activity upon which all industrial companies are based: mining.
Several reports have warned of the amount of minerals needed to make the energy transition, which could increase annual demand for some metals by a factor of 15 (Gregoir & van Acker, 2022; International Energy Agency, 2021). Among these, copper (Cu) stands out because of its ubiquitous use and the difficulty of its substitution. By way of illustration, the production of photovoltaic panels requires around four tonnes of copper per megawatt (MW), wind turbines between one and five tonnes depend on copper-based technology (Carrara et al., 2020), and electric vehicles use up to four times more copper than internal combustion engine vehicles (International Copper Study Group, 2023).
Furthermore, copper is also a key metal for digitalisation: its content in electronic equipment varies between 3–10 % and can reach up to 30 % in printed circuit boards (PCBs) (Torrubia et al., 2022). In addition to the energy and digital sectors, copper is also essential in other sectors of the economy, accounting for 60–75 % of global consumption (Gregoir & van Acker, 2022; International Energy Agency, 2021). The annual demand for copper is therefore rising sharply and by 2050, it could be 30 million tonnes higher than in 2019 (Hund et al., 2020). If these projections are realised, we would need 550 million tonnes of copper in the next 25 years, as much as has been mined since the activity began more than 7,000 years ago (Tabelin et al., 2021).
Primary copper mining
The trends described in the previous section are not new, but the energy and digital transition may accentuate them. Copper mining has been on an upward trend for the past 120 years, with an average annual growth rate of 3.8 %. So much so that in the first 20 years of the 21st century, humans mined as much copper as in the entire 20th century. By 2020, we had already mined 21 million tonnes. These mining trends have led to a steady decline in ore grade, i.e., the concentration of ore in the mined deposits. Whereas at the beginning of the 20th century copper mines could have a copper concentration of up to 2.5 %, by 2015 this had fallen to 0.6 % (Figure 1). This situation meant that between 2001 and 2014, 80 % of new copper reserves came from the reclassification of old ores previously considered unusable (Mills, 2022).
The decrease in ore grade has also led to an exponential increase in the energy consumption of mining operations (Calvo et al., 2016). Moreover, this consumption is highly dependent on fossil fuels, with an average carbon footprint of 5 kg of CO2 per kg of copper extracted – although the values can reach peaks of up to 65 kg of CO2 per kg of copper, depending on the technologies used and the refinery location (Torrubia, Valero, & Valero, 2023). Therefore, the increasing demand for copper and its increasing environmental impact highlight the importance of developing secondary and more sustainable production through recycling.
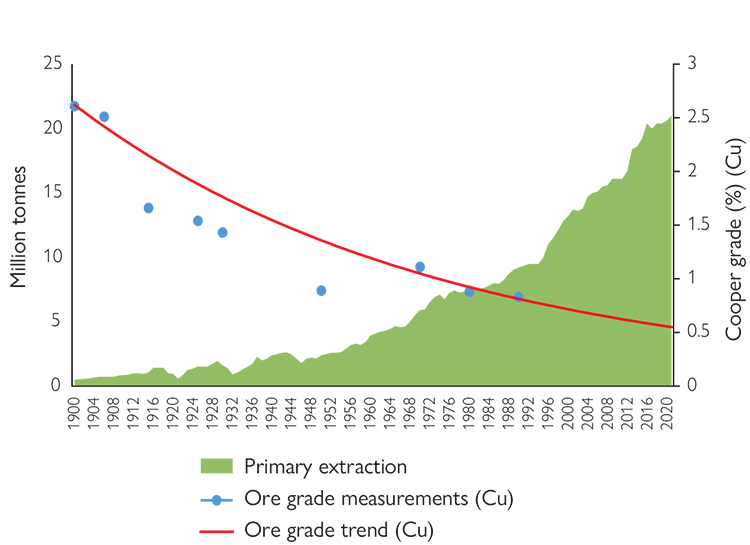
Figure 1
Primary copper extraction (in million tonnes) and the decline in ore grade (as a percentage concentration) between 1900 and 2020.
The blue dots indicate the ore grade (concentration of ore in the deposit) of various deposits in Australia over the 20th century; the red line shows the downward trend. Created by the authors based on data from the US Geological Survey (USGS) and Van der Voet et al. (2019).
Alternative sources and recovery options
While primary copper mining will continue to be necessary (Hund et al., 2020), increasing its recycling rate can limit its environmental impact in countries in which it is mined, thereby conserving resources for future generations and reducing the consumption of fossil fuels required to obtain copper.
Indeed, secondary copper resources are vast. The amount of available scrap metal was estimated at around 250 million tonnes in 2005, with a clear upward trend, and this figure is now likely to be even higher (Figure 2). This means that approximately 30 % of current copper demand could be met from secondary sources (Loibl & Tercero Espinoza, 2021).
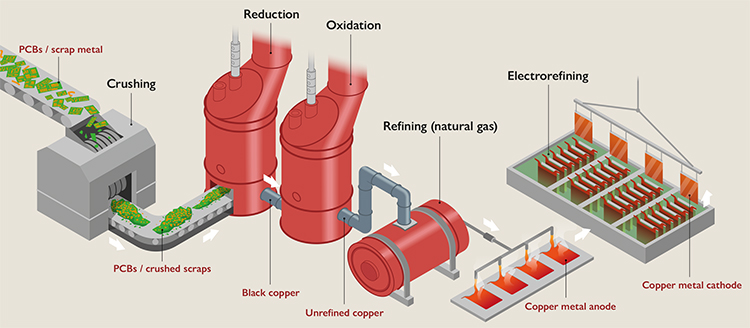
Figure 2
Simplified diagram of the processes required to recover copper from electronic waste.
The copper-rich waste is first crushed and then subjected to a pyrometallurgical (reduction, oxidation, and natural gas refining) and hydrometallurgical (electrorefining) processes to obtain a high-purity copper cathode. Adapted from Torrubia et al. (2024).
Xavi SepúlvedaIn fact, the waste electrical and electronic equipment (WEEE) generated worldwide between 1980 and 2010 represents the largest group of copper-containing waste and has become one of the most promising secondary sources of copper. For example, its availability is very high in Europe, where about 16.2 kg of such waste is generated per person per year (Forti et al., 2020). One of the reasons for this is the short life cycle, of an average of two to 10 years, of many products (Torrubia, Valero, Valero, & Lejuez, 2023). This has made copper the fastest-growing type of household waste, with a global annual increase of 3–5 % (Valero et al., 2021).
WEEE contains copper in various components, mainly in cables, alloys, and PCBs, although the concentration in each of these components varies. For example, cables are virtually pure copper. Conversely, alloys contain 60–90 % copper, while the content of PCBs is very variable, ranging from 5–65 % (Torrubia et al., 2022). According to the ProSUMs project (Huisman et al., 2017), between the year 2000 and 2020, about 7.5 million tonnes of cables, 760,000 tonnes of brass, 450,000 tonnes of bronze, 4.5 million tonnes of other copper alloys, and about 4.4 million tonnes of PCBs went onto the European market in the form of WEEE alone.
Industrial recovery of copper from waste electrical and electronic equipment
The most common technology for copper recovery follows the pyrometallurgical route, by using heat to refine metals. However, the processes differs depending on the concentration of copper, which varies according to the component in which the metal is embedded. Of the 8.72 million tonnes of copper recycled in 2018, 5.43 million tonnes were recovered by direct smelting. However, this route is only possible when the copper has very few impurities, such as in the case of cables. The remaining 3.29 million tonnes require more complex pyrometallurgical processes to extract the copper because of the higher chemical complexity of alloys and PCBs (Loibl & Tercero Espinoza, 2021).
Pyrometallurgical processes are more efficient than hydrometallurgical processes but require higher capital investment and have high operating costs, especially to avoid the emission of toxic products. They are therefore used by large companies traditionally involved in primary copper production, such as Boliden (Sweden), Umicore (Belgium), Aurubis (Germany), Xstrata (Canada), and Dowa (Japan). Furthermore, in the case of copper recovery from PCBs, these processes emit dioxins and furans, highly toxic products resulting from the combustion of the plastics contained in the boards. Therefore, scrubbers must be installed to limit the emission of these gasses. However, such equipment is very expensive and requires large capital investments. For example, the Umicore plant in Antwerp invested more than €200 million in gas treatment equipment (Valero Navazo et al., 2014).
The most common pyrometallurgical configuration is called Reverse Knudsen (Figure 2), which consists of a reduction stage, because copper scrap is usually highly oxidised, followed by an oxidation stage to remove the remaining impurities. In both processes, coke (from coal) is usually used as a reducing agent and a fuel, because the furnaces must be kept at a temperature of about 1,300 °C. The product of this oxidation is called black copper and has a copper concentration of about 85 %. This intermediate product is then refined using natural gas as a fuel (fire refining) to remove the remaining oxygen and sulphur impurities. Finally, the copper anode produced in the previous process is cooled and electrorefined to obtain a high-purity copper cathode (99.99 % copper). During this last stage, the main inputs are electricity and sulphuric acid, which are essential for the process (Torrubia et al., 2024).
Interestingly, after electrorefining (when PCBs are fed into the process), certain amounts of precious metals such as gold, silver, platinum, or palladium can also be found in the sludge because these metals are embedded in the PCBs. While their quantities are not negligible – they can represent up to 70 % of the price of the metals on PCBs (Torrubia et al., 2024) – further processes, usually hydrometallurgical, are required to recover them.
Table 1 shows the consumption of the described copper recycling process from scrap, PCBs, or a mixture of both waste streams. The energy requirements shown in the table range from 2.9 to 33.8 GJ per tonne of recycled copper, which is between 42 % and 95 % less compared to the energy that would be required for primary extraction, 58 GJ per tonne of copper (Torrubia, Valero, & Valero, 2023). However, despite this reduction, most of the energy consumption of this recycling process still comes from the use of fossil fuels. The coke reduction, oxidation, and natural gas refining stages consume 62–96 % of this energy, while the remaining energy is required by the electrorefining process in the form of electricity.
Thus, despite the reduced use of fossil fuels in recycling, the dependency continues. This raises the question: to what extent can these recycling processes be considered part of a truly circular economy when their main inputs are non-renewable and therefore cannot be recycled, and they are irreversibly consumed in the process? Thus, we might wonder whether if it is possible to minimise our dependence on fossil fuels by using renewable energy.
Energy consumption and emissions of secondary copper recycling processes (Torrubia et al., 2024).
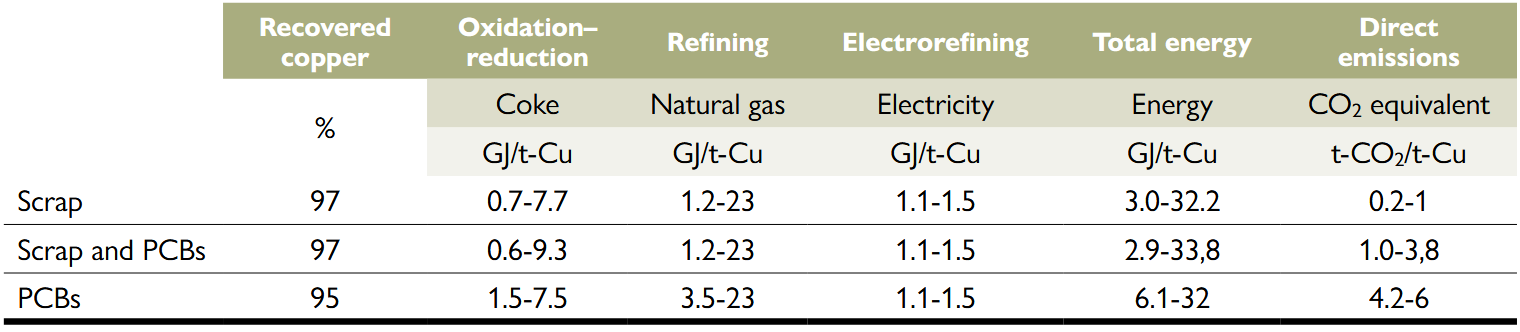
PCB stands for printed circuit board. The starting products in the recycling process are listed from lowest to highest CO2 emissions. Note: The percentages indicate the amount of copper recovered in relation to the copper input. Direct emissions refer only to the combustion of coke and natural gas, not to emissions related to electricity generation.
Use of renewable energy in copper recovery
The use of renewable energy is important not only to decarbonise and reduce CO2 emissions, but also to move towards a truly circular economy. As we have seen, although copper recovery from waste has two advantages – it does not involve primary extraction and it uses less fossil energy resources – it still depends on fossil fuels. Therefore, in our study (Torrubia et al., 2024) we simulated the substitution of coke and natural gas by hydrogen (Table 2).
Despite a slight increase in energy consumption in some cases (up to 5 GJ more per tonne of recycled copper), the energy can be supplied entirely by renewable energy as it is possible to produce renewable hydrogen, also called green hydrogen, by electrolysis of water using electricity from renewable energy sources. In this way, the entire energy consumption of the process could be carried out without the direct use of fossil fuels. However, Table 2 still shows that in the case of PCBs there are direct CO2 emissions (from 0.8 to 3.8 tonnes of CO2). These emissions are caused when the plastics contained in WEEEs are burned. Therefore, the only way to avoid these emissions would be to separate the plastics before introducing them into the pyrometallurgical process. Nevertheless, the combustion of plastics makes it possible to dispense with the use of fuels (such as coke or hydrogen) during reduction and oxidation, thereby making it more attractive for the recycling industry from an economic point of view (Valero Navazo et al., 2014).
Energy consumption and emissions from secondary copper recycling by replacing coke and natural gas with hydrogen (Torrubia et al., 2024).
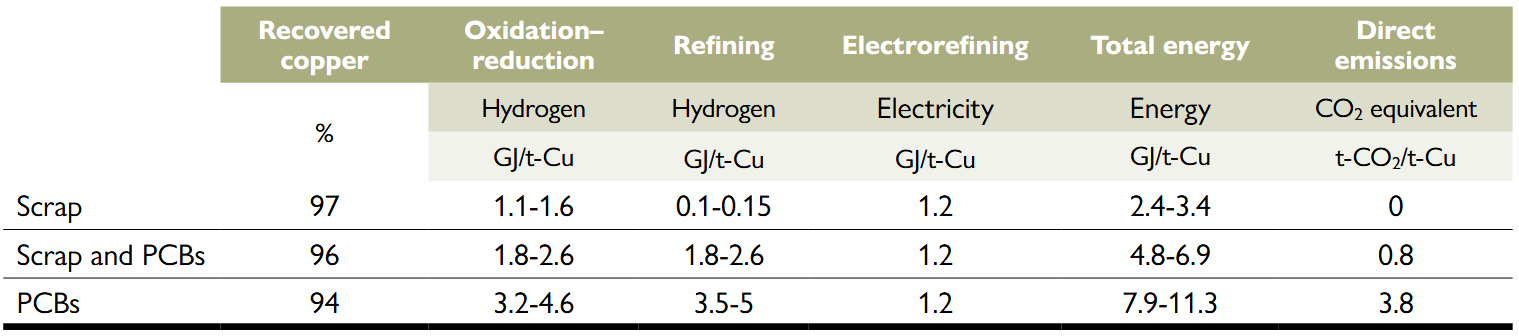
PCB stands for printed circuit board. Note: The percentages indicate the amount of copper recovered in relation to the copper input. Direct emissions refer only to emissions due to combustion of plastics embodied in PCBs, not to emissions related to electricity production.
Challenges for the circular copper economy
There are therefore three technical challenges that could be addressed to improve the circular copper economy. Firstly, the use of fossil fuels should be replaced by green hydrogen and electricity from renewable sources should be used in recycling processes. Secondly, to limit CO2 emissions when recovering copper from PCBs, the plastics must be separated beforehand. However, this practice is not common because plastics can be used as a fuel, thus contributing to cost savings. Therefore, this step, although desirable from the point of view of circularity, could reduce economic viability. Finally, 3 to 6 % of the copper is lost in the slag, along with other valuable metals such as tin, zinc, and precious or critical metals. It is therefore important to design new processes to recover as many metals as possible, not just the most economically valuable ones. In this sense, the role of chemistry is fundamental in maximising not only the percentage of metal recovered, but also the range of metals, in order to achieve maximum efficiency.
In conclusion, although it is technically impossible to achieve a truly circular economy (because of the chemical and thermodynamic limits outlined above), we must strive to get closer to this ideal in order to reduce environmental impacts. As a result, non-technological factors are very important and so measures such as reducing the demand for metals (prioritising more essential uses), designing products for subsequent repair and recycling, extending their useful lifespans, or improving collection channels are also essential to achieving the objectives of the energy and digital transition.
References
Calvo, G., Mudd, G., Valero, A., & Valero, A. (2016). Decreasing ore grades in global metallic mining: A theoretical issue or a global reality? Resources, 5(4), 36. https://doi.org/10.3390/resources5040036
Carrara, S., Alves Dias, P., Plazzotta, B., & Pavel, C. (2020). Raw materials demand for wind and solar PV technologies in the transition towards a decarbonised energy system. Publications Office of the European Union. https://doi.org/10.2760/160859
Deetman, S., de Boer, H. S., Van Engelenburg, M., van der Voet, E., & van Vuuren, D. P. (2021). Projected material requirements for the global electricity infrastructure – generation, transmission and storage. Resources, Conservation and Recycling, 164, 105200. https://doi.org/10.1016/j.resconrec.2020.105200
Forti, V., Baldé, C. P., Kuehr, R., & Bel, G. (2020). The global e-waste monitor 2020: Quantities, flows, and the circular economy potential. United Nations University (UNU), United Nations Institute for Training and Research (UNITAR), International Telecommunication Union (ITU) & International Solid Waste Association (ISWA).
Gregoir, L., & van Acker, K. (2022). Metals for clean energy: Pathways to solving Europe’s raw materials challenge. KU Leuven. https://eurometaux.eu/media/rqocjybv/metals-for-clean-energy-final.pdf
Huisman, J., Leroy, P., Tertre, F., Ljunggren Söderman, M., Chancerel, P., Cassard, D., Løvik, A. N., Wäger, P., Kushnir, D., Susanne Rotter, V., Mählitz, P., Herreras, L., Emmerich, J., Hallberg, A., Habib, H., & Wagner, M. (2017). ProSUM final report: Prospecting secondary raw materials in the urban mine and mining wastes. ProSUM. https://prosumproject.eu/sites/default/files/DIGITAL_Final_Report.pdf
Hund, K., La Porta, D., Fabregas, T. P., Laing, T., & Drexhage, J. (2020). Minerals for climate action: The mineral intensity of the clean energy transition. World Bank. https://pubdocs.worldbank.org/en/961711588875536384/Minerals-for-Climate-Action-The-Mineral-Intensity-of-the-Clean-Energy-Transition.pdf
International Copper Study Group. (2023). The world copper factbook. ICSG. https://icsg.org/copper-factbook
International Energy Agency. (2021). The role of critical minerals in clean energy transitions. IEA. https://www.iea.org/reports/the-role-ofcritical-minerals-in-clean-energy-transitions
Loibl, A., & Tercero Espinoza, L. A. (2021). Current challenges in copper recycling: Aligning insights from material flow analysis with technological research developments and industry issues in Europe and North America. Resources, Conservation and Recycling, 169, 105462. https://doi.org/10.1016/j.resconrec.2021.105462
Mills, R. (2022, 27 October). Copper: The most important metal we are running short of. Ahead of the herd. https://aheadoftheherd.com/copperthe-most-important-metal-were-running-short-of-richard-mills/
Tabelin, C. B., Park, I., Phengsaart, T., Jeon, S., Villacorte-Tabelin, M., Alonzo, D., Yoo, K., Ito, M., & Hiroyoshi, N. (2021). Copper and critical metals production from porphyry ores and e-wastes: A review of resource availability, processing/recycling challenges, socio-environmental aspects, and sustainability issues. Resources, Conservation and Recycling, 170, 105610. https://doi.org/10.1016/j.resconrec.2021.105610
Torrubia, J., Parvez, A. M., Sajjad, M., Paz, F. a. G., & Van Den Boogaart, K. G. (2024). Recovery of copper from electronic waste: An energy transition approach to decarbonise the industry. Journal of Cleaner Production, 485, 144349. https://doi.org/10.1016/j.jclepro.2024.144349
Torrubia, J., Valero, A., & Valero, A. (2022). Thermodynamic rarity assessment of mobile phone PCBs: A physical criticality indicator in times of shortage. Entropy, 24(1), 100. https://doi.org/10.3390/e24010100
Torrubia, J., Valero, A., & Valero, A. (2023). Energy and carbon footprint of metals through physical allocation. Implications for energy transition. Resources, Conservation and Recycling, 199, 107281. https://doi.org/10.1016/j.resconrec.2023.107281
Torrubia, J., Valero, A., Valero, A., & Lejuez, A. (2023). Challenges and opportunities for the recovery of critical raw materials from electronic waste: The Spanish perspective. Sustainability, 15(2), 1393. https://doi.org/10.3390/su15021393
Valero, A., Torrubia, J., Anía, M. Á., & Torres, A. (2021). Assessing urban metabolism through MSW carbon footprint and conceptualizing municipal-industrial symbiosis—the case of Zaragoza city, Spain. Sustainability, 13(22), 12724. https://doi.org/10.3390/su132212724
Valero Navazo, J. M., Villalba Méndez, G., & Talents Peiró, L. (2014). Material flow analysis and energy requirements of mobile phone material recovery processes. The International Journal of Life Cycle Assessment, 19, 567–579. https://doi.org/10.1007/s11367-013-0653-6
Van der Voet, E., Van Oers, L., Verboon, M., & Kuipers, K. (2019). Environmental implications of future demand scenarios for metals: Methodology and application to the case of seven major metals. Journal of Industrial Ecology, 23(1), 141–155. https://doi.org/10.1111/jiec.12722
Notas de autor
jtorrubia@unizar.es
Información adicional
redalyc-journal-id: 5117
