Everything is chemistry: Challenges for a sustainable future
Chemistry for next-generation diagnostics: Key factors in the development of medical biosensors
Chemistry for next-generation diagnostics: Key factors in the development of medical biosensors
Mètode Science Studies Journal, vol. 15, núm. 2, e27225, 2025
Universitat de València

Esta obra está bajo una Licencia Creative Commons Atribución-NoComercial-SinDerivar 4.0 Internacional.
Recepción: 04 Agosto 2023
Aprobación: 16 Noviembre 2023
Abstract: Biosensors have been profiled as potential next-generation diagnostic technologies, offering excellent clinical performance, wide versatility, and integration in miniaturized devices for on-site and portable analysis. But the sensor biofunctionalization, the way bioreceptors are immobilized on the sensor chip, is still an unresolved challenge that demands for specific research in surface chemistry strategies and the use of novel nanomaterials. We provide a brief overview of the key factors driving the improvement of medical biosensors, with a special focus on the current limitations in sensor surface modification and the direct analysis of human samples. We conclude the successful implementation of cutting-edge diagnostic biosensors will only be possible through the collaborative synergy of different sciences, including physics, biology, engineering, and certainly chemistry.
Keywords: sensor functionalization, label-free biosensors, surface chemistry, nanotechnology, clinical diagnostics.
Biosensors as medical technologies
In the rapidly advancing field of healthcare, biosensors have emerged as revolutionary tools poised to transform medical diagnostics and clinical monitoring. These technologies, capable of detecting and analyzing biological information, offer unprecedented opportunities for personalized and precision medicine, paving the way for timely disease diagnosis and personalized treatment interventions that improve the overall patient outcomes and well-being. In the context of public health emergencies, the access to quick biosensors can be crucial for an effective population screening and early detection of infectious agents, aiding in the transmission chain control. Furthermore, the use of affordable and portable diagnostic biosensors in low-resource locations with limited access to clinical equipment, accounting for the 47% of the world population, could ultimately bridge the gap between underserved communities and quality healthcare. But despite these promising advantages and potential, and after decades of research, the full adoption of these biosensors still remains a challenge where chemistry could serve as a driving force and solution-provider.
A biosensor is a compact analytical device that combines biological components, such as enzymes, antibodies, or nucleic acids, with a physical transducer to detect and quantify specific biological or chemical analytes (the substance or component we are trying to measure) in a sample, including proteins, pathogens, drugs, toxins, or genomic markers. The biological component acts as a selective recognition element, capturing and interacting with the analyte of interest. This interaction induces a series of physicochemical changes in the transducer, which can be electrical, optical, thermal, or mechanical in nature, and are readily converted into a measurable signal. Among the different types of transducers, electrochemical and optical sensors are the most employed for medical applications, mainly due to their high sensitivity, versatility, label-free analysis capability, and their miniaturization and integration potential (Figure 1) (Altug et al., 2022; Kim et al., 2021).

Figure 1
A biosensor is a compact analytical device that combines biological components, with a physical transducer to detect and quantify specific biological or chemical analytes in a sample, including proteins, pathogens, drugs, toxins, or genomic markers. The biological component acts as a selective recognition element, capturing and interacting with the analyte of interest. In the picture, an optical interferometric biosensor integrated in a hand-held cartridge, designed to detect bacteria in the blood of patients with sepsis.
M. Soler / L. M. LechugaIn fact, the first biosensor fully implemented in clinics was the well-known glucometer, an electrochemical biosensor proposed by Clark and Lyons in 1962 for the rapid measurement of glucose levels in blood (Jing et al., 2022). This biosensor employs the glucose oxidase (GOx) enzyme as receptor immobilized on a working electrode. Upon interaction with glucose, the enzyme releases gluconic acid and hydrogen peroxide (H2O2), which is immediately oxidized at the electrode generating a measurable current directly related to the glucose concentration. The commercialization of the glucose biosensor in the late 1970s allowed, for the first time, a frequent and less invasive monitoring of blood sugar levels for diabetic patients, reducing the risks and potential complications of the disease, boosting their quality of life, and empowering the patients to become more involved in their own care.
Another interesting application of electrochemical transducer technologies are the ion-sensitive field-effect transistor (ISFET) sensors, with a working principle similar to glucose sensors and widely employed for blood gas analysis and pH monitoring in critical care settings.
On the other hand, the most popular and well-known optical biosensor is the lateral-flow assay (LFA) test. These devices have been broadly used by society for decades, for example for pregnancy detection and, more recently, for self-diagnosis of COVID-19 (Figure 2). LFAs are essentially based on the optical properties of nanoparticles conjugated to specific antibodies, which in presence of the target molecule, are immobilized on the test line, forming a visible signal (colored line).
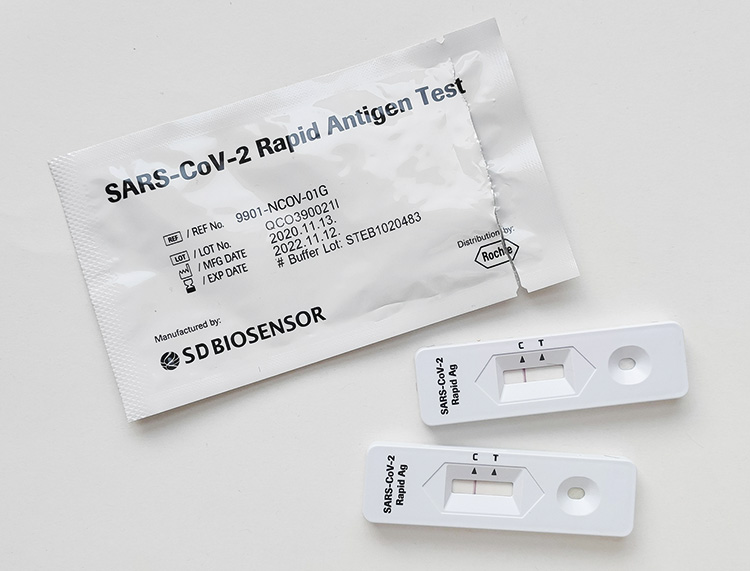
Figure 2
One of the most popular and well-known optical biosensors is the lateral-flow assay (LFA) test. These devices have been broadly used by the society for decades, for example for pregnancy detection and, more recently, for self-diagnosis of COVID-19.
Steve Nomax / UnsplashNonetheless, despite the simplicity, affordability, and suitability of LFAs for many rapid diagnosis applications, they still suffer certain limitations in terms of sensitivity, multiplexing (simultaneous detection of several analytes), and specific quantification of the analyte levels. To that, a different type of optical biosensors, based on label-free analysis (without fluorescent compounds or changes in colour), were proposed as a powerful technique for the detection, study, and monitoring of biomolecular interactions. It is the case, for example, of Surface Plasmon Resonance (SPR, light waves generated on the surface of certain metals) biosensors, commercialized since the 1990s and routinely employed today by pharmaceutical or biomedical laboratories worldwide (Soler & Lechuga, 2021). To date, SPR instruments are sold as bare analytical equipment with gold sensor chips ready to be functionalized with a desired recognition element, commonly an antibody, that will interact with the target analyte (e.g., protein, small-molecule), producing a variation in one of the light properties, such as the angle of incidence, the reflected intensity, or the wavelength of the plasmonic resonance peak position. By interrogating this signal in real time, SPR sensorgrams provide accurate information about the affinity and kinetic parameters of the biomolecular interaction, which is primarily relevant in the design and development of drug candidates.
Amid the rise of nanotechnology and microelectronics technologies, biosensors have seen a boost in miniaturization and integration, expanded their capability for multiplexing (simultaneous detection of different analytes), and greatly enhanced their sensitivity. For instance, glucose biosensors have terrifically evolved until being fully integrated in wearable devices for continuous glucose levels monitoring with highly accurate performance (Figure 3), wireless connection and customized alerts, or even automated treatment administration (Saha et al., 2023). Likewise, plasmonic sensors incorporating nanostructures (nanoslits, nanodisks, nanoholes, etc.) in their transducers can avoid the need of complex optical-coupling schemes, allowing for their integration in handheld devices or even as accessories for common smartphones (Lopez et al., 2017).
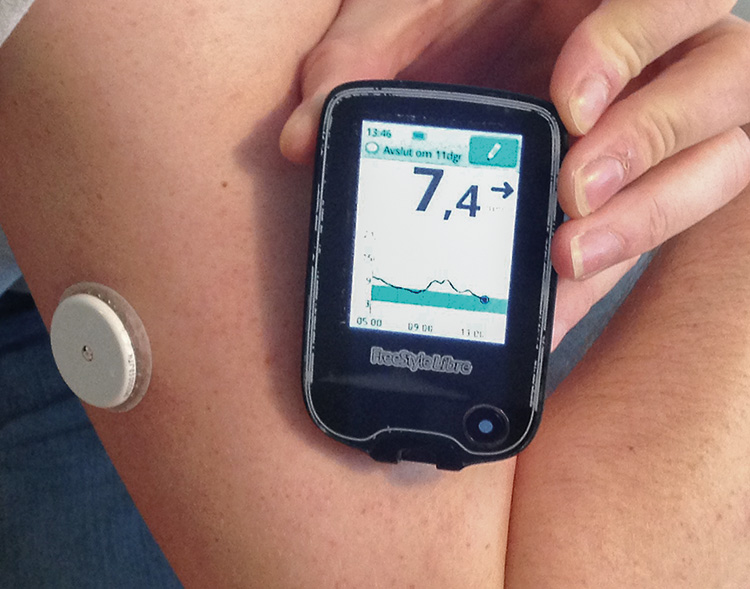
Figure 3
Amid the rise of nanotechnology and microelectronics technologies, biosensors have seen a boost in miniaturization and integration. For instance, glucose biosensors have terrifically evolved until being fully integrated in wearable devices for continuous glucose levels monitoring with highly accurate performance, wireless connection and customized alerts, or even automated treatment administration.
Sjö / WikipediaThese technological advances also promote the expansion to more complex medical biosensor applications, such as early cancer detection, identification of viral and bacterial infection, or the individualized monitoring of therapies, ultimately attempting a point-of-care precision diagnosis and personalized medicine. Indeed, during the last decade, research in biosensors has vastly demonstrated their unique value as medical technologies, reporting ultrasensitive and multiplexed detection of novel tumor biomarkers (e.g., microRNAs, autoantibodies, or epigenomic alterations), on-site detection of infectious viruses and bacteria, rapid analysis and determination of antibiotic resistance, or non-invasive control of life-long treatments (Cesewski & Johnson, 2020; Sadighbayan et al., 2019; Soler et al., 2019; Yoo & Lee, 2016).
However, although excellent results are achieved in academic laboratories – often equivalent or superior to the traditional analytical techniques – the technology transfer of such biosensors to the clinical practice has not been fully achieved yet. Among others, a major limitation relies on the need of robust sensor chemical biofunctionalization strategies that ensure an optimal and stable bioreceptor immobilization, maximizing the detection efficiency and reproducibility, and the antifouling coating of the sensor surface that prevents and minimizes non-specific adsorptions and interferences coming from the complex biological samples. And herein, surface chemistry plays a critical and fundamental role.
The role of surface chemistry
Sensor biofunctionalization is a key process for achieving sensitive, selective, and robust biosensors, especially for those working in a label-free scheme, where the assay specificity and detection efficiency greatly depends on the biorecognition layer coating the transducer. Three main factors should be considered for anchoring the biorecognition elements onto the sensor surface: the receptor orientation, exposing the analyte binding sites to the sample; the grafting density, minimizing steric hindrance effects for analyte capture; and the immobilization stability, ensuring reliable and reproducible assays with different sensor chips.
Surface chemistry is indisputably the best approach to firmly attach the bioreceptor molecules to the sensor surface, but there are still challenges to be addressed, especially for the functionalization of certain sensor materials (Figure 4) (Soler & Lechuga, 2022). In particular, for medical biosensors, the most common sensor materials are gold, carbon, and silicon. Gold transducers are widely employed for both plasmonic sensors and for working electrodes in electrochemical sensors, due to their excellent electrical conductivity, chemical stability, and optical properties (e.g., plasmonic resonances in the visible range).

Figure 4
Surface chemistry is indisputably the best approach to firmly attach the bioreceptor molecules to the sensor surface, but there are still challenges to be addressed, especially for the functionalization of certain sensor materials. Above, illustration of an interferometric sensor functionalized with DNA strands (pink) and antibodies (green), allowing the simultaneous detection of different cancer markers (genomic and protein). Inset figure represents the chemical modification of the silicon-based sensor surface through silanization. The silanization method allows for coating an hydroxil-activated material with a monolayer of organic molecules (silanes) carrying specific terminal functional groups that can be employed for covalently anchoring different biomolecules.
M. Soler / L. M. LechugaGold can be readily modified by chemisorption of thiol (SH)-functional molecules, a rapid and efficient reaction that creates strong and stable covalent bonds. The simplest approach is to directly immobilize thiolated receptors, such as thiol-functional DNA probes or antibody fragments. However, for antibodies and other protein receptors, the commonest procedure is the formation of functional self-assembled monolayers (SAMs) for subsequent crosslinking of the native biomolecule or affinity linkers (e.g., streptavidin, protein A/G) that might help in orientation. The formation of SAMs is also a well-established procedure that typically utilizes short-chain alkanethiols bearing a functional end-group (COOH, NH2, OH, etc.) that spontaneously attach on gold by chemisorption creating a highly ordered and dense matrix. Furthermore, SAMs can be formed by mixing different functionalities at the desired ratio, offering good control of the biomolecule grafting density. In this regard, some companies have specialized in the production of chemical compounds for SAM formation, including different chain lengths and reactive groups, to facilitate customized sensor surface functionalization. Also, the commercial availability of ready-to-use biomolecule conjugation kits (e.g., biotinylation kits) can greatly simplify the bioreceptor immobilization procedure, enhancing the reliability, reproducibility and scalability for industrialization.
Unfortunately, the chemical functionalization of sensor transducers based on carbon or silicon is more complex and challenging. Carbon-based materials, like carbon nanotubes or graphene, are widely employed for electrochemical sensors because of their excellent electrical conductivity and fast electron transfer kinetics, high surface area, chemical stability and mechanical strength. The chemical functionalization of these carbon-based electrodes usually relies on the oxidation of the carbon structure to introduce carboxylic or epoxy groups, which will be crosslinked to the biomolecule. But this oxidation process is highly irregular, limiting the formation of an ordered biorecognition layer with optimum grafting density, and can also potentially damage the native properties of the carbon structure, leading to a decrease in the electrical conductivity and therefore worsening the analytical performance of the biosensor.
On the other hand, silicon and silicon-derived materials, like silicon oxide (SiO2) or silicon nitride (Si3N4), are utilized as a substrate material for nanophotonic waveguide-based biosensors due to their excellent optical properties (e.g., low propagation losses, high refractive index contrast, broad wavelength working range, etc.) and, importantly, the compatibility with standard microelectronic component production techniques, which are essential for the cost-effective fabrication of integrated biosensor devices. Indeed, these microchip-based biosensors are envisioned as the future diagnostic devices with impressive capabilities in terms of sensitivity and miniaturization, but still a few hurdles must be resolved. Silicon-based surfaces are modified by the so-called silanization procedure that basically consists in creating a functional SAM – similar to alkanethiol SAMs on gold – through covalent binding of organosilanes to reactive hydroxyl groups released on the silicon surface. Although silanization is a vastly used technique in various fields, it is far from being standardized and highly depends on the silicon substrate characteristics, the type of organosilane and its chemical properties, and the specific environment conditions and parameters employed during the reaction. In fact, nowadays there is no consensus on the optimum parameters and protocols to achieve a stable and homogeneous silane monolayer for silicon-based sensor functionalization, so it is a process that needs to be optimized for each sensor and application purpose.
Hence, considering that both carbon and silicon-based sensors are profiled as leading-edge technologies for next-generation medical diagnostics, research endeavors should prioritize improving, optimizing, and establishing standard sensor chemistry methods for both of them. These efforts may aim not only to attain the utmost analysis accuracy and reliability but also facilitate scalability and industrialization of ready-to-use biosensors for point-of-care applications.
Towards whole blood analysis
As previously mentioned, the ultimate goal of medical biosensors is to perform an accurate diagnostic analysis directly from the human sample, eliminating laboratory treatment and processing steps to enable rapid, decentralized, and user-friendly operation. Most common diagnostic samples include blood and urine, although saliva, mucosal fluids, cerebrospinal fluids, or even tears or sweat are also target specimens for specific disease diagnostics. All these biological fluids contain large amounts of proteins, cells, and other compounds that can get adsorbed on the sensor surface during the assay interfering with the analysis, either producing a false-positive signal or hampering the biorecognition interaction. To prevent, avoid, or minimize these undesired matrix effects, sensor surfaces should be coated with antifouling compounds, capable of repelling the nonspecific adsorption of molecules while guaranteeing an efficient analyte detection.
Since nonspecific matrix adsorptions are mostly governed by electrostatic and Van der Waals interactions, the most efficient mechanisms to repel them consist in increasing the surface hydrophilicity, generating a hydration layer (an organized layer of water molecules at the sensor interface), as well as altering the effective surface charges of the functional chemical scaffold to reduce the ionic interactions (Soler & Lechuga, 2022). A popular approach is to use polyethylene glycol (PEG) compounds, such as thiol-PEG-R or silane-PEG-R (where R is the functional reactive group), for the monolayer formation, which are known to offer a high hydrophilicity that attracts water molecules, increasing the resistance to protein fouling.
This strategy can work well for urine or semi-processed samples, with a relatively low content on proteins, but it results insufficient for attempting direct evaluation of serum, plasma, or other complex matrices. Better antifouling properties have been achieved with zwitterionic layers, formed by polymers containing both cationic and anionic groups that provide a charge balance on the sensor surface. Besides, zwitterionic compounds also attract water molecules via hydrogen bonds and ionic solvation, creating a hydration layer more stable than PEG derivatives. However, the direct use of current zwitterionic polymers as functional scaffolds for bioreceptor immobilization is not recommended due to the low control on the bioreceptor positioning and grafting density, the high probability of hindering the biorecognition interaction due to the large polymer brushes, and the frequent incompatibility with a strong chemical attachment to the sensor surface.
This unresolved challenge therefore opens a new avenue for surface chemistry and materials researchers to develop innovative surface coatings with a high antifouling capacity but ensuring a highly stable anchoring to the sensor surface and an aligned exposure of functional groups for bioreceptor crosslinking and immobilization.
Novel 2D materials and supramolecular chemistry
By addressing two main limitations of current biosensors, the standardization of transducer functionalization protocols and the design of novel antifouling materials for an effective reduction of nonspecific adsorptions, both organic and inorganic chemistry research may play a crucial role. But furthermore, the emerging fields of 2D materials and supramolecular chemistry can significantly provide fresh and attractive opportunities for biosensor applications and clinical implementation.
For example, interesting graphene nanostructures, such as nanoporous graphene (NPG) or functional graphene nanoribbons (GNR), have been recently synthesized with atomic precision incorporating specific reactive groups (NH2, COOH, OH, etc.) with controlled distribution as well as enhancing the electrochemical and mechanical properties of conventional graphene (Moreno et al., 2018). The use of this novel material as sensing transducers could solve many challenges in electrochemical carbon-based biosensor technologies, by providing both a highly efficient sensor electrodes and highly-organized scaffold for bioreceptor immobilization. Besides, certain graphene nanostructures have also showed exceptional photonic properties that could be exploited for optical sensing (Grigorenko et al., 2012).
Other pioneering and fascinating materials are the metal-organic frameworks (MOFs) and their derivatives, the covalent-organic frameworks (COFs) and the metal-organic polyhedra (MOPs). These porous materials have shown exceptional properties for capturing specific small molecules, and recently also biomolecules, through mechanisms similar to the lock-and-key technique or host-guest interactions, in which the framework cavities are specifically designed to match with the analyte dimensions, electrostatic and hydrophobic nature (Avci et al., 2017; Wang et al., 2022). The application of these materials as biorecognition elements in biosensor devices could offer new possibilities for the specific detection of certain small molecules, like ions, drugs, or peptides, with higher affinity than traditional antibodies and, importantly, with higher robustness and scalability toward cost-effective mass production.
To conclude, it is important to remark that in the pursuit of designing and implementing novel diagnostic biosensors, interdisciplinary science becomes indispensable, encompassing the fields of biology, physics, engineering, and also chemistry. In particular, surface chemistry holds a huge potential for enhancing the accuracy and reliability of clinical biosensors and for expanding their applicability, but it is, unfortunately, an often-overlooked factor in the harnessing of these new technologies. The claim towards the biosensor community is therefore clear: collaborative science and multidisciplinary research will be the key factor in the success of biosensors as next-generation diagnostics.
References
Altug, H., Oh, S. H., Maier, S. A., & Homola, J. (2022). Advances and applications of nanophotonic biosensors. Nature Nanotechnology, 17(1), 5–16. https://doi.org/10.1038/s41565-021-01045-5
Avci, C., Imaz, I., Carné-Sánchez, A., Pariente, J. A., Tasios, N., Pérez-Carvajal, J., Alonso, M. I., Blanco, A., Dijkstra, M., López, C., & Maspoch, D. (2017). Self-assembly of polyhedral metal–organic framework particles into three-dimensional ordered superstructures. Nature Chemistry, 10(1), 78–84. https://doi.org/10.1038/nchem.2875
Cesewski, E., & Johnson, B. N. (2020). Electrochemical biosensors for pathogen detection. Biosensors and Bioelectronics, 159, 112214. https://doi.org/10.1016/J.BIOS.2020.112214
Grigorenko, A. N., Polini, M., & Novoselov, K. S. (2012). Graphene plasmonics. Nature Photonics, 6(11), 749–758. https://doi.org/10.1038/nphoton.2012.262
Jing, Y., Chang, S. J., Chen, C. J., & Liu, J.-T. (2022). Review—Glucose monitoring sensors: History, principle, and challenges. Journal of the Electrochemical Society, 169(5), 057514. https://doi.org/10.1149/1945-7111/AC6980
Kim, J. H., Suh, Y. J., Park, D., Yim, H., Kim, H., Kim, H. J., Yoon, D. S., & Hwang, K. S. (2021). Technological advances in electrochemical biosensors for the detection of disease biomarkers. Biomedical Engineering Letters, 11(4), 309–334. https://doi.org/10.1007/S13534-021-00204-W
Lopez, G. A., Estevez, M. C., Soler, M., & Lechuga, L. M. (2017). Recent advances in nanoplasmonic biosensors: Applications and lab-on-a-chip integration. Nanophotonics, 6(1), 123–136. https://doi.org/10.1515/nanoph-2016-0101
Moreno, C., Vilas-Varela, M., Kretz, B., Garcia-Lekue, A., Costache, M. V., Paradinas, M., Panighel, M., Ceballos, G., Valenzuela, S. O., Peña, D., & Mugarza, A. (2018). Bottom-up synthesis of multifunctional nanoporous graphene. Science, 360(6385), 199–203. https://doi.org/10.1126/science.aar2009
Sadighbayan, D., Sadighbayan, K., Tohid-kia, M. R., Khosroushahi, A. Y., & Hasanzadeh, M. (2019). Development of electrochemical biosensors for tumor marker determination towards cancer diagnosis: Recent progress. TrAC Trends in Analytical Chemistry, 118, 73–88. https://doi.org/10.1016/J.TRAC.2019.05.014
Saha, T., Del Caño, R., Mahato, K., De la Paz, E., Chen, C., Ding, S., Yin, L., & Wang, J. (2023). Wearable electrochemical glucose sensors in diabetes management: A comprehensive review. Chemical Reviews, 123(12), 7854–7889. https://doi.org/10.1021/acs.chemrev.3C00078
Soler, M., Huertas, C. S., & Lechuga, L. M. (2019). Label-free plasmonic biosensors for point-of-care diagnostics: A review. Expert Review of Molecular Diagnostics, 19(1), 71–81. https://doi.org/10.1080/14737159.2019.1554435
Soler, M., & Lechuga, L. M. (2021). Principles, technologies, and applications of plasmonic biosensors. Journal of Applied Physics, 129(11), 111102. https://doi.org/10.1063/5.0042811
Soler, M., & Lechuga, L. M. (2022). Biochemistry strategies for label-free optical sensor biofunctionalization: Advances towards real applicability. Analytical and Bioanalytical Chemistry, 414, 5071–5085. https://doi.org/10.1007/S00216-021-03751-4
Wang, J., Imaz, I., & Maspoch, D. (2022). Metal−organic frameworks: Why make them small? Small Structures, 3(1), 2100126. https://doi.org/10.1002/SSTR.202100126
Yoo, S. M., & Lee, S. Y. (2016). Optical biosensors for the detection of pathogenic microorganisms. Trends in Biotechnology, 34(1), 7–25. https://doi.org/10.1016/j.tibtech.2015.09.012
Notas de autor
maria.soler@icn2.cat
Información adicional
redalyc-journal-id: 5117