Artículos

Esta obra está bajo una Licencia Creative Commons Atribución-SinDerivar 4.0 Internacional.
Recepción: Febrero , 28, 2021
Aprobación: Marzo , 15, 2021
Publicación: Agosto , 10, 2021
DOI: https://doi.org/10.5281/zenodo.5449803
Abstract:
Objective: This work aims to estimate serum ferritin levels of the B-thalassemia major (BTM) patients in one of the Iraqi thalassemia centers and to evaluate its relationship with their retarded growth.
Methods: this was a cross-sectional study that included 163-patients diagnosed with BTM, undergoing continuous red cells transfusion and chelation therapy. The patients' serum ferritin, height, weight, and body mass index (BMI) was measured. The patients were classified into three groups based on their growth states. All the qualitative parameters were displayed as numerical data and percentages, though the quantitative parameters had displayed as mean ± SD. The variations amongst the 3-groups were analyzed by ANOVA test. The regression fit curves between serum ferritin and growth variables were evaluated. The ROC was examined for ferritin value and growth failure. A .<5% was considered as significant.
Results: The mean age of the studied subjects were 12.6±3.5 years (range 3–19 years). The mean serum ferritin levels were 1812 ±1024.9 ng/mL, which showed a significant correlation with height, weight, and age. The mean height Z-score was -2.1±1.1, and 70% of the patients had growth failure (GF). The mean weight Z-score was -2.0 ±1.3. Both height and weight had a significant correlation with ferritin levels in BTM patients. ROC-curves revealed a sensitivity, specificity, and CI% of [72%,78% & (0.703-0.858)], respectively for predictability of serum ferritin for GF in BTM patients.
Conclusion: In BTM patients, the overall incidence of GF (both mild and severe form) was more than 70%. The mean serum ferritin levels correlated significantly with the growth retardation of BTM patients.
Keywords: growth retardation, β-thalassemia major, ferritin, iron overload.
Resumen:
Objetivo: Este trabajo tiene como objetivo estimar los niveles de ferritina sérica de pacientes con B-talasemia mayor (BTM) en uno de los centros de talasemia iraquíes y evaluar su relación con su crecimiento retardado.
Métodos: se trata de un estudio transversal que incluyó a 163 pacientes diagnosticados de MTB, sometidos a transfusión continua de hematíes y terapia quelante. Se midieron la ferritina sérica, la altura, el peso y el índice de masa corporal (IMC) de los pacientes. Los pacientes se clasificaron en tres conjuntos según sus estados de crecimiento. Todos los parámetros cualitativos se mostraron como datos numéricos y porcentajes, aunque los parámetros cuantitativos se mostraron como media +/- DE.
Las variaciones entre los 3 conjuntos se habían mostrado mediante la prueba ANOVA. Se evaluaron las curvas de ajuste de regresión entre ferritina sérica y variables de crecimiento.
La República de China había examinado el valor de ferritina y el retraso del crecimiento. Se había planificado como significativo un valor de p <5%.
Resultados: La edad media de los sujetos estudiados fue de 12,6 ± 3,5 años (rango 3-19 años). Los niveles medios de ferritina en suero fueron 1812 ± 1024,9 ng / ml, lo que mostró una correlación significativa con la altura, el peso y la edad. La media de la puntuación z de la estatura fue de -2,1±1,1 y el 70% de los pacientes tenía FG. La puntuación z del peso medio fue -2,0±1,3. Tanto la altura como el peso tuvieron una correlación significativa con los niveles de ferritina en pacientes con BTM. Las curvas ROC revelaron una sensibilidad, especificidad e IC% de [72%, 78% y (0,703-0,858)], respectivamente, para la predictibilidad de la ferritina sérica para GF en pacientes con BTM.
Conclusión: En los pacientes con BTM, la incidencia global de FG (tanto de forma leve como grave) fue de más del 70%. Los niveles medios de ferritina sérica se correlacionaron significativamente con el retraso del crecimiento de los pacientes con BTM.
Palabras clave: retraso del crecimiento, β-talasemia mayor, ferritina, sobrecarga de hierro.
INTRODUCTION
Thalassemia is the most common monogenic inherited disorder that disrupts the hemoglobin β-chain and poses an essential health burden in Iraq1,2. Beta-thalassemia major (BTM) patients may suffer chronic anemia, owing to hemolysis and erythropoiesis' dysfunction. Therefore, regular red blood corpuscles (RBCs) transfusions to preserve the hemoglobin level at 9.5-10.5 g/dL are crucial for patients' survival3. Consequently, continual iron-overload, surpassing the body's ability to bind with plasma-free iron, leads to the deposition of iron in the main organs, specifically if proper lifelong iron chelators had not been introduced4,5. BTM patients with poor amenability to chelation treatment will develop complications of iron-overload include multiorgan failure and growth failure (GF), which represents a challenge for the patients and clinicians similarly. The evidence indicates a significant association between high serum ferritin and GF of the BTM patients5. These results confirm that regular evaluation of iron status by serial measurements of ferritin levels is critical for proper management and applied universally as an informal and reliable way of following up the iron status in BTM patients.
Of note, significant well-conducted statistical data about the correlation of serum ferritin with the GF in BTM patients is not published in Babylon yet. For that reason, the aim of this work was to establish the possible association between serum ferritin levels with the growth retardation in β-thalassemia patients.
MATERIALS AND METHODS
Study Design
This cross-sectional study was conducted on 163 children aging 3-19 years with transfusion dependent BTM patients at Babylon Hereditary Blood Disorders Center in Babylon during the period from 10 December 2019 to February 2020.
Subjects
The study population comprised 163 patients of both sexes with an already confirmed diagnosis of BTM upon repeated RBCs transfusion and iron chelation regime. The whole research was approved by the Ethical committee of local authorities. A written consent had obtained from all patients or their attendants.
Methodology
A uniform and authenticated survey formula had been used by the questionary to gather data from the BTM patients during their admission for consistent RBC transfusion or follow-up visits.
Assessment of Anthropometric Parameters
Demographics (age, gender, age at the time of diagnosis and at first transfusion), anthropometric parameters (weight, height, and body mass index, BMI) were collected. The height, weight, and (BMI) of each participant was calculated using a standard stadiometer with minimal clothes and a classical ground scale. BMI was measured depending on height (m) and weight (kg) were BMI=weight/height2. The height, weight, and BMI Z-scores was considered by standard growth chart calculations provided by the World Health Organization (WHO) and Centers for Disease Control and Prevention (CDC)6. A Z-scores less than minus two heights for age was labeled as GF, and a Z-scores lower than minus two weights for age was considered as underweight. The BTM patients were classified according to their height measures into three groups: the 1st group with height Z-scores < -2, the 2nd with height Z-scores > -2 & normally growing subjects regarded as the 3rd group2,4,5.
Analysis of Circulatory Ferritin Values
Blood samples were collected and centrifuged (at how many rpm?). Serum was separated and stored at -60oC, until assessed biochemically. Serum ferritin was determined by the immunoturbidometric method (Spectrum®, Egypt).
Statistical analysis
Data were collected, managed, edited, entered, and analyzed by statistical package of social sciences (SPSS) software. All the qualitative parameters were displayed as numbers and percentage, and the results were expressed as mean ± standard deviation (SD). The statistical variations amongst the 3-gropus were analyzed with the ANOVA-test. The regression fit curves between serum ferritin and growth variables were analyzed. The receiver operating curve had examined for ferritin value and GF. A .<5% was considered as significant.
RESULTS
Demographic characteristics of studied participants (Table 1 and figure 1)
This study comprised 163 BTM patients, 79 (48.5%) females and 84 (51.5%) males. The mean age was 12.6±3.5 years (3-19 range). The median age of the initial blood transfusion was 13 months (10-19 range), and the duration range of regular RBCs transfusion was 11–174 months (results not shown). The mean sera ferritin concentrations were 1812±1024.9 ng/ml (675-4970 range). There were significant variations in the height, weight, and sera ferritin concentrations between the genders, being worse in males (p=0.004, p=0.006, p-0.003), respectively. Two third of all studied patients suffered GF either in mild or severe form.
Demographic characteristics of study parameters among participants
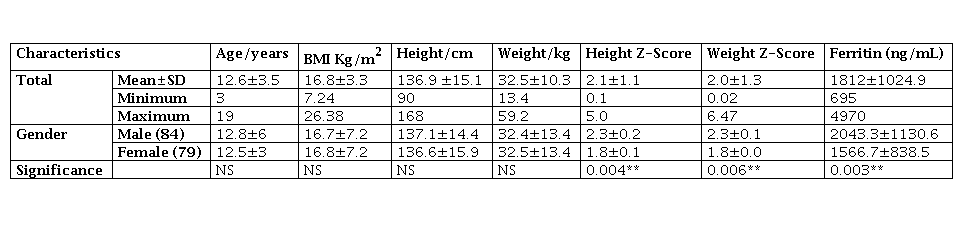
* *P 001
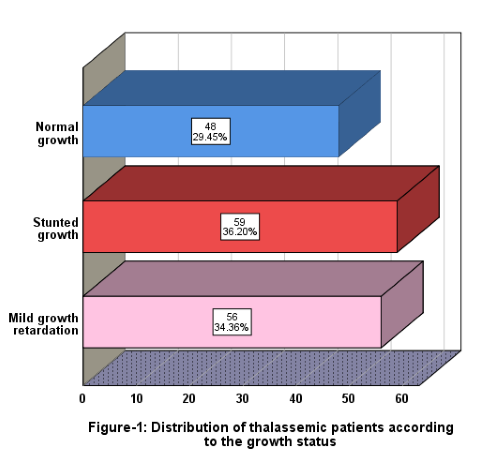
Figure 1.
Distribution of thalassemic patients according to their growth status
There was a positive weakly significant correlation between the ages of the BTM patients with serum levels of ferritin (Figure 2). Likewise, a positive weakly significant correlation between the serum ferritin levels and both height and weight Z-scores (Figures 3 and 4).
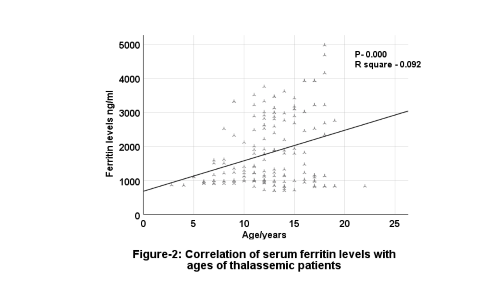
Figure 2.
Correlation of serum ferritin levels with the ages of thalassemic patients
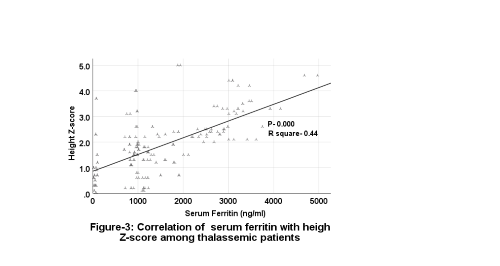
Figure 3.
Correlation of serum ferritin with height Z-score of thalassemic patients
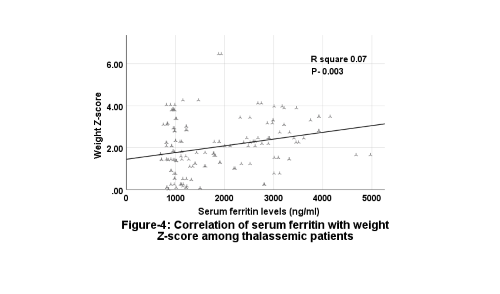
Figure 4.
Correlation of serum ferritin with weight Z-score of thalassemic patients
The receiver operating characteristic curve of blood ferritin concentrations with growth failure (GF) is shown in figure 5. The accuracy of serum ferritin was 87% for detecting GF in BTM patients. Ferritin cutoff point was 1335.5 ng/mL for GF. The patients had classified based on this point, and the authors detected a significant correlation between ferritin values and the incidence of GF (P=0.001) with sensitivity, specificity, and CI% to predict GF of [72%,78%, and 0.703-0.858] respectively (Figure 5).
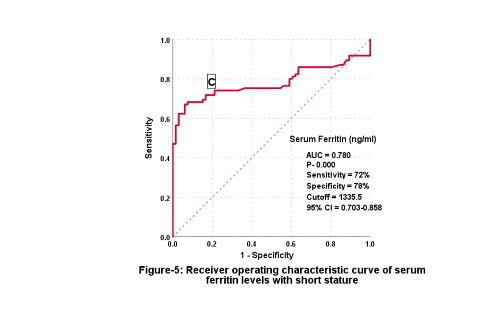
Figure 5.
Receiver operation characteristic curve of serum ferritin with short stature (C=cutoff point)
DISCUSSION
In Iraq, BTM is not uncommon inherited hemoglobinopathy. Iron-overload and GF are common secondary problems in multi-transfused BTM. Hence, intensive chelation therapy and adequate monitoring of iron load are critical in these patients. The detection of blood ferritin levels is used in this study to evaluate the iron burden in BTM as it is an informal, valuable, and non-traumatic pointer of iron overload. Notwithstanding much therapeutic advance in the last couple of decades, GF still is a substantial challenge in BTM, frequently affecting the economic, social, and hence life-quality of families. There is few information about this issue in Iraq, thus we were motivated to conduct this study.
Growth Failure and Thalassemia
Our data support the concept that mean heights of BTM patients were significantly lower than the expected height-for-age. Equally, the studied subjects were wasted. Both findings are in line with what had been reported by other studies7. A presence of GF either in mild (34.4%) or severe form (36.2%) or more than 2/3rd of patients is consistent with previous studies in the middle, capital, south, and Northeastern Iraq that observed a GF ranging from 27.8 - 34%1,4,5,7-9. Conversely, an Iranian scholar was found a higher prevalence rate of GR (43%) among BTM patients10. In the other hand, De Sanctis et al. found that the prevalence of GF in only 7% of BTM patients11. The inconsistencies in the prior results might be cause by malnutrition, dysregulated follow-up, or compliance, altered socioeconomic state, and shortage of iron chelators. As well, the Iraqi conditions and the events that was escorted ISIS, violence, assassination, displacement, and the consequences of such on the thalassemic families as regards financial and social aspects were visible. Such events negatively mirrored the attitude of many families to follow up their regime regularly.
The exact pathophysiology of GF in BTM patients appears to be multifactorial3, 5. Many factors including iron overload causes growth delay. High‑serum ferritin leads to short stature. The delayed growth has been attributed to growth hormone deficit, iron-overload, multi-endocrine dysfunctions, diabetes mellitus, zinc deficit, and long-lasting hypoxia2. Added higher ferritin values can impair growth amongst BTM patients1,12 that is manifested much through and after teenage years13.
Thalassemia patients with mild or advanced GF showed higher circulatory levels of ferritin1,4,5. A similar phenomenon to what was shown in this study, concerning ferritin levels in patients with BTM, had also been published by other researchers9,11-13.
Iron-overload in Thalassemia
Although repeated RBCs transfusions are crucial to survival in BTM patients. With current chelation therapy, the most prominent endocrine complications are failure of normal pubertal development and growth retardation, whereas other disorders such as hypothyroidism, hypoparathyroidism, multiorgan failure and diabetes mellitus are less frequent1,3-5. Progressive deposition of iron causes multiorgan dysfunction and failure including bone marrow, heart, liver, and glands including hypophysis, thyroid, parathyroid, suprarenal as well as pancreas2. Circulatory iron excess can directly upsurge blood ferritin (iron-binding molecule) concentrations. In this regard, the high ferritin levels (1812±1024.9 ng/mL) found in our study support the claim that the management of BTM patients was sub ideal, since the values were double of the recomended ferritin values of 1000 ng/mL. The data of the study suggests that most of the patients had poor chelation treatment, as documented by high circulatory ferritin levels more than accepted.
Ferritin and Growth Failure
It is well documented the high prevalence of GF in BTM related to the effective iron chelation with raised ferritin values5,14. A similar phenomenon was shown in our work. Such association continued in BTM even after they were classified depending on their growth status. These can be more clarified by a proven impact of iron-chelators on cellular division, collagen synthesis, and DNA-molecule, besides trace mineral deposits of zinc and copper. These intricate pathways affect vertebral bodies with ultimate shortening of the spinal and truncal length14,15. Additional plausible explanations might be endocrinopathies induced by iron buildup in different endocrine glands, liver fibrosis, and perhaps owing to the disparity of puberty onset in some BTM patients5,16,17.
Worth mentioning, the transforming growth factor-β (TGF-β) which is a cytokine of the ''TGF-β superfamily'' that possesses a multicellular physiological action1,5,18,19. High TGF-β1 values in BTM could be an unexpected source of dysregulated iron uptake in terms of high plasma ferritin measurements5. Higher plasma TGF-β1 measures were well-correlated with GF in BTM patients1,5.
Age with Ferritin
A closer look at the data indicates a positive correlation of ferritin with age increments. The evidence indicate that iron buildup in BTM is age‑related, indicating that it occurs even in BTM patients not on transfusion regimen20, 21. Even though GF starts in the early years of life in BTM, the retarded growth becomes more evident with increasing age22. Pituitary magnetic resonance imaging (MRI) demostraste that severe hypophysial iron buildup can develop as early as 4-years of age in patients with BTM23.
Sex and ferritin
Our data exhibit some gender variation in some of the growth parameters and ferritin. There was a variable sex influence in the occurrence of GF among BTM, similar to the one reported by other investigations5,24, though other authors revealed significant differences between the two sexes16,21.
This work described the GF and its association with plasma ferritin, which remains the primary study to be directed in our province, where there is a progressive incidence of thalassemias. It is not only imperative to transfuse the packed RBCs to preserve the desired hemoglobin values suitable for the growth and development of BTM patients, but as well the clinicians need to screen the iron state in patients using plasma ferritin levels. Since high blood iron and ferritin will be mutually harmful and will lead to GF. Consequently, wide ranging multicenter systematic researches with a larger population size are recommended.
Conclusion
In BTM patients, the overall incidence of GF (mild and severe form) was more than two-third. The mean serum ferritin levels correlated significantly with the linear growth of BTM patients.
Acknowledgments
The authors would like to thank the medical staff of Babylon Hereditary Blood Disorders Center for their immense cooperation and advice during the study time.
References:
1. Fouad Shareef Dleikh, A.J.A.-A., Rebee Mohsin, Mazin Jaafar Mousa, Hayder Abdul-Amir, et al. Possible cause-and-effect linkage of transforming growth factor-beta1 and platelets derived growth factor-AB with delayed anthropometric parameters in adolescent patients with Cooley’s anemia: Cases vis control research strategy. EurAsian Journal of BioSciences, 2020. 14: 7.1119-1125.
2. Mohsin R., B.A., Shakeer W., Makki H., Mazin J., Fouad S., A study of Linear Growth in β-Thalassemia Major Patients: A cross-sectional study from Babylon Hereditary Blood Disorders Center in Babylon. Indian Journal of Forensic Medicine and Toxicology, 2021. 12(2).
3. Bhavana Singhal, N.S.a. R.M., Iron-overload and Growth of Thalassemic Patients in Marwar Region. International Journal of Pharmaceutical Sciences and Research, 2012. 20(3):6.
4. Hayder M., Mazin. J., Shaker A., No Significant Relationship of Ferritin Levels to the Levels of Platelet-derived Growth Factor (PDGF) in the Peripheral Blood of Transfusion-dependent ß-Thalassemia Major Patients with Growth Retardation. International Journal of Pharmaceutical Research, 2020. 12(3): 258-65.
5. Makki H., M.M., Asseel K., Al-Saad R., AL-Dujaili W., Relationship of levels of transforming growth factorbeta1 (TGF-β1) to the levels of ferritin in blood of transfusion dependent β-thalassemia major patients with growth retardation: A case-control study. EurAsian Journal of BioSciences, 2020. 14: 7, 521-527.
6. de Oliveira GJ, B.S., Cesa CC, Pellanda LC Comparison of NCHS, CDC, and WHO curves in children with cardiovascular risk. . Revista da Associação Médica Brasileira (English Edition) 2013. 59(4): 6.
7. Rahul Jahagirdar, S.P., Ruma Deshpande, Sanjay Lalwani, Growth profile of children with thalassemia major. Pediatrics, 2017..(1).
8. Abdulwahid, D.A. and M.K. Hassan, β- and α-Thalassemia intermedia in Basra, Southern Iraq. Hemoglobin, 2013. 37(6): 553-63.
9. Amin, S.S., et al., Beta-Thalassemia Intermedia: A Single Thalassemia Center Experience from Northeastern Iraq. BioMed Research International, 2020. 2020: 2807120.
10. Karamifar, H., et al., Endocrine function in thalassemia intermedia. International Journal Of Biomedical Science: IJBS, 2006. .(3): 236-240.
11. De Sanctis, V. Tangerini, A. Testa, M. R. Lauriola, A. L. Gamberini, M. R. Cavallini, A et al., Final height and endocrine function in thalassemia intermedia. J Pediatr Endocrinol Metab, 1998. 11 Suppl 3: 965-71.
12. Rathaur VK, I.A., Pathania M., Growth pattern in thalassemic children and their correlation with serum ferritin.2020;9:1166-9. J Family Med Prim Care 2020. .: 4.
13. Hashemi, A., Ghilian, R., Golestan, M., Akhavan G. Mahvash, Z., Dehghani, M., The Study of Growth in Thalassemic Patients and its Correlation with Serum Ferritin Level %J Iranian Journal of Pediatric Hematology and Oncology. 2011. .(4): 147-151.
14. Rodda, C. P., Reid, E. D., Johnson, S., Doery, J., Matthews, R., Bowden, D. K., Short stature in homozygous beta-thalassaemia is due to disproportionate truncal shortening. Clin Endocrinol (Oxf), 1995. 42(6): 16.
15. Low, C.K., et al., The effect of platyspondyly and pubertal growth spurt on the stature of patients with beta-thalassaemia major. Chin Med J (Engl), 1998. 111(8): 5.
16. Saxena, A., Growth Retardation in Thalassemia Major Patients. International Journal of Human Genetics, 2003. .(4): 237-246.
17. Kyriakou, A. and N. Skordis, Thalassaemia and aberrations of growth and puberty. . Mediterranean journal of hematology and infectious diseases, 2009. .(1).
18. Raghdan Z. Al-Saad, A.K., Fouad S., Hayder A.A., Is There Any Association Between Highly Sensitive C-Reactive Protein and Dental-Status in Ischemic Heart Diseases? Comparative-Study Biochemical and Cellular Archives, 2020. 20(2), 6069-6075.
19. Abbas A., A-A. Hayder, Chabuk S., Mousa M., Conicity index as an Anthropometric Index of Central Obesity in the Prediction of Adult Bronchial Asthma; Correlation with Fractional Exhaled Nitrous Oxide Tests. Medico-Legal Update, 2021. 21(2).
20. Buonanno G., V.A., Gonnella F., Cantore N, de Bellis G., Serum ferritin in beta‑thalassaemia intermedia. Scand J Haematol 1984. 32: 5.
21. Karunaratna AMDS, R.J., Mudiyanse RM., Iron-overload in beta thalassemia major patients. Int J Blood Transfus Immunohematol 2017. .: 8.
22. Shuvraneel B., T.K.D., Pranab K., Rajib De, Prakas K., Prantar C., Does Thyroid Dysfunction Correlates with Iron-overload in Eβ Thalassemia Patients? A Study from a Tertiary Care Thalassemia Center in India. Arch Med Health Sci 22019. .: 6.
23. Bushra M., A.H., Sobiya S., Ahmed R., Bilal H. & Manesh G., Anthropometric measurements in children having transfusion-dependent beta thalassemia. Hematology, 2018. 23(4): 6.
24. Shalitin, S., Carmi, D., Weintrob, N. Phillip, M. Miskin, H. Kornreich, L., et al., Serum ferritin level as a predictor of impaired growth and puberty in thalassemia major patients. Eur J Haematol, 2005. 74(2): 93-100.
Notas de autor
phar.hayder.abdul@uobabylon.edu.iq
Información adicional
Conflicts of interest: The authors of this manuscript declare that they have no conflicts of interest, real or perceived, financial or nonfinancial in this article.
Strengths and Limitations: Some limitations of this study were verified. First, the BTM patients were not evaluated for their sexual growth. As hypogonadism is the most element of GF in BTM which leads to progressive GF with increasing age. Second, the effect of other factors like endocrinopathies on growth status and their exact link with plasma ferritin must be evaluated frequently. This was not completed in our work owing to the multifactorial and costly nature of hormonal tests.
