Artículos

Esta obra está bajo una Licencia Creative Commons Atribución-SinDerivar 4.0 Internacional.
Recepción: 09 Septiembre 2020
Aprobación: 15 Octubre 2020
Publicación: 09 Diciembre 2020
DOI: https://doi.org/10.5281/zenodo.4544035
Abstract: Background: Daytime sleepiness refers to the propensity of falling asleep in a variety of situations, maybe due to insufficient sleep (major cause), or fragmented sleep. Objective: Measure lipid peroxidation and evaluate the effect of antioxidant vitamin intake in medicine interns with daytime sleepiness in Peruvian hospitals. Methods: Fluorometric assays were used to determine the concentration of Malondialdehyde, glucose, total cholesterol, triglycerides, HDL, LDL, and VLDL. We also evaluated Body Mass Index and age. The values of these variables were compared between groups of sleepy (n=33) and normal interns (n=33). Then vitamin C and E were given, for 10 weeks, to 17 sleepy interns and the remaining sample (n=16) were considered the control group. Results: sleepy interns had more MDA than the non-sleepy group (greater lipid peroxidation) due to poor sleep. Vitamins C and E decrease MDA levels in sleepy inmates and levels of lipid peroxidation. These results were similar to those reported by other scientists. Conclusions: Drowsy interns have almost double of lipid peroxidation, compared to control. Vitamins C and E reduce the level of Malondialdehyde in inters with Daytime sleepiness.
Keywords: Sleep deprivation, Malondialdehyde (MDA), Lipid Peroxidation.
Introduction
Daytime sleepiness refers to the propensity of falling asleep in a variety of situations1, may be due to insufficient sleep (major cause), or fragmented sleep2. The first case, lack of sleep, maybe due to poor sleep hygiene or socially imposed sleep deprivation, as is the case of those jobs that involve making night guards2. While fragmented sleep is caused mainly by Obstructive Sleep Apnea, which is a syndrome caused by multiple respiratory cessations during sleep that induces partial arousals and interferes with the maintenance of sleep. It may be central, obstructive, or mixed central - obstructive apnea3.
Sleep problems have been associated with the imbalance of many systems such as lipid peroxidation (LPO), which is peroxidase-catalyzed lipid oxidation and having hydrogen peroxide as the electron acceptor.
Reactive species oxidizes phospholipids of cell membranes, the oxidized phospholipids may play an important role in inflammatory and pro-inflammatory processes, and even participate in programmed cell death4,5. LPO is related to the severity of depression and the number of depressive episodes6.
Malondialdehyde (MDA) is an organic molecule with formula CH2 (CH0) 2 resulted from lipid peroxidation of polyunsaturated fatty acids (PUFA)7, and has been evaluated as an excellent measurement of lipid peroxidation8. and as a good marker of LPO for sleep problems, specifically Obstructive Apnea syndrome9.
Since 1994, the theory that sleep is an antioxidant was put forward10, and since then, research carried out in Russia and first world countries have focused on the relationship of oxidation and sleep problems, mainly focused on Obstructive Sleep Apnea (OSA)1,11-14. Although OSA is a cause of Daytime DS, this is not the only one. Investigations focused on Daytime Sleepiness, measured with the Epworth scale are few, highlighting the Russian investigations, focused mainly on pregnant or menopausal women, in which it has been found that this is dependent on ethnic origin1,15. Also, researchers recommended exploring the effect of sleep in a population whose training is to stay awake16 - such is the case of medical interns.
To our knowledge there is no research about the relationship of lipid peroxidation and daytime sleepiness in Peruvian population, considering that previous studies found variation according to ethnicity, it would be interesting to evaluate this relationship in the local population subjected to sleep disturbance due to their job’s requirements. Concurrently the effect of vitamins C and E over lipid peroxidation in this population could be explored.
Consequently, it is proposed to evaluate the level of lipid peroxidation in inmates who lack daytime sleepiness and apply vitamins D and E to evaluate its effect on the level of lipid peroxidation.
Methods
Population and sample
The population consisted of 94 medical interns in hospitals in the department of Lambayeque-Perú. The sample size was calculated using the Epidat program, version 4.2, based on previous research17. It was included Medical interns registered at the Hospitals who agreed to participate in the study and to donate a blood sample, it was excluded medical interns who suffered from kidney disease, AIDS, cancer, diabetes, high blood pressure, smoking, morbid obesity and who had received drug treatment in the last two weeks. Out of the 94 medical interns, it was selected, randomly, 33 suffering from Daytime Sleepiness, and 33 without this condition. The 33 presenting DS were divided into two groups, 17 of them received vitamins C and E during, and the 16 remaining were controls.
Techniques and instruments for data collection
Epworth Daytime Sleepiness Scale was used to detect sleepy individuals. the overall score has a range of 0 to 24. A score greater than 10 was considered positive for the characteristic of excessive daytime sleepiness18.
For both groups of 33 medical interns, MDA, glucose, and lipid profile were measured; likewise, blood pressure and body mass index (BMI) were evaluated. The evaluation of the concentration of MDA in blood was carried out following the Northwest kit for lipid peroxidation and the determination of glucose and lipid profile.
Thirty-three interns suffering daytime sleepiness were randomly distributed to two groups (17 interns with treatment and 16 controls). The first group was given vitamin C and E for 12 weeks in doses of 1 g and 400 IU, respectively.
At the end of the treatment, the concentration of MDA, glucose, lipid profile, blood pressure, and BMI were measured again, mean plus standard deviation was calculated and the Difference of Unpaired Means test was applied, to determine the role of chance in the difference stockings.
Statistical analysis
The results were recorded as continuous variables (MDA, Glucose, Cholesterol, Triglycerides, HDL, LDL, VLDL, PAS, PAD, IMC, Age) and expressed as mean and standard deviation (SD) depending on their normal distribution. The Mann–Whitney U test was employed for the asymmetrically distributed variables. Statistical analysis was performed using SPSS 20.0 software (SPSS Inc., Chicago, IL, USA). Two-tailed significance values were used, and the significance levels were set at 0.05.
The research protocol was approved by the Ethics Committee of the Lambayeque Regional Hospital. All participating interns sign an individual informed consent, after explanation and information for the study.
Results
The concentration of various analytes such as glucose, cholesterol, triglycerides, HDL, LDL, VLDL, PAS, PAD, and BMI (Table 1) was compared and a significant difference was found only for the concentration of MDA between the two groups (p<0.01). Finding that the group of drowsy interns had a higher concentration of MDA (7.7±2.1) than the group of students without daytime sleepiness (4.8±2.2). Also, it should be noted that the concentration of cholesterol and triglycerides did not have a significant difference.
Cholesterol levels remained close to the upper limit of reference values (200 mg / dL) in both groups, the same happened with values for LDL and VLDL, whose values remained around 120 and 24 mg / dL, respectively.
Depending on the reference levels, HDL greater than 50 mg / dL10, the values for both groups were close to the lower limit than recommended (Table 1). The BMI was close to the upper limit for what is considered "normal" or referential.
Of the 66 medical interns who participated in the study, 33 drowsy identified had a higher concentration of MDA (x̅ = 7.7 µM), compared to non-drowsy (x̅ =4.8 µM), with a significant difference (p<0.01). For the other analytes, no significant differences were observed, (Table 1).
Comparison of the levels of different analytes between sleepy and nonsleepy groups before treatment
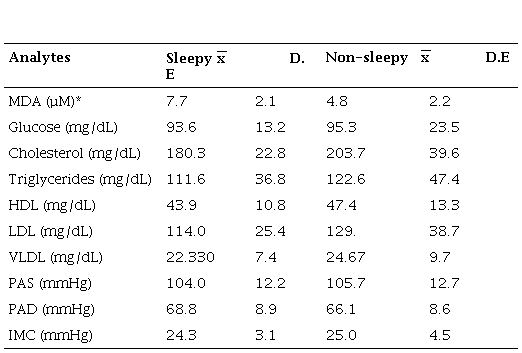
* p001 Mann Whitney U nonparametric test
When the values of the various variables (analytes) were compared, before and after, in the group that did not receive the vitamin treatment, it was observed that the concentration of MDA increased slightly after the end of the experiment (12 weeks), the difference was significant (p=0.027). It was also observed that the concentration of glucose, cholesterol, triglycerides, HDL, VLDL, PAS, and PAD did not show significant differences, although before and after the BMI was significantly different (p=0.004), the decrease was mildly observed (Table 2).
Comparison of the biological indicators of the control group 16 interns before and after the experiment
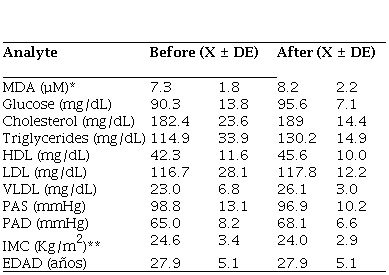
* p005 Mann Whitney U nonparametric test ** p001 Mann Whitney U nonparametric test
From the group of drowsy inmates who were given vitamin C and E, it was observed that the concentration of MDA decreased below half after the end of treatment (Table 3). Glucose and SBP decreased slightly, while PAD remained stable and the rest of the variables did not have a significant difference between their values before and after treatment (Table 3).
Comparison of the biological indicators of the experimental group before and after treatment
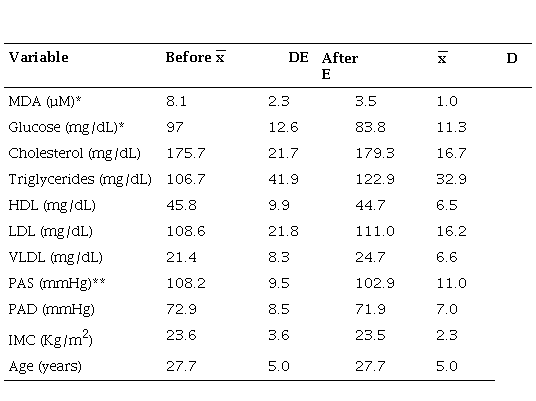
* p005 Mann Whitney U nonparametric test ** p001 Mann Whitney U nonparametric test
When the experimental and control groups were compared after 12 weeks of treatment, it was observed that there was a significant difference (p <0.01) for the concentration of MDA, the experimental group had a lower concentration of MDA. The glucose concentration was also lower in the experimental group than the control group (p = 0.03). On the contrary, the value of the rest of the variables had no significant differences (Table 4).
Comparison of the biological indicators of the control group and the experimental group after treatment
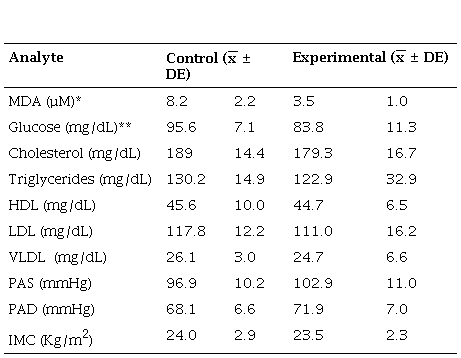
* p001 Mann Whitney U nonparametric test ** p005 Mann Whitney U nonparametric test
Discussion
The group of drowsy interns has more MDA (greater lipid peroxidation) than the group of non-sleepy due to Daytime Sleepiness. The results are similar to those found for research in the Russian population. Two ethnic groups were investigated, the Russian (n=45) and the Buryat (n=28) concerning the LPO, it was found that the women with daytime sleepiness suffered from higher lipid peroxidation, and that also the level varied according to an ethnic group, affecting to a greater extent the Buyat group, Coefficient of Oxidative Stress 2.2 versus 3.8, respectively11. In the same line, in 45 perimenopausal women, Epworth scale 12.2±0.42, lipid peroxidation side products increased 2.2. times, compared to the control group15. What was found in the present investigation would help to evaluate that in a different ethnic population the result is the same, also, the LPO (MDA concentration) remains close to double, compared to the non-sleepy group.
In the case of humans, nocturnal apnea increases oxidative stress in patients with Obstructive Sleep Apnea as tested with 8-isoprostane oxidative marker19. In patients suffering from depression, the concentrations of MDA were significantly higher in those with insomnia than in patients without insomnia (Average =2.87 (2.04-3.72) and 2.31 (1.62-3.17) nmol/mL respectively, (p=0.004)20. When the level of MDA was compared in women with high sleep deprivation and women with low sleep deprivation, a significant difference was found for MDA levels (p<0.01) 6.1±1.8 and 4.1±1.2, respectively. There was also a significant difference for other oxidative stress markers such as Carbonyl Protein (PCO) and High Sensitivity Protein C (hsCRP) Babies of mothers with high sleep deprivation showed anthropometric differences concerning those of mothers with low sleep deprivation21. Although other factors could explain the increase in the level of BMI or cholesterol there was no significant difference when these values were compared before and after 12 weeks of treatment for both control and experimental groups. Also, other values that remained similar were LDL, VDLD, and HDL.
This differs from a study in the Colombian pediatric population where a significant statistical association was found between serum LDL and total cholesterol concentrations with plasma MDA concentrations, indicating that the higher the concentration of serum lipids, the greater the lipid peroxidation. The hypercholesterolemic children had concentrations of MDA 125% higher than controls of similar age and sex22. Others have reported that the level of MDA increased with the increase in BMI and the level of insulin resistance, significantly correlated with abdominal obesity23.
The vitamins C and E decrease MDA levels in drowsy interns, then lower levels of lipid peroxidation. When the level of lipid peroxidation was compared among the 17 interns who were given vitamins C and E and the control group, the level of peroxidation of the treated group decreased below half. It goes in accordance, with previous results, vitamin C together protects from oxidative damage in the mitochondria and lipids in neural circuits. However, this has been studied mainly in people experiencing depression and suicidal behavior24, and here we add more literature about the effect of these vitamins in people suffering from SDiurna.
These results were similar to those found in elderly hemodialysis patients supplemented with vitamin C and E who improved antioxidant status and decreased plasma lipid peroxides. For those who were supplemented with vitamin C and E, the concentration of MDA + 4-hydroxynonenal (µmol / L) decreased from 53.1±28.4 (week 0) to 32.3±21.6 (week 10)25.
Vitamins C and E act as antioxidants disrupting the free radical formation chain, act as electron donors for the irreversible reduction of peroxide hydrogen which is a source of free radicals26. Studies have found that vitamin C can protect DNA from oxidative damage induced by radiation.
In the case of vitamin C, it is considered an important radiation-induced radical scavenger and could be used to develop new radiotherapy protocols especially in cancer patients, since radiation-induced tissue damage is one of the largest complications in cancer radiotherapy27, meanwhile, vitamin E reduces hydroperoxides protecting membrane lipids. The concentration of the vitamin in the lipid bilayer determines the length of the chain of fatty acids affected by an oxidation event, vitamin E then acts by reducing peroxyl radicals by breaking the radical chain and therefore decreasing the rate of lipid peroxidation28.
In this research, it is suggested that the supply of vitamin C and E to reduce the levels of lipid damage in people suffering from daytime sleepiness could be a good alternative to compensate for the damages suffered at the cellular level.
Conclusions
The group of drowsy interns has almost the double of lipid peroxidation (MDA), compared to control, and this cipher is similar to the one found for perimenopausal Russian women with Daytime sleepiness. Vitamins C and E reduce the level of MDA in inters with Daytime sleepiness.
Conflict of interest
The authors declare no financial or other conflicts of interest.
Highlights
1. The group of sleepy inmates had a higher MDA (x̅ = 7.7 µM) compared to the non-sleepy MDA (x̅ = 4.8 µM).
2. MDA decreased more than half from 8.1 µM to 3.5 µM in medical inmates with vitamin treatment which was not observed in inmates who did not receive treatment.
3. The vitamins C and E did not cause any alteration in glucose, cholesterol, triglyceride, and HDL values.
4. Vitamin C and E therapy could be a good alternative to reduce levels of cell lipid damage in people suffering from daytime sleepiness.
Acknowledgments
Acknowledgments
To all the medical interns who participated in this research and to the Catholic University Santo Toribio de Mogrovejo for the use of their laboratory to perform the processing of the sample.
References
1. Semenova N V., Madaeva IM, Kolesnikova LI. Free Radical Oxidation and Sleep Disorders in Andro- and Menopause (Literature Review). Acta Biomed Sci. 2020;5(1):31–41.
2. Dauvilliers Y. Differential diagnosis in hypersomnia. Curr Neurol Neurosci Rep. 2006;6(2):156–62.
3. Berry RB, Budhiraja R, Gottlieb DJ, Gozal D, Iber C, Kapur VK, et al. Rules for Scoring Respiratory Events in Sleep: Update of the 2007 AASM Manual for the Scoring of Sleep and Associated Events. J Clin Sleep Med. 2012;8(5):597–619.
4. Su LJ, Zhang JH, Gomez H, Murugan R, Hong X, Xu D, et al. Reactive Oxygen Species-Induced Lipid Peroxidation in Apoptosis, Autophagy, and Ferroptosis. Vol. 2019, Oxidative Medicine and Cellular Longevity. 2019.
5. Ramos-Tovar E, Muriel P. Free radicals, antioxidants, nuclear factor-E2-related factor-2 and liver damage. Vol. 40, Journal of Applied Toxicology. 2020. p. 151–68.
6. Sowa-Kućma M, Styczeń K, Siwek M, Misztak P, Nowak RJ, Dudek D, et al. Are there differences in lipid peroxidation and immune biomarkers between major depression and bipolar disorder: Effects of melancholia, atypical depression, severity of illness, episode number, suicidal ideation and prior suicide attempts. Prog Neuro-Psychopharmacology Biol Psychiatry. 2018;81:372–83.
7. Draper HH, Hadley M. Malondialdehyde determination as index of lipid Peroxidation. Methods Enzymol. 1990 Jan;186(C):421–31.
8. de Oliveira Ulbrecht MO, Gonçalves DA, Zanoni LZG, do Nascimento VA. Association Between Selenium and Malondialdehyde as an Efficient Biomarker of Oxidative Stress in Infantile Cardiac Surgery. Biol Trace Elem Res. 2019 Jan;187(1):74–9.
9. Lira AB, de Sousa Rodrigues CF. Evaluation of oxidative stress markers in obstructive sleep apnea syndrome and additional antioxidant therapy: a review article. Vol. 20, Sleep and Breathing. Sleep and Breathing; 2016. p. 1155–60.
10. Reimund E. The free radical flux theory of sleep. Med Hypotheses. 1994;43(4):231–3.
11. Kolesnikova LI, Semenova N V., Solodova EI, Madaeva IM. Oxidative stress in women with insomnia in different stages of menopause. Ter Arkh. 2017;89(8):50–6.
12. Kolesnikova LI, Madaeva IM, Semenova N V, Vlasov BY, Grebenkina LA, Darenskaya MA, et al. Antioxidant potential of the blood in men with obstructive sleep breathing disorders. Bull Exp Biol Med. 2013;154(6):731–3.
13. Semenova N V., Madaeva IM, Kolesnikova LI. The role of melatonin as a component of the antioxidant defense system in perimenopausal women with insomnia. Zhurnal Nevrol i Psihiatr Im SS Korsakova. 2019;119(7):7–13.
14. Eisele HJ, Markart P, Schulz R. Obstructive sleep apnea, oxidative stress, and cardiovascular disease: Evidence from human studies. Oxid Med Cell Longev. 2015;2015:1–9.
15. Kolesnikova LI, Madaeva IM, Semenova N V., Solodova EI, Grebenkina LA, Darenskaya MA. Evaluation of lipid peroxidation - Antioxidant protection in perimenopausal women with sleep disorders. Vestn Ross Akad Meditsinskikh Nauk. 2014;69(11–12):11–6.
16. Van Liempt S, Vermetten E, Geuze E, Westenberg HGM. Pharmacotherapy for disordered sleep in post-traumatic stress disorder: A systematic review. Vol. 21, International Clinical Psychopharmacology. 2006. p. 193–202.
17. Rajendiran S, Swetha Kumari A, Nimesh A, Soundararaghavan S, Ananthanarayanan PH, Dhiman P. Markers of oxidative stress in pregnant women with sleep disturbances. Oman Med J. 2015;30(4):264–9.
18. Carpagnano GE, Kharitonov SA, Resta O, Foschino-Barbaro MP, Gramiccioni E, Barnes PJ. 8-Isoprostane, a Marker of Oxidative Stress, Is Increased in Exhaled Breath Condensate of Patients with Obstructive Sleep Apnea after Night and Is Reduced by Continuous Positive Airway Pressure Therapy. Chest. 2003;124(4):1386–92.
19. Adibhatla RM, Hatcher JF. Altered lipid metabolism in brain injury and disorders. Subcell Biochem. 2008;49:241–68.
20. Jouyban A, Khoubnasabjafari M. Comments on “Malondialdehyde: A novel predictive biomarker for post-stroke depression.” J Affect Disord. 2018;225:52–3.
21. Céspedes Miranda E, Castillo Herrera J. La peroxidación lipídica en el diagnostico del estrés oxidativo del paciente hipertenso. ¿Realidad o mito? Vol. 27, Revista Cubana de Investigaciones Biomedicas. 2008. p. 1–13.
22. Velásquez Rodríguez C, Uscátegui Peñuela R, Burgos Herrera L. Peroxidación lipídica y concentración de vitamina E plasmática en niños hipercolesterolémicos de Medellín (Colombia). An Pediatría. 2004;61(1):16–22.
23. Jia XJ, Liu LX, Tian YM, Wang R, Lu Q. The correlation between oxidative stress level and intra-abdominal fat in obese males. Med (United States). 2019 Feb;98(7):e14469.
24. Du J, Zhu M, Bao H, Li B. The Role of Nutrients in Protecting Mitochondrial Function and Neurotransmitter Signaling: Implications for the Treatment of Depression, PTSD, and Suicidal Behaviors. Physiol Behav. 2016;56(15):2560–78.
25. Chao JCJ, Yuan MD, Chen PY, Chien SW. Vitamin C and E supplements improve the impaired antioxidant status and decrease plasma lipid peroxides in hemodialysis patients. J Nutr Biochem. 2002;13(11):653–63.
26. Zequeira DEB. Vitaminas y oxidorreductasas antioxidantes: Defensa ante el estrés oxidativo. Vol. 25, Revista Cubana de Investigaciones Biomedicas. 2006.
27. Epperly MW, Osipov AN, Martin I, Kawai KK, Borisenko GG, Tyurina YY, et al. Ascorbate as a “redox sensor” and protector against irradiation-induced oxidative stress in 32D CL 3 hematopoietic cells and subclones overexpressing human manganese superoxide dismutase. Int J Radiat Oncol Biol Phys. 2004;58(3):851–61.
28. Marnett LJ. Lipid peroxidation - DNA damage by malondialdehyde. Mutat Res - Fundam Mol Mech Mutagen. 1999 Mar;424(1–2):83–95.
