Artículos
Use of calcium channel blockers and NSAIDS as alternative to anticholinergics in the treatment of urge urinary incontinence in women
Uso de bloqueadores de los canales de calcio y AINE como alternativa a los anticolinérgicos en el tratamiento de la incontinencia urinaria de urgencia en mujeres
Use of calcium channel blockers and NSAIDS as alternative to anticholinergics in the treatment of urge urinary incontinence in women
Archivos Venezolanos de Farmacología y Terapéutica, vol. 40, núm. 4, pp. 418-423, 2021
Sociedad Venezolana de Farmacología Clínica y Terapéutica

Esta obra está bajo una Licencia Creative Commons Atribución-SinDerivar 4.0 Internacional.
Recepción: 28 Enero 2021
Aprobación: 15 Febrero 2021
Publicación: 10 Junio 2021
Abstract: Objectives: The present study aimed to investigate the effect of use of calcium channel blockers and non-steroid anti-inflammatory drugs as alternative to the anticholinergics therapy in cases of urge urinary incontinence in women. Methods: A total of 63 women with urge incontinence were included in the study after inclusion and exclusion criteria and randomized into three treatment groups and their clinical and urodynamic parameters were recorded and analyzed comparatively. Results: Significant differences were found in the means of patients’ urinary incontinence parameters including numbers of voidings and of leakage episodes per day as well as their urodynamic parameters including maximum bladder capacity, volume at 1st contraction and residual volume after using Solifenacine, calcium channel blocker (Nifidipine. or Non-steroid anti-inflammatory (Ponstan) drugs (.<0.05). Furthermore, significant differences were found among study’s groups regarding their response for treatment indicated by improvement in clinical and urodynamic parameters (.<0.05).Conclusion: The study demonstrated that CCBs seem to be having some beneficial effects, with a different adverse effect profile and so may be used to complement a lower dose of anticholinergic drug.
Keywords: Urinary incontinence, anticholinergics, calcium channel blockers, NSAIDs.
Resumen:
Objetivos: El presente estudio tuvo como objetivo investigar el efecto del uso de bloqueadores de los canales de calcio y antiinflamatorios no esteroides como alternativa a la terapia anticolinérgica en casos de incontinencia urinaria de urgencia en mujeres. Métodos: Se incluyó en el estudio un total de 63 mujeres con incontinencia de urgencia después de los criterios de inclusión y exclusión y se asignaron al azar en tres grupos de tratamiento y sus parámetros clínicos y urodinámicos se registraron y analizaron comparativamente. Resultados: Se encontraron diferencias significativas en los promedios de los parámetros de incontinencia urinaria de los pacientes, incluido el número de micciones y de episodios de fugas por día, así como sus parámetros urodinámicos, incluida la capacidad máxima de la vejiga, el volumen en la primera contracción y el volumen residual después de usar solifenacina, bloqueador de los canales de calcio. (Nifidipina) o medicamentos antiinflamatorios no esteroides (Ponstan) (P <0.05). Además, se encontraron diferencias significativas entre los grupos de estudio con respecto a su respuesta al tratamiento indicada por la mejora en los parámetros clínicos y urodinámicos (P <0,05). Conclusión: El estudio demostró que los BCC parecen tener algunos efectos beneficiosos, con un perfil de efectos adversos diferente, por lo que pueden utilizarse para complementar una dosis más baja de fármaco anticolinérgico.
Palabra clave: incontinencia urinaria, anticolinérgicos, bloqueadores de los canales de calcio, AINE.
Introduction
Urinary incontinence is estimated to affect >50% of women aged more >65 years, (only 2/3 of those seek medical advice), and one of its main types is urge incontinence: involuntary leak of urine associated with urgency and attributed to involuntary detrusor muscle contraction1. In the pharmacological treatment perspective, two main drugs are used: anticholinergics and B3 adrenergic agonists. The latest (Mirabegron) though typically well tolerated but is used less often because of high cost and the reports stating the need to combine or follow initiation with anticholinergic to reach maximum efficacy.
Anticholinergics though have long been the 1st choice for urgency medical treatment but findings have raised concerns about central adverse effects as brain atrophy and cognitive decline in elderly patients2, beside the peripheral side effects as dry mouth, constipation, tachycardia, etc.3. Managing those side effects sometimes necessitates dose reduction though on the expense of efficacy4. For this reason, searching for alternatives to reduce the bladder contractions deemed necessary. Of such drugs: are the Ca channels blockers (CCBs), since the regulation of smooth muscle tone depends on Ca influx through L-type calcium channels, an effect which has been demonstrated in many humans and animals’ studies5.
Non-steroidal anti-inflammatory drugs (NSAIDs) have also been implicated since prostaglandins (PG) are important in provoking detrusor muscle contractions6, and some studies reported their role in causing urinary retention when used in higher than the recommended dose7. This study aimed to investigate the effect of use of calcium channel blockers and non-steroid anti-inflammatory drugs as alternative to the anticholinergics in treatment of urge urinary incontinence among women.
Patients and methods
This is a prospective study where 63 women diagnosed with urge incontinence (by history and urodynamic study), and with no contraindications to any drugs in the study as peptic ulcer, low or high blood pressure profile were included in the study. Data about study variables were reported at the study beginning, then patients were randomized into three treatment groups (anticholinergic alone: Solifenacine 10 mg/day, NSAIDs: Diclofenac 100 mg/day, CCBs: Nifidipine 10 mg/day) and data about similar variables assessed at six-week interval post treatment. Variables studied were those reflecting clinical response (total number of voidings /day, number of leakages /day), urodynamic response (volume at 1st contraction, maximum bladder capacity, and residual volume) as well as data about withdrawal from study due to adverse effects. Ethical Approval was obtained from Ethical Committee/ Department of Family and Community Medicine/ College of Medicine/ Al-Iraqia University.
Data were entered and analyzed using IBM-Computer Software Programs of Statistical Package of Social Science (SPSS) version 26 and STATISTICA version 12. The descriptive statistics of frequency distribution tables, number and percentage were used for qualitative data, whereas, the mean, standard deviation and range were used for quantitative data. Paired t-test were used to compare the effect of drug regime on the patients’ parameters for each study’s group and the Multivariate analysis of TWO Way MANCOVA test with Scheffe post-hoc tests were used to Identify the significant differences among study’s groups and its source as the main drugs’ effect on patient’s clinical and urodynamic parameters. Additionally, Chi-square test was used to find the differences regarding withdrawal adverse effects among study’s groups. A P-value of <0.05 was used as the criterion for determining statistical significance throughout study.
Results and discussion
The age of study sample ranged between 35-65 years old with means of study’s groups were 48.14±11.222, 48.24±11.207 and 47.76±10.867 years old of Solifenacine, Nifidipine and Ponstan respectively, with no significant differences among groups (F: 0.434, P= 0.974).
Anticholinergic usage was found to be associated with the best improvement in patients’ variables. Regarding the use of Solifenacine, there were significant differences in the means of urinary incontinence parameters between pre-and post-usage of drug, as the mean of numbers of voidings and of leakage episodes per day were significantly lower in post- Solifenacine usage than in pre-usage (3.2381±0.49038 vs. 5.5714±0.48181) (t: 17.863, P=0.000) and (2.190±0.51177 vs. 2.5810±0.57586) (t: 2.262, P=0.035) respectively. On the other hand, the means of urodynamic parameters of maximum bladder capacity, volume at 1st contraction and residual volume were found to be significantly increased after Solifenacine usage as compared to before its usage (366.2857±28.11075 vs. 289.333±6.05255) (t: 11.475, P=0.000), (245.2381±39.19427 vs. 159.1905±15.89849) (t: 10.334, P=0.000) and (86.5238±5.63577 vs. 60.8095±1.74983) (t: 19.801, P=0.000) respectively (Table 1).
| Group Parameters | Solifenacine treatment | |||
| Pre- use | Post- use | Mean differences | Significance* | |
| Mean ± Sd | Mean ± Sd | |||
| Clinical Parameters | ||||
| Number of voidings/ day | 5.5714± 0.48181 | 3.2381± 0.49038 | - 2.33333 | t: -17.863 P= 0.000 |
| Number of leakage episodes/day | 2.5810± 0.57586 | 2.190± 0.51177 | - 0.39048 | t: -2.262 P= 0.035 |
| Urodynamic Parameters | ||||
| Maximum bladder capacity (cc) | 289.333± 6.05255 | 366.2857± 28.11075 | 76.95238 | 11.475 P= 0.000 |
| Volume at 1st contraction(cc) | 159.1905± 15.89849 | 245.2381± 39.19427 | 86.04762 | t: 10.334 P= 0.000 |
| Residual volume (cc) | 60.8095± 1.74983 | 86.5238± 5.63577 | 25.71429 | t: 19.801 P= 0.000 |
Similarly, significant differences were found in the means of patients’ urinary incontinence parameters after using calcium channel blocker (Nifidipine) or Non-steroid anti-inflammatory (Ponstan) drugs (P<0.05) (Table 2) and (Table 3) respectively.
Nifidipine, clinically it was associated with reduced number of voidings /day but less than that induced by anticholinergics (may be due to the diuretic and natriuretic effect) 8.
| Group Parameters | Ponstan treatment | |||
| Pre- use | Post- use | Mean differences | Significance* | |
| Mean ± Sd | Mean ± Sd | |||
| Clinical Parameters | ||||
| Number of voidings/ day | 5.5714± 0.48181 | 3.2381± 0.49038 | - 2.33333 | t: 17.863 P= 0.000 |
| Number of leakage episodes/day | 2.5810± 0.57586 | 2.190± 0.51177 | - 0.39048 | t: 2.262 P= 0.035 |
| Urodynamic Parameters | ||||
| Maximum bladder capacity (cc) | 289.333± 6.05255 | 366.2857± 28.11075 | 76.95238 | 11.475 P= 0.000 |
| Volume at 1st contraction(cc) | 159.1905± 15.89849 | 245.2381± 39.19427 | 86.04762 | t: 10.334 P= 0.000 |
| Residual volume (cc) | 60.8095± 1.74983 | 86.5238± 5.63577 | 25.71429 | t: 19.801 P= 0.000 |
| Group Parameters | Ponstan treatment | |||
| Pre- use | Post- use | Mean differences | Significance* | |
| Mean ± Sd | Mean ± Sd | |||
| Clinical Parameters | ||||
| Number of voidings/ day | 5.5714± 0.48181 | 3.2381± 0.49038 | - 2.33333 | t: 17.863 P= 0.000 |
| Number of leakage episodes/day | 2.5810± 0.57586 | 2.190± 0.51177 | - 0.39048 | t: 2.262 P= 0.035 |
| Urodynamic Parameters | ||||
| Maximum bladder capacity (cc) | 289.333± 6.05255 | 366.2857± 28.11075 | 76.95238 | 11.475 P= 0.000 |
| Volume at 1st contraction(cc) | 159.1905± 15.89849 | 245.2381± 39.19427 | 86.04762 | t: 10.334 P= 0.000 |
| Residual volume (cc) | 60.8095± 1.74983 | 86.5238± 5.63577 | 25.71429 | t: 19.801 P= 0.000 |
The above results concerning NSAIDs were comparable to results of similar previous studies 9- 11.
Furthermore, significant differences were found among study’s groups regarding their response for treatment represented by improvement in clinical and urodynamic parameters (P<0.05) (Figure 1-5), and such differences were illustrated to be attributed significantly to the use of Solifenacine; as Solifenacine treated groups show more improvement scales as compared with Nifidipine and Ponstan groups regarding majority of clinical and urodynamic parameters (P< 0.05) (Table 4).
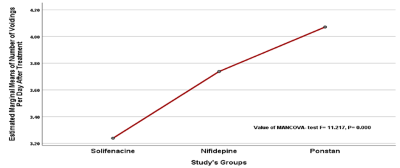
Figure 1
Comparison between study’s groups regarding treatment regime on number of voidings per day
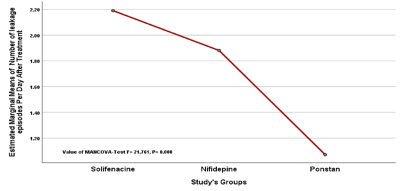
Figure 2
Comparison between study’s groups regarding treatment regime on number of leakage episodes per day
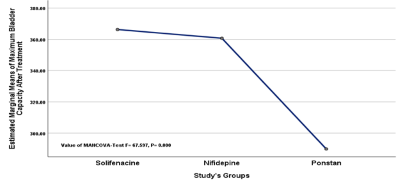
Figure 3
Comparison between study’s groups regarding treatment regime on maximum bladder capacity
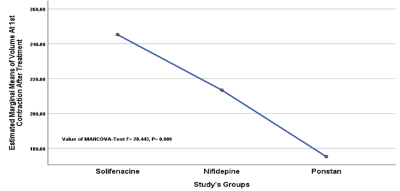
Figure 4.
Comparison between study’s groups regarding treatment regime on volume at 1st contraction
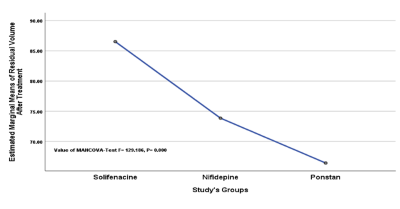
Figure 5
Comparison between study’s groups regarding effect on residual volume
| Patients’ Parameters | Study’s groups Comparison | Mean difference | P -value | 95% C.I. |
| Number ofvoidings/ day | Solifenacine -- Nifidepine | 0.5000 | 0.024* | 0.0554 - 0.9446 |
| Solifenacine -- Ponstan | 0.8333 | 0.000** | 0.3888 - 1.2779 | |
| Nifidepine -- Ponstan | 0.3333 | 0.179 | -0.1112 - 0.7779 | |
| Number ofLeakageepisodes/day | Solifenacine -- Nifidepine | 0.3095 | 0.218* | -.1302 - 0.7492 |
| Solifenacine -- Ponstan | 1.1190 | 0.000** | 0.6793 - 1.5588 | |
| Nifidepine -- Ponstan | 0.8095 | 0.000** | 0.3698 - 1.2492 | |
| Maximum bladder capacity | Solifenacine -- Nifidepine | 5.5714 | 0.750 | -12.8257 - 23.9686 |
| Solifenacine -- Ponstan | 76.4286 | 0.000** | 58.0314 - 94.8257 | |
| Nifidepine -- Ponstan | 70.8571 | 0.000** | 52.4600 - 89.2543 | |
| Volume at 1stcontraction | Solifenacine -- Nifidepine | 31.8571 | 0.005* | 8.5291 - 55.1852 |
| Solifenacine -- Ponstan | 70.0000 | 0.000** | 46.6720 - 93.3280 | |
| Nifidepine -- Ponstan | 38.1429 | 0.001* | 14.8148 - 61.4709 | |
| Residual volume | Solifenacine -- Nifidepine | 12.6667 | 0.000** | 9.4932 - 15.8401 |
| Solifenacine -- Ponstan | 20.0952 | 0.000** | 16.9218 - 23.2687 | |
| Nifidepine -- Ponstan | 7.4286 | 0.000** | 4.2551 - 10.6020 |
Those findings support the findings of animal studies12, however in humans few focused on the ability of CCBs in reducing the irritative symptoms and the results produced were mixed13,14.
Moreover, no significant differences were found among study’s groups regarding developing adverse effects causing withdrawals (P> 0.05) (Table 5).
| Study’s Groups | Withdrawal (side effect) | |||
| Yes | No | |||
| No. | % | No. | % | |
| Solifenacine | 7 | 33.3 | 14 | 66.7 |
| Nifidepine | 6 | 28.6 | 15 | 71.4 |
| Ponstan | 3 | 14.3 | 18 | 85.7 |
| Total | 16 | 25.4 | 47 | 74.6 |
 : 2.178, df: 2, P: 0.337
: 2.178, df: 2, P: 0.337
Conclusion
In trying to search for alternatives of anticholinergics for the treatment of urge incontinence, to avoid the inherent risks of appreciable peripheral and central adverse effects that may be debilitating in susceptible patients; our results demonstrated that CCBs seem to be having some beneficial effects, with a different adverse effect profile and so may be used to complement a lower dose of anticholinergic drug; on the other hand, NSAIDs were having minimal effect on response. One of the expected problems is blood pressure disturbance though this can be tailored by measuring patient's blood pressure before allocated to any treatment.
Competing interests
Authors declare that no potential conflicting of interests is exist.
Funding
This study is self-funding project.
Acknowledgments
The authors would like to thanks all women who participated voluntarily in this study.
References
1. Urinary incontinence: An inevitable part of aging? National Poll on healthy aging. 2018. Downloaded from: https://www.healthyagingpoll.org/sites/default/files/2018-11/NPHA_Incontinence-Report_FINAL-110118.pdf
2. Reisacher SL, MacDonald BC, Tallman EF, West JD, Farlow MR, Unverzagt FW, et al. Association between anticholinergic medication and cognition, brain metabolism and brain atrophy in cognitively normal older adults. JAMA Neurol. 2016; 73(6):721-32
3. Talbot JA, Halesk E, and Yudofsky SC. The American Pyschiatric Press textbook. Washington DC: American Pyschiatric Press, Inc.1988. Available at: https://www.worldcat.org/title/american-psychiatric-press-textbook-of-psychiatry/oclc/17353807
4. Lieberman JA. Managing anticholinergic side effects. Primary Care Companion to the Journal of Clinical Psychiatry. 2004; 6(suppl 2): 20–23. Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC487008/
5. Andersson KE, Arner A. Urinary bladder contraction and relaxation: Physiology and Pathophysiology. Physiol Rev. 2004 Jul;84(3):935-86. doi: 10.1152/physrev.00038.2003.48: 935 -986
6. Gruenefelder J, McGuire EJ, Faeeber GJ. Acute urinary retention associated with the use of cyclooxygenase – 2 inhibitors [case report]. J Urol. 2002; 168 (3): 1106. doi: 10.1016/S0022-5347(05)64591-0
7. Verhamme KMC, Dieleman JP, Van Wijk MAM, Der Lei JV, Bosch JLH, Stricker BHC, et al. Nonsteroidal anti-inflammatory drugs and increased risk of Acute Urinary Retention. Arch Internet Med J. 2005; 165(13): 1547-1551. doi:10.1001/archinte.165.13.1547
8. Pinnock C, Marshall VR. Troublesome lower urinary tract symptoms in the community: a prevalence study. Med J Aust. 1997; 167(2): 72-5. doi: 10.5694/j.1326-5377.1997.tb138783.x.
9. Gates MA, Hall SA, Chiu GR, Kupelian V, fitzgerald MP, Link CL, et al. Nonsteroidal anti-inflammatory drug use and lower urinary tract symptoms: results from the Boston Area Community Health survey. American Journal of Epidemilogy. 2011; 173(9): 1022-31. doi: 10.1093/aje/kwq473
10. Antonelli D, Koltun B. Excessive nifedipine diuretic effect. Br Med J. 1984; 288: 760. Available at: https://scholar.google.com/scholar_lookup?title=Excessive%20nifedipine%20diuretic%20effect%20%5Bletter%5D&journal=BMJ&volume=288&publication_year=1984&author=Antonelli%2CD&author=Koltun%2CB
11. Mattiasson A, Ekstorm B, Anderson KE. Effects of Intravesical instillation of verapamil in patients with detrusor hyperactivity. J Urol. 1989; 141(1):174-7. doi: 10.1016/s0022-5347(17)40635-5.
12. Frochlich G, Burmeister S, Wiedemann A, Bulitta M. Intravesical instillation of trospium chloride, oxybutinin and verapamil for relaxation of bladder deteusor muscle. A placebo controlled randomized clinical test. Arzneimittelforscung. 1998; 48(5): 486-91. Available at: https://pubmed.ncbi.nlm.nih.gov/9638316/
13. Hughes JD, Coles MA, Joyce A. Calcium channel blocker associated lower urinary tract symptoms in males: An Australian retrospective observational study. Qual prim Care. 2011; 19(4): 223-31. Available at: https://pubmed.ncbi.nlm.nih.gov/21902901/
Australian statistics on medicine 2008. Department of Health and aging. Canberra (Australia). Australian Government. 2009; 74 – 75. Available at: https://www.pbs.gov.au/info/statistics/asm/asm-2008