Febrile neutropenia management in pediatric onco-hematologic patients: a systematic review
Manejo da neutropenia febril em pacientes pediátricos onco-hematológicos: uma revisão sistemática
Manejo de la neutropenia febril en pacientes pediátricos oncohematológicos: una revisión sistemática
Febrile neutropenia management in pediatric onco-hematologic patients: a systematic review
Revista de Epidemiologia e Controle de Infecção, vol. 11, núm. 1, pp. 46-53, 2021
Universidade de Santa Cruz do Sul

Esta obra está bajo una Licencia Creative Commons Atribución 4.0 Internacional.
Recepción: 12 Octubre 2019
Aprobación: 23 Agosto 2020
Abstract: Background and Objectives: cancer cases are gradually increasing, and most treatments still cause several adverse reactions, such as myelosuppression. When neutrophils decline, febrile neutropenia (FN) can be triggered, considered an oncological emergency, leaving patients susceptible to infections. Therefore, it is necessary to determine the best treatment, seeking to reduce the risk of complications. The purpose of this review is to identify, in literature, randomized clinical studies that compare different treatments for FN in pediatric onco-hematological patients. Content: a systematic search was carried out on the PubMed database, for randomized clinical studies, from 2009 to 2019, in English, using “Febrile Neutropenia”, “Pediatric”, and “Therapeutics” as descriptors. A total of 233 articles were found, seven of which were selected for review. The most described antimicrobial for FN treatment was Piperacillin/Tazobactam (PIP/TAZ) and its use is justified by its spectrum of action to cover the most frequent microorganisms in patients with FN. The possibility of using oral antimicrobials may be an alternative and should be analyzed. The description of the risk classification criteria is essential to guide the therapy, and new tools, such as the stewardship, add safety to patient care. Conclusion: the most used antimicrobial to treat FN was PIP/TAZ, and the establishment of standardized risk classification scores in pediatric onco-hematological patients is essential to guide clinical management in FN treatment.
Keywords: Febrile Neutropenia, Pediatrics, Therapy.
INTRODUCTION
It is estimated that 420 thousand new cases of cancer will occur in Brazil for the 2018-2019 biennium, excluding non-melanoma skin cancer from this number. Considering that the median percentage of childhood and juvenile tumors observed is 3%, it is expected that 12,500 new cases of cancer will occur in children and adolescents (up to 19 years old). The Southeast and Northeast regions are those with the highest numbers, 5,300 and 2,900, respectively, followed by the Center-West (1,800), South (1,300) and North (1,200)1.
Despite advances in cancer treatment, the main drugs used to treat neoplasms, hematology and solid tumors still cause numerous adverse reactions. One of the main ones that competes at great risk to patients is myelosuppression, characterized by a decrease in the elements of the immune system, leaving patients exposed to various infections. When there is a decline in neutrophils, the first line of defense against some pathogens, it can trigger febrile neutropenia (FN), considered an oncological emergency2.
The severity of FN can vary due to the type and cycle of therapy, type of cancer, sex and clinical conditions of patients. The incidence of FN in the United States is estimated at 60,294 cases per year, 7.83 cases per 1,000 cancer patients, and 43.3 cases per 1,000 hematological tumor patients. The epidemiology of FN is related to some factors, which may be responsible for 50% of deaths in patients receiving chemotherapy for solid tumors and 75% for leukemias. In relation to Brazil, there are no general data; however, we can use as a basis the results of a study carried out in a hospital in northeastern Brazil with onco-hematological children and adolescents, which out of 180 occurrences, 87 were FN, giving rise to the 74 cases of infection reported in the study.2-4
According to the guideline published by the European Society for Medical Oncology (ESMO) in 2016, FN is defined as oral temperature ˃ 38.3ºC or two consecutive measurements ˃ 38.0ºC for 2h and absolute neutrophil count (ANC) of 500 cells/mm³, or an expectation that it will decrease to below 500 cells/mm³ 5. When FN is detected, the rapid onset of broad-spectrum empirical antibiotics is necessary, as the permanence of patients with FN can lead to delay in treatment, which directly or indirectly affects morbidity and mortality6.
Cancer patients have a higher risk of infection, not only due to chemotherapy treatment that induces immunosuppression and neutropenia, but also due to hypogammaglobulinemia and loss of normal physiological barriers. This increases the risk of infections by bacteria, viruses, fungi and parasites, as well as complications or spread of common pathogens from normal flora and latent viral infections7 .
The clinical identification of infections can contribute to the diagnosis in more than 30% of the cases of FN and thus guide the treatment. However, a relevant portion (10-60%) of FN in patients is treated due to fever of unknown origin, without elucidating the pathogen8
The purpose of this review is to identify, in the literature, randomized clinical trials that compare different treatments for FN in pediatric onco-hematological patients.
METHODS
The question used to guide the research was: what is the most used therapy to treat FN in pediatric onco-hematological patients?
This is a literature review based on PICO strategy. The population consisted of pediatric patients with solid or hematological tumors who developed FN and who needed treatment with antimicrobial or antifungal agents. They were randomized to studies that aimed to compare the effectiveness of each treatment and, with that, to define the best conduct for this population.
The search was carried out in literature through combination of “Febrile Neutropenia”, “Pediatric” and “Therapeutics” descriptors using the Boolean operators (OR and AND) and limited to the English language. These criteria were defined after a search for articles was carried out in other databases, such as Scielo and LILACS, in Brazilian Portuguese, English and Spanish, not finding studies that fit the defined criteria, so it was decided to include only the articles found on PubMed.
Articles published from 2009 to 2019, a randomized clinical trial comparing treatments for FN in pediatric onco-hematological patients (maximum age up to 18 years) were included. Review articles, guidelines, case reports, non-pediatric population (over 18 years old), duplicate articles, which addressed FN prophylaxis or which did not address the comparison of treatments for FN were excluded. The search was limited to pediatric onco-hematological patients, as information on treatment and management of FN in this population is scarce when compared with adult patients, with no standard risk classification score.
After selecting the articles by two reviewers screened using the keywords, the titles and abstracts were read, focusing on their methodology. The data of the selected articles were compiled and presented in a table, according to the objectives of this review.
RESULTS AND DISCUSSION
The search in the database resulted in a total of 233 articles that met the descriptors used. Of these, 217 were excluded after reading the summary. A total of 16 articles were selected for detailed analysis. Nine articles were excluded because they did not address the comparison between treatments for FN in randomized controlled trials. Thus, seven articles presented information relevant to the research objective for this review, as described in Figure 1.
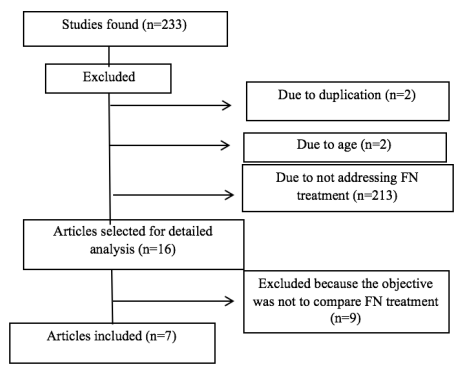
Figure 1
Flowchart of articles selected for review.
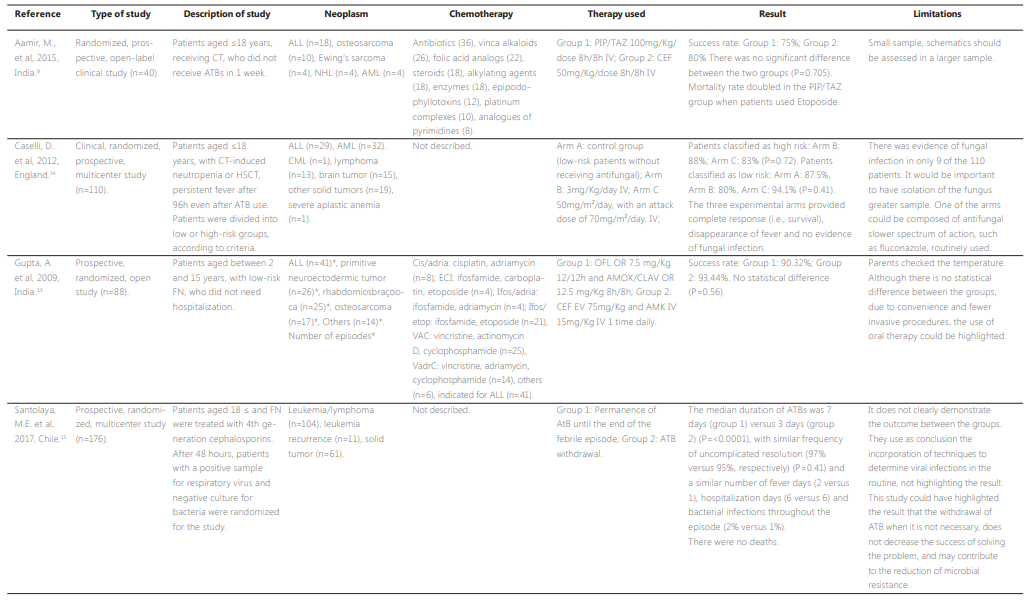
Of the seven articles selected, five sought to compare antimicrobial regimens, an antifungal regimen and a treatment for viral infections.9-15 In clinical practice, most infections that affect patients with FN are caused by bacteria and because of this, studies mainly seek to define the best antimicrobial scheme (ATB) to be used in this clinical situation. Only one article used a different definition for FN, characterized by ANC of ≤ 1000 cells/mm³, the others used the ESMO criterion.10The most frequent ATB in the regimens was piperacillin/tazobactam (PIP/TAZ) and amoxacillin/clavulanate (AMOX/CLAV).9-13 Only two articles described the chemotherapeutic drugs used in the treatment of pediatric onco-hematological patients. 9,13 The most frequent neoplasm in the cases of FN in the articles assessed was acute lymphoid leukemia, with a high prevalence of other hematological neoplasms compared to solid tumors. Three articles described the FN classification criteria, but they were not the same criteria.10,13-14 There was evidence of infection in the seven articles.9-15
FN is a clinical condition originated by chemotherapy, which can make an individual susceptible to infections. Patients’ characteristics (age, neoplasia, treatment, type of infection, performance status) need to be considered when choosing the best therapy.
Bacterial infections are generally associated with FN in onco-hematological patients, and the identification of the pathogen causing the infection is important for choosing the appropriate ATB. The use of empirical PIP/TAZ in the management of patients with FN is explained by its spectrum of action, which includes the microorganisms most associated with FN in pediatric patients (Enterobacteriaceae (30%) and Coagulase-negative staphylococci (24%), and Pseudomonas aeruginosa (5%)).16,17
No statistically significant differences were found in clinical outcomes when compared to therapy with PIP/TAZ versus cefepime, drugs routinely used in bacterial infections in neutropenic patients. However, an increase in mortality was observed in the PIP/TAZ group, when patients have a previous history of treatment with etoposide, a chemotherapy used in several protocols for childhood and juvenile neoplasms. This reinforces the importance of knowing patients’ history and assessing the medications they use so that the choice of ATB is adequate, as this information may interfere with patients’ clinical evolution.9,18
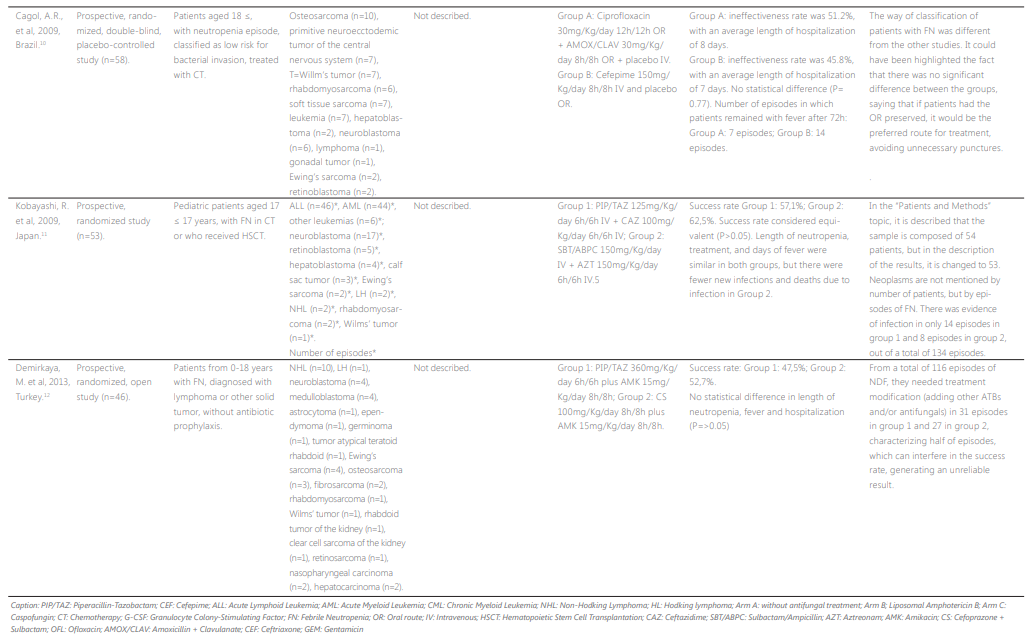
The combination of PIP/TAZ with other ATBs can help to improve the clinical condition of patients with FN, however, it is essential to define the best association. One study found that sulbactam/ampicillin associated with aztreonam (62.5%) is equivalent to or higher than PIP/TAZ associated with ceftazidime, (57.1%), with fewer cases of new infections and deaths.11 However, another study that assessed the association of
Part 1
Part 2
PIP/TAZ and amikacin compared to the association of cefoperazone/sulbactam and amikacin, found no statistical difference between the groups (47.5% versus 52.6%, respectively). The search for different combinations of ATB is essential, especially in cases of microbial resistance or non-response to PIP/TAZ, requiring the use of other effective alternatives, such as schemes with broad-spectrum ATB that are not routinely used.12
In practice, the concern with the rapid evolution of the infection, makes the use of ATB often occur empirically. In this context, the use of PIP/TAZ for the empirical treatment of FN is of great value, as demonstrated in a study, in which the results found showed that the decrease in body temperature in patients occurred in 62.5% on the fourth day, 57.1% on the seventh day and 75.0% at the end of treatment, suggesting that the applicability of the empirical form of PIP/TAZ is satisfactory for the resolution of FN.19
Comparing the use of oral ATB with intravenous ATB in patients with FN can be an effective alternative, especially for those who have a low risk of infection and with the opportunity to perform treatment at home. The use of oral ATB can facilitate treatment in pediatrics, allowing greater autonomy for family members and patients, decreasing the number of punctures, in addition to being also considered the alternative of hospital discharge, reducing the possibility of exposure to nosocomial infection and providing reduction of hospitalization costs.13,17,20–22
However, it is important to classify the severity of the risk of infection for this patient, considering several factors that directly affect the conduct to be taken. There is no internationally accepted risk stratification, requiring each hospital to choose a validated stratification and adapt to its reality. Risk score for children with FN was validated in a hospital in India and compared with other models already published. The authors concluded that this model demonstrated applicability, however, a multicenter study is needed to verify the possibility of employment in practice in developed countries, in which health conditions differ from those in development, such as malnutrition.23-25
Despite the fact that most infections in FN patients are of microbial origin, other etiologic agents such as fungi and mycobacteria can also be the cause, especially in more severe and prolonged episodes of neutropenia, in a period longer than 10 days of hospitalization. 26
Fungi can be considered responsible for 30-40% of infections after the fifth day of neutropenia, the most common being Candida albicans and Aspergillus, but there is an increase in documentation related to Non-albicans candida. A study compared the effectiveness of two antifungals, caspofungin and amphotericin B liposomal in patients divided into low and high-risk groups. The results found, demonstrated that both treatments are effective for management of fungal infections in patients with FN. However, it is important to develop studies that compare broad-spectrum antifungals with those of minors, most used in routine.14,27
In addition to bacterial or fungal infections, viral infections, especially respiratory infections, appear as potential pathogens in this specific population. Viral respiratory infections can induce morbidity and mortality, being detected in more than 57% of FN episodes in children with cancer. 28,29
It is essential to determine the type of pathogen that causes the infection for the appropriate choice of therapy, since ATBs are ineffective in viral infections and their indiscriminate use can induce antimicrobial resistance. The suspension of ATB after confirming the results of negative cultures, does not interfere in the final outcome, with the resolution rates in both groups without significant difference (97% in the group with ATB and 95% in the group without ATB). 15
The use of adjuvant therapies to decrease patients’ neutropenia time can be considered a useful tool, such as the use of granulocyte growth stimulating factors (G-CSF), adopted in the onco-hematological routine. Its use contributes to the increase of ANC and, with that, it decreases the time that patients would be susceptible to opportunistic infections, also reducing the time of use of ATB.30,31
The guidelines for initial management of FN in children and adolescents with cancer, published by the Brazilian Society of Pediatrics, indicate the initial empirical use of monotherapy with B-lactam antipseudomonal, fourth-generation cephalosporin or carbapenem.32These indications corroborate the studies included in this review, in which most studies used PIP/TAZ and/or cefepime as ATB compared to other ATB.
Although the empirical use of PIP/TAZ is widespread in the treatment of FN in pediatric onco-hematological patients, it is necessary to consider the infectious focus and modify the therapy as necessary. For this, it can be considered that the stewardship program, a term used to describe an integrated strategy that seeks to reduce the irrational use of ATB, helps in choosing the best treatment. This program has acquired some objectives over the years, including cost reduction, optimization of therapeutic results and reduction of antimicrobial resistance, associated with tools that are characterized by the restrictions of ATB classes, rotation in the use of ATB, support in clinical decisions, education of the team of prescribers.33,34
Recent systematic reviews and meta-analysis on stewardship differ in some points, however they converge in the fact that the application of this tool reduces the occurrence of nosocomial infections caused by drug-resistant bacteria. Moreover, it increases control and reduces the time of using ATB, without increasing the mortality rate, reducing hospital length of stay.35,36
Establishing protocols of the stewardship program in pediatric onco-hematological patients with FN, can be a useful resource for decreasing microbial resistance and the unnecessary use of broad-spectrum ATB. As they are patients who often use protocols that cause severe myelosuppression, when they have neutropenia, they seek to protect themselves from serious infections, but that can often be treated simply, and in some cases, on an outpatient basis.
CONCLUSION
Treatment with ATB was the most used, with PIP/TAZ being the most frequent in the regimens, followed by AMOX/CLAV. There was no statistically significant difference between treatment outcomes. However, length of stay, ANC, presence of fever and other clinical conditions must be taken into account when choosing the most appropriate ATB. Viral and fungal infections need to be considered to determine treatment with the correct class of drugs, and to avoid the irrational use of ATB. The establishment of standardized risk classification scores in pediatric onco-hematological patients is essential to guide clinical management in FN treatment.
REFERENCES
1. Instituto Nacional de Câncer José Alencar. Inca estimativa 2018[Internet]. Rio de Janeiro: INCA, 2017 [citado out 2019]. Available from: http://www1.inca.gov.br/estimativa/2018/estimativa-2018.pdf
2. Rasmy A, Amal A, Fotih S, et al. Febrile Neutropenia in Cancer Patient: Epidemiology, Microbiology, Pathophysiology and Management. J Cancer Prev Curr Res .2016 Aug 23;5(3). 00165. http://dx.doi.org/10.15406/jcpcr.2016.05.00165
3. Stephanos K, Picard L. Pediatric Oncologic Emergencies. Emerg Med Clin North Am. 2018;36(3):527–35. http://dx.doi.org/10.1016/j.emc.2018.04.007
4. Silva SEM, Silva IB. Epidemio. Perfil das urgências onco-hematológicas em crianças e adolescentes atendidos em um hospital público de referência. Enferm Bras. 2017;16(5):293–302. http://dx.doi.org/10.1017/CBO9781107415324.004
5. Klastersky J, Naurois J, Rolston K, Rapoport B, Maschmeyer G, Aapro M, et al. Management of febrile neutropaenia: ESMO clinical practice guidelines. Ann Oncol. 2016;27(October 2008):v111–8. http://dx.doi.org/10.1093/annonc/mdw325
6. Lehrnbecher T, Robinson P, Fisher B, Alexander S, Ammann RA, Beauchemin M, et al. Guideline for the management of fever and neutropenia in children with cancer and hematopoietic stem-cell transplantation recipients: 2017 update. J Clin Oncol. 2017;35(18):2082–94. http://dx.doi.org/10.1200/JCO.2016.71.7017
7. Silva D; Andrea MLM, Lins AGN. Emergências oncológicas. In: Tratado de pediatria. 4th ed. 2017. p. 1564–1470.
8. Kara Ö1, Zarakolu P, Aşçioğlu S, Etgül S, Uz B, Büyükaşik Y AM. Epidemiology and emerging resistance in bacterial bloodstream infections in patients with hematologic malignancies. Infect Dis (Auckl). 2015;47(10):686–93. http://dx.doi.org/10.3109/23744235.2015.10511053
9. Aamir M, Abrol P, Sharma D, Punia H. A clinical evaluation of efficacy and safety of cefepime monotherapy versus piperacillin-tazobactam in patients of paediatric age group with febrile neutropenia in a tertiary care centre of north India. Trop Doct. 2016;46(3):142–8. http://dx.doi.org/10.1177/0049475515617571
10. Cagol ÂR, Castro Junior CG, Martins MC, Machado AL, Ribeiro RC, Gregianin LJ, et al. Oral vs. intravenous empirical antimicrobial therapy in febrile neutropenic patients receiving childhood cancer chemotherapy. J Pediatr (Rio J). 2009;85(6):531–5. http://dx.doi.org/10.2223/JPED.1956
11. Kobayashi R, Sato T, Nakajima M, Kaneda M, Iguchi A. Piperacillin/tazobactam plus ceftazidime versus sulbactam/ampicillin plus aztreonam as empirical therapy for fever in severely neutropenic pediatric patients. J Pediatr Hematol Oncol. 2009;31(4):270–3. http://dx.doi.org/10.1097/MPH.0b013e31819daf4a
12. Demirkaya M, Çelebi S, Sevinir B, Hacimustafaoglu M. Randomized comparison of piperacillin-tazobactam plus amikacin versus cefoperazone-sulbactam plus amikacin for management of febrile neutropenia in children with lymphoma and solid tumors. Pediatr Hematol Oncol. 2013;30(2):141–8. http://dx.doi.org/10.3109/08880018.2012.756565
13. Gupta A, Swaroop C, Agarwala S, Pandey RM, Bakhshi S. Randomized control trial comparing oral amoxicillin-clavulanate and ofloxacin with intravenous ceftriaxone and amikacin as outpatient therapy in pediatric low-risk febrile neutropenia. J Pediatr Hematol Oncol. 2009;31(9):635–41. http://dx.doi.org/10.1097/MPH.0b013e3181acd8cd
14. Caselli D, Cesaro S, Ziino O, Ragusa P, Pontillo A, Pegoraro A, et al. A prospective, randomized study of empirical antifungal therapy for the treatment of chemotherapy-induced febrile neutropenia in children. Br J Haematol. 2012;158(2):249–55. http://dx.doi.org/10.1200/JCO.2016.71.7017
15. Santolaya ME, Alvarez AM, Acuña M, Avilés CL, Salgado C, Tordecilla J, et al. Efficacy and safety of withholding antimicrobial treatment in children with cancer, fever and neutropenia, with a demonstrated viral respiratory infection: a randomized clinical trial. Clin Microbiol Infect. 2017;23(3):173–8. http://dx.doi.org/10.1016/j.cmi.2016.11.001
16. Mikulska M, Viscoli C, Orasch C, Livermore DM, Averbuch D, Cordonnier C, et al. Aetiology and resistance in bacteraemias among adult and paediatric haematology and cancer patients. J Infect. 2014;68(4):321–31. http://dx.doi.org/10.1016/j.jinf.2013.12.006
17. Santos, L., Torriani, M.S., Barros E. Medicamentos na prática da farmácia clínica. 1ª ed. Porto Alegre: Artmed; 2013
18. Patel P, Lavoratore SR, Flank J, Kemp M, Vennettilli A, Vol H, et al. Chemotherapy-induced nausea and vomiting control in pediatric patients receiving ifosfamide plus etoposide : a prospective, observational study. 2019. http://dx.doi.org/10.1007/s00520-019-04903-0
19. Tamura K, Akiyama N, Kanda Y, Saito M. Efficacy and safety of tazobactam/piperacillin as an empirical treatment for the patients of adult and child with febrile neutropenia in Japan. J Infect Chemother. 2015;21(9):654–62. http://dx.doi.org/10.1016/j.jiac.2015.05.00920
20. Loeffen EAH, Poele EM, Tissing WJE, Boezen HM, Bont ESJM. Very early discharge versus early discharge versus non-early discharge in children with cancer and febrile neutropenia. Cochrane Database Syst Rev. 2016;2016(2). http://dx.doi.org/10.1002/14651858.CD008382.pub2
21. Gil-Veloz M, Pacheco-Rosas DO, Solórzano-Santos F, Villasís-Keever MA, Betanzos-Cabrera Y, Miranda-Novales G. Early discharge of pediatric patients with cancer, fever, and neutropenia with low-risk of systemic infection. Bol Med Hosp Infant Mex. 2018;75(6):352–7. http://dx.doi.org/10.24875/BMHIM.18000015
22. Miedema KGE, Tissing WJE, Abbink FCH, Ball LM, Michiels EMC, Van Vliet MJ, et al. Risk-adapted approach for fever and neutropenia in paediatric cancer patients - A national multicentre study. Eur J Cancer. 2016;53:16–24. http://dx.doi.org/10.1016/j.ejca.2015.10.065
23. Silva DB, Barreto JHS, Cordoba JC, et al. Diretrizes Para O manejo inicial da neutropenia febril, após quimioterapia, em crianças e adolescentes com câncer. Soc Bras Pediatr. 2018.Disponível em : https://www.sbp.com.br/fileadmin/user_upload/Oncologia_-_20942d-Diretrizes_manejo_inicial_neutropenia_febril_pos_quimio__003_.pdf
24. Rivas-Ruiz R, Villasis-Keever M, Miranda-Novales G, Castelán-Martínez OD, Rivas-Contreras S. Outpatient treatment for people with cancer who develop a low-risk febrile neutropaenic event. Cochrane Database Syst Rev. 2019;2019(3). http://dx.doi.org/10.1002/14651858.CD009031.pub2
25. Das A, Trehan A, Oberoi S, Bansal D. Validation of risk stratification for children with febrile neutropenia in a pediatric oncology unit in India. Pediatr Blood Cancer. 2017;64(6):1–8. http://dx.doi.org/10.1002/pbc.26333
26. Hagag AA, Hassan SM, Elgamasy MA, Afifi IK. Study of Common Bacterial and Fungal Pathogens in Children with Hematological Malignancies during Febrile Neutropenia: Single Center Egyptian Study. Infect Disord Drug Targets . 2016;16(1):54–62. http://dx.doi.org/0.2174/1871526516666151230124333
27. Kumar J, Singh A, Seth R, Xess I, Jana M, Kabra SK. Prevalence and Predictors of Invasive Fungal Infections in Children with Persistent Febrile Neutropenia Treated for Acute Leukemia – A Prospective Study. Indian J Pediatr. 2018;85(12):1090–5. http://dx.doi.org/10.1007/s12098-018-2722-0
28. Benites ECA, Cabrini DP, Silva ACB, Silva JC, Catalan DT, Berezin EN, et al. Acute respiratory viral infections in pediatric cancer patients undergoing chemotherapy. J Pediatr (Rio J). 2014;90(4):370–6. http://dx.doi.org/10.1016/j.jped.2014.01.006
29. Wong-Chew RM, Espinoza MA, Taboada B, Aponte FE, Arias-Ortiz MA, Monge-Martínez J, et al. Prevalence of respiratory virus in symptomatic children in private physician office settings in five communities of the state of Veracruz, Mexico. BMC Res Notes. 2015;8(1):1–8. http://dx.doi.org/10.1186/s13104-015-1239-0
30. Dwivedi P, Greis KD. Granulocyte colony-stimulating factor receptor signaling in severe congenital neutropenia, chronic neutrophilic leukemia, and related malignancies. Exp Hematol. 2017;46:9–20. http://dx.doi.org/10.1016/j.exphem.2016.10.008
31. Younis T, Rayson D, Jovanovic S, Skedgel C. Cost-effectiveness of febrile neutropenia prevention with primary versus secondary G-CSF prophylaxis for adjuvant chemotherapy in breast cancer: a systematic review. Breast Cancer Res Treat. 2016;159(3):425–32. http://dx.doi.org/10.1007/s10549-016-3954-1
32. Akova M. Antimicrobial Stewardship in Hematology Patients 1st ed. Vol. 2, Antimicrobial Stewardship. Elsevier Inc.; 2017. 205–217. http://dx.doi.org/10.1016/B978-0-12-810477-4.00017-9.33
33. Sociedade Brasileira de Pediatria. Diretrizes para o manejo inicial da neutropenia febril, após quimioterapia, em crianças e adolescentes com câncer. Departamento Científico de Oncologia. 2018; (2).
34. Rice LB. Antimicrobial Stewardship and Antimicrobial Resistance. Med Clin North Am [Internet]. 2018;102(5):805–18. http://dx.doi.org/10.1016/j.mcna.2018.04.004
35. Davey P, Scott CL, Brown E, Charani E, Michie S, Ramsay CR, et al. Interventions to improve antibiotic prescribing practices for hospital inpatients (updated protocol). Cochrane Database Syst Rev. 2017;2017(2). http://dx.doi.org/10.1002/14651858.CD011236.pub2
36. Karanika S, Paudel S, Grigoras C, Kalbasi A, Mylonakis E. Systematic review and meta-analysis of clinical and economic outcomes from the implementation of hospital-based antimicrobial stewardship programs. Antimicrob Agents Chemother. 2016;60(8):4840–52. http://dx.doi.org/10.1128/AAC.00825-16