Artigo Original
Severe COVID-19 in the context of a vaccinated population: Case-control study
COVID-19 grave no contexto de uma população vacinada: estudo caso-controle
COVID-19 grave en el contexto de una población vacunada: Estudio de casos y controles
Severe COVID-19 in the context of a vaccinated population: Case-control study
Revista de Epidemiologia e Controle de Infecção, vol. 14, núm. 1, pp. 66-74, 2024
Universidade de Santa Cruz do Sul

Esta obra está bajo una Licencia Creative Commons Atribución 4.0 Internacional.
Recepción: 13 Diciembre 2023
Aprobación: 16 Febrero 2024
Abstract: Background and Objectives: Since its appearance in 2019, multiple risk factors have been identified for presenting a severe form of COVID-19 and different vaccines have also been developed to prevent severe manifestations. However, despite a vaccination history, some cases progress to complications or even death. The objective of this study was to determine the strength of the association between the severity of COVID-19 and the history of vaccination in patients treated at a public reference hospital in Mexico City. Methods: This was a non-experimental, retrospective, and analytical epidemiological study of cases and controls. The study population was people treated at a concentration hospital for COVID-19 care between July 1, 2021, and June 30, 2022, in Mexico City. Results: 132 participants (44 cases and 88 controls) were included in the study. The risk factors most strongly associated with COVID-19 severity were age greater than or equal to 60 years, presenting 22 breaths per minute at the first medical evaluation, systolic blood pressure greater than or equal to 140 millimeters of mercury, and a history of at least one chronic comorbidity. However, vaccination history was associated with 94% (OR 0.06) lower odds of developing severe COVID-19 compared to those without a history of vaccination, regardless of the presence of associated risk factors. Conclusion: Lacking a history of vaccination and presenting any of the identified risk factors confer higher odds of developing severe forms of the disease.
Keywords: COVID-19 vaccines, Mass vaccination, Patient acuity, Public Health.
Resumo: Justificativa e Objetivos: Desde o seu aparecimento em 2019, foram identificados múltiplos fatores de risco para a apresentação de uma forma grave de COVID-19 e foram desenvolvidas diferentes vacinas para prevenir o aparecimento de manifestações graves. No entanto, apesar de um histórico de vacinação, alguns casos podem evoluir para complicações ou mesmo para a morte. O objetivo deste estudo foi determinar a força de associação entre a gravidade da COVID-19 e o histórico de vacinação em pacientes atendidos em um hospital público de referência na Cidade do México. Métodos: Estudo epidemiológico não-experimental, retrospectivo e analítico, de casos e controles. A população do estudo foram indivíduos atendidos em um hospital de concentração para atendimento à COVID-19 entre 1 de julho de 2021 e 30 de junho de 2022, na Cidade do México. Resultados: 132 participantes (44 casos e 88 controles) foram incluídos no estudo. Os fatores de risco mais fortemente associados à gravidade da COVID-19 foram idade superior ou igual a 60 anos, apresentar 22 respirações por minuto na primeira avaliação médica, pressão arterial sistólica superior ou igual a 140 milímetros de mercúrio e histórico de pelo menos uma comorbidade crônica. No entanto, histórico de vacinação foi associado a uma probabilidade 94% (OR 0,06) menor de desenvolver COVID-19 grave em comparação com aqueles sem histórico de vacinação, independentemente da presença de fatores de risco associados. Conclusão: A ausência de histórico de vacinação e a presença de algum dos fatores de risco identificados conferem maiores probabilidades de desenvolver formas graves da doença.
Palavras-chave: Vacinas contra COVID-19, Vacinação em massa, Gravidade do paciente, Saúde Pública.
Resumen: Justificación y Objetivos: Desde su aparición en 2019, se han identificado múltiples factores de riesgo para presentar una forma grave de COVID-19 y también se han desarrollado distintas vacunas que previenen la aparición de manifestaciones de gravedad. Sin embargo, a pesar del antecedente de vacunación, algunos casos se complican o incluso fallecen. El objetivo del este estudio fue determinar la fuerza de asociación entre la gravedad de la COVID-19 con el antecedente de vacunación en pacientes atendidos en un hospital público de referencia de la Ciudad de México. Métodos: Estudio epidemiológico no experimental, retrospectivo y analítico, de casos y controles. La población de estudio fueron personas atendidas en un hospital de concentración para la atención de COVID-19 entre el 1 de julio de 2021 y el 30 de junio de 2022 en la Ciudad de México. Resultados: 132 participantes (44 casos y 88 controles) fueron incluidos en el estudio. Los factores de riesgo más fuertemente asociados con la gravedad de la COVID-19 fueron la edad mayor o igual a 60 años, presentar 22 respiraciones por minuto en la primera valoración médica, tensión arterial sistólica mayor o igual a 140 milímetros de mercurio y el antecedente de al menos una comorbilidad crónica. No obstante, el antecedente de vacunación se asoció con 94% (RM 0.06) menos posibilidades de desarrollar COVID-19 grave con respecto a aquellos sin antecedente vacunal, independientemente de la presencia de los factores de riesgo asociados. Conclusión: carecer del antecedente de vacunación y presentar alguno de los factores de riesgo identificados confieren las mayores posibilidades de presentar formas graves de la enfermedad.
Palabras clave: Vacunas contra la COVID-19, Vacunación masiva, Gravedad del paciente, Salud Pública.
INTRODUCTION
Coronaviruses (CoV) are a group of multiple viruses that commonly infect animals and humans, and can cause manifestations with a wide range of severity, from asymptomatic to fatal.1 In late 2019, the first cases of infection with SARS-CoV-2, the etiologic agent of the respiratory disease that was later named COVID-19, appeared.2,3
The World Health Organization (WHO) has established a definition of a severe case of COVID-19: “Adolescent or adult with clinical signs of pneumonia (fever, cough, dyspnea, rapid breathing) plus one of the following: Respiratory rate (RR) >30 breaths/min; severe respiratory distress; or SpO2 <90% on room air; or acute respiratory distress syndrome (ARDS), sepsis, septic shock, or death”.4
Risk factors for the development of severe COVID-19 can be studied in at least 5 different groups: Demographic factors (age and sex), symptoms (clinical manifestations), comorbidities, complications, and laboratory indicators.5
In response to the high number of severe cases of the disease, vaccines were developed to reduce the occurrence of severe cases of COVID-19. Mexico initiated its national vaccination strategy on December 24, 2020.6 The prioritization for the use of vaccines followed the recommendations of the Vaccine Technical Advisory Group (GTAV), which included 4 guiding principles: Age of individuals (over 60 years of age), comorbidities, priority groups (including health personnel), and the behavior of the epidemic.7
The vaccines used in Mexico during the study period were those developed by Pfizer-BioNTech (BNT162b2 mRNA), Moderna (mRNA-1273), AstraZeneca (ChAdOx1 nCoV-19), Centro NITsEM N. F. Gamaleya (rAd26-rAd25), CanSino (Ad5-nCoV), Sinovac (PiCoVacc) and Janssen (Ad.26.COV2.S).8The vaccination strategy avoided commercialization in order to provide equity in access to immunizations in an environment of limited availability whose access to the type of vaccines was within the framework of the COVAX initiative.9 Mexico City reached a vaccination coverage of 95% of its adult population on December 30, 2021.10 However, between July 1, 2021, and June 30, 2022, 11,858 deaths due to COVID-19 occurred in the country’s capital.11,12 This suggests that deaths continue to occur despite vaccination coverage rates approaching 100%, and it should be emphasized that death is not the only indicator of severity in COVID-19. The aim of this study was to determine the strength of the association between the severity of COVID-19 and the history of vaccination in patients assisted at a public reference hospital in Mexico City.
METHODS
A non-experimental, epidemiological, retrospective, analytical, and case-control study was conducted to determine the strength of the association between a history of vaccination against COVID-19 and the severity of COVID-19. In addition, sociodemographic, clinical, and biochemical covariates that have been associated with COVID-19 severity were analyzed to establish estimates of magnitude and significance regarding how they may influence the disease.
Study population
People treated at the General Regional Hospital #1 "Dr. Carlos Mac Gregor Sánchez Navarro" (HGR 1), during the period from July 1, 2021, to June 30, 2022. Data were collected from clinical, physical, and electronic records. Inclusion criteria were people aged ≥18 years, of any sex, who requested a medical evaluation at HGR1 during the aforementioned period and had valid medical insurance at the Mexican Social Security Institute (IMSS), with polymerase chain reaction (PCR) test or positive rapid antigenic test (RAP) for SARS-CoV-2. Exclusion criteria were evidence of receiving the vaccine against COVID-19 in a country other than Mexico and participants with habitual residence outside Mexico City. The elimination criterion was documented evidence of having had SARS-CoV-2 infection at a time prior to requesting medical care.
Case Definition: A person who meets the screening criteria and who, at the time of seeking care, presented severe COVID-19 criteria suggested by WHO.4 Control Definition: A person who meets the screening criteria and who, at the time of seeking care, did not present severe COVID-19 criteria suggested by WHO.4
The sample size for case-control studies was calculated with the aid of the Epi Info 7.2.5.0 program, considering a safety of 95% and a statistical power of 80%, as well as a ratio of 2 controls for each case. The final sample consisted of 132 participants, 44 cases and 88 controls. The type of sampling was simple random probability. The data source was secondary (clinical record).
The variables of interest were collected in a database. Descriptive analysis was performed to report frequencies, proportions, and percentages for qualitative variables; and measures of central tendency and dispersion for quantitative variables. Chi2 and Fisher's exact tests were performed to establish whether there were significant differences in proportions between cases and controls. Bivariate analysis employed Student’s t-test for independent samples or the Mann-Whitney U test to determine differences between means or between medians, respectively, and in congruence with the distributional nature of the data. Crude odds ratios (OR) were estimated with their respective 95% confidence intervals (CI) and finally, multivariate analysis was performed using multiple logistic regression. The analyses corresponding to the normality, univariate, and bivariate tests were performed with the IBM SPSS version 23 program. The multivariate analysis was performed with the STATA program (College Station, TX, USA) version 14.0. In all cases, a p-value <0.05 was considered statistically significant.
This study used secondary source data and was approved by the local ethics and research committee, under registration number R-2023-3609-002.
RESULTS
Table 1 shows the qualitative characteristics of the 132 participants in this study, grouped according to whether they belonged to the case or control group. The history of vaccination was considered present if the record explicitly mentioned it, even if it was not possible to identify the brand or specific name of the vaccine. Only 34.1% of the cases had evidence of having been vaccinated, while this history was found in 81.8% of the controls (p< 0.001). All cases were complete schedules, whose second dose was administered at least 14 days before the onset of COVID-19 symptoms.
The differences between sexes were not significant (p = 1.000) because the design and selection of participants ensured that the proportion of men and women were equal between cases and controls, considering that there were 2 controls for each case.
Most of the symptoms and comorbidities related to the severity of COVID-19 had statistically significant differences depending on the study group (p<0.05). Among complications (coagulopathy or acute kidney injury), there were no statistically significant differences.
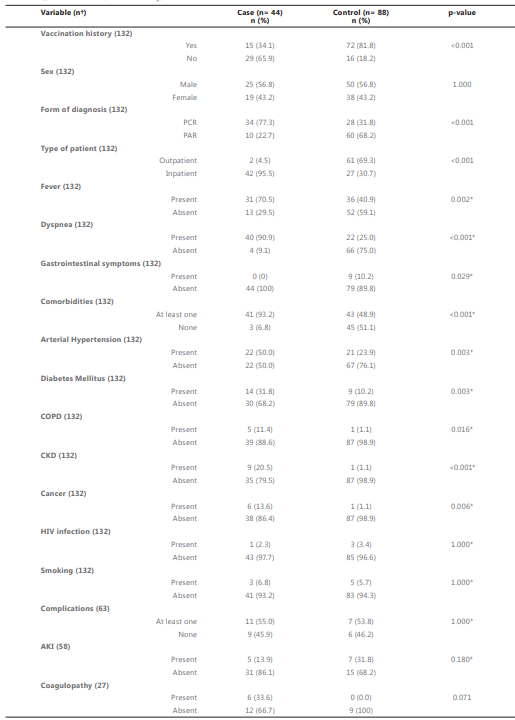
†Values may vary because the variable was not measured in all participants.
*Fisher’s exact test; COPD: Chronic Obstructive Pulmonary Disease; CKD: Chronic Kidney Disease; AKI: Acute Kidney Injury.
Table 2 shows the quantitative characteristics of the 132 participants in this study. Cases had a median age of 71 years old with an interquartile range (IQR) of 63.00 - 79.50; whereas controls had a significantly lower median age (41 years, IQR 25 - 58.5; p <0.001).
No statistically significant differences were found between the median BMI of the cases (25.64 kg/m2) and that of the controls (25.33 kg/m2; p = 0.572). Systolic blood pressure (SBP) recorded during the first medical evaluation showed significant differences (129.77 ± 13.10 mmHg) between cases and controls (115.93 ± 16.62 mmHg), p < 0.001; as did diastolic blood pressure (DBP) in cases (95, IQR 90 - 100 mmHg) compared to controls (89, RIC 71 - 97 mmHg); p = 0.003.
In the white formula, significant differences were only found in lymphocytes and eosinophils (both absolute and relative), as well as in the percentage of basophils. There was also a significant difference between the medians of the neutrophil/lymphocyte index (NLI) which was 10.90 (IQR 6.29 - 22.30) for the cases and 5.40 (IQR 2.09 - 15.13) for the controls, p = 0.019. The rest of the determinations of the white formula and the platelet count were not statistically significant.
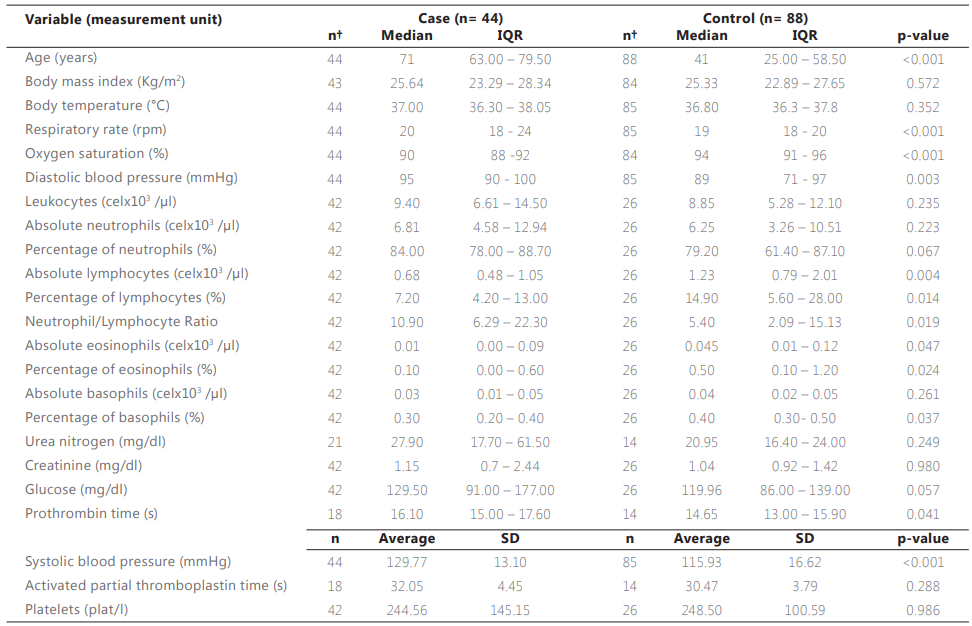
Table 3 shows the crude ORs for the different variables that were significant in Tables 1 and 2. It can be seen that, in this study, a history of vaccination is associated with 89% (95% CI 71.8 - 95.4) less chance of belonging to the group of cases (calculated as 1 - OR x 100) and, therefore, of having presented more severe manifestations of COVID-19.
Age older than 60 years was associated with an almost 17-fold increased probability of belonging to the case group (OR 16.86, 95% CI 6.55 - 43.38). A history of fever (OR of 3.44, 95% CI 1.59 - 7.47), respiratory rate (RR) ≥22 breaths per minute (rpm) (OR 34.58, 95% CI 7.54 - 158.57), an SBP ≥140 mmHg (OR 9.45 95% CI 2.88 - 30.99) and a DBP ≥90 mmHg (OR 3.07 95% CI 1.38 - 6.86) measured at the first medical assessment, were also associated with a greater chance of belonging to the case group.
The presence of at least one comorbidity increased the odds of having a severe case of COVID-19 with an OR of 14.30 (95% CI 4.12 - 49.65), as did lymphopenia (OR 4.72, 95% CI 1.65 - 13.49), eosinopenia (OR 3.00, 95% CI 1.07 - 8.43) and neutrophil/lymphocyte ≥3 (OR 3.92, 95% CI 1.14 - 13.97).
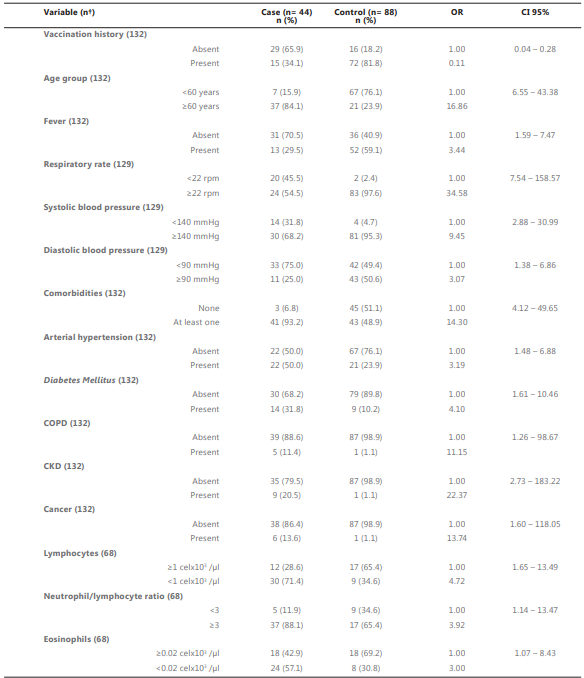
†Values may vary because the variable was not measured in all participants.
OR: Odds ratio; CI 95%: Confidence interval at 95%; COPD: Chronic Obstructive Pulmonary Disease; CKD: Chronic Kidney Disease; rpm: respirations per minute; mmHg: millimeters of mercury
Table 4 presents the multiple logistic regression model with the highest explanatory power (pseudo r2 = 0.57, n = 129) for the variables associated with severe forms of COVID-19. This model shows that a history of vaccination still reduces the chances of presenting severe COVID-19 by 94% (OR 0.06, 95% CI 0.01 - 0.28), even when adjusting for age groups, RR, SBP, and presence of comorbidity.
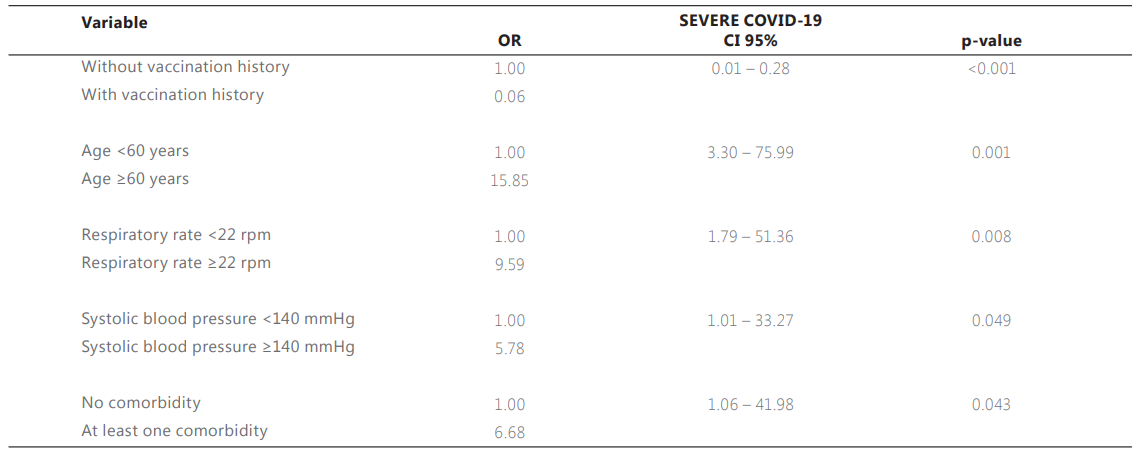
n= 129; Pseudo r2 = 0.57 Importar tabla
CI 95%: Confidence interval at 95%; rpm: respirations per minute; mmHg: millimeters of mercury
DISCUSSION
In this study, vaccination history was associated with up to 94% lower odds of developing severe COVID-19 compared to those without vaccination history, regardless of the presence of the risk factors most strongly associated with severe manifestations, highlighting the importance of vaccination against COVID-19 and the need to prioritize older adults to benefit from this strategy.
These findings are consistent with results obtained in various settings, for example, an investigation in the United States measured the joint efficacy for the prevention of severe forms of COVID-19 of two different vaccines (BNT162b2 mRNA and mRNA-1273), reporting a decrease of 86.9% (95% CI 80.4 - 91.2).13 Similar results were reported by other researchers in the United Kingdom, who determined that prior vaccination with BNT162b2 mRNA and ChAdOx1 nCoV-19 mRNA was associated with lower odds of being hospitalized among persons older than 80 years (71.4% and 80.4%, respectively).14
Similarly, a case-control study conducted in Israel reported that vaccination with two doses of BNT162b2 mRNA, with the second dose being given at least 7 days before a SARS-CoV-2 infection, was able to prevent hospitalization in 87% (95% CI 55-100) and severe disease in 92% (95% CI 75-100).15
Regarding lower-income countries, in India, receiving two doses of ChAdOx1 nCoV-19 vaccine, the last dose being at least 14 days before the onset of symptoms of SARS- CoV-2 infection, was found to be associated with 81.5% (95% CI 79.9 - 99) lower odds of moderate to severe disease.16 Meanwhile, in Argentina, a case-control study that measured the effectiveness of three different vaccines (rAd26-rAd25, ChAdOx1 nCoV-19, and BBIBP- CorV) in preventing deaths from COVID-19 in people over 60 years of age reported that these vaccines were associated with 93.1% (95% CI 92.6 - 93.5), 93.7% (95% CI 93.2 - 94.3), and 85.0% (95% CI 84.0 - 86.0) lower chances of death.17
It has been published that age over 60 years is a risk factor for severe COVID-19.1,5,18 In this study, age was statistically different between cases and controls (this study is not age-matched), which is consistent with the reported findings.
Respiratory rate greater than 30 breaths per minute (rpm) is part of the operational case definition4 and therefore, considering this cutoff point would result in significant associations due to collinearity. However, if the cutoff point is reduced to 22 rpm, a parameter considered in the q-SOFA scale,19 it is possible to determine whether less marked RR elevations may be associated with the presence of COVID-19 severity. In this work, RR ≥22 rpm was associated with greater chances of belonging to the group of cases.
While systemic arterial hypertension has been linked to COVID-19 severity20-22, the findings presented in this study suggest that systolic blood pressure figures at first medical assessment ≥140 mmHg are associated with increased risk of severity regardless of history of arterial hypertension.
Regarding comorbidities, the results presented here are consistent with those reported by a study conducted in China (n = 1,099), which identified that the presence of at least one comorbidity out of 9 possible (including COPD, diabetes, hypertension, cancer, and CKD) was significantly present among those with severe COVID-19 (38.72%, 95% CI 31.43 - 46.41) compared to those with a non-severe case (20.95%, 95% CI 18.37 - 23.71).23
The primary limitation of this study is that the main independent variable, i.e., vaccination history, was obtained from the clinical record, since Mexico does not have an open database that allows health professionals to access confirmation of such history through official records endorsed by the health authority. It should be noted that this is a retrospective study whose source of information consisted of clinical records (physical and/or electronic), so the variables were not collected for the specific purposes of this research, which implies a decrease in the precision of the measurement and, above all, the availability of some variables, particularly in the group of controls, a significant proportion of whom received outpatient management, which leads to an underreporting of laboratory indicators that could have been useful to improve the quality of the results presented here.
In contrast, the selection criteria and the randomization of the sample made it possible to avoid systematic biases, mainly selection bias, and at the same time, the sample obtained was representative of the source population and exposure. Among the strengths, this study made it possible to establish the magnitude and meaning of the associations found between the severity of COVID-19 and different covariates that, despite having already been recognized as risk factors for presenting manifestations of severity, to the knowledge of the researchers involved in this study, this is the first time that they have been explored in the context of mass vaccination, This is relevant because it has been carried out in a developing country, demonstrating the importance of the vaccination strategy and adding evidence to raise public awareness of the need to have a complete vaccination schedule against COVID- 19 and to set a precedent for future cases of other emerging or reemerging diseases that could present scenarios similar to the one recently presented.
It has been documented that the population of several Latin American countries faced fear and distrust of receiving hastily developed vaccines due to the state of emergency, nuanced by diverse political and social contexts.24 However, studies such as this one highlight the benefits of mass vaccination and support the intersectoral efforts made by international agencies, governments, the scientific community, and the private sector to promote health protection through the development, implementation, and equitable accessibility of immunizations.
REFERENCES
1. Hu B, Guo H, Zhou P, Shi ZL. Characteristics of SARS-CoV-2 and COVID-19. Nat Rev Microbiol. 2021;19(3):1. doi:10.1038/S41579-020-00459-7
2. Hui DS, Azhar E, Madani TA, et al. The continuing 2019-nCoV epidemic threat of novel coronaviruses to global health — The latest 2019 novel coronavirus outbreak in Wuhan, China. Int J Infect Dis. 2020;91:264. doi:10.1016/J.IJID.2020.01.009
3. Ochani RK, Kumar Ochani R, Asad A, et al. COVID-19 pandemic: from origins to outcomes. A comprehensive review of viral pathogenesis, clinical manifestations, diagnostic evaluation, and management. Le Infez Med n. 2021;20:20–36.
4. OPS. Evaluación de la efectividad de las vacunas contra la COVID-19 orientación provisional.; 2021. Consultado marzo 24, 2022. https://iris.paho.org/handle/10665.2/54270
5. Gao YD, Ding M, Dong X, et al. Risk factors for severe and critically ill COVID- 19 patients: A review. Allergy. 2021;76(2):428–455. doi:10.1111/ALL.14657
6. Secretaría de Salud. 78% de población adulta en México cuenta con al menos una vacuna contra COVID-19 Secretaría de Salud | Gobierno | gob.mx. 78% de población adulta en México cuenta con al menos una vacuna contra COVID-19. Published el 20 de octubre de 2021. Consultado septiembre 18, 2022. https://www.gob.mx/salud/prensa/78-de-poblacion-adulta-en-mexico-cuenta-con-al- menos-una-vacuna-contra-covid-19
7. Secretaría de Salud. Política nacional de vacunación contra el virus SARS-CoV-2, para la prevención de la COVID-19 en México. Documento Rector. 2021 p. 1–68. https://coronavirus.gob.mx/wp- content/uploads/2021/04/28Abr2021_13h00_PNVx_COVID_19.pdf
8. Secretaría de Salud. Información de la vacuna – Vacuna Covid. Consultado mayo 2, 2022. http://vacunacovid.gob.mx/wordpress/informacion-de-la-vacuna/
9. OMS. COVAX: colaboración para un acceso equitativo mundial a las vacunas contra la COVID-19 [Internet]. [citado el 10 de febrero de 2024]. Disponible en: https://www.who.int/es/initiatives/act-accelerator/covax
10. Gobierno de la Ciudad de México. INFORME COVID-19 30 de diciembre de 2021.; 2021. https://www.salud.cdmx.gob.mx/storage/app/uploads/public/61f/d90/dce/61fd90dce 4f2f151470382.pdf
11. Secretaría de Salud. COVID-19 México. Informe Técnico Diario 1 de Julio de 2021.; 2021. https://www.gob.mx/cms/uploads/attachment/file/649747/Comunicado_Tecnico_Dia rio_COVID-19_2021.07.01.pdf
12. Secretaría de Salud. Informe Técnico Diario COVID-19 México 30 de junio 2022.; 2021. https://www.gob.mx/cms/uploads/attachment/file/738376/Informe_Tecnico_Diario_ COVID-19_2022.06.30.pdf
13. Tenforde MW, Patel MM, Ginde AA, et al. Effectiveness of SARS-CoV-2 mRNA Vaccines for Preventing Covid-19 Hospitalizations in the United States. medRxiv. Published online el 8 de julio de 2021. doi:10.1101/2021.07.08.21259776
14. Hyams C, Marlow R, Maseko Z, et al. Effectiveness of BNT162b2 and ChAdOx1 nCoV-19 COVID-19 vaccination at preventing hospitalisations in people aged at least 80 years: a test-negative, case-control study. Lancet Infect Dis. 2021;21(11):1539. doi:10.1016/S1473-3099(21)00330-3
15. Dagan N, Barda N, Kepten E, et al. BNT162b2 mRNA Covid-19 Vaccine in a Nationwide Mass Vaccination Setting. N Engl J Med. 2021;384(15):1412–1423. doi:10.1056/NEJMOA2101765
16. Thiruvengadam R, Awasthi A, Medigeshi G, et al. Effectiveness of ChAdOx1 nCoV- 19 vaccine against SARS-CoV-2 infection during the delta (B.1.617.2) variant surge in India: a test-negative, case-control study and a mechanistic study of post- vaccination immune responses. Lancet Infect Dis. 2022;22(4):473. doi:10.1016/S1473-3099(21)00680-0
17. Rearte A, Castelli JM, Rearte R, et al. Effectiveness of rAd26-rAd5, ChAdOx1 nCoV- 19, and BBIBP-CorV vaccines for risk of infection with SARS-CoV-2 and death due to COVID-19 in people older than 60 years in Argentina: a test-negative, case-control, and retrospective longitudinal study. Lancet (London, England). 2022;399(10331):1254. doi:10.1016/S0140-6736(22)00011-3
18. Harrison AG, Lin T, Wang P. Mechanisms of SARS-CoV-2 Transmission and Pathogenesis. Trends Immunol. 2020;41(12):1100. doi:10.1016/J.IT.2020.10.004
19. Singer M, Deutschman CS, Seymour C, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016;315(8):801–810. doi:10.1001/JAMA.2016.0287
20. Tian J, Yuan X, Xiao J, et al. Clinical characteristics and risk factors associated with COVID-19 disease severity in patients with cancer in Wuhan, China: a multicentre, retrospective, cohort study. Lancet Oncol. 2020;21(7):893. doi:10.1016/S1470- 2045(20)30309-0
21. Li X, Xu S, Yu M, et al. Risk factors for severity and mortality in adult COVID-19 inpatients in Wuhan. J Allergy Clin Immunol. 2020;146(1):110–118. doi:10.1016/J.JACI.2020.04.006
22. Wang D, Hu B, Hu C, et al. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA. 2020;323(11):1061–1069. doi:10.1001/JAMA.2020.1585
23. Guan WJ, Ni ZY, Hu Y, et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N Engl J Med. 2020;382(18):1708–1720. doi: 10.1056/NEJMoa2002032
24. Tonelli P, Parada-Lezcano M, Ismael B. La pandemia por COVID 19: el derecho a la salud en tensión. Un estudio cualitativo exploratorio en Chile, Colombia, Costa Rica y México [Internet]. 2021. Disponible en: https://www.flacso.org/sites/default/files/2022-03/la_pandemia_por_covid19.pdf
Información adicional
redalyc-journal-id: 5704