ABSTRACT
Introduction: Type 1 Diabetes mellitus (T1DM) is an autoimmune disorder which arises from the destruction of insulin-producing pancreatic β-cells. Currently, Brazil’s advanced therapy medicinal products (ATMP), developed for clinical research and therapeutic purposes, take place in the so-called Cellular Technology Centers (CTC), according to the Resolution nº. 9/2011 of the Collegiate Board of Directors (RDC), enacted by the National Health Surveillance Agency (Anvisa).
Objective: This study was conducted with the main objective of describing and discussing the development of ATMP for T1DM treatment.
Method: A qualitative research, narrative review and critical discussion of the literature were under taken.
Results: ATMP promote new therapeutic approaches for Diabetes, holding great potential to restore the patients’ endogenous insulin secretion, improving their life quality, overcoming the chronic complications of Diabetes and reducing the socioeconomic burden. Nowadays, ATMP in T1DM comprise: a) cell therapy; b) gene therapy products; c) tissue engineering and d) ATMP associated to biopharmaceutical products.
Conclusions: Further research should contribute to stimulate public and private organizations to effectively act towards reducing the impact of Diabetes on individuals and the society as a whole. It is essential that Brazilian legislation closely follows the biotechnological developments, supporting the scientific progress and benefiting T1DM patients with modern and cutting-edge therapies.
Keywords: Advanced Therapy Medicinal Products (ATMP, Type 1 Diabetes Mellitus (T1DM, Pancreatic Islet Transplantation and Encapsulation, Regulatory Legislation for Cell and Gene Therapy, Biopharmaceuticals.
RESUMO
Introdução: O diabetes mellitus do tipo 1 (T1DM) é uma desordem autoimune que culmina na destruição das células β-pancreáticas produtoras de insulina. Atualmente, Produtos Medicinais de Terapia Avançada (ATMP) são desenvolvidos no Brasil, para fins de pesquisa clínica e terapia celular, pelos chamados Centros de Tecnologia Celular (CTC), de acordo com a Resolução da Diretoria Colegiada nº 9/2011, emitida pela Agência Nacional de Vigilância Sanitária (Anvisa).
Objetivos: Esse estudo foi conduzido com o objetivo principal de descrever e discutir o desenvolvimento de produtos medicinais em terapia avançada (ATMP) para o tratamento de T1DM.
Método: Esse estudo foi realizado através de pesquisa qualitativa, revisão narrativa e discussão critica.
Resultados: Os ATMP promovem abordagens terapêuticas para o diabetes demonstrando grande potencial para restaurar a secreção endógena de insulina dos pacientes, aumentando sua qualidade de vida, superando as complicações crônicas do Diabetes e reduzindo o impacto socioeconômico desta doença. Atualmente, os ATMP para T1DM compreendem: a) terapia celular; b) terapia gênica; c) engenharia tecidual e d) ATMP associados a produtos biofarmacêuticos.
Conclusões: Pesquisas adicionais deverão contribuir para mobilizar governos e organizações para ações que promovam, de forma eficiente, a redução do impacto do diabetes nos indivíduos e na sociedade. Para isso, é essencial que a legislação brasileira acompanhe de perto os desenvolvimentos biotecnológicos, dando suporte para o progresso científico e beneficiando pacientes de T1DM com terapias modernas.
Palavras chave: Produtos Medicinais de Terapia Avançada, Diabetes Mellitus do Tipo 1, Transplante e Encapsulamento de Ilhotas Pancreáticas, Legislação Regulatória de Terapias Celulares e Genéticas, Biofármacos.
ARTICLE
Advanced Therapy Medicinal Products in type I diabetes mellitus: technological and regulatory challenges
Produtos Medicinais de Terapia Avançada no diabetes mellitus tipo I: desafios tecnológicos e regulatórios
INCQS-FIOCRUZ
Received: 03 October 2017
Accepted: 16 January 2018
Diabetes mellitus (DM) belongs to a class of metabolic disorders characterized by impairment of the insulin regulatory activity, due to a combined deficiency in hormone synthesis, secretion and/or activity itself. According to the International Diabetes Federation, 415 million adults were estimated to have Diabetes worldwide in 2015, of which 318 million adults display impaired glucose tolerance 1 . Brazil has the fourth highest DM incidence and the National Health Surveyor ( Pesquisa Nacional de Saúde – PNS) estimated that, in 2013, 6.2% of the Brazilian population, aged over 18 years, were diabetic 2 . In 2015, DM caused 5.0 million deaths, costing between 673 and 1,197 billion dollars in healthcare spending 1 . It is estimated that by 2040 there will be 642 million people living with the disease, with a 19% increase in healthcare spending 1 . Tackling this global epidemic is a monumental task, which requires further knowledge about the disease, in addition to the development of more effective therapeutic strategies.
Type 1 Diabetes mellitus (T1DM) is an autoimmune disorder, which arises from destruction of insulin-producing pancreatic β-cells, present in the islets of Langerhans, a cluster of hormones-producing cells, which constitutes the endocrine pancreas. β-cell destruction results in severe complications associated with episodes of hyperglycemia, hypoglycemic unawareness, and ketoacidosis 3 . The chronic health complications associated with T1DM, include retinopathy, nephropathy, neuropathy, as well as cerebrovascular and cardiovascular diseases 4 , with many of these long-term complications being associated with high mortality 5 . Furthermore, this condition affects T1DM patients’ quality of life and greatly increases the burden on healthcare costs.
The current therapy for T1DM is focused on maintaining a balanced diet along with lifelong insulin administration 6 . However, although a tight glucose control through insulin therapy can delay the long-term complications for numerous patients with T1DM, this treatment often renders many of the patients prone to severe hypoglycemic episodes, and thus far it has not yet been possible to adequately mimic the endogenous insulin secretion levels 7 .
Pancreas transplantation, the only clinically approved therapeutic approach for T1DM, performed almost worldwide, is able to establish prolonged normoglycemia by restoring endogenous insulin secretion in response to appropriate stimuli 8 . However, it is noteworthy that this prolonged surgical procedure exposes T1DM patients to a high risk and, in addition, patients are doomed to lifelong immunosuppression. For these reasons, this procedure is most commonly applied in combination with kidney transplantation or in diabetic patients with chronic kidney failure due to nephropathy. At a lower frequency, isolated pancreas transplantation is performed in patients with very unstable (brittle) Diabetes presenting severe episodes of asymptomatic hypoglycemia. In these patients, the risk of death from DM and poor quality of life justify the risk of pancreas transplantation and chronic immunosuppression. Therefore, given the current epidemiological proportions of T1DM and the associated socio-economic cost, the scientific community has been seeking for technically and economically feasible therapeutic alternatives to reestablish endogenous glycemic control for the majority of T1DM patients.
Advanced therapy medicinal products (ATMP) constitute a new category of products, including genetic and cell therapy, as well as tissue engineering, all of which may be associated with biopharmaceutical products. The products are based on biomedical research progress and on the use of new and sophisticated technologies, aiming to establish patient-specific designed therapeutic interventions and effective biomarkers to predict and monitor their clinical response. ATMP development promotes new therapeutic approaches for many diseases, including cancer, Diabetes, neurodegenerative and cardiovascular, among others 9 . For Diabetes, the development of ATMP holds great potential to restore the patients’ endogenous insulin secretion, increasing their quality of life, overcoming the chronic complications of Diabetes and reducing the socioeconomic burden.
In Brazil, no specific regulation regarding clinical research or therapy with ATMP is yet available; therefore, no legislation establishes the requirements to ensure product efficiency and safety. However, on September 9 th , 2016, the National Health Surveillance Agency ( Agência Nacional de Vigilância Sanitária – Anvisa), through Ordinance n o . 1731, established the Technical Chamber for Advanced Therapies (CAT), a permanent collegiate, technically linked to Anvisa’s Blood, Tissues, Cells and Organs Management (GSTCO), with the purpose of advising Anvisa’s Board of Directors (Dicol) on the regulation, evaluation, registration and post-registration procedures for ATMP, mainly to guide their production, quality control, efficacy and safety, including clinical trials 10 . Gathering of knowledge and deliberation about several aspects of cell therapy, considering the scientific, ethical and legal aspects, as well as intellectual property commercialization and the results already obtained from clinical research, should support the scientific community to establish new regulations to ensure safe and efficient production and use of ATMP. This article aims to discuss the status of ATMP in T1DM treatment in the context of the Brazilian legislation.
This study was performed through qualitative research 11 . A traditional literature review (or literature narrative) is presented, aiming to describe and discuss the development of ATMP for T1DM treatment. The subject was not restricted and the search tools were not pre-established, allowing the protocol to be flexible 12 . The literature was surveyed for references related to the subject, using the following databases: Scientific Electronic Library Online (SciELO), National Center for Biotechnology Information (NCBI/PubMed) and the National Virtual Library ( Biblioteca Virtual em Saúde – Bireme). Specific Brazilian legislation was also surveyed, using the Federal Government Legislation Portal and the Anvisa website. In addition, online database systems, divulgation articles published in electronic journals and biotechnological companies’ websites were also referenced.
Currently, Brazil’s ATMP are developed for clinical research and therapeutic purposes at the so-called Cell Technology Centers ( Centros de Tecnologia Celular – CTC), according to the Resolution of the Collegiate Board of Directors ( Resolução da Diretoria Colegiada – RDC) nº. 9, enacted by Anvisa 13 . To fulfill their purposes, the CTC must follow minimum technical and sanitary requirements for sampling, processing, packaging, storage, quality control evaluation, disposal, approval and transportation of human cells and their derivatives, in order to ensure quality and safety of the therapeutic products.
ATMP constitute a new category of products, comprising gene therapy and cell therapy, as well as tissue engineering, all of which may be associated with biopharmaceutical products. For T1DM treatment, several ATMP are under development ( Table ).
Advanced Therapy Medicinal Products (ATMP) currently under development and their potential use in type 1 diabetes.
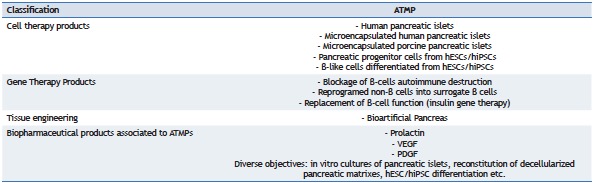
hESCs: human embryonic stem cells; hiPSCs: human induced pluripotent stem cells; VEGF: vascular endothelial growth factor; PDGF: platelet-derived growth factor.
Regarding the cell type, cell source and the degree of manipulation complexity, cell therapy products can be very highly variable. The cells may be adult, fully differentiated, self-renewing stem cells (SC) or progenitor cells, manipulated or expanded in vitro , in order to exert a specific physiological function. These cells may be of autologous, allogeneic or xenogeneic origin. In addition, cells may also be genetically modified. Another possibility is to use either cells alone or associated with biomolecules and chemicals, or combined with structural materials classified as medical devices 9 .
Currently, pancreatic islet transplantation is considered as one of the most promising cell therapy approaches to achieve insulin independence in T1DM patients. Pancreatic islet isolation and purification requires a 5–7 h multi-step process to extract the islet fraction, which represents hundreds of thousands of clustered cells (Langerhans’s islets), which comprise only 1%–2% of the pancreas volume 14 . This procedure involves mechanical and enzymatic digestion to isolate pancreatic islet cells from the pancreatic parenchyma of a deceased organ donor. Following isolation and purification, the pancreatic islets are cultured in vitro prior to their transplantation into a T1DM recipient patient 15 .
Detailed standard operating procedures (SOP) and master production batch records for manufacturing of purified human pancreatic islet lots suitable for clinical transplantation are key to ensure that a viable mass of insulin-producing cells can be safely infused into the recipients 14 . These SOP detail each procedural step, from donor procurement/selection and islets isolation and purification to pre-transplantation in vitro culture, quality controls and product release criteria for transplantation, such as: assessment of identity, viability, potency and sterility of the final islet product 14 . Using a minimally invasive surgery, the pancreatic islets are then infused into the liver through the portal vein system of the T1DM patient. The implanted islets become lodged within the liver capillaries, engrafting and functioning within the liver tissue. Patient recovery is rapid, with a very low risk of infection and minimal morbidity. Actually, pancreatic islet transplantation is considered to be among the safest of all transplantation procedures, when performed in dedicated centers 16 .
Islet transplanted T1DM patients present marked improvement in both short-term and long-term outcomes, and insulin independence after initial or subsequent transplantation was achieved in up to 80% of patients one year post-transplantation 15 , 17 . Approximately 50% of the patients remained insulin independent at five years after receiving the transplant. Islet transplantation is already approved and reimbursable by insurance companies or covered by National Health Systems in several countries, including Canada, Australia, the UK, Switzerland, Italy, France and other parts of Europe, in a total of 40 countries 14 . Over 1,500 procedures have been performed worldwide, including 864 allogeneic transplants and 480 autologous transplants, in cases of total pancreatectomy of non-diabetic patients 17 .
An assessment of the Clinical Trials website (www.clinicaltrials.gov), in September 2017, using the keyword “islet transplantation”, identified 155 studies. Among these, eight were withdrawn, two were suspended, three are not yet recruiting patients, 16 are active, but not recruiting patients, 35 are currently recruiting patients, four are enrolling patients, 57 were clinically completed, 11 are finished and 19 have unknown status. One is at early phase I, 60 are in phase I, 74 are in phase II, 15 studies in phase III, and three studies in phase IV. These studies include investigations of different immunosuppressive drugs, anti-inflammatory therapy, sites of implantation, islet encapsulation and device development, autologous islet transplantation in cases of total pancreatectomy, effect of dietary interventions, cord blood infusion, hematopoietic stem cell transplantation and mesenchymal stem cell transplantation. Recently, the Clinical Islet Transplantation Consortium completed the first multi-center phase III study to evaluate the efficacy and safety of clinical grade islets for treatment of T1DM complicated by severe hypoglycemia and hypoglycemic unawareness. Islet transplantation was shown to be safe and to result in elimination of severe hypoglycemia and restoration of normal or near-normal glycemic control in 87.5% of participants at the one year follow-up 18 .
In Brazil, unfortunately, pancreatic islet transplantation is still considered as an experimental procedure. In addition, access to this procedure is limited by unsustainable costs, based only on limited academic research funding. The first Brazilian pancreatic islet transplantation was performed in 2002 by our group, the Cell and Molecular Therapy Center (Nucel), São Paulo University, the procedure being performed in a T1DM patient with recurrent and severe hypoglycemia episodes and metabolic instability 19 . The efforts of our multidisciplinary group have made the procedure possible. The cell biology team, responsible for islet isolation, purification and quality control, maintains close contact with a team of specialized surgeons and clinicians 20 . By the time this article was submitted, eleven islet infusions had been performed in five patients (unpublished data). Currently, two Brazilian groups are interested in islet isolation research, namely, our own group and that associated with PUC-Paraná and the Pro-Kidney Foundation, who reported an islet transplantation in 2005 21 .
Despite the success of islet transplantation, widespread utilization of the procedure remains hampered by the shortage of good quality donor pancreas 22 . This limited supply of organs makes it very challenging to reach a target islet cell mass of approximately 10,000 islet equivalents (IEQ) per kilogram of recipient body weight from one organ alone 15 , 23 , 24 . Moreover, up to three individual islet infusions from multiple allograft donors are often necessary to reach a sufficient islet mass to achieve normoglycemia 25 . Additionally, following islet cell transplantation, the patient is placed on an immunosuppressive regimen in order to prevent islet cells rejection. Potential islet transplant recipients are selected for this type of therapy only if they are deemed capable of tolerating long-term immunosuppressive medication 26 . The side effects of immunosuppressant drugs are widely known, including mouth ulceration, nausea, anemia, leukopenia and vomiting, as well as certain cancers 24 .
Islet encapsulation involves coating cells with an artificial membrane so as to preserve their physical and functional integrity. The membrane functions as a barrier with selective permeability, allowing nutrients to enter the cells and metabolic products to be discarded, while preventing the influx of components of the immune system (cells and cytokines) 27 . The material should be biocompatible and mechanically stable, while providing a suitable environment for cell survival, growth and differentiation. In recent years, the field of islet microencapsulation has demonstrated the potential to overcome the need for immunosuppression and to expand the donor pool so as to include other cell sources 28 .
In addition to acting as a barrier to the immune system, capsules can also act as a therapeutic factor to increase cell viability. Pancreatic islets isolation is still associated with loss of cell viability, which is due to disruption of the extracellular matrix (ECM), leading to β-cells apoptosis 29 . Our group has developed a novel biomaterial, called Bioprotect® 30 , 31 to tackle this issue. Bioprotect, based on alginate, chondroitin sulfate and laminin, has been used for microencapsulation of rat islets which were implanted into mice rendered diabetic, causing complete reversion of T1DM over a long period of time 30 , 31 . Addition of laminin to the microcapsules composition leads to modulation of gene expression and functionality of microencapsulated pancreatic islets, providing greater cell viability and normoglycemia in diabetic animals, for extended periods of time. Translation of this technology into the clinical practice will require encapsulation compounds in adequate purity and observation of GMP standards in order to obtain the adequate therapeutic product for humans.
Numerous clinical trials involving microencapsulation as a treatment for T1DM have been performed in the past few decades 32 . The safety of encapsulated islet allotransplantation was evaluated in a procedure performed in a 38 year T1DM patient 33 . Cadaveric human islets were encapsulated in alginate microcapsules and infused intraperitoneally. The patient was already on anti-rejection medications due to a renal transplant, but was able to discontinue all exogenous insulin for nine months. Following progress in capsule design has led to two additional encapsulated human islet transplantations to T1DM recipients 34 , 35 . Also, studies carried out with T1DM patients transplanted with microencapsulated xenograft islets provided a significant reduction in exogenous insulin requirements for a certain period, but no complete insulin independence was achieved 36 , 37 . Transplantation of encapsulated human islets has been clinically evaluated and is generally recognized as safe. Since T1DM is an autoimmune disease, the capsules may still be required regardless of the cell source for protection of the graft from destruction by the recipient’s own immune system.
As previously stated, two major challenges preventing islet cell transplantation from becoming available to a larger cohort of patients with T1DM are: the requirement for immunosuppressive medication following transplantation, which could be overcome with islet encapsulation, and the shortage of islet cells donors. Since islet transplantation is based on cell isolation and purification from pancreas of cadaveric donors, which are frequently in great shortage and demand, it is essential to develop approaches to increase the supply of insulin-producing cells for β-cell replacement therapy, such as stem-cell-derived β-cells and xenogeneic sources of pancreatic endocrine cells. Islet microencapsulation allows for safe and efficacious use of xenogeneic material for T1DM treatment. A constantly growing number of clinical studies point to the use of microencapsulated porcine pancreatic islets. There are numerous advantages in using porcine islets, which include: homology between porcine and human insulin, ample availability of large quantities of islets from porcine donors, and reduced ethical issues in their production 37 , 38 , 39 , 40 .
Notably, in 2007, a large clinical study using commercial microencapsulated pig islets (also called “Diabecell”) was performed by the Living Cell Technologies (LCT) Company 41 . Six of eight patients demonstrated a reduced exogenous insulin requirement and reduced glycated hemoglobin (HbA1c) levels, with two of the patients demonstrating insulin independence for up to 32 weeks 41 . Further studies (phase II clinical study, performed in New Zealand and ongoing phase II clinical trial in Argentina 42 , 43 ) showed greater benefit of using encapsulated porcine islets, however, no clinical trials involving porcine tissue have resulted in excellent metabolic control to date 32 . These initial studies have demonstrated the potential use of this technology as a safe and effective treatment option for T1DM, but also raised concerns about the potential risk of zoonosis. Islet xenografts will demand rigorous regulation and should be obtained from specific pathogen-free (SPF) pig breeds.
Another possible solution for the organ donor shortage is generation of β-cells or islet tissue from pluripotent stem cells, such as human embryonic stem cells (hESCs) 44 and human induced pluripotent stem cells (hiPSCs) 45 , 46 . Directed differentiation of pancreatic lineage cells from hESCs/iPSCs has been vigorously studied as ATMP for T1DM therapy 47 . Substantial progress in this research field was made in recent years. In the United States of America (USA), phase I/II clinical trials for T1DM patients have already been started with the use of hESC-derived pancreatic progenitors cells 47 .
In order to induce differentiation of hESCs/iPSCs into the pancreatic lineage cells, the normal developmental stages of the pancreas are mimicked and reproduced in vitro using overexpression of key transcription factors involved in pancreas development. The pancreatic developmental stages are induced using a combination of growth factors and chemical compounds to activate or inhibit key signal pathways. Until recently, most investigators have generated pancreatic β-like cells that produce and secrete insulin in response to stimuli, however, these cells do not secrete suitable amounts of insulin in response to changes in blood glucose levels and usually co-express other hormones, such as glucagon and somatostatin, rendering them inferior to adult β-cells 48 , 49 .
More recently, two groups have succeeded in generating functionally mature β-like cells from hESCs/iPSCs, however, details on the maturation mechanism remain to be elucidated 50 , 51 . Some issues, such as the stability and high cost of the differentiation process, still remain to be improved. In 2014, ViaCyte Inc. announced a clinical trial for T1DM patients’ treatment using an immunoprotective device which carried pancreatic progenitor cells differentiated from hESCs 52 . The immature β-cells are designed to further differentiate in vivo into mature pancreatic β-cells, which synthesize and secrete insulin and other hormones. This trial represents an initial and important step for the development of a stem cell-based ATMP therapy for T1DM.
The use of hiPSCs has a number of advantages over hESCs, such as the possibility of autologous cell transplantation and fewer ethical problems; therefore, hiPSC-based therapy is expected to lead the β-cell replacement therapy in the future. However, so far, no clinical trials have been carried out with transplantation of hiPSC-derived pancreatic cells 47 . Achieving economically and technologically viable SC therapy still constitutes a great challenge, since this ATMP requires strict rules for handling and production, as well as the use of compounds and factors produced under appropriate Good Manufacturing Practice (GMP) conditions.
Transfer of genetic sequences (DNA, RNA) to cells using different transducing agents, including plasmids, virus or bacterial vectors, is called Gene Therapy. The genetic sequences are designed to modify, control, inhibit, or overexpress a specific target sequence. The main objective is to induce genetic modification of somatic cells in vivo31 . Any modification of germ cells is strictly prohibited by Anvisa. Somatic cells can also be modified ex vivo or in vitro54 , 55 . The goal of future treatments for T1DM is to restore dynamic control over blood glucose levels without requiring daily insulin injections, surgical procedures, or lifelong immunosuppressive regimens. Additionally, future therapies must be affordable for all patients. Gene therapy has emerged as a promising alternative for T1DM treatment, which could meet all of the aforementioned criteria. The in vivo delivery of therapeutic genes can improve the clinical outcome of diabetic individuals by preventing the autoimmune destruction of β-cells prior to the onset of the disease, reprogramming non-β cells into replacement β-cells, or simply replacing the function of lost β-cells.
Two main strategies are envisaged to prevent the autoimmune destruction of β-cells through gene therapy, namely: edition of the immune system so as to prevent recognition of β-cell antigens as foreign and modification of residual β-cells to withstand the autoimmune attack. However, these approaches are limited because they rely on early detection of Diabetes, while more than 80% of the patient’s β-cells have already been destroyed by the time they become symptomatic. In addition, T1DM is a multifactorial disease, rendering it difficult to predict whether an individual will ever become diabetic, causing early intervention risky 56 , 57 .
Gene therapy may be used to reprogram non-β cells to generate replacement β-cells, which are as similar as possible to native β-cells. Many cell types have been targeted for this purpose, including pancreatic exocrine cells 58 , 59 , keratinocytes 60 and hepatocytes 61 , 62 , 63 , the latter being the most common target due to their close developmental relationship with β-cells. To obtain reprogramed cells, the expression of transcription factors is induced, mainly the pancreatic and duodenal homeobox gene 1 (PDX1) transcription factor, which regulates pancreatic development during embryogenesis and controls β-cell function in adults 61 . The β-cells produced from reprogramming are able to ameliorate hyperglycemia in diabetic models, however, the long-term efficacy of this strategy requires the absence of recurring autoimmunity and might require either lifelong immunosuppression or selective immunomodulation to prolong the survival of the newly generated β-cells 56 .
The strategy known as insulin gene therapy aims to replace the key functions of β-cells without substantially altering the phenotype of the host cell, by expressing insulin alone in non-β cells without the risk of autoimmune destruction 56 . An appropriate insulin-sensing target organ and an effective gene delivery method, both of which have to be safe and effective, must be selected. Both viral and non-viral vector-based gene delivery methods have been used for insulin gene therapy applications, each of which showing successful amelioration of Diabetes-associated hyperglycemia in small animal models 64 , 65 , 66 . Viral vectors have been more commonly used due to efficient delivery to target cells and chromosomal integration. Adenoviruses 65 , 67 , 68 , adeno-associated viruses 69 , 70 , 71 , oncoretroviruses 72 , 73 and lentiviruses 74 , 75 , 76 all have been used to deliver insulin to hepatocytes. Lentiviral vectors can transduce both dividing and non-dividing cells, are non-immunogenic, and can induce long-term expression of insulin, being thus considered the optimal gene delivery vehicle for long-term correction of T1DM 74 , 75 , 76 . Many advances in hepatic insulin gene therapy have been made, yielding promising results in mouse and rat models of T1DM, however, these models do not always accurately replicate the human disease and long-term safety of lentiviral vector-mediated insulin gene therapy still needs to be evaluated. Safety mechanisms, such as self-inactivating vectors and suicide genes, are alternative options to improve vectors safety.
Human tissue engineering products combine several aspects of cellular and molecular biology, medicine, materials science and engineering. The aim is to regenerate, repair or replace unhealthy or absent tissues and organs, acting by replacing the missing tissue, restoring tissue function or replacing unhealthy tissues. These products are characterized by a three-dimensional structural complexity. Development of an artificial pancreas, through an appropriate combination of cells and biologically active molecules in a scaffold, constitutes an attractive alternative therapy for T1DM.
Each organ or tissue is composed of an unique ECM, with particular microstructures and biomechanical properties, which is able to support the resident cells 77 . This matrix influences cell proliferation and chemotaxis, in addition to directing cell differentiation and inducing tissue remodeling 78 , 79 , 80 , 81 , 82 , 83 . The three-dimensional ultrastructure, surface topology and extracellular matrix composition are likely to contribute to these effects 84 . Matrices derived from fully decellularized organs constitute an attractive scaffold to be repopulated with different kinds of cells, aiming at the generation of an engineered new pancreas 85 , 86 , 87 .
Some studies have showed increased functionality of pancreatic islets when they were cultured in decellularized matrices derived from submucosa of the small intestine 88 and pancreas 85 , 89 . For mouse pancreas, it is possible to generate a 3D scaffold from the whole organ by the perfusion-decellularization technique and to reconstitute the scaffold with acinar and endocrine insulinoma cell lines, producing an engineered organ 86 . For human pancreas, a study was able to generate a 3D scaffold from the entire acellular organ, by the perfusion-decellularization technique with detergents 87 . The scaffold was used in 2D cultures of human pancreatic islets and in 3D assays in a bioreactor, demonstrating scaffold cytocompatibility 87 . Despite notable progress, significant challenges are still posed in the field. Thus, it is still necessary to scale-up the techniques for human-sized organs, to find clinically relevant cell types for recellularization, and to completely rebuild the vasculature and parenchyma of the scaffolds in order to support long-term function after transplantation 90 .
Biopharmaceuticals or biological medical products are those obtained from a biological source or process. The active substance is obtained through the industrial use of microorganisms or genetically modified cells. These biotechnological processes may be carried out both in vitro and in vivo , allowing for production of complex proteins with improved biological activity, longer half-lives and fewer side effects. Biological drugs for direct treatment of T1DM patients have been extensively pursued. However, biopharmaceutical products can also be used in association with ATMP to improve their viability, functionality and quality, as to enhance and expand in vitro cultures of pancreatic islets, improve cellular reconstitution of decellularized pancreatic matrixes, differentiate hESC/hiPSC into insulin producing cells and as active compounds of immunoprotective microcapsules.
The in vitro expansion of endocrine pancreatic cultures is an alternative strategy to obtain a greater β-cells mass to be transplanted. It is well known that β-cells rarely proliferate and the proliferation rate is very low, except during the gestational and the neonatal periods 91 , 92 . Biopharmaceutical molecules involved with cell cycle regulation, such as cyclins and hormones, have been evaluated as means to stimulate β-cell proliferation 93 , 94 . Our group has shown that Prolactin, a hormone produced during pregnancy, not only has a mitogenic activity in β-cells, but, also, induces insulin production and secretion 94 . β-cell proliferation can also be induced in bioreactors 95 associated with proliferation-inducing molecules.
Regarding the reconstitution of decellularized pancreatic matrices, biological drugs can create an adequate ambient for matrix repopulation. Growth factors added to scaffolds and to biomaterials, such as the immunoprotective microcapsules, can increase pancreatic islets vascularization, cell viability, function and glucose responsiveness 96 . Pancreatic islets are extremely vascularized 97 , therefore, it is crucial to reestablish the vasculature network before attempting to recellularize pancreatic matrices. Formation of a vascular network is highly dependent on growth factors, such as the vascular endothelial growth factor A (VEGF-A), which is also able to prevent β-cell apoptosis 96 , 98 , 99 , 100 , and platelet-derived growth factor subunit BB (PDFG-BB). Biopharmaceutical products may also be used to induce hESC/hiPSC differentiation into insulin producing cells, allowing the use of a chemically defined culture medium and maintenance of GMP specifications.
In Brazil, no specific regulation regarding clinical research or therapy with ATMP is yet available; therefore, no legislation establishes which procedures and assays are required to ensure product efficiency and safety. However, any clinical research protocol must be previously analyzed and approved by the Ethics Research Committee System and the National Commission on Research ( Sistema Comitê de Ética em Pesquisa/Comissão Nacional de Ética em Pesquisa – CEP/Conep), as a way to protect the population 101 . In addition, ANVISA enacted RDC n o . 09 in 2011, to establish the minimum technical and sanitary requirements for CTC 13 , which are the research centers in charge of ATMP development for clinical use. Anvisa is also deliberating, in a broad and democratic manner, about several aspects of cell therapy, considering the scientific, ethical and legal aspects, as well as intellectual property commercialization. From these discussions, manuals of Good Practices in Cell Therapy have been elaborated, combined with new resolutions involving ATMP 13 , 102 , 103 . Development of new regulations, gathering of knowledge on relevant Brazilian policies and the results already obtained from clinical research, support the scientific community in the establishment of new regulations to ensure safe and efficient production and use of ATMP.
Article 199 from the 1988 Brazilian Federal Constitution, prohibits commercialization of organs, tissues and human substances for the purpose of transplantation, research and treatment 104 . The first Brazilian law (n o . 4,280) regulating organs transplantation, was created in November 6 th , 1963, specifying the guidelines for removal of organs or tissues from deceased individuals 105 . This law was subsequently revoked by Law n o . 5,479 (August 10 th , 1968), which regulates organ and tissue donation and transplantation, including living donors, for therapeutic and scientific purposes 106 . In November 18 th 1992, Law n o . 8,489, also called the Brazilian Law of Transplants, was enacted, overruling and updating the previous law 107 . Subsequently, a new law (n o . 9,434) was enacted (February 4 th , 1997) 108 , which was also overruled by the most recent one, Law n o . 10,211 (March 23 rd 2001) 109 .
In May 28 th 1992, the Federal Medical Council ( Conselho Federal de Medicina – CFM) issued Resolution n o . 1,358, forbidding human embryos destruction, but allowing the altruistic and anonymous embryos donation for assisted reproduction purposes 110 . On February 4 th 1997, Law n o . 9,434 overruled this CFM resolution 111 . After 18 years, a new CFM Resolution (n o . 1,957) was enacted (December 15 th , 2010), updating regulations regarding assisted reproduction procedures 112 .
In 1995, Law n o . 8,974, also known as Biosafety or Genetic Engineering Law, was enacted, regulating the use of transgenic, genetic and genomic therapies and forbidding “production, storage or manipulation of human embryos intended to serve as biological material”. The National Technical Biosafety Commission ( Comissão Técnica Nacional de Biossegurança – CTNBio) was also created to inspect ATMP-related activities 113 .
The Industrial Property Law, also known as Industrial Property Code or Patent Law, n o . 9,279 was enacted on May 14 th , 1996 114 , regulating SC-related inventions and considering as patentable any invention that meets the novelty, inventive and industrial application requirements.
Also, in October 10 th , 1996, the National Health Council (Conselho Nacional de Saúde, CNS), from the Ministry of Health (Ministério da Saúde, MS), created Resolution nº. 196, dictating guidelines and regulatory standards for research involving human beings 115 . A mandatory research tool was established, the Informed Consent Terms, to be completed and signed by patients or family members 101 .
The Copyright Law n o . 9,610 was enacted in February 19 th , 1998 116 , protecting intellectual work as creations of the spirit, expressed by any means or support, tangible or intangible, known or future. In sciences, the copyright law does not cover scientific or technical content, meaning that the industrial or commercial use of the ideas is not subject to copyright protection.
At the end of the 1990s, Regulatory Agencies were created in Brazil. Anvisa was created by Law n o . 9,782 (January 26th, 1999) 117 , promoting consistent incorporation of standardized procedures and protocols to control health services. On February 21 st , 2002, Anvisa enacted RDC n o . 50 118 , this resolution establishes technical standards on physical infrastructure, including planning, preparation and evaluation of physical projects for health care establishments. On July 18 th , 2003, Anvisa enacted RDC n o . 190, which establishes technical standards for the operation of umbilical and placental cord blood banks 119 . Also, on August 4 th of the same year, Anvisa enacted RDC n o . 210 to establish GMP technical standards for drugs 120 , this RDC served as a guide for laboratories working with the ATMP, at a time when no specific/relevant Brazilian legislation existed.
On December 2 nd , 2004, Law n o . 10,973, called the Innovation Law, was enacted 121 , regulating the relationship between Universities, Research Institutions and Companies. This Law promotes patents on research results developed with public resources and the creation of Specialized Medical Services (SMEs) and scientific spin-off companies. The law also regulates how the profits of licensing patent-protected technologies, jointly developed by companies and Universities/researchers, should be shared.
Also in 2004, Anvisa enacted two other RDCs, namely: n o . 153 determining the technical regulation for hemotherapy procedures, including collection, processing, test, storage, transportation, quality control and human use of blood and its components, obtained from venous blood, umbilical cord, placenta and bone marrow 122 and n o . 306, describing the technical regulation required for management of health services waste 123 .
In January 13 th , 2005, the CNS enacted Resolution nº. 347 to regulate storage and use of human biological material in the scope of research projects 124 . In that same year, the Biosafety Law (nº. 11,105) was enacted (March 24 th , 2005), establishing safety standards and mechanisms to supervise human embryonic stem-cells related activities and, also, created the National Biosafety Council ( Conselho Nacional de Biossegurança – CNBS), restructured CTNBio and the National Biosafety Policy ( Política Nacional de Biossegurança – PNB) 54 . This law has also delegated to Anvisa the establishment of rules for the collection, processing, test, storage, transport, quality control procedures for ATMP and, to the Health Ministry (MS), the regulation of cellular research and therapies 125 .
On October 26 th , 2005, Anvisa enacted RDC nº. 315, regulating the technical registration, post-registration modifications and registry revalidation of finished biological products 126 . This RDC was later updated by RDC nº. 55, in 2010 127 , which could be used as a guide for ATMP technical registration, regulation and association with biopharmaceutical products. Also in 2005 (November 21 st ), Law nº. 11,196, the so called Good Law, was enacted 128 .
On December 16 th , 2010, Anvisa approved RCD nº. 56, which reviewed the regulation regarding the collection, processing, test, storage, transport, quality control and human use of hematopoietic SC obtained from bone marrow, peripheral blood, cord blood and placental blood for the purpose of conventional transplantation of hematopoietic progenitor cells 129 . This resolution was subsequently amended by RDC nº. 19 (March 23 rd 2012) 130 .
In 2011, a major milestone was the publication of RDC nº. 9, dated March 14 th , by Anvisa. Minimum technical and sanitary requirements for CTC operation were established and, also, rules for collection, processing, packaging, storage, quality control evaluation, disposal and approval for release of human cells and their derivatives were published, contemplating clinical research and SC therapy 13 . Also in 2011, Anvisa published RDC nº. 23 (May 27 th ), which establishes the technical regulation for operation of germinative tissue and cell biobanks and provides other regulations 131 . This resolution was subsequently amended by RDC nº. 72 (March 30 th , 2016) 132 .
It is important to note that, in Brazil, hematopoietic stem cells (HSC) transplants are the only routinely and clinically regulated cell therapies. These transplants, which consist of the intravenous infusion of HSCs, were initiated in 1979, but it was only in 1992 that Law nº. 8,489 was sanctioned, mentioning, for the first time, transplantation of HSCs as a therapeutic procedure 133 . Still, only in 2010, ANVISA reviewed this matter in RCD nº. 56 129 , 134 , which was later updated by RDC nº. 19, in 2012 130 .
The Biobanks and the Biorepositories are meant to store biological samples, usually from human source, such as: body fluids, cells, tissues, intracellular substances and DNA. Nowadays, there is a worldwide concern to establish a coordinated Biobank Network, following ethical, legal and technical principles 135 . In Brazil, the National Cell Therapy Network ( Rede Nacional de Terapia Celular – RNTC www.rntc.org.br) was created, in 2008, comprising eight CTC, located in five Brazilian States and, also, by 52 research groups selected by the National Council for Scientific and Technological Development ( Conselho Nacional de Desenvolvimento Científico e Tecnológico – CNPq), supported by the Ministry of Science, Technology and Innovation ( Ministério de Ciência, Tecnologia e Inovação – MCTI) and the Ministry of Health (MS-Decit). The RNTC was created to increase the integration among researchers from all over Brazil, to enable information exchange regarding scientific knowledge and technological competence in Regenerative Medicine, as well to establish a national Stem Cells Biobank 136 . The MS has also enacted Decree nº. 2,201, establishing the National Guidelines for Biorepository and Biobank of Human Biological Material for Research Purpose 137 .
As described above, Brazilian regulations have been established over the past few years in the face of several discussions regarding laws and decrees, ethics, sanitary issues, and procedural approvals. Much has been learned from pre-existing documents from related areas, which provide information that serve as a regulatory basis to establish up-to-date guidelines to deal with ATMP and future challenges. This task is not simple, since the cellular response mechanisms are extremely complex and not always completely elucidated. Nevertheless, no specific Brazilian regulation is yet available to determine which procedures and trials should be followed with ATMP to warrant their safe and effective use in humans.
Another issue is whether the ATMP should be regulated as personalized treatments or as medicines, both of which pose obstacles. If ATMP are to be regulated as personalized procedures/treatments, they no longer have industrial attractiveness, rendering them more time-consuming and costly. On the other hand, if the ATMP become regulated as medicines, the current Federal Constitutional impediment of human organs, tissues and substances commercialization for the purpose of transplantation, research and treatment, will limit the development of new therapies, reducing the interest of pharmaceutical companies in the development of Brazilian ATMP-based technology 125 .
Another important issue to be considered is the need for large-scale production of ATMP. A large-scaled, profitable, efficient and safe bioprocess involves a high cost of execution and analysis. The cost issue is an important question to be addressed, since high cost represents an obstacle to the promotion of ATMP-based therapy products, however, it is important to keep in mind that the process should be developed mainly to be consistent with the regulatory agencies requirements.
DM is a metabolic disorder that will affect 642 million people by 2040, 10% of which will be T1DM patients. In addition to placing a large financial burden on individuals and their families, due to the cost of insulin and other essential medicines, Diabetes also has a substantial economic impact on countries (particularly the less favored ones) and National Health Systems. The majority of countries spend between 5% and 20% of their total health expenditure on Diabetes 1 . Tackling this global epidemic is a monumental task that will require increasing knowledge about the disease and the development of more effective therapeutic strategies.
The world market for Diabetes medications generated US$ 35.6 billion in 2012, and the revenues indicate strong growth for 2023, reaching US$ 55.3 billion in 2017 138 . The Brazilian market for diabetic drugs was valued as US$ 0.65 billion in 2011 138 . Since 2006, and the introduction of Federal Law nº. 11347, medications and supplies needed for the control and monitoring of Diabetes, are now freely distributed by the government 139 . The Brazilian Diabetes drug market revenue forecast is of US$ 1.18 billion for 2023, with a 1% annual growth 138 . These data highlight the fact that Diabetes treatment has become an industrial goal with high profits and great impact on public health and the economy. ATMP development promotes new therapeutic approaches for many diseases, including Diabetes, holding great potential to restore patients’ endogenous insulin secretion, increasing the patients’ life quality, overcoming the chronic complications of Diabetes and reducing the socioeconomic burden.
Islet cell transplantation is currently viewed as one of the most promising cell therapy approaches to achieve insulin independence in T1DM patients. Despite the success of islet transplantation, widespread utilization of this procedure remains limited. Only a reduced number of procedures have been performed in Brazil, a fact explained by the unsustainable costs, based only on limited academic research funding. Additionally, two major challenges prevent islet cell transplantation from becoming available to a larger cohort of T1DM patients, namely: the need for immunosuppressive medication following transplantation and the shortage of pancreatic islet tissue donor.
Since islet transplantation is based on cell isolation and purification from pancreas of cadaveric donors, which are frequently in great shortage and demand, it is essential to develop approaches to increase the supply of insulin-producing cells for β-cell replacement therapy, such as xenogeneic or SC-derived sources of pancreatic endocrine cells. Initial studies have demonstrated the potential use of xenogeneic pancreatic islet as a safe and effective treatment option for T1DM, but also raised concerns about the potential risk of zoonosis. SC therapy, despite some issues, such as the stability and cost of the differentiation, is expected to overcome the major obstacles for Regenerative Medicine for Diabetes treatment. The use of hiPSCs has advantages over hESCs, such as feasibility of autologous cell transplantation and fewer ethical issues, therefore, hiPSC-based therapy is expected to lead the replacement therapy in the future. Rendering SC therapy economically and technologically viable is a major a challenge. Production of this ATPM will require strict rules of handling and production, as well as the use of compounds produced under appropriate GMP conditions.
Regarding the second obstacle for clinical pancreatic islet transplantation, the need for immunosuppressive medication following transplantation, transplantation of encapsulated human islets has been clinically evaluated and is generally recognized as safe, similarly to transplantation of encapsulated xenogeneic pancreatic islets. But, since hiPSC-based therapy holds great promise for T1DM treatment and allows autologous transplantation, it is important to emphasize that, due to the T1DM autoimmune mechanism, encapsulation may still be required regardless of the cell source, to protect the graft from an autoimmune attack.
A very promising future perspective is to reconstitute the whole pancreas, however, despite notable progress, significant challenges still lie ahead in the field. It is necessary to scale-up the techniques for human-sized organs, to find clinically relevant cell types for recellularization, and to completely rebuild the vasculature, following GMP requirements. In this process, biopharmaceutical products will be essential to improve cellular reconstitution of decellularized pancreatic matrixes. The field of biopharmaceutical development holds great promise to drive many other ATMP protocols, by providing the necessary molecules to enhance and expand in vitro cultures of pancreatic islets, to induce hESC/hiPSC differentiation into insulin producing cells, to improve immunoprotective microcapsules, allowing the use of defined culture media and maintenance of GMP specifications, while also acting, through an appropriate combination of cells and biologically active molecules, in the development of an artificial pancreas.
Still, the main goal for future T1DM treatments will be to restore dynamic control over blood glucose levels, without requiring daily insulin injections, surgical procedures, or lifelong immunosuppressive regimens. More important, future therapies must be affordable for all patients. In this sense, gene therapy is a promising alternative for T1DM treatment and one that could meet all of the aforementioned criteria. However, it will require much developmental advancement to ensure its safety and efficacy.
Further research should contribute to stimulate public and private organizations to effectively act towards reducing the impact of Diabetes on individuals and the society as a whole. Several regulatory challenges still remain to be addressed and efforts should be directed to optimize the regulatory processes. Brazilian regulations should contemplate the needs of the scientific community and of patients, preparing for good manufacture practices, attracting financial resources, promoting the development of new ATMP-based technologies and increasing the patient’s access to these products. It is essential that the Brazilian legislation promote biotechnological development, supporting the scientific progress and benefiting T1DM patients with modern and cutting-edge therapies.
The CTC (Cell Technologies Center) named Nucel (Cell and Molecular Therapy Center), São Paulo University Medical School, seeks alternative treatments for T1DM and is partially supported by BNDES, Capes, CNPq, Fapesp, Finep, MCTI and MS-Decit. We would like to express our gratitude to the National Technical Commission on Biosafety (CTNBio) and the National Health Surveillance Agency (Anvisa) for sharing their knowledge with us and for ensuring safety and efficacy of our TDM1 research.
Authors have no potential conflict of interest to declare, related to this study’s political or financial peers and institutions.
* E-mail: mcsoga@iq.usp.br
Advanced Therapy Medicinal Products (ATMP) currently under development and their potential use in type 1 diabetes.

hESCs: human embryonic stem cells; hiPSCs: human induced pluripotent stem cells; VEGF: vascular endothelial growth factor; PDGF: platelet-derived growth factor.
