Articles
Received: 01 June 2018
Accepted: 09 January 2019
Abstract:
Taxco de Alarcon is an important mining site in Mexico. The town is a well-known tourist destination where the potable water quality is important for guaranteeing the health of the users. Due to the altitude of the city, the surface and rain water are the main sources of water. Thus, the aims of this study were to characterize i) the composition and chemical speciation of particulate matter suspended in water to determine the content of potentially toxic elements, and ii) the chemical and bacteriological composition of the potable water of Taxco de Alarcon, Guerrero, Mexico, according to the Mexican official norm NOM-127-SSA1-1994.
The analyzed representative samples were taken from the municipal water distribution network, the water treatment plant, the Chacuhalco spring and a mixture of the waters of the Chontalcuatlan river, the Teneria spring, and the San Marcos and El Sombrerito dams. According to the results, 94 % of the analyzed samples did not comply with the standard NOM-127-SSA1-1994. Moreover, the distribution of the analyzed elements in the different chemical fractions of the particulate matter showed that the highest concentration of Cd (4.17 mg∙kg-1) is associated with the carbonate fraction. This means Cd is moderately bioavailable. On the other hand, the highest concentrations of Pb, Zn, Mn and Cu (62.2, 42.3, 243 and 1026 mg∙kg-1, respectively) were detected in the Fe-Mn oxyhydroxide fraction, also considered as moderately bioavailable, whereas the highest concentrations of As, Fe and V (24.0, 7583 and 31.3 mg∙kg-1, respectively) were associated with the residual fraction, considered as non-bioavailable.
The results indicate that, to improve the quality of potable water, modifications must be incorporated along the water treatment processes, and in the distribution network and management.
Keywords: Chemical speciation, Hydrogeochemistry, Mexico, particulate matter, potable water, Taxco de Alarcon, water quality.
Resumen:
Taxco de Alarcón es una importante ciudad con una amplia tradición minera en México. La ciudad es uno de los destinos turísticos más importantes del país donde la calidad del agua es importante para garantizar la salud de los usuarios. Debido a la altitud de la ciudad, el agua de lluvia y agua superficial son las principales fuentes de abastecimiento. Por esta razón, los objetivos de este estudio fueron: i) caracterizar la composición y especiación química del material particulado suspendido en el agua para determinar el contenido de elementos potencialmente tóxicos y ii) analizar química y bacteriológicamente el agua potable de Taxco de Alarcón, Guerrero, México de acuerdo con la norma oficial mexicana NOM-127-SSA1-1994.
Las muestras representativas fueron tomadas y analizadas a partir de la red de distribución de agua municipal, la planta potabilizadora, el manantial Chacuhalco y una mezcla de agua compuesta por agua del río Chontalcuatlán, el manantial de Tenería y las presas San Marcos y El Sombrerito.
Los resultados muestran que el 94 % de las muestras analizadas no cumplen con la NOM-127-SSA1-1994. Además, la distribución de los elementos analizados en las diferentes fracciones químicas del material particulado mostró que la concentración más alta de Cd (4.17 mg∙kg-1) está asociada con la fracción de carbonatos; es decir, el Cd es moderadamente biodisponible. Por otro lado, las concentraciones más altas de Pb, Zn, Mn y Cu, de 62.2, 42.3, 243 y 1026 mg∙kg-1, respectivamente, fueron detectadas en la fracción de oxihidróxidos de Fe-Mn, también considerada como moderadamente biodisponible, mientras que las concentraciones más elevadas de As, Fe y V (24.0, 7583 y 31.3 mg∙kg-1, respectivamente) se encuentran asociadas con la fracción residual, considerada como no biodisponible.
En general, los resultados indican que se deben realizar modificaciones a lo largo del proceso de tratamiento, en la red de distribución y en el almacenamiento para poder mejorar la calidad del agua suministrada en la Ciudad de Taxco de Alarcón, Guerrero.
Palabras clave: Agua potable, calidad del agua, especiación química, Hidrogeoquímica, material particulado. México, Taxco de Alarcón.
INTRODUCTION
For many centuries, the provision of good quality water has been a determining factor for both subsistence and the society economy (Qu et al., 2013). Nevertheless, although the percent of freshwater available is low (0.5%), their unsuitable use and the detriment of the water quality is a common phenomenon around the world (CONAGUA, 2016; Sun et al., 2017). Water pollution depicts one of the most alarming environmental problems in the contemporary world (Pulido-Bosch, 2007; Hernandez-Flores et al., 2017). In many countries, particularly those with emerging economies, anthropogenic activities related to population growth, industrial development, mining, the excessive use of fertilizers, pesticides and insecticides makes the water unusable for any human activity (Cifuentes et al., 1994; Literathy, 1996; GomezAlvarez et al., 2004; Sjomander Magnusson and van der Merwe, 2005; Gordillo-Martinez et al., 2010; Lu, 2011).
In Mexico, some states with important mining activity such as Zacatecas, San Luis Potosi, Guerrero and Sonora have been affected by the pollution by heavy metals (Hg, Ar, Pb and Cr) attributed to the mining industry and effected by the inadequate management of solid mining wastes (Covarrubias and Pena Cabriales, 2017; Corona-Chavez et al., 2017). On the other hand, INEGI (2013) reported that the surface water or shallow groundwater represents the main source of domestic water supply for a large number of rural populations and many cities in Mexico. Even raw or superficial shallow groundwater without any treatment is still used for human consumption, which may represent a risk to the health of the inhabitants.
Taxco de Alarcon (Tx-A), Guerrero, Mexico (18° 33' 24" N; 99° 36' 17" W), recognized as a city of mining origin founded in the midseventeenth century is considered a major tourist destination (CRM, 1999; Des Rivieres and Beals, 2011). From the 20th century on, mining in Taxco has been centered on base metals Pb, Cu, Zn and precious metals such as Ag and Au. Unfortunately, linked to their production processes, some elements such as As and heavy metals (potentially toxic elements) were produced and released into the environment in waste. Those pollutants are present in the liquid and solid mining waste disposed in the surrounding environment of Tx-A, where the wind, animals and the rain are some of the possible dispersion factors, altering the properties of the soil and water (Romero et al., 2007; Talavera-Mendoza et al., 2016; Corrales-Perez and Martin-Romero, 2018).
The city has several different water sources. The main source of supply is the surface water of the Chontacuatlan river captured at 18° 39' 15" N; 99° 34' 27" W. Other supply sources of Tx-A are the springs of Chacuhalco and Teneria, and the San Marcos and El Sombrerito dikes (known as dams). These dikes are located in the recharge zone and, therefore, their capturing capacity is very low. In addition, these dikes are located in areas where the hydraulic load is very high, i.e. where the difference between the level of stored water and the water flow of the Chontalcuatan River is almost 1 km.
In most cities, potable water is treated in treatment plants that aim to eliminate pathogenic contaminants and microbes that may potentially harm the health of users. The methods used as well as the efficiency of the process determine the quality of the water that is sent to different places, and finally determine the use for which it is destined. The evaluation of the chemical and microbiological composition of potable water for human consumption, as well as of the chemical composition of suspended particulate matter is an imperative need, not only for determining the water quality but also to evaluate the efficiency of the treatment methods used.
The city of Tx-A has a water treatment plant (WTP) (Figure 1). The water processed in the WTP comes from the Chontalcuatlan river, the Chacuhalco and Teneria springs, and the San Marcos and El Sombrerito dams. According previous analysis, the water of the Chacuhalco spring shows good quality and presents levels of turbidity below the NOM-127-SSA1-1994 standard (Secretaria de Salud, 2000); therefore, water enters directly to the chlorination stage of the WTP. The water from the other sources is collected in a mixing box before being treated. Tx-A treatment plant uses the pitcher method to determine the amount of flocculant aluminum sulphate Al2(SO4)3 to be added by adjusting, for this, the pH of the water between 6.5 and 7.5 with 20 % CaO and HCl solutions.
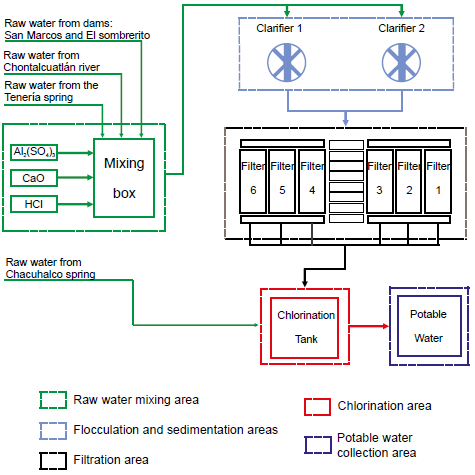
Figure 1
Schematic diagram of the purification process in the Taxco de Alarcon water treatment plant.
The WTP, as many others around the world, is not designed to treat the pollution caused by mining waste and the risk of the presence of this type of pollutants is high. The pollution of water by As, Cd, Pb, Hg, Tl, among other toxic elements, related to biomagnification of these elements from solid and liquid mining wastes close to urban settlements is an issue of public health concern (Smith and Huyck, 1999; Massoudieh et al., 2010; Talavera-Mendoza et al., 2016).
Thus, due to the importance of this city, its proximity to the mining solid wastes, and the fact that surface water is the main source for supply, we performed this study with the aim of characterize i) the composition and chemical speciation of particulate matter suspended in water to determine the content of potentially toxic elements, and ii) the chemical and bacteriological composition of the potable water of Tx-A, according to the Mexican official standard NOM-127-SSA1-1994.
MATERIALS AND METHODS
Thirty-three water samples from the Taxco de Alarcon city wereanalyzed (Figure 2). One raw water sample comes from the Chacuhalcospring (sample 1, Figure 2), and the second sample is a mixture of waterof the Chontalcuatlan river, the Teneria spring, and the San Marcosand El Sombrerito dams (sample 2, Figure 2). The third sample wastaken from the WTP output (sample 3, Figure 2). Finally, thirty samplescome from the water distribution network collected directly from thetap (samples 4-33, Figure 2). The source of the treated water, and thetreatment method have remained the same for a decade. Samplingwas carried out in July 2007, at the beginning of the rainy season. Theselection of sampling points was random; in Figure 2, the samplingpoints are located on a map of the city of Tx-A.
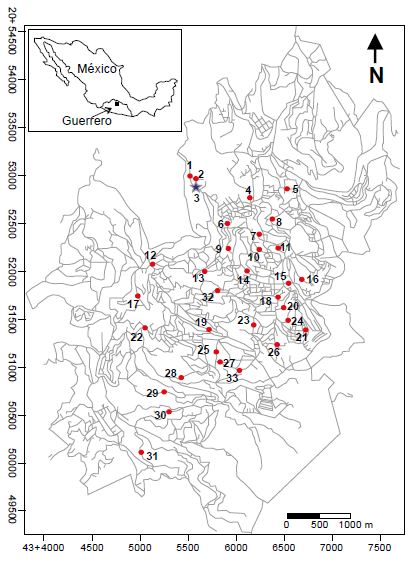
Figure 2
Map of the urban area of Taxco de Alarcon, Guerrero, showing the location of the municipal water treatment plant (bluestar), collection sites of samples of raw water (red circles labeled 1 and 2) and household tap water (red circles labeled 3-33).
At each sampling site, the temperature, pH, Eh and electrical conductivitywere determined. Temperature and pH were measured by potentiometrywith an Oakton portable pH meter. The Eh was measured with a Cole-Parmer model waterproof ORPtestr Double Junction.Conductivity was measured with a Horiba Model ES-14 E conductivitymeter with automatic temperature compensation and stabilization.
Three aliquots were taken at each point, two of them in Nalgene™high density polypropylene bottles prewashed with diluted HNO3,deionized water and tempered with the sample water. For the bacteriologicalanalyzes, a 250 mL aliquot was taken in sterile containers. Thecollected samples were placed in a cooler at <15 °C and transportedto the laboratory for analysis at the end of the day. To determine theanions, the aliquot was 1 L, where as for the analysis of the metals the aliquot was 500 mL acidified with 2 mL of ultrapure concentrated HNO3 (Ultrex II).
The alkalinity was determined by the titration method with0.02 M HCl using phenolphthalein, bromocresol green, and methyl redas colorimetric indicators. The chlorides were determined by titrationwith 0.02 M AgNO3 and 0.04 M K2CrO4. The sulfate (SO42-), nitrate(NO3- as N), fluorine (F-) and turbidity contents were analyzed bycolorimetry using a HACH model DR/890. BaCl2 and citric acid wereused for the sulfates; for the nitrates a cadmium reduction reagent wasused, whereas for the fluoride SPANDS was used. Only absorbence wasmeasured for turbidity, comparing it with a target.
Bacteriological analysis was performed by the 0.45 μm celluloseacetate membrane filtration method using ampoules with Hach'sm-ColiBlue24®- culture medium. The bacteria were incubated in a Cole-Parmer 52100-100 convection oven at a temperature of 37 oC for 24 h.
The cations concentration was determined in acidified and filteredsamples (0.45 μm cellulose acetate) by the technique of inductivelycoupled plasma atomic emission spectroscopy (ICP-AES) in a PerkinElmer, Optima 3200 DV. Sodium was analyzed on a 100 Perkin ElmerAAnalyst 800 Atomic Absorption Spectrometer. During the analysis,four certified High-Purity water standards (CWW-TM-D; CWWTM-H; CWW-TM-A and CWW-TM-E) were used.
On the other hand, to assess the presence, concentration and bioavailabilityof toxic metals in the water, the extractable metal contentwas determined treating 1.0 g of suspended particulate matter obtainedfrom sample 13 (Figure 2) with concentrated HNO3 (assuming that thenature of the particulate is the same in all the samples). Furthermore, toestablish the potential release effect of toxic elements of suspended materialin potable water with greater precision, a sequential extraction ofmetals from the particulate was carried out following the methodologyproposed by Tessier et al. (1979). It has been shown that the presence oftoxic metals (total or partial concentration) in particulate matter doesnot necessarily mean that they are bioavailable (Tessier et al., 1979) and, therefore, have harmful effects on biota or man. The bioavailabilityof an element depends largely on the chemical species in which it isfound. For example, high concentrations of Cd in a sulfur phase withlow solubility will be less bioaccessible than very low concentrationsof Cd in a water-soluble phase. For this reason, the understanding ofthe fraction in which a particular metal is associated and its concentrationprovides more information on the bioavailability of the metaland, therefore, a better approximation to the true impact on health.According to Tessier et al. (1979), the metals can be separated into fivechemical fractions: 1) exchangeable fraction (soluble + adsorbed); 2)fraction bound to carbonates; 3) fraction associated with oxyhydroxidesof Fe-Mn; 4) fraction bound to sulfides and organic matter, and 5) residual fraction (insoluble). Total metals and sequential extractionsolutions were analyzed in an ICP-AES Perkin-Elmer, optima 3200 DV (Talavera-Mendoza et al., 2016).
RESULTS
Physicochemical and bacteriological characteristics of watersamples
Raw water
The real detection limits (DL) reported by Talavera-Mendoza et al. (2006) were used for water samples analyzed under the same analyticalconditions, on the same equipment and using the same samplepreparation procedure (Table 1, Table 2).
Physicochemical characteristics of water samples from Taxco de Alarcon, Guerrero.
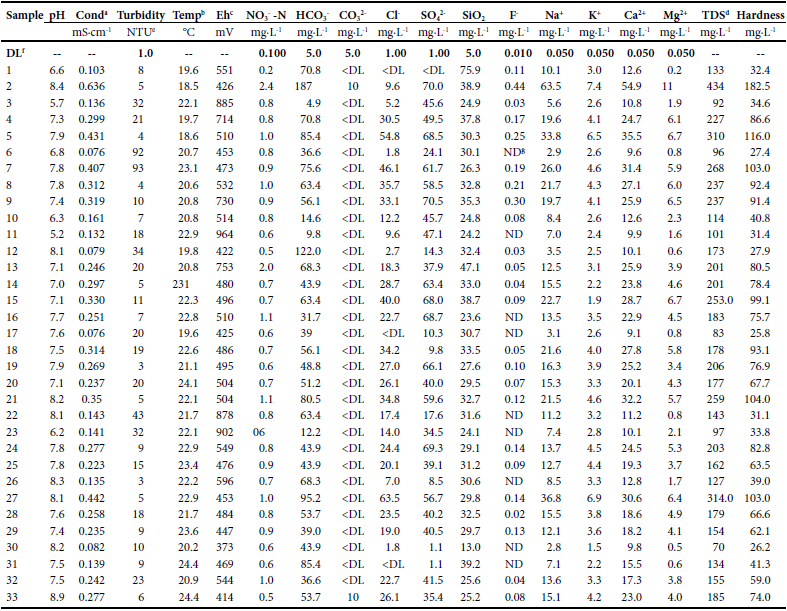
Data values have astandard deviation less than 5 %
a Conductivityb Temperaturec Redox potentiald Total dissolved solidse Nephelometric transfer unitsf Detection limitg Not detected
Concentration of trace elements in water samples from Taxco de Alarcon, Guerrero.
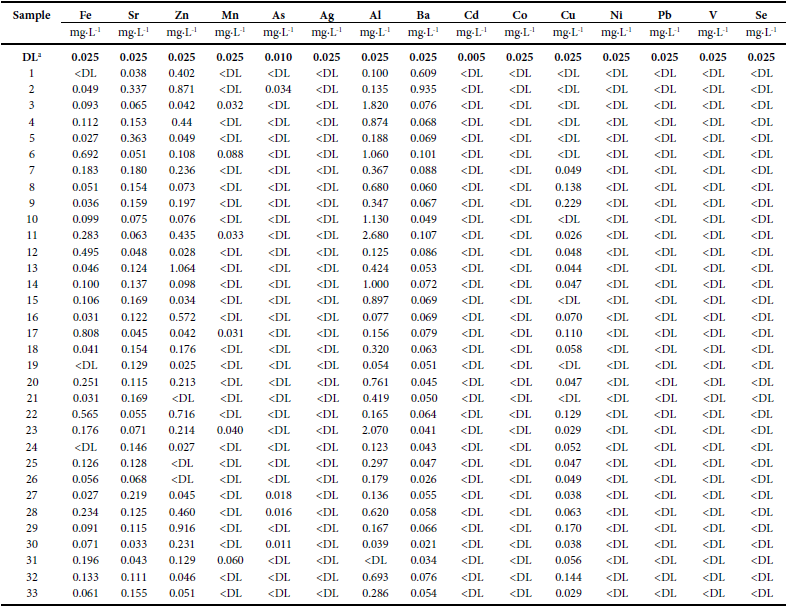
a Detection limit; Data values have a standard deviation less than 5 %.
The temperature of the water of Chacuhalco spring (sample 1; Figure 2) was 19.6 °C; the pH was almost neutral (6.6) and the Eh was551 mV indicating high oxygenation. The conductivity of Chacuhalcowater was 0.103 mS cm-1 and reflects low dissolved solids content.Water has a low turbidity (8 NTU) and is characterized by contentsof SO42- and Cl- <DL and low values of HCO3-, NO3-N, F-, Na+, Ca2+,Mg2+ and K+ (Table 1). The hydrogeochemistry of the Chacuhalcospring shows that the water has values of turbidity just above that establishedin NOM-127-SSA1-1994. The concentration of the majorityof the measured metals is below the DL except for Sr (0.038 mg∙L-1),Zn (0.402 mg∙L-1), Al (0.100 mg∙L-1) and Ba (0.609 mg∙L-1) (Table 2).Bacteriological analysis indicates that Chacuhalco spring water presentseight colony-forming units (CFU) of total coliforms (TC) (Table 3).This means that the water is not suitable for human consumption, and yet, this water is not treated in the WTP and is only chlorinated beforesending it to the homes.
Bacteriological analyzes of water samples from Taxco de Alarcon, Guerrero. Fecal coliforms correspond to E. coli.

ND: No detected.
On the other hand, the mixed sample of the Chontalcuatlanriver, the Teneria spring, and the San Marcos and El Sombreritodams (sample 2) has a slightly basic pH (8.4) and an Eh of 426 mV,which indicates an oxidizing environment. The conductivity of thiswater was moderate (0.636 mS∙cm-1) and therefore the total dissolvedsolids (TDS) are also moderate (434 mg∙L-1). Turbidity is low(5 NTU). Sample 2 has low anion and cation contents (Table 1).With the exception of Ba (0.935 mg∙L-1), all metals are within theNOM-127-SSA1-1994, and even Cu, Ag, Mn, Cd, Co, Ni, Pb, Se,and V are found <DL of the method (Table 2). In the bacteriologicalanalysis, more than 20 CFU/100 mL of E. coli and more than 100CFU/100 mL of TC were found (Table 3). The results indicate thatthis water does not meet the specifications of NOM-127-SSA1-1994.
Treated water
The water sample treated at the WTP (sample 3, Figure 2) showeda slightly acid pH (5.7). The Eh was 885 mV which indicates that thewater is highly oxygenated. It also has low conductivity (0.136 mS cm-1)and consequently low TDS (92 mg∙L-1), whereas the turbidity is high(32 NTU).
Sample 3 is characterized by low content of HCO3-, Cl-, SO42-,NO3-N, F-, Na+, K+, Ca2+, and Mg2+ (Table 1). Moreover, concentrations of As, Ag, Cd, Co, Cu, Ni, Pb, Se and V are <DL of the method,whereas the contents of Fe, Sr, Zn, Mn and Ba were low (Table 2).Furthermore, bacteriological analysis indicates that the treated waterdoes not contain E. coli or TC (Table 3). Therefore in sample 3, theparameters that do not meet the NOM-127-SSA1-1994 standard arepH, turbidity and Al content. The acidity of the water is probably dueto the chlorination process. On the other hand, the high values ofturbidity and Al content are due to the inefficiency of the flocculationprocess during the treatment process.
Water from the distribution network
The temperatures of the household samples 4 to 33 (Figure 2) rangebetween 18.6 and 24.4 °C. The pH in the household samples displayssignificant variations, ranging from 5.2 to 8.8. Half of the sampleshave slightly basic pH values (7.5–9.0), 40 % have circum-neutral pHvalues (7.5 – 6.5) and only 10 % have slightly acidic pH values (6.5–5.5).The Eh values are variable and range from 373 to 964 mV (Table 1).Conductivity varies between 0.076 and 0.442 mS∙cm-1, which translatesinto TDS between 70 and 314 mg∙L-1. The turbidity of the samples isalso variable and ranges from 3 to 93 NTU.
The HCO3- contents were variable and range from 9.7 to 122.0 mg∙L-1.The contents of SO42- (1.1 – 70.5 mg∙L-1) and of Cl- (0.9 – 63.5 mg∙L-1) were low. The contents of NO3-N range from 0.5 to 2.0 mg∙L-1. Theconcentrations of F- were very low between <DL and 0.3 mg∙L-1.
The content of Ca2+ varied from 9.1 to 35.5 mg∙L-1, whereas theconcentration of Mg2+ varied between 0.452 and 6.690 mg∙L-1. Theselow concentrations of Ca2+ and Mg2+ translate into a low hardness (<116mg∙L-1 as CaCO3). According to the Durfor and Becker (1964) classification,the household tap water is classified as soft to moderately hardwater. In general, all samples are characterized by low concentrationsof Na+ (2.7–36.7 mg∙L-1) and potassium K+ (1.4 – 6.9 mg∙L-1).
The concentrations of Ag, Cd, Co, Ni, Pb, Se and V in all samplesof household water were very low (<DL of the analytical method used).The concentrations of Zn range between 0.027 and 0.916 mg∙L-1, onlythree samples were <DL. In most samples, the Mn content was low or <DL (Table 2), and only in five samples was above the detection limit,but with concentrations that are consistently low (0.031–0.088 mg∙L-1).On the other hand, the contents of As were equally low since in mostsamples were <DL. Samples 27, 28 and 30 had concentrations exceedingthis limit which varied between 0.011 and 0.018 mg∙L-1. The contents of Sr varied between 0.033 and 0.363 mg∙L-1. Concentrations of Fe rangebetween <DL and 0.808 mg∙L-1, whereas those of Al varies between<DL and 2.68 mg∙L-1. The concentrations of Ba were low (0.021–0.107mg∙L-1). Finally, the Cu contents ranged from 0.026 to 0.230 mg∙L-1, except in six samples which had concentrations <DL.
The household water samples contain a number of CFUs of variableTC and fall in the range of 0 to16 CFU/100 mL. The number ofE. coli CFUs in the samples ranges from 0 to 8 CFU/100 mL (Table 3).
The chemical composition of water reaching households variesgreatly and shows that during water distribution and storage, it undergoesimportant transformations. Comparing the household sampleswith sample 3 from the output of the WTP, the household water showsimportant variations in pH, turbidity, Fe and Al contents as well as inTC and fecal coliforms (FC). Sample 3 does not present FC or TC butup to 8 CFU of E. coli and up to 16 CFU of TC were detected in thehousehold water.
Chemical composition of particulate matter
Taxco potable water shows a notorious turbidity. Turbidity inhousehold samples is generated by suspended particles generally ofthe size of clays (<2 μm) and colloids (<0.3 μm) resulting from an incomplete and/or inefficient flocculation process in the WTP. Theresults of partial extraction with HNO3 are presented in Table 4.
Concentration of metals extractable with HNO3 in particulate matter of household tap water sample 13.

Sequential extraction
The distribution of the analyzed metals (Cd, Co, Cu, Fe, Mn, Pb,V, Zn and As) in the sequential extraction is quite variable indicatingthat the metals are associated with different chemical fractions (Figure 3, Table 5). Cd, As and Pb are the metals of major ecotoxicologicalinterest due to the effects they have on biota, including humans (Fergusson, 1990).
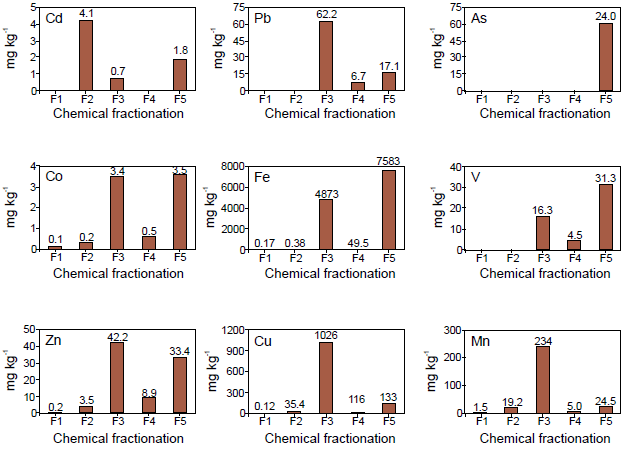
Figure 3
Chemical speciation of the potentially toxic elements detected in the particulate matter of one household tap water sample from Taxco de Alarcon.
F1: Exchangeable fraction (soluble + adsorbed); F2: Fraction bound to carbonates; F3: Fraction associated with Fe-Mn oxyhydroxides; F4: Fraction bound to sulfidesand organic matter; F5: Residual fraction (insoluble).
Chemical fractionation in particulate matter of household tap water sample 13.
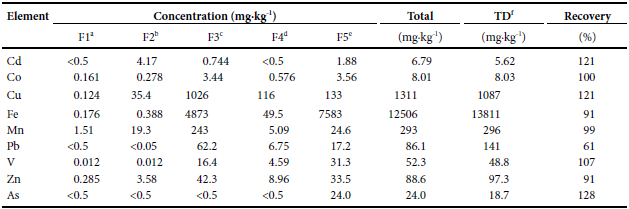
a Exchangeable fraction (soluble + adsorbed)b Fraction bound to carbonatesc Fraction associated with oxyhydroxides of Fe-Mnd Fraction bound to sulfides and organic mattere Residual fraction (insoluble)f Metal content in total digestion
The element Cd is mainly associated with fraction 2 or carbonatedfraction (4.17 mg∙kg-1), followed by fraction 5 or residual fraction(1.88 mg∙kg-1). The fraction of Cd bound to Fe-Mn oxyhydroxides (fraction 3) is lower at 0.744 mg∙kg-1, and the content in the exchangeablefraction (fraction 1) and those linked to sulfides and organic matter (fraction 4) is very low or <DL of the method. Although Cd was notdetected in fraction 1, which is the most available, the presence of thismetal adsorbed to fractions 2 and 3 indicates that there is a moderatepotential for releasing them. A change to lower pH (<5.5), as occursin sample 11, will release Cd from the particulate matter.
The highest concentration of Pb (62.2 mg∙kg-1) is associated with the Fe-Mn oxyhydroxides fraction followed by the residual fraction(17.15 mg∙kg-1) and, to a lesser extent, in the fraction associated withsulfides and organic matter (6.75 mg∙kg-1). The exchangeable fractionand that associated with carbonates are below the DL of the method.Similar to Cd, the fraction associated with Fe-Mn oxyhydroxides willbe released if the pH of the water decreases below 5.5. The Pb fractionassociated with sulfides and organic matter will be more difficult torelease since it requires values of pH <2.0, conditions not registeredin the samples studied.
Fortunately, total arsenic is associated with the residual fraction,so the likelihood of its release is extremely low.
Co, Fe, V and Zn have a similar speciation and differ only by theabsolute concentrations in each fraction. These metals are mostly bound to the Fe-Mn oxyhydroxides fraction followed by the residualfraction and the fraction associated with sulfides and organic matter. The most available fractions (1 and 2) have the lowest concentrationsin these metals (0.161 and 0.278 mg∙kg-1, respectively). In particular,the concentration of Co in fraction 3 and 5 (3.44 and 3.56 mg∙kg-1,respectively) are similar, whereas the concentration in fraction 4 ismuch lower (0.576 mg∙kg-1). The lowest Fe concentration was foundin the exchangeable fraction (0.176 mg∙kg-1) and in that associatedwith carbonates (0.388 mg∙kg-1), whereas the highest concentrationwas found in the residual fraction (7583 mg∙kg-1), followed by fraction 3 (4873 mg∙kg-1), and finally fraction 4 (49.53 mg∙kg-1). The lowestconcentrations of V were found in fractions 1 and 2 (0.012 mg∙kg-1)and the highest concentration was found in fraction 5 (31.34 mg∙kg-1).In fraction 3 the concentration of V is 16.4 mg∙kg-1. Finally, in fraction4 the content of V is 4.59 mg∙kg-1. The content of Zn is variable;the fraction with the highest content is 3 followed by fraction 5. Thelowest content of Zn was found in the exchangeable fraction. The Znconcentrations in the rest of the fractions are less than 10 mg∙kg-1. Cuand Mn have a similar distribution. The highest concentration of Cuand Mn is found in fraction 3, followed by fraction 5 and the lowestconcentration is found in fraction 1 (Figure 3).
DISCUSSION
Household water quality with respect to NOM-127-SSA1-1994 standard
Table 6 shows a comparison between the physicochemical andbacteriological parameters analyzed in household water samplesand the permissible limits established in the NOM-127-SSA1-1994 standard. Samples 19 and 26 are the only two samples that met thestandard. Figure 4 shows the spatial distribution of samples that exceedpermissible limits in turbidity, Al, pH, Fe, FC and TC. Figure 4a–4f, respectively), established in the NOM-127-SSA1-1994 standard.
Range values of physicochemical parameters of Taxco de Alarcon household tap water, compared with the permissible limits inNOM-127–SSA1-1994 standard.
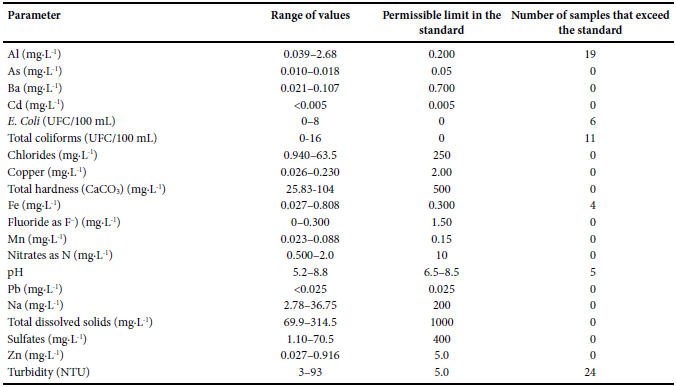
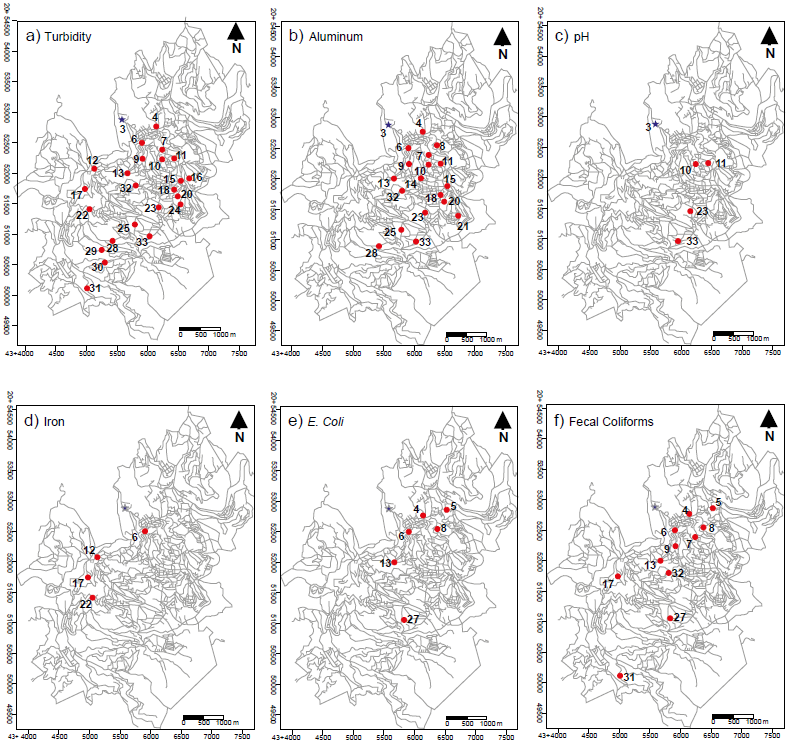
Figure 4
Location of sampling sites of household water samples that do not meet the permissible limits established in the NOM-127-SSA1-1994
Blue star is thelocation of the water treatment plant.
The pH is one of the physicochemical parameters with the greatestinfluence on water chemistry. It controls the conductivity, TDS content,alkalinity, hardness and metal content, among other parameters. In thisstudy, the pH plays an important role in the interaction of water withthe distribution pipes, an acid pH causes the dissolution of metallicmaterials of the pipe; in the case of pipes of different materials, e.g.plastic or cement, the effect is less. Also, a high pH causes the accumulationof precipitates.
Turbidity is important due to the fact that suspended particlesare mainly composed of clays and organic matter, which have a highretention capacity of potentially toxic elements such as metals and As.Under reducing conditions, the toxic elements retained in the claysbecome soluble; and therefore, their concentration in water increases (Romero, 2001).
On the other hand, the high Al contents recorded in the potablewater samples has its origin in the Al2(SO4)3 used in the WTP forthe flocculation process. At least 19 samples exceed the maximumpermissible limit in the standard (0.20 mg∙L-1) reaching up to 13.4 times the permissible limit, as in sample 11 (Figure 5a). Any excess ofthis reagent will be solubilized and ionic aluminum will be released.
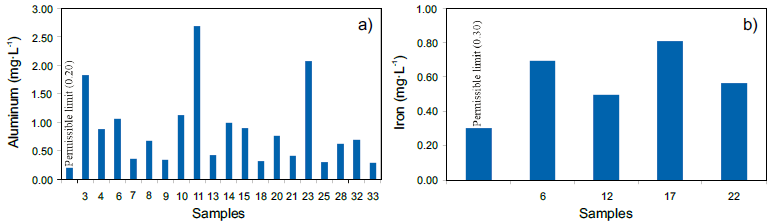
Figure 5
Samples that exceed the permissible limits established in the Official Mexican standard NOM-127-SSA1-1994. a) Aluminum, and b) iron.
The accumulation of aluminum in the brain correlates with the onsetof Alzheimer's (Emsley, 1998). On the other hand, the presence ofiron (Fe) can be explained by the fact that the water from the WTPhas a slightly acidic pH, probably dissolving Fe from the distributionpipelines. At least four samples exceed the permissible limit of 0.30mg∙L-1 and reach up to 2.7 times the permissible limit (Figure 5b).Although Fe is an essential element for humans in small doses(10–30 mg∙d-1), high doses can cause health problems (Emsley, 1998).
Finally, fecal coliforms (FC) are subdivided into subgroups andone of them is E. coli which is a bacteria found in human and animalfeces, therefore, when the water contains E. coli, it indicates that thewater could have been contaminated with human or animal feces, and this represents a health risk (Edberg et al., 2000). The treated waterof the WTP did not show the presence of TC or E. coli. There aresome factors that can lead to the pollution with coliforms. The watertreated in the WTP is distributed to the households using the gravityforce. Nevertheless, the difference of height between the WTP andthe households results in high pressure in the water network, thuspressure reducing boxes are necessary. Those boxes are found outsidethroughout the city and are not hermetically sealed and represent asource of contamination for the treated water. On the other hand, mostof the households store the water in tanks that not receive maintenance.Thus, the contamination is related to water transportation or storagein tanks at homes.
Potential affectation by the toxic elements contained in thesuspended particles
The distribution of the analyzed elements (Cd, Co, Cu, Fe, Mn, Pb,V, Zn and As) in the different chemical fractions of the particulates varygreatly. Although the chemical behavior of these elements is variableand depends on the chemical fraction, their presence in the suspendedparticles in the household water represents a potential risk to the healthof the users if the polluted water is drank.
Under acidic conditions and reduced Eh, the metals are soluble (Tessier et al., 1979; Fergusson, 1990). If the metals are in a basic mediumand with an oxidizing Eh, they will oxidize and become insoluble.In the case of Taxco de Alarcon, 90 % of the water that reaches thehouses presents a circum-neutral to slightly basic pH (6.5–9.0) andan oxidizing Eh. Therefore, it is very likely that the metals remaininsoluble and are retained in the suspended particles.
The elements contained in the exchangeable fraction represent themost soluble elements and, therefore, have greater bioavailability. Theamount of metals adsorbed or retained on the surface of a mineral dependson the surface area of each mineral. On the other hand, the typeof adsorbed metals depends on the Point Zero Charge (PZC) of eachmineral and the pH of the medium in which they are found (Manningand Goldberg, 1996). For example, the PZC of Fe oxides ranges from 5 to 8. When the pH of the water is lower than the PZC of Fe oxides,the surface of this mineral is positively charged, thus attracting allnegatively charged compounds and retaining them on its surface. Incontrast, when the pH of the water is higher than the PZC, the surfaceof this mineral is negatively charged releasing the negatively chargedelements and retaining positively charged metals. The particulate matterhas low but significant concentrations of Co, Cu, Fe, Mn, V andZn in the exchangeable fraction, which will become soluble with smallchanges in the physicochemical conditions of the water.
The elements associated with the carbonate fraction are consideredto be moderately bioavailable. A pH<6.5 is required to release the elementsassociated with this fraction. The matter has high concentrations of Cd, Cu and Mn, and smaller but significant in Co, Fe, V and Zn, inthe fraction associated with carbonates. Samples 3, 10, 11 and 23 havepH values <6.5 and represent the highest potential for releasing theseelements associated with the carbonate fraction.
In the Fe-Mn oxyhydroxide fraction, the associated elements areconsidered to be moderately bioavailable. In this case, the elementscontained could be released into the environment when the pH of themedium reach values <5.5. In the analyzed particulate matter, highconcentrations of Cu, Fe, Mn, Pb, V, and Zn, and lower concentrationsof Co, were detected in the fraction of oxyhydroxides. Sample11 is the only one that has a pH value <5.5. However, the elementscontained in this fraction will become bioavailable when the waterundergoes additional important external transformations (Fergusson,1990).
The elements bound to the fraction of sulfides and organic matterare considered to have low bioavailability. Extreme pH conditions(<2.0) are necessary for release the elements. This situation is unlikelyto occur in both the distribution system and the homes. As an example,an acid pH (~1) is reached in gastric juices. In environments likethese, the elements associated to sulfides and organic matter fractionrepresent a potential hazard. Fortunately, tap water is not usually usedfor drinking. The elements associated to the fraction of sulfides andorganic matter in the particulate matter are Cu and Fe and in smallerproportion Co, Mn, Pb, V and Zn.
Finally, the elements associated with the residual fraction areconsidered non-bioavailable because they are associated with silicateminerals with a very low solubility product, and require extremephysicochemical conditions over a long period of time. The elementsassociated with this fraction do not represent a potential danger forbiota. In the particulate matter, all the analyzed elements are presentin this fraction and stand out for their concentration: Fe (7583 mg∙L-1)and, due to its toxicological importance, Pb (17.15 mg∙L-1) and As(24.0 mg∙L-1). The latter was only detected in residual fraction so itdoes not represent a potential danger to the inhabitants.
CONCLUSIONS
The water supply to the city of Taxco de Alarcon comes from surfacewater from rivers, dams and springs of the zone. The main supplycomes from the Chontalcuatlan river, the springs of Chacuhalco andTeneria within the city, and from the San Marcos and El Sombreritodams. The obtained and analyzed results indicate that the waterquality from these sources is not adequate for potable use becausethe Chacuhalco spring presents turbidity levels above the NOM-127-SSA1-1994. In addition, eight CFU/100 mL of TC were detected.Mixed water from Chontalcuatlan river water, the Teneria spring, andthe San Marcos and El Sombrerito dams is not suitable for humanconsumption since they do not comply with Mexican regulations;they exceed the limits in barium and have more than 20 FC and morethan 100 CFU/100 mL TC. For these reasons, the water from thesesources is treated in the WTP before sending the water to the homes.Unfortunately, the chemical analyzes carried out in the plant's outputwater indicate that the treatment process is inefficient because the watersent to the homes is slightly acidic (pH 5.7), below the range establishedin the Mexican standard (6.5–8.5). Furthermore, suspended particleswere observed, which results in a very high turbidity value (32 NTU)compared to that established in the standard (5 NTU). On the otherhand, the output water contains high concentrations of Al (1.82 mg∙L-1)that exceed the maximum permissible limit in the standard (0.200mg∙L-1)in up to 9 times.
Particulate matter is characterized by high concentrations of total metals (Al, Cu, Fe, Pb, As, Cr, V, Zn, Sr, Ni). The speciation showedthat Cu, Mn, Zn, Co, V and Fe have different concentrations in theexchangeable fraction and carbonate-associated fraction. Cd is associatedwith the carbonate fraction; the Pb is in the fraction associatedwith Fe-Mn oxyhydroxides, whereas the As is in the residual fraction,which is the most stable chemical fraction.
Finally, samples 19 and 26 from households were the only two samplesthat meet the Official Mexican Standard (NOM-127-SSA1-1994)in all measured parameters. However, overall the results provideinformation on the quality of potable water in the city of Taxco deAlarcon and indicate that modifications must be carried out in thewater treatment processes, especially in the distribution network andwater storage tanks, so that the water meets the standards of qualityestablished by current regulations.
Acknowledgments
The authors gratefully acknowledge the input and suggestions ofthe Editor and Reviewers, which allowed us to significantly improvethe manuscript. Part of this contribution was part of a bachelor thesisby Guadalupe Sanchez Montoya. The authors also thank Travis DeaneAshworth for reviewing the English version of the paper.
REFERENCES
Cifuentes, E., Blumenthal, U.J., Ruiz-Palacios, G., Bennett, S., Peasey, A., 1994,Escenario epidemiologico del uso agricola del agua residual: el Valle delMezquital, Mexico: Salud Publica, Mexico, 36(1), 3-9.
CONAGUA (Comision Nacional del Agua), 2016, Situacion del SubsectorAgua Potable, Drenaje y Saneamiento Edicion 2016: Ciudad de Mexico,Secretaria de Medio Ambiente y Recursos Naturales, Comision Nacionaldel Agua, 189 pp.
CRM (Consejo de Recursos Minerales) 1999, Monografia geologico-mineradel Estado de Guerrero: Pachuca, Hgo., Mexico, Secretaria de Comercioy Fomento Industrial, Consejo de Recursos Minerales, PublicacionM-19e, 262 pp.
Corona-Chavez, P., Maldonado, R., Ramos-Arroyo, Y.R., Robles-Camacho,J., Lozano-SantaCruz, R., Martinez-Medina, M., 2017, Geoquimicay mineralogia de los jales del distrito minero Tlalpujahua-El Oro,Mexico, y sus implicaciones de impacto ambiental: Revista Mexicanade Ciencias Geologicas, 34(3), 250-273. doi: http://dx.doi.org/10.22201/cgeo.20072902e.2017.3.540.
Corrales-Perez, D., Martin-Romero, F., 2018, Adecuaciones para mejorarla aplicacion del metodo D3987-85 en la extraccion de EPT de losantiguos residuos mineros El Fraile, Guerrero, Mexico: Revista Mexicanade Ciencias Geologicas, 35(1), 1-17. doi: http://dx.doi.org/10.22201/cgeo.20072902e.2018.1.536.
Covarrubias, S.A., Pena Cabriales, J.J., 2017, Contaminacion ambiental pormetales pesados en Mexico: problematica y estrategias de fitorremediacion:Revista Internacional de Contaminacion Ambiental, 33, 7-21. doi10.20937/RICA.2017.33.esp01.01.
Des Rivieres, J., Beals, D., 2011, The Taxco mining District. Mineralogical: Record,42(5), 401-445. doi: http://dx.doi.org/10.22201/cgeo.20072902e.2018.1.536.
Durfor, C.N., Becker, E., 1964, Public Water Supplies of the 100 Largest Citiesin the United States, US Geog. Sur: Water Supply Paper, 1812, 364 pp.
Edberg, S.C., Rice, E.W., Karlin, R.J., Allen, M.J., 2000, Escherichia coli: the bestbiological drinking water indicator for public health protection: Journalof Applied Microbiology, 88, 106S-116S.
Emsley, J., 1998, The elements: Oxford University Press, Oxford ChemistryGuides, v. 2, 292 pp.
Fergusson, J.E., 1990, The heavy elements: Chemistry, environment impact andhealth effects: Oxford, England, Pergamon Press, 614 pp.
Gomez-Alvarez, A., Villalba-Atondo, A., Acosta-Ruiz, G., Castaneda-Olivares,M., Kamp, D., 2004, Metales pesados, en el agua superficial del Rio SanPedro durante 1997 y 1999: Revista Internacional de ContaminacionAmbiental, 20(1), 5-12.
Gordillo-Martinez, A.J., Cabrera-Cruz, R.B.E., Hernandez-Mariano, M.,Galindo, E., Otazo, E., Prieto, F., 2010, Evaluacion regional del impactoantropogenico sobre, aire, agua y suelo. Caso: Huasteca Hidalguense,Mexico: Revista Internacional de Contaminacion Ambiental,26(3), 229-251.
Hernandez-Flores, G., Solorza-Feria, O., Poggi-Varaldo, H.M., 2017,Bioelectricity generation from wastewater and actual landfill leachates:A multivariate analysis using principal component analysis: InternationalJournal of Hydrogen Energy, 42(32), 20772-20782, http://dx.doi.org/10.1016/j.ijhydene.2017.01.021.
INEGI (Instituto Nacional de Estadistica, Geografia e Informatica), 2013, Perfilsociodemografico, Estados Unidos Mexicanos, Censo de Poblacion yVivienda 2010: Mexico, INEGI, 110 pp.
Literathy, P., 1996, Industrial wastes and water pollution: Regional Approachesto Water Pollution and Environment, Nato ASI Series, 20, 21-32.
Lu, J.L., 2011, Insecticide residues in eggplant fruits, soil, and water in thelargest eggplant-producing area in the Philippines: Water, Air and SoilPollution, 220(1-4), 413-422.
Manning, B.A., Goldberg, S., 1996, Modeling Arsenate Competitive Adsorptionon Kaolinite, Montmorillonite and Illite: Clays and Clay Minerals, 44(5),609-623.
Massoudieh, A., Bombardelli, F.A., Ginn, T.R., 2010, A biogeochemical modelof contaminant fate and transport in river waters and sediments: Journalof contaminant hydrology, 112, 103-117.
Secretaria de Salud, 2000, Modificacion a la Norma Oficial Mexicana NOM-127-SSA1-1994, Salud ambiental. Agua para uso y consumo humano. Limitespermisibles de calidad y tratamientos a que debe someterse el agua parasu potabilizacion: Ciudad de Mexico, Diario Oficial de la Federacion, 22de noviembre de 2000, 73-79.
Pulido-Bosch, A., 2007, Nociones de hidrogeologia para ambientologos: Espana,Universidad de Almeria, 492 pp.
Qu, X., Alvarez, P.J.J., Li, Q., 2013, Applications of nanotechnology in water andwastewater treatment: Water research, 47, 3931-3946.
Romero, M.F., 2001, Procesos Geoquimicas de liberacion y trasporte de metalespesados y metaloides en residuos mineros (jales): Mexico D.F., Mexico,Universidad Nacional Autonoma de Mexico, tesis de doctorado, 76 pp.
Romero, F.M., Armienta, M.A., Gonzalez-Hernandez, G., 2007, Solid-phasecontrol on the mobility of potentially toxic elements in an abandonedlead/zinc mine tailings impoundment: Applied Geochemistry, 22, 109-127.
Sjomander Magnusson, T., van der Merwe, B., 2005, Context driven policydesign in urban water management. A case study of Windhoek, Namibia:Urban Water Journal, 2(3), 151-160.
Smith, K.S., Huyck, H.L.O., 1999, An overview to the abundance, relativemobility, bioavailability and human toxicity of metals, in Plumlee, G.S.,Logsdon, M.J. (eds), The Environmental Geochemistry of MineralDeposits: Reviews in Economic Geology, 6A, 29-70.
Sun, H., Shi, B., Yang, F., Wang, D., 2017, Effects of sulfate on heavy metalrelease from iron corrosion scales in drinking water distribution system:Water Research, 114, 69-77.
Talavera-Mendoza, O., Armienta-Hernández,, M.A., Garcia-Abundis, J.,Flores-Mundo, .N., 2006, Geochemistry of leachates from the El Frailesulfide tailings piles in Taxco, Guerrero, southern Mexico: EnvironmentalGeochemistry and Health, 28, 243-255.
Talavera-Mendoza, O., Ruiz J, Diaz-Villasenor, E., Ramirez-Guzman, A.,Cortes A., Salgado-Souto, S.A., Dotor-Almazan, A., Rivera-Bustos,. R.,2016, Water-rock-tailings interactions and sources of sulfur and metalsin the subtropical mining region of Taxco, Guerrero (southern Mexico):A multi-isotopic approach: Applied Geochemistry, 66, 73-81.
Tessier, A., Campbell, P.G.C., Bisson, M., 1979, Sequential Extraction Procedurefor the speciation of particulate trace Metals: Analytical Chemistry, 51(7),845-851.
Author notes
