Article / Autopsy Case Report
Advanced chronic lymphoid leukemia with severe bronchopneumonia: an Autopsy Case Report
Advanced chronic lymphoid leukemia with severe bronchopneumonia: an Autopsy Case Report
Autopsy and Case Reports, vol. 6, no. 1, pp. 11-20, 2016
São Paulo, SP: Universidade de São Paulo, Hospital Universitário
Received: 11 November 2015
Accepted: 15 January 2016
DOI: 10.4322/acr.2016.022
Abstract: Chronic lymphocytic leukemia (CLL) is a lymphoid neoplasia with the B immunophenotype, which corresponds to the leukemic form of lymphocytic lymphoma. This entity is characterized, in most cases, by immunosuppression due to impaired function of immune cells, hypogammaglobulinemia, bone marrow infiltration, and immune dysfunction due to the neoplasia and the chemotherapy, when prescribed. We describe the case of a 63-year-old woman with a previous diagnosis of advanced CLL, refractory to treatment, who presented respiratory failure at the emergency department and died soon after hospital admission. The autopsy examination showed a large retroperitoneal mass compressing large vessels and abdominal and pelvic organs; generalized lymphadenopathy; and liver, spleen, bone marrow, heart and kidney infiltration. A Gram-negative bacilli bronchopneumonia with diffuse alveolar damage was detected, which was likely to be the immediate cause of death.
Keywords : Leukemia, Lymphocytic, Chronic, B-Cell, Respiratory Insufficiency, Pneumonia, Autopsy.
CASE REPORT
A 63-year-old Caucasian woman, who was previously diagnosed with type-2 diabetes, hypertension, and hypothyroidism, presented a 2-year history of weight loss (nearly 20 kg) intermittent fever, nocturnal sweating, progressive abdominal distension, and generalized lymphadenopathy. Her first complete blood count showed a hemoglobin of 8.0 g/dL (reference value [RV]: 12.3-15.5 g/dL), neutrophils of 3.510/mm 3 (RV: 4.4-11.3 × 10 3/mm 3), lymphocytes of 112,320/mm 3 (RV: 1.2-4.0 × 10 3/mm 3, and platelets of 83,000/mm 3 (RV:150-400 × 10 3/mm 3). The flow cytometric immunophenotyping of the peripheral blood, bone marrow aspirate, and a needle biopsy on the right axillary lymph node rendered the diagnosis of chronic lymphoid leukemia (CLL)/lymphocitic lymphoma. The bone marrow aspirate showed more than 90% of mature lymphocytes CD20-positive, CD5-positive, CD10-negative, and immunoglobulin M (IgM)-negative. The lymph node histology showed complete replacement of the lymph node architecture by small lymphoid cells ( Figure 1A). The immunohistochemistry was positive for CD-20 (Cell Marque™ L26), CD-23 (Cell Marque™ 1B12), and CD-5 (Cell Marque™ 4C7); and negative for CD-3 (Cell Marque™ MRQ-39), cyclin-D1 (Spring™ SP4), and CD-10 (Cell Marque™ 56C6) ( Figure 1B, D). Zeta-chain-associated protein kinase-70 (ZAP-70) (Ventana™ 2F3.2) was positive ( Figure 2) and p53 (Cell Marque™ DO7) was negative. Anti- LEF-1 was not available. The Ki-67 (Spring™ SP6) showed a low index of positivity ( Figure 1E). The bone marrow biopsy was hypercellular due to CLL interstitial infiltration. The peripheral blood karyotypes test was positive for trisomy 12. The positron emission tomography was not consistent with signs of high-grade non-Hodgkin's lymphoma transformation. The clinical evaluation set the disease as an advanced stage (Rai stage IV and Binnet stage C with a bulky abdominal mass measuring more than 10.0 cm and the presence of “B symptoms”). A transthoracic echocardiogram showed concentric left ventricle hypertrophy, with an ejection fraction of 63%.
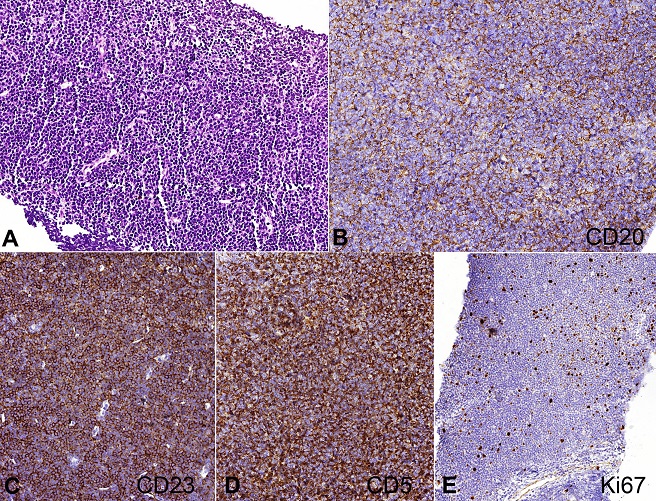
Figure 1
Photomicrography of the lymph node showing typical histology and immunophenotype of a chronic lymphoid leukemia diagnosis. A - Lymph node obliterated by small and mature lymphoid cells (H&E, 100X); B - CD20 membranous positivity (IH-Cell Marque™ L26 clone, 200X); C - CD23 positivity (IH-Cell Marque™ 1B12, 200X); D - CD5 aberrant positivity (IH-Cell Marque™ 4C7 clone, 200X); E - Ki67 low index positivity (IH-Cell Marque™ SP6 clone, 200X).
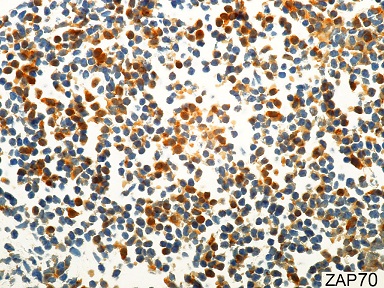
Figure 2
Photomicrography of the lymph node showing partial cytoplasmic positivity for ZAP-70 of the neoplastic cells with moderate or strong intensity (Immunohistochemistry-Ventana™ 2F3.2, 600X).
The patient received four cycles of fludarabine-based chemotherapy associated with cyclophosphamide and dexamethasone. Unfortunately, the rituximab was not available. The patient developed pneumonia and episodes of febrile neutropenia. A discrete renal dysfunction was detected, which was probably due to infection; however, normal renal function was restored after fluid replacement. After 3 months of treatment, the patient was brought to the emergency facility with respiratory failure, signs of circulatory shock, and altered mental status. According to her family, she had recently complained of dyspnea and a cough with bloody sputum, which worsened on the way to the hospital. On admission she presented cardiac arrest, which did not respond to advanced cardiac life support maneuvers. During orotracheal intubation, a large amount of frothy bloody secretion was drained from the airways. The autopsy was performed.
AUTOPSY FINDINGS
The corpse weighted 46.0 kg and measured 149.0 cm in length (body mass index 20.72). During the external examination, an abdominal bulging on the left side was noticed.
At the opening of thoracic cavity, the lungs were heavy and hardened showing pleuropulmonary adhesions. The right lung weighed 554.0 g (mean [m] RV: 465 g) and the left lung: 702.0g (mRV: 400 g). At the cut surface, a moderate amount of foamy and serohemorrhagic fluid drained after parenchymal compression. Acute bronchopneumonia and histological signs of diffuse alveolar damage, represented by the formation of hyaline membrane, septal necrosis, and foci of pulmonary hemorrhage, were disclosed at microscopy. Septal vessels had fibrinoid necrosis and mild bronchiolar inflammation. Gram-negative bacilli (Brown-Hopps staining), permeating the bronchi wall as well as the small septal vessels, extending into the adjacent pulmonary parenchyma, were found. Septal congestion, recent alveolar edema, and siderophages (hemosiderin-containing macrophages) within alveolar spaces were other evident features ( Figure 3).
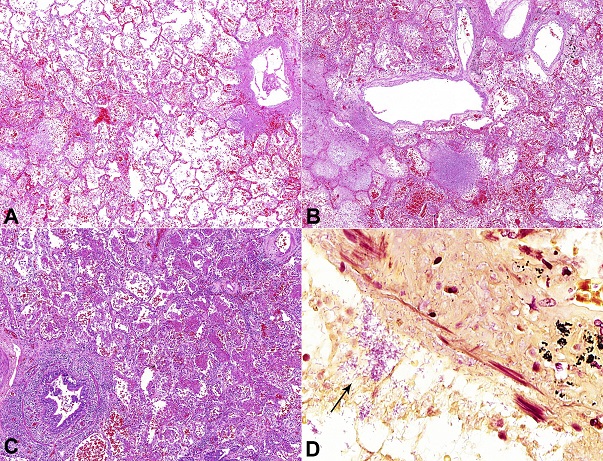
Figure 3
Photomicrographs of the lungs showing acute lung damage due to Gram-negative bacilli pneumonia: A - Septal congestion, alveolar edema, and foci of hemorrhage (H&E, 50X); B - Foci of hemorrhage and septal alveolar necrosis with cellular debris. Note the inflammatory reaction is not exuberant (H&E, 50X); C - Congestion and organizing pneumonia (H&E, 50X); D - Gram-negative bacilli (arrow) within the lumen and permeating the wall of anthracotic bronchi, indicating the airways as the route of acquisition of the pneumonia. The ciliated columnar epithelium was slaughtered (Brown-Hopps stain, 400X).
The heart weighted 260.0 g (mRV: 260 g); the coronary arteries were tortuous and obstructions did not exceed 40%; mild/moderate calcification of the aortic valve was observed. Microscopically, cardiomyocyte hypertrophy and rounded foci of small lymphocytes in the epicardial fat layer compatible with CLL infiltration ( Figure 4) were observed.
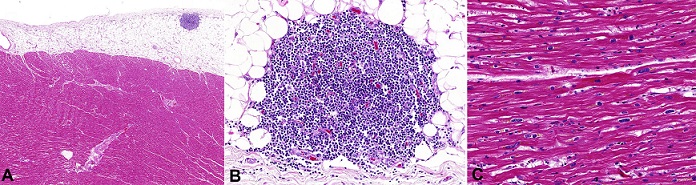
Figure 4
Photomicrography of the heart: A and B - A nodular lymphoid infiltrate in the epicardial fat composed of small lymphocytes compatible with CLL infiltration (H&E: A 20X; B 200X); C - Cardiomyocyte hypertrophy containing lipofuscin pigment (H&E, 200X).
At the opening of the peritoneal cavity, a bulging, lobulated, and well-defined retroperitoneal mass measuring 24.0 × 20.0 × 13.0 cm and weighing 3076.0 g was found, which compressed the large vessels, and the abdominal and pelvic viscera ( Figure 5A, B). Generalized lymphadenopathy (para-aortic, paracaval, mesenteric, mesocolic, hepatic hilum, splenic hilum, peripancreatic, iliac, mediastinal, bilateral cervical, supraclavicular, axillary, and inguinal) was observed, with lymph nodes measuring from 2.0 to 10.0 cm at their longest axis. At the cut surface, the retroperitoneal mass and the lymph nodes were light-brown, bright, homogeneous, and fleshy, with firm-elastic consistency on palpation ( Figure 5C). Microscopic examination revealed diffuse proliferation of small lymphocytes, without necrosis or mitotic figures, alternating with some vaguely nodular areas composed by slightly larger cells ( Figure 5D, E).
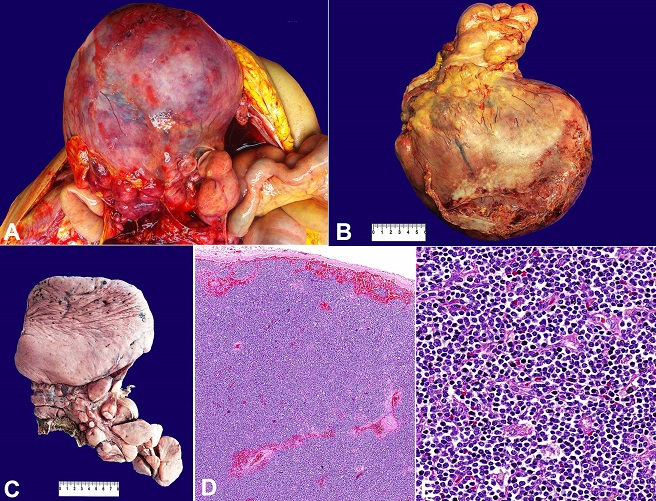
Figure 5
Gross finding of the retroperitoneal mass. A, B and C - The mass was formed by a conglomerate of lymph nodes, measuring up to 24.0 × 20.0 × 13.0 cm (C, after fixation in formalin 10%); D - Photomicrography of the abdominal mass showing the obliteration of lymph nodes by small lymphocytes, in a similar pattern to the biopsy at Figure 1 (H&E, 50X); E - Histologic detail of the neoplasia formed by small mature lymphocytes without large cells (H&E, 400X).
The bone marrow sampled from the iliac crest was infiltrated by small lymphocytes representing more than 90% of marrow cellular tissue ( Figure 6A, B). The spleen had a marked expansion of the white pulp due to atypical lymphoid cells ( Figure 6C, D).
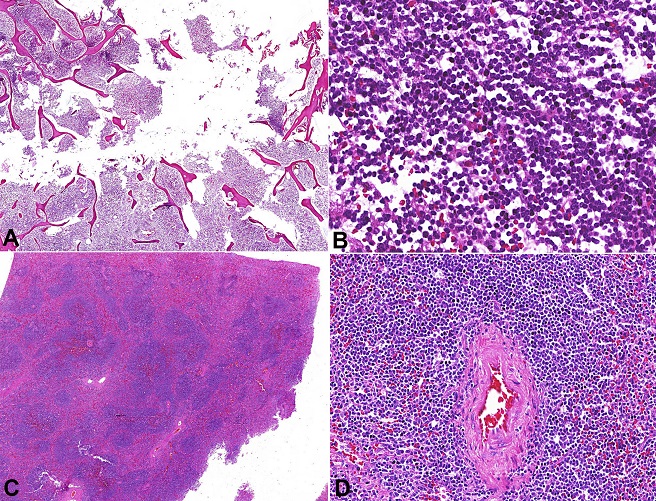
Figure 6
Photomicrographs of the bone marrow and the spleen infiltrated by chronic lymphoid leukemia (CLL). A - Interstitial and nodular patterns of CLL infiltration, replacing almost the entire marrow space (H&E, 10X); B - Detail of the nodule in the marrow space formed by small lymphocytes (H&E, 400X); C - Expansion of the splenic white pulp due to small mature lymphocytes (H&E, 10X); D - A detail of the splenic central artery surrounded by small mature lymphocytes (H&E, 200X).
The liver had a sinusoidal dilatation around the central vein, while the portal tracts were extensively infiltrated by atypical lymphocytes, which were morphologically similar to those described in the lymph nodes ( Figure 7A, B).
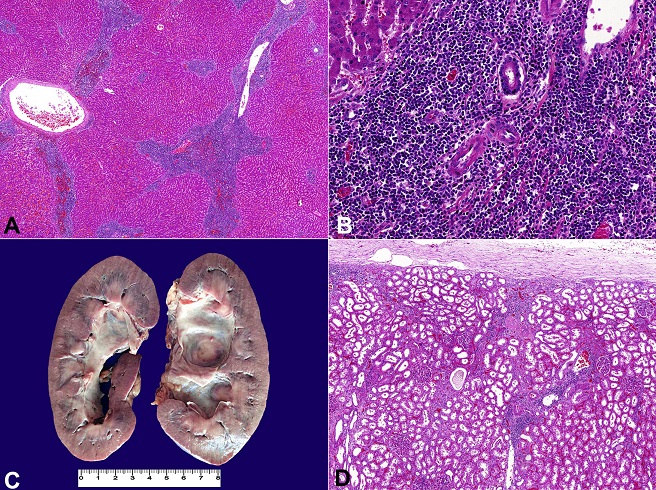
Figure 7
Liver and renal involvement in chronic lymphoid leukemia. A - photomicrography of the liver showing expanded portal tracts due to infiltrative small mature lymphocytes (H&E, 20X); B - Photomicrography showing a detail of the portal tract infiltrated by small mature lymphocytes (H&E, 200X); C - Gross appearance of the kidneys showing bilateral dilated renal pelvis due to ureteral compression by enlarged lymph nodes (kidneys fixed with formalin 10%); D - Photomicrography showing interstitial neoplastic infiltration of the renal parenchyma (H&E, 50X).
The aorta and its branches had multiple atherosclerotic plaques, some of them ulcerated, including the ostia of the renal arteries. The kidney volumes were mildly increased (right kidney weighed 164.0 g; left kidney 158.0 g [RV of both: 288 g]) exhibiting a focus of CLL infiltration, nephrosclerosis, and acute tubular necrosis ( Figure 7C, D). There was no evidence of high-grade transformation or association with Hodgkin's lymphoma in any of the organs examined after extensive sampling. The remaining organs and structures did not show unusual histological features.
The findings of the autopsy point towards the septic shock and acute respiratory failure due to Gram-negative pneumonia and diffuse alveolar damage as the immediate cause of death in a patient with advanced CLL. The pulmonary edema observed was likely caused by a synergistic pathogenic mechanism, involving the infection and left ventricular diastolic dysfunction due to chronic arterial hypertension.
DISCUSSION
The case reported herein describes a woman who died of a leukemized and refractory non-Hodgkin's lymphoma. The CLL is a B-immunophenotype lymphoid neoplasia that represents the leukemic form of lymphocytic lymphoma. 1,2 CLL corresponds to 5-10% of non-Hodgkin's lymphomas. 90% of CLL, in the United States and Europe, affects patients older than 50 years (usually between 55 and 65 years of age) and is twice as common in men. It is the most common form of leukemia in adults. 2-5
The criteria used for differentiating lymphoma and leukemia is based on the number of circulating lymphocytes in the peripheral blood. Thus, when the monoclonal lymphocytes count is greater than 5000 cells/mm 3, the leukemia diagnosis is established. In these cases, the histological and immunohistochemical methods are not essential for this purpose. 1,2 CLL is characterized by an indolent-course generalized lymphadenopathy. When the lymphocytic lymphoma becomes leukemic, extra-nodal involvement becomes more frequent. Over 90% of patients present advanced-stage disease (stage III and IV) at the time of diagnosis, with bone marrow involvement in 70-80% of the cases, and liver and/or spleen infiltration in 25–55% of cases. 6,7 The “B symptoms” (weight loss, fever, and night sweats) occur in up to 10% of the cases. 1 Hemolytic anemia, thrombocytopenia, and other autoimmune phenomena are observed in fewer than one-third of cases. 8 Immune dysfunction leading to immunosuppression is described in a relatively high proportion of cases, even when there is no bone marrow failure. Some authors 8,9 consider the CLL immunosuppression arising from the dysfunction of T-cells, NK-cells, neutrophils, and macrophages stemming from the deregulation in interleukin production (IL-6 and IL-10), the tendency to Th2 polarization of the immune response, the cell-to-cell interactions, and the direct cytotoxicity. Hypogammaglobulinemia is described in up to 40% of CLL cases and contributes to the vulnerability to infection. 8 The majority of infections are due to bacteria ( Streptococcus pneumoniae, Staphylococcus aureus, Haemophillus influenza, and Gram-negative bacilli) causing a lower respiratory tract infection, followed by viruses ( Herpes simplex, Herpes zoster). The immunosuppressive drugs used for the treatment may also predispose the patient to opportunistic infections caused by fungi, protozoa, bacteria ( Listeria monocytogenes), and virus (cytomegalovirus). The risk of infection increases over time and is usually the cause of death. 8 Batlle et al. 10 reported 30 episodes of pneumonia in CLL with etiological confirmation in 53% of the episodes of pneumonia in a series comprising 17 patients. The most common agent was Pneumococcus (five cases) followed by Pseudomonas aeruginosa (four cases), Pneumocystis jirovecii (two cases) and Aspergillus fumigatus (two cases). Of note, the pneumonia caused by opportunistic agents was associated with fludarabine and three out of four cases of P. aeruginosa pneumonia died (75%).
The clinical course of CLL is usually indolent and generally asymptomatic with deaths not related to the neoplasia. However, some patients may present a rapid course, dying in 2 or 3 years after the diagnosis due to treatment refractoriness and complications (infections, hemorrhage, and cachexia). CLL is considered an incurable disease with an overall median survival of 7 years. 1,2
The histology of the lymph nodes in CLL presents, in general, the complete replacement of the nodal architecture by a diffuse proliferation of small round lymphocytes (6-12 microns), which may enfold some residual germinal centers (mimicking a marginal zone lymphoma) extending to the capsule and adjacent adipose tissue. 2 In addition, there are larger cells with loose chromatin, called pro-lymphocytes (medium-sized cells with small eccentric nucleoli), and para-immunoblasts (the largest cells in the neoplasia with a central and large nucleoli). 2 These larger cells are found isolated or in a small grouping – the pseudofollicles – which are more frequent in lymph nodes than extranodal sites and have an increased proliferative index. The disease has an accelerated course and a poorer prognosis when a predominance of prolymphocytes and/or para-immunoblasts in peripheral blood is present. 2,11 Other less common histological patterns may be found; namely, the presence of Reed-Sternberg-like CD30-positive; Epstein-Barr virus-infected cells; plasma cell differentiation with IgM overproduction (5% of the cases); Richter's syndrome, which represents the transformation to diffuse large B cell lymphoma (5-20% of the cases); and Hodgkin's lymphoma (0.5-2% of the cases). 2 The bone marrow involvement may be nodular, interstitial, and/or diffuse. The diffuse pattern is characterized by the complete replacement of the bone marrow by the leukemic cells and is associated with advanced disease, as observed in our case. 2
In this case, the involvement of the kidney and the heart was mild. It is not an infrequent finding in CLL, especially in advanced disease, but it is usually asymptomatic in both. 12-15 Kidney infiltration is bilateral and involves both the cortex and the medulla. 15 Acute renal failure is extremely rare, being described in about 25 autopsy cases, 16-18 all having massive CLL kidney infiltration. 16 Heart involvement can be endocardial, myocardial, and/or epicardial. 19 However, heart symptomatic disease is rare, which is described as ischemic syndrome, and is secondary to leukemic infiltration of the wall of the coronary arteries, 20 severe endocardial fibroelastosis, 19 and constrictive pericarditis. 21
The classical immunophenotype of CLL is the positivity for CD23 (mainly in the pseudofollicles); a weak and irregular positivity for B cell markers (CD20, CD19 and CD79a); T-cell aberrant expression (CD5 and CD43); and the negativity for cyclin D1 and CD10. The proliferative index tends to be low (nearly 5%), but may be higher in the accelerated phase of the disease and is associated with treatment refractoriness. 2 Recently, LEF-1 was described as a specific marker for CLL and its expression can be accessed by immunohistochemistry, described as strong and diffuse in the neoplastic cells, especially in cases of Richter transformation. 22 The LEF-1 – a nuclear mediator of the WNT signaling pathway with an important role in lymphopoiesis and expressed in pro-B cells – is especially useful in cases without a typical immunophenotype, but is not always necessary for the diagnosis. In this case, LEF-1 was not performed because of its unavailability, but it was considered an unnecessary marker for the CLL diagnosis, because the immunophenotype and the morphology were typical of CLL. In addition, LEF-1-negativity does not exclude the diagnosis because its sensitivity and specificity is not 100% (it is approximately 70% and 90%, respectively). 23 The immunohistochemical profile should always be analyzed in conjunction with the morphological aspects for the final diagnosis and should differentiate between other small-cell lymphoid tumors. 2
At the molecular level, some karyotype abnormalities are observed in about 80% of the cases, which include: the gain of chromosome 12 and the loss of the long arms of chromosomes 13 (the most common abnormality), 11, 17, and 6. Some biological markers associated with worse prognosis in the CLL are the expression of ZAP-70 and CD38, and the deletion of the short arm of chromosome 17. 1,2,24,25 The case reported herein had the trisomy of chromosome 12, but other genetic abnormalities were not tested. ZAP-70 was expressed in the neoplastic cells and can explain, in part, the poor prognosis. However, the advanced stage at the time of diagnosis and the difficulties in providing some medications to the patient also can be considered as contributors to the unfavorable outcome.
Some CLL autopsy series are reported in the medical literature. Of note, Möller et al. 26 described the case of an old man whose CLL transformed to Richter's syndrome and a large retroperitoneal and intra-abdominal mass, similar to that observed in our patient. However, we did not find the transformation to large-cell lymphoma. The large lymph nodes, forming large masses, were probably due to advanced and refractory disease.
ACKNOWLEDGEMENTS
The authors are thankful for Mrs. Deryn Pompeia and Mr. Reginaldo Silva do Nascimento for their technical assistance in the preparation of this manuscript.
REFERENCES
Hallek M, Cheson BD, Catovsky D, et al. Guidelines for the diagnosis and treatment of chronic lymphocytic leukemia: a report from the International Workshop on Chronic Lymphocytic Leukemia updating the National Cancer Institute-Working Group 1996 guidelines. Blood. 2008;111(12):5446-56. http://dx.doi.org/10.1182/blood-2007-06-093906. PMid:18216293.
Swerdlow SH, Campo E, Harris NL, et al, editors. World Health Organization classification of tumours of haematopoietic and lymphoid tissues. 4th ed. Lyon: IARC Press; 2008.
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA Cancer J Clin. 2015;65(1):5-29. http://dx.doi.org/10.3322/caac.21254. PMid:25559415.
Smith A, Howell D, Patmore R, Jack A, Roman E. Incidence of haematological malignancy by sub-type: a report from the Haematological Malignancy Research Network. Br J Cancer. 2011;105(11):1684-92. http://dx.doi.org/10.1038/bjc.2011.450. PMid:22045184.
Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA Cancer J Clin. 2014;64(1):9-29. http://dx.doi.org/10.3322/caac.21208. PMid:24399786.
Rai KR, Sawitsky A, Cronkite EP, Chanana AD, Levy RN, Pasternack BS. Clinical staging of chronic lymphocytic leukemia. Blood. 1975;46(2):219-34. PMid:1139039.
Binet JL, Auquier A, Dighiero G, et al. A new prognostic classification of chronic lymphocytic leukemia derived from a multivariate survival analysis. Cancer. 1981;48(1):198-206. http://dx.doi.org/10.1002/1097-0142(19810701)48:1<198::AID-CNCR2820480131>3.0.CO;2-V. PMid:7237385.
Dearden C. Disease-specific complications of chronic lymphocytic leukemia. Hematology. 2008;2008(1):450-6. http://dx.doi.org/10.1182/asheducation-2008.1.450 PMid:19074125.
DiLillo DJ, Weinberg JB, Yoshizaki A, et al. Chronic lymphocytic leukemia and regulatory B cells share IL-10 competence and immunosuppressive function. Leukemia. 2013;27(1):170-82. http://dx.doi.org/10.1038/leu.2012.165. PMid:22713648.
Batlle M, Ribera JM, Oriol A, et al. Pneumonia in patients with chronic lymphocytic leukemia: study of 30 episodes. Med Clin. 2001;116(19):738-40. http://dx.doi.org/10.1016/S0025-7753(01)71970-7. PMid:11412696.
Nowakowski GS, Hoyer JD, Shanafelt TD, et al. Using smudge cells on routine blood smears to predict clinical outcome in chronic lymphocytic leukemia: a universally available prognostic test. Mayo Clin Proc. 2007;82(4):449-53. http://dx.doi.org/10.4065/82.4.449. PMid:17418074.
Viadana E, Bross ID, Pickren JW. An autopsy study of the metastatic patterns of human leukemias. Oncology. 1978;35(2):87-96. http://dx.doi.org/10.1159/000225262. PMid:274679.
O’Mahony D, Piekarz RL, Bandettini WP, Arai AE, Wilson WH, Bates SE. Cardiac involvement with lymphoma: a review of the literature. Clin Lymphoma Myeloma. 2008;8(4):249-52. http://dx.doi.org/10.3816/CLM.2008.n.034. PMid:18765314.
Schwartz JB, Shamsuddin AM. The effects of leukemic infiltrates in various organs in chronic lymphocytic leukemia. Hum Pathol. 1981;12(5):432-40. http://dx.doi.org/10.1016/S0046-8177(81)80023-8. PMid:7250955.
Barcos M, Lane W, Gomez GA, et al. An autopsy study of 1206 acute and chronic leukemias (1958 to 1982). Cancer. 1987;60(4):827-37. http://dx.doi.org/10.1002/1097-0142(19870815)60:4<827::AID-CNCR2820600419>3.0.CO;2-A. PMid:3474054.
Aymard B, Beghoura R, Molina TJ. Renal infiltrate by a plasmocytoïd chronic B lymphocytic leukaemia and renal failure: a rare occurrence in nephropathology: a case report and review of the literature. Nephrol Ther. 2011;7(6):479-87. http://dx.doi.org/10.1016/j.nephro.2011.02.001. PMid:21439927.
Erten N, Saka B, Caliskan YK, Besisik S, Karan MA, Tascioglu C. Acute renal failure due to leukaemic infiltration in chronic lymphocytic leukaemia: case report. Int J Clin Pract Suppl. 2005;59(147):53-5. http://dx.doi.org/10.1111/j.1368-504X.2005.00049.x. PMid:15875623.
Xiao JC, Walz-Mattmüller R, Ruck P, Horny HP, Kaiserling E. Renal involvement in myeloproliferative and lymphoproliferative disorders. A study of autopsycases. Gen Diagn Pathol. 1997;142(3-4):147-53. PMid:9065578.
Applefeld MM, Milner SD, Vigorito RD, Shamsuddin AM. Congestive heart failure and endocardial fibroelastosis caused by chronic lymphocytic leukemia. Cancer. 1980;46(6):1479-84. http://dx.doi.org/10.1002/1097-0142(19800915)46:6<1479::AID-CNCR2820460631>3.0.CO;2-R. PMid:7417948.
Assiri AH, Lamba M, Veinot JP. Chronic lymphocytic leukemia involving the coronary arteries with accompanying acute myocardial infarction. Cardiovasc Pathol. 2005;14(6):324-6. http://dx.doi.org/10.1016/j.carpath.2005.07.006. PMid:16286042.
Habboush HW, Dhundee J, Al Okati D, Davies AG. Constrictive pericarditis in B cell chronic lymphatic leukaemia. Clin Lab Haematol. 1996;18(2):117-9. http://dx.doi.org/10.1046/j.1365-2257.1996.00164.x. PMid:8866146.
Tandon B, Peterson L, Gao J, et al. Nuclear overexpression of lymphoid-enhancer-binding factor 1 identifies chronic lymphocytic leukemia/small lymphocytic lymphoma in small B-cell lymphomas. Mod Pathol. 2011;24(11):1433-43. http://dx.doi.org/10.1038/modpathol.2011.103. PMid:21685909.
Menter T, Dirnhofer S, Tzankov A. LEF1: a highly specific marker for the diagnosis of chronic lymphocytic B cell leukemia / small lymphocytic B cell lymphoma. J Clin Pathol. 2015;68(6):473-8. http://dx.doi.org/10.1136/jclinpath-2015-202862. PMid:25713417.
Schlette EJ, Schlette EJ, Admirand J, et al. p53 expression by immunohistochemistry is an importantdeterminant of survival in patients with chronic lymphocytic leukemiareceiving frontline chemo-immunotherapy. Leuk Lymphoma. 2009;50(10):1597-605. http://dx.doi.org/10.1080/10428190903165241. PMid:19863337.
Admirand JH, Knoblock RJ, Coombes KR, et al. Immunohistochemical detection of ZAP70 in chronic lymphocytic leukemia predicts immunoglobulin heavy chain gene mutation status and time to progression. Mod Pathol. 2010;23(11):1518-23. http://dx.doi.org/10.1038/modpathol.2010.131. PMid:20657554.
Möller P, Feichter GE, Fritze D, Haag D, Schüle B. J-chain-producing immunoblastic lymphoma in a case of Richter’s syndrome. Immunohistochemical evidence for a gradual malignant transformation of a single B-cell clone and flow cytophotometric data. Virchows Arch A Pathol Anat Histol. 1982;396(2):213-24. PMid:6812283.
Notes
Author notes
Correspondence Amaro Nunes Duarte Neto Departamento de Patologia - Faculdade de Medicina - Universidade de São Paulo (USP) Avenida Dr. Arnaldo, 455 – Cerqueira Cesar – São Paulo/SP – Brazil CEP: 01246-903 Phone: +55 (11) 98546-4225 E-mail: amaro.ndneto@hc.fm.usp.br
Conflict of interest declaration