Article / Autopsy Case Report
Acute kidney injury and cardiac arrhythmia as the presenting features of widespread diffuse large B-cell lymphoma
Acute kidney injury and cardiac arrhythmia as the presenting features of widespread diffuse large B-cell lymphoma
Autopsy and Case Reports, vol. 9, no. 3, e2019114, 2019
São Paulo, SP: Universidade de São Paulo, Hospital Universitário
Received: 20 May 2019
Accepted: 02 August 2019
DOI: 10.4322/acr.2019.114
Abstract: Diffuse Large B-cell Lymphoma, the most common adult non-Hodgkin lymphoma, is a proliferative neoplasm of enlarged B cells. Patients may be asymptomatic on presentation, but if present, symptoms often correlate with direct organ dysfunction resulting from the site of involvement. While the gastrointestinal system is the most common site of extranodal involvement, virtually any part of the body can be infiltrated by malignant lymphocytes. Here, we present an unusual case of cardiac and bilateral renal involvement by Diffuse Large B-cell Lymphoma in a 78-year-old male with a relatively unremarkable medical history. This combination of organ involvement and the resulting clinical symptoms are uncommonly described in the literature. The patient was treated for his symptoms prior to death, but the underlying cause that explained his presentation was not identified until performance of an autopsy. As such, this case demonstrates the utility of the medical autopsy, a gold standard in diagnostic medicine that can provide a variety of benefits in today’s healthcare system.
Keywords: Lymphoma, Acute Kidney Injury, Arrhythmias, Cardiac, Autopsy.
CASE REPORT
A 78-year-old male with a history of hypertension and hypothyroidism presented to an outside hospital with edema, anorexia, and abdominal pain. Computed tomography (CT) imaging demonstrated focal hypoperfusion of the medial right kidney with bilateral perinephric stranding and suspected phlegmon/abscess formation, which was interpreted as being consistent with bilateral pyelonephritis. Additional imaging findings included pleural effusions, colonic wall thickening adjacent to the suspected perinephric abscess, thought to be a reactive process, and a small pelvic fluid collection. Antibiotics were initiated to treat the working diagnosis of pyelonephritis, and the patient was transferred to our tertiary care facility for surgical consideration in the setting of worsening respiratory function.
On arrival, the patient endorsed worsening edema and pain. Bloodwork was sent to the laboratory for testing, and notable values included the following: white blood cell count 20.1 K/mm 3 (reference range [RR]; 4.8-10.8 K/mm 3), hemoglobin 7.4 g/dL (RR; 14-18 g/dL), platelet count 37 K/mm 3 (RR; 140-440 K/mm 3), lactic acid 15 mmol/L (RR; 0.4-2.3 mmol/L), urea nitrogen 67 mg/dL (RR; 7-20 mg/dL), and creatinine 2.5 mg/dL (RR; 0.7-1.3 mg/dL). He was found to be nonreactive for HIV-1 and HIV-2 antibodies. These lab values, which were indicative of acute kidney injury, further supported the clinical impression of pyelonephritis. The renal status continued to worsen both clinically and based on laboratory values, and the patient ultimately became anuric and required continuous renal replacement therapy (CRRT). Concurrently, increasing difficulty of breathing necessitated intubation and mechanical ventilation. Surgical intervention was declined due to the patient’s poor condition and unclear benefit, and the clinical teams continued to administer broad-spectrum antibiotics and supportive care. Over the next several days, his condition deteriorated with refractory shock thought to be secondary to sepsis, nonfunctioning kidneys, and frequent ectopic arrhythmias requiring cardioversion. Care was withdrawn due to severe multisystem organ failure, and the patient was pronounced dead six days after initial presentation to the outside hospital. An autopsy was requested by clinicians and approved by the patient’s next-of-kin due to his rapid decline and several unexplained clinical features at death, such as frequent ectopic beats and severe, refractory shock. An autopsy limited to the chest and abdomen was performed two days following death.
AUTOPSY FINDINGS
External examination demonstrated a well-nourished adult male with marked anasarca and bullae formation on the hip and knee. Internally, significant serosanguineous fluid collections in the pericardial (150 mL), right (950 mL) and left (800 mL) pleural, and peritoneal (800 mL) cavities were documented. The heart was mildly enlarged (460 grams, RR; 233-383 g) 1 with a normally developed coronary artery system that was affected by mild to moderate calcific atherosclerosis. On cut surface, the myocardium was found to be heterogeneous, with prominent tan-yellow mottling noted at the junction of the posterior left ventricle and the interventricular septum ( Figure 1). The lungs were diffusely edematous and weighed 1570 grams, combined. No mediastinal mass was appreciated, and thoracic lymph nodes were grossly unremarkable.
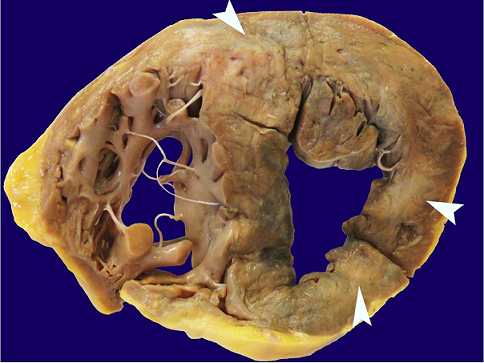
Figure 1
Gross image of the heart, obtained at autopsy, demonstrates tan-yellow mottling of the myocardium of the left ventricle, interventricular septum, and, to a lesser extent, the right ventricle (arrowheads).
The right and left kidneys weighed 200 and 180 grams (RR; 79-223 for right kidney and 74-235 for left kidney), 2 respectively, with adherent, opaque capsules. The renal parenchyma was grossly orange in color ( Figure 2); the radiologically suspected abscess was not present.
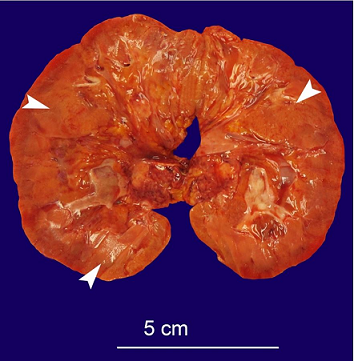
Figure 2
Gross view of the kidney, obtained at autopsy (bisected), demonstrates diffuse yellow-orange discoloration of the renal parenchyma. This discoloration was apparent in both kidneys. Representative areas of discernible discoloration are marked with stars.
A 1.5 cm mucosal plaque was present in the ileum; on sectioning, it was found to be tan-white and fibrotic. The remainder of the intestines was unremarkable, as were the liver (1870 grams, RR; 838-2584), 2 pancreas, and adrenal glands. The spleen was enlarged (480 grams, RR; 43-344) 2 and appeared congested, but no grossly identifiable infiltrate was appreciated. Abdominal lymph nodes did not appear enlarged. Sections of the abnormal findings described above, as well as representative sections from the remaining organs, were submitted for histologic review.
Microscopic examination of the heart, kidneys, perinephric fat and lungs, demonstrated an infiltrate of large, pleomorphic lymphocytes with scattered associated apoptotic debris ( Figures 3- 5). The liver, adrenal glands and periadrenal fat, and a small bowel plaque were also involved by this same infiltrate ( Figure 6).
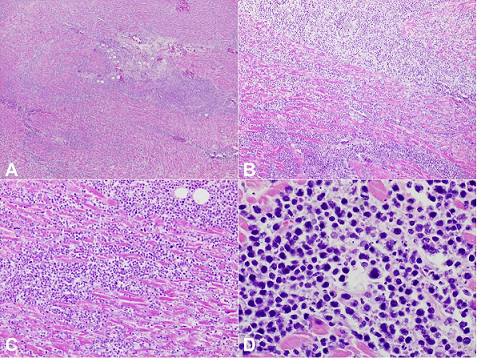
Figure 3
Photomicrographs of the heart with diffuse infiltrate of enlarged, atypical cells. These malignant cells were later determined to be of B-cell lineage (see Figure 7). On higher power, high-grade cellular morphology is discernible, including nuclear pleomorphism, large, irregularly shaped nuclei, and high nuclear to cytoplasmic ratio. Scattered apoptotic debris can be seen in the background (H&E, A – 40X; B – 100X; C – 200X; D – 600X).
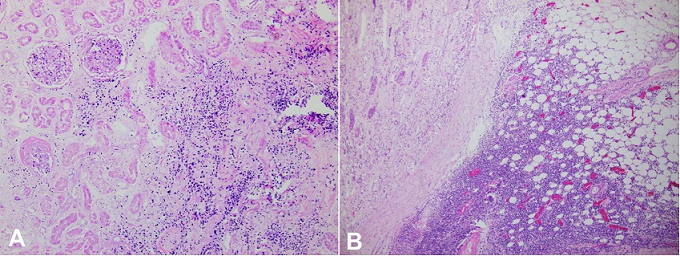
Figure 4
Photomicrographs of the kidney demonstrate in A – renal involvement by a diffuse infiltrate of enlarged, atypical cells (H&E, 100X); B – infiltration into perinephric fat, which may have contributed to the findings that were seen on radiology and interpreted as pyelonephritis with perinephric abscess formation (H&E, 40X).
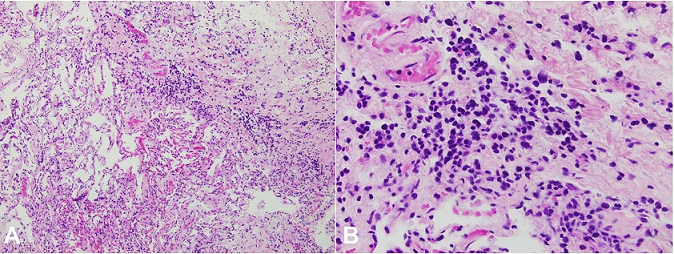
Figure 5
Photomicrograph of the lung showing a diffuse infiltrate of enlarged, atypical cells (H&E, A – 100X; B – 400X).
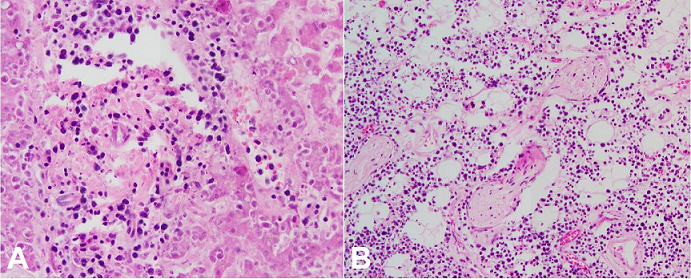
Figure 6
Photomicrographs of the liver ( A – H&E, 200X) and periadrenal fat ( B – H&E, 100X) showing a diffuse infiltrate of enlarged, atypical cells.
While areas suggestive of a cellular infiltrate were identified in the spleen, particularly in a perivascular location, the organ was markedly autolyzed, precluding definitive histomorphologic assessment. In the affected tissues, the infiltrating lymphocytes disrupted and focally effaced the normal architecture. There were no germinal centers or follicles appreciated, and overall, the cells were dyscohesive. Of note, variable autolysis affected all sampled tissues. The bone marrow was found to be hypercellular for age, although no definitive areas of cellular infiltrate were identified.
Discernible morphologic features of the infiltrates in all affected organs included the previously mentioned large size, round to oval nuclei with irregular contours, and high nuclear to cytoplasmic ratio. Nucleoli were present in a majority of the neoplastic cells. The findings on hematoxylin- and eosin-stained sections were suggestive of a diffusely infiltrative neoplastic process with intermediate to high grade nuclei, and immunohistochemical stains were performed to further characterize the lymphoid infiltrate. Negative staining for CD3 (T-cell marker) and positive staining for CD20 (B-cell marker) confirmed B-cell lineage ( Figure 7A and 7B). Kappa and Lambda were both negative in the neoplastic cells. A Ki-67 stain was weakly positive in over 50% of the cells of interest ( Figure 7C).
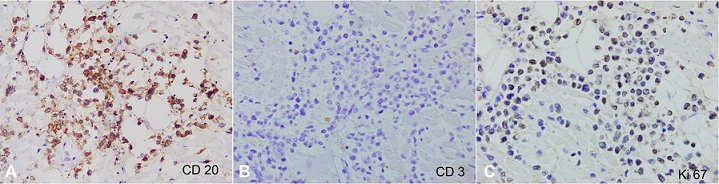
Figure 7
Photomicrographs of the heart, immunohistochemical profile. These findings confirm B-cell lineage of the cells of interest via positive CD20 ( A – 400X), negative CD3 ( B – 400X), and the high (albeit weak) positivity with Ki-67 ( C – 400X).
No further stains were pursued, in large part due to the poor yield commonly encountered with stains performed on autopsy specimens. Based on the clinical picture, the gross and microscopic findings at autopsy, and the immunohistochemical staining profile, the cause of death was documented as Diffuse Large B-cell Lymphoma (DLBCL) with multi-organ involvement; the manner of death was natural.
DISCUSSION
Diffuse Large B-cell Lymphoma is the most common adult non-Hodgkin lymphoma, representing approximately 30% of cases in developed countries. The disease is a proliferative neoplasm of enlarged B cells with no established etiology. DLBCL commonly affects lymph nodes, but disseminated disease is frequent; as many as 40% of cases may initially be exclusively extranodal in nature. Though the gastrointestinal tract represents the most common site of extranodal disease, practically any part of the body can be involved. Many patients are asymptomatic with a growing mass or abnormal lab values on presentation, but symptoms, when present, often correlate directly with these sites of extranodal involvement. Generalized B-cell symptoms, such as fever, lethargy, and weight loss, may also be seen. 3
The prognosis of this disease is largely dependent upon the age and stage at diagnosis. An enhanced National Comprehensive Cancer Network-International Prognostic Indicator (NCCN-IPI) prognostic tool stratifies patients based upon these and other factors, yielding a 5-year overall survival ranging from 38% (high risk) to 96% (low risk). 4 This patient unfortunately met all criteria for the high-risk group, including age >75 years, Lactate Dehydrogenase >3x Upper limit of Normal, stage III/IV disease at diagnosis, presence of extranodal disease, and performance status >2 (according to the ECOG scale). 5 Accordingly, his clinical picture was fulminant.
Characteristic histologic features include a diffusely infiltrative growth pattern of enlarged lymphocytes that express pan-B-cell markers by flow cytometry or immunohistochemistry, including CD19, CD20, CD22, CD45, and kappa or lambda light chain immunoglobulin. A cellular proliferation index marker, such as Ki-67, is usually high. With time, the B-cell infiltrate can disrupt and entirely replace the normal architecture of involved organs. 6 Despite the fact that autopsy series have demonstrated direct lymphoma involvement in unusual locations such as the heart and kidneys, patient findings at presentation may be nonspecific, making it unlikely that clinical teams would immediately suspect a process such as widespread lymphoma. The findings of this case – heart and bilateral kidney involvement with clinically significant symptoms – are still only rarely described in the literature. In 2014, Hafeez et al. described the case of an 18-year-old male who presented with a similar cardiac and renal distribution of DLBCL with associated severe left ventricular failure. This group noted that this particular presentation had not been previously described based on their literature review. 7 Other publications have reported symptomatic cardiac involvement and attempts made for treatment purposes, but the findings are still poorly studied overall. 8,9,10 Encouragingly, some of these studies outline successful treatment with varied chemotherapy regimens.
This case contributes to the body of literature by describing DLBCL with cardiac and renal involvement, with a constellation of symptoms that can be attributed to the malignant lymphoid infiltrate. This patient initially presented with gastrointestinal complaints and quickly developed widespread end-organ failure. The clinical workup largely focused on an erroneous diagnosis of pyelonephritis with abscess formation and apparent sepsis; however, it is probable that the initial radiologic findings were due to a malignant, not reactive, lymphoid infiltrate. In other words, the bilateral perinephric stranding and other features which were interpreted as early abscess formation more likely represented fibroadipose tissue infiltration of malignant B-lymphocytes. In this case, chemotherapy would have been preferred over antibiotics. Similarly, his frequent ventricular ectopy remained unexplained prior to autopsy, but knowledge of an underlying infiltrative process and treatment targeting the malignancy may have improved this finding as well. Enhanced clinical awareness of such cases, achieved in part by published case reports, could help refine the treatment protocols seen in the literature. 8, 11
The presented case also demonstrates the importance of pursuing the medical autopsy, even in today’s healthcare system. For decades, the rate of non-forensic autopsies has been declining precipitously. Studies indicate that hospital deaths in the United States were sent for autopsy approximately 40-60% of the time prior to 1970, while the current rate is below 5%. 12 This trend is not unique to the United States; according to a 2013 study published in the Journal of Clinical Pathology, less than 1% of all hospital deaths in the United Kingdom were followed by an autopsy. 13 A review of medical autopsies performed over a six-year period at our academic institution demonstrates a marginally higher rate of 7.4 to 9.5%, but this is still far below historic values.
The decline is multifactorial in nature, but autopsy pathology remains a diagnostic gold standard that can provide many benefits to the medical system. As in this case, autopsy findings can yield an unexpected cause of death that provides closure for loved ones as well as valuable instruction for resident and attending physicians. In a study of almost 2,500 autopsies performed at 248 institutions, Zarbo et al. 14 found that 93% of the overall clinical questions were able to be answered by autopsy performance. Moreover, a high percentage of autopsies revealed unexpected findings; in fact, as many as 40% of the reviewed autopsies yielded a major, unexpected finding that contributed to the patient’s death. 14 Again, review of autopsies performed at our institution over a recent six-year period show the discovery of major, unexpected findings between 19 and 50% of the time. This unpublished data indicate that autopsies still routinely provide valuable teaching points despite the more advanced imaging and other diagnostic techniques available today. Our findings in this case documented widespread organ involvement by DLBCL, which accounted for his varied clinical symptoms including refractory cardiac ectopy and perinephric changes mimicking pyelonephritis with abscess formation radiologically. Although the rapid progression in the present case would have likely stymied antemortem treatment efforts, more timely consideration of a hematologic malignancy could have allowed an attempt at chemotherapy with palliative or curative intent. By studying the findings of this (and other) autopsy report(s), clinicians can be better prepared to recognize unusual, but significant, manifestations of a disease process such as DLBCL in the future.
CONCLUSION
This case demonstrates the importance of keeping a broad differential, to include malignancy, despite a lack of classic symptoms, particularly when multi-organ involvement by a single neoplasm can unite an otherwise disparate clinical picture. Additionally, autopsy, a gold standard in diagnostic medicine, is a valuable tool that is often able to contribute to quality of assurance protocols, improve patient care, provide ongoing medical education, and assist in providing closure to families.
The authors are grateful for the willingness of the patient and family to contribute to the body of literature for this disease.
REFERENCES
Molina K, DiMaio V. Normal organ weights in men: part 1 – the heart. Am J Forensic Med Pathol. 2012;33(4):362-7. http://dx.doi.org/10.1097/PAF.0b013e31823d298b. PMid:22182983.
Molina K, DiMaio V. Normal organ weights in men: part 2 – the brain, lungs, liver, spleen, and kidneys. Am J Forensic Med Pathol. 2012;33(4):368-72. http://dx.doi.org/10.1097/PAF.0b013e31823d29ad. PMid:22182984.
Swerdlow SH, Campo E, Harris NL, et al. WHO classification of tumours of haematopoietic and lymphoid tissues (4th ed). Lyon: IARC; 2017.
Zhou Z, Sehn L, Rademaker A, et al. An enhanced international prognostic index (NCCN-IPI) for patients with diffuse large B-cell lymphoma treated in the rituximab era. Blood. 2014;123(6):837-42. http://dx.doi.org/10.1182/blood-2013-09-524108. PMid:24264230.
QxMD Medical Incorporated. Calculate by QxMD. Diffuse large B-cell lymphoma prognosis (NCCN-IPI). 2018 [cited 2019 Feb 9]. Available from: https://qxmd.com/calculate/calculator_311/diffuse-large-b-cell-lymphoma-prognosis-nccn-ipi
Medeiros L, Vega F, Miranda R. Diffuse large B-cell lymphoma, not otherwise specified. USA: Elsevier; 2018 [cited 2019 Feb 9]. Available from: https://app.expertpath.com/document/diffuse-large-b-cell-lymphoma-not--/8fd8916c-f615-4004-8aae-d969294ec1ba?searchTerm=Diffuse%20Large%20B-Cell%20Lymphoma
Hafeez I, Alai MS, Iqbal K, et al. Lymphoma presenting as severe left ventricular systolic dysfunction: a case report. Oman Med J. 2014;29(3):223-5. http://dx.doi.org/10.5001/omj.2014.55. PMid:24936276.
Amirimoghaddam Z, Khoddami M, Nayeri ND, Molaee S. Hodgkin’s lymphoma presenting with heart failure: a case report. J Med Case Rep. 2010;4(1):14. http://dx.doi.org/10.1186/1752-1947-4-14. PMid:20205760.
Al-Mehisen R, Al-Mohaissen M, Yousef H. Cardiac involvement in disseminated diffuse large B-cell lymphoma, successful management with chemotherapy dose reduction guided by cardiac imaging: a case report and review of literature. World J Clin Cases. 2019;7(2):191-202. http://dx.doi.org/10.12998/wjcc.v7.i2.191. PMid:30705895.
Ban-Hoefen M, Zeglin MA, Bisognano JD. Diffuse large B cell lymphoma presenting as a cardiac mass and odynophagia. Cardiol J. 2008;15(5):471-4. PMid:18810725.
Jeudy J, Burke A, Frazier A. Cardiac lymphoma. Radiol Clin North Am. 2016;54(4):689-710. http://dx.doi.org/10.1016/j.rcl.2016.03.006. PMid:27265603.
Hamza A. Declining rate of autopsies: implications for anatomic pathology residents. Autops Case Rep. 2017;7(4):1-2. http://dx.doi.org/10.4322/acr.2017.036. PMid:29259925.
Turnbull A, Osborn M, Nicholas N. Hospital autopsy: endangered or extinct? J Clin Pathol. 2015;68(8):601-4. http://dx.doi.org/10.1136/jclinpath-2014-202700. PMid:26076965.
Zarbo RJ, Baker PB, Howanitz PJ. The autopsy as a performance measurement tool: diagnostic discrepancies and unresolved clinical questions. Arch Pathol Lab Med. 1999;123(3):191-8. PMid:10086506.
Notes
Author notes
Correspondence Anthony Jacob Emanuel 165 Ashley Ave, MSC 908, Charleston, South Carolina, USA Zip code: 29425 Phone: +1 (843) 792-1086/Fax: +1 (843) 792-8974 emanuela@musc.edu
Conflict of interest declaration
