Article / Clinical Case Report
Fibrous Dysplasia treated with virtual planning – report of two cases
Fibrous Dysplasia treated with virtual planning – report of two cases
Autopsy and Case Reports, vol. 9, no. 3, e2018086, 2019
São Paulo, SP: Universidade de São Paulo, Hospital Universitário
Received: 06 December 2018
Accepted: 28 March 2019
DOI: 10.4322/acr.2018.086
Abstract: Fibrous dysplasia (FD) is part of a rare group of bone dysplasia. It exhibits benign behavior and can lead to osteolytic lesions, deformities, and fractures. The treatment is challenging, and accurate removal of the lesion is necessary to restore function and esthetics. Here we present two cases of FD where virtual planning with presurgical computed tomography (CT) was used for the production of a surgical guide for bone contouring. First, CT image reconstruction was performed to mirror the patient’s original anatomy. Then, three surgical guides that determined the area and depth of bone wear were prepared and used in the relevant sequence during the actual surgeries, which were successfully performed in both patients. This technique is termed the template guide holes (TGH) technique. The findings from this report suggest that presurgical virtual planning and guide preparation allows direct and objective measurement of the level of bone wear and improves the functional and esthetic outcomes of surgery for FD. In particular, the TGH technique is safe and allows adequate preoperative surgical simulation, reduces the surgical duration, and increases the predictability of the final result.
Keywords: Surgery, oral, Surgery, plastic, Pathology, oral, Reconstructive Surgical Procedures, Software.
INTRODUCTION
Fibrous dysplasia (FD) is part of a rare group of bone dysplasia, exhibiting benign behavior and characterized by focal proliferation of fibrous tissue in the bone marrow, which leads to osteolytic lesions, deformities, and fractures. 1 It is a nonhereditary congenital bone disorder that occurs with similar frequencies in both sexes. 2
Although early symptoms generally appear during childhood or adolescence, the condition may remain asymptomatic and could be incidentally diagnosed by radiography during adulthood.
Clinically, FD commonly presents with painless and slowly progressive expansion of the involved skeletal structure. Generally, lesions develop in childhood and grow until the end of the bone growth period. 3 In some cases, spontaneous regression occurs after stabilization of bone growth. 4 Although it is a benign condition, rare cases of malignancy have been reported (0.5%-4%). 5 Blindness and deafness are two severe consequences of the associated bone expansion and nerve and artery compression. 4,6
The radiographic features of FD are varied and depend on the degree of ossification (proportion of bone and fibrous tissue in each lesion) in the lesions, although the ground glass appearance is the most characteristic pattern observed on radiographs. 7,8
FD can involve any bone and represents 2% of all bone tumors and 7.5% of benign bone tumors. 9 There are two clinical types, namely monostotic FD (MFD; 70%) and polyostotic FD (PFD; 30%), 10 and there is no report of transformation from one form to another. 11 MFD is classically defined by the unilateral involvement of a single bone and/or the adjacent structures such as the craniofacial skeleton. On the other hand, multiple foci involving different bones characterize PFD, although it typically affects only one side of the body. 11
Less than 5% patients present with endocrine dysfunction associated with the bone lesions. 7 This association is characteristic of McCune–Albright syndrome, which is generally unilateral and classically defined by the clinical triad of PFD, precocious puberty, and cutaneous pigmentation known as café-au-lait spots. 7,11
The treatment of FD is challenging, with options including medications and/or surgery. Bisphosphonates are used as modulators of the inflammatory response because of their antiresorptive action and ability to control osteoclastic activity. 7 Surgery is indicated for the management of esthetic and/or functional deformities, treatment or prevention of fractures, and release of optic and auditory channels in order to overcome neurological lesions. 7 There is no consensus for the best surgical approach, although it is routine practice to wait until the completion of bone growth, which is confirmed by bone scintigraphy. 5
The challenge of surgical planning and treatment for the correction of deformities resulting from FD is the achievement of appropriate postoperative facial symmetry and contour. Accordingly, virtual planning based on three-dimensional (3D) reconstruction of computed tomography (CT) images is used to aid the surgical procedure. 12 Here we describe the successful use of presurgical virtual planning and 3D printed models by way of a novel technique called the template guide holes (TGH) for the surgical treatment of FD in two patients.
CASE REPORTS
The two patients reported here presented to our Maxillofacial Surgery Service with chief complaints of progressive hemifacial bulging that caused asymmetry and functional alterations. In both cases, the patient and a family member provided written informed consent to undergo the procedures and for publication of this report.
Patient 1
Patient 1 was a 23-year-old man who was diagnosed with MFD at the age of 5 years, when he developed right-sided facial asymmetry ( Figure 1). Endocrinopathies and associated syndromes had been ruled out. At that time, he was advised to wait until growth completion for the initiation of treatment. In addition to facial and cranial asymmetry, he presented with 95% vision loss and exophthalmia.
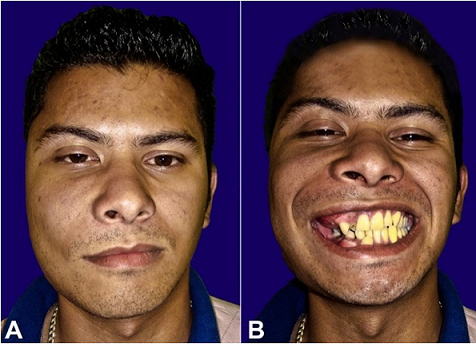
Figure 1
Gross examination of the patient before the surgical treatment. A – normal position with the mouth closed and B – smiling with the mouth opened.
Patient 2
Patient 2 was a 7-year-old girl referred to our department for further evaluation of a hard tumor involving the right side of the face ( Figure 2). She was placed on an observation protocol after a thorough diagnostic workup ruled out any endocrinopathies or syndromes.

Figure 2
Gross examination of the patient before the surgical treatment (A), and 2 years after the surgical treatment (B).
Neither patient exhibited trauma and/or associated infections, and both had normal laboratory test findings. The facial CT images of patient 1 showed an expansive lesion with the ground glass appearance that involved the skull base and the right maxillary, zygomatic, sphenoidal, temporal, frontal, and mandibular bones ( Figure 3).
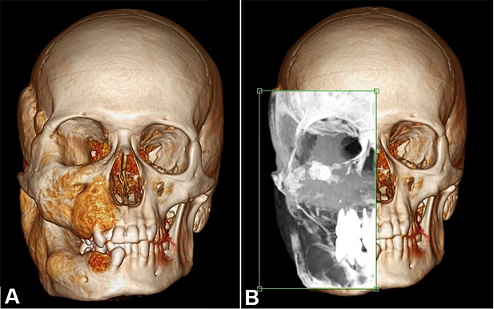
Figure 3
A – 3D reformation of the facial and cranium computed tomography of patient number 1. B – Analysis of the ground glass appearance in the involved area.
Involvement of the orbital bone resulted in a decrease in the socket volume and optic canal diameter, which explained the exophthalmia and vision loss. Bone scintigraphy showed osteogenic activity in the right craniofacial region, without involvement of other skeletal structures. The clinical features and laboratory and radiographic findings confirmed the diagnosis of MFD. The patient underwent osteoplasty of the affected maxillary, zygomatic, mandibular, and temporal bones for the restoration of facial harmony. The surgery was preceded by virtual planning ( Figure 4).
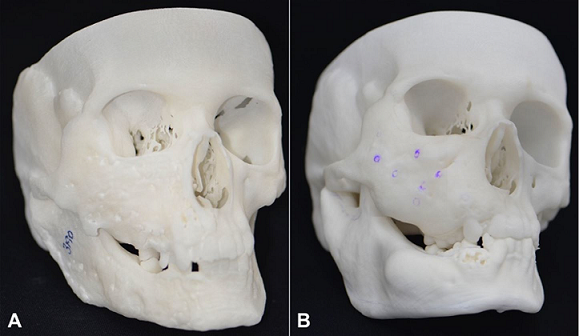
Figure 4
A – Lateral view of the stereolithography model, before surgery; B – Lateral view of the stereolithography model, after surgery.
In case of patient 2, because of the early development and slight facial deformity, a clinical follow-up protocol was selected. During the growth spurt between 12 and 13 years of age, she developed significant occlusal alterations, facial asymmetry, and difficulty in supporting her eyeglass stem because of sharp growth in the right mastoid apophysis. Laboratory test findings were within normal limits, and bone scintigraphy showed intense osteogenic activity on the right side of the skull. CT revealed volumetric osteofibrous thickening of the maxillary, mandibular, zygomatic, sphenoidal, temporal, mastoid, and parietal bones, with preservation of the internal acoustic meatus, optic canal, and stylomastoid foramen.
At the age of 15 years, the patient underwent osteoplasty of the right mastoid and zygomaticomaxillary bones for esthetic comfort and orthodontic treatment. Second surgery for the correction of facial deformities was subsequently performed with virtual planning, which allowed precise esthetic bone remodeling. Subsequently, she underwent orthodontic treatment for future complementary surgeries, including orthognathic surgery and surgeries for the correction of remaining deformities.
Both patients underwent periodic postoperative examinations, including CT evaluations. In the initial postoperative period, both used a face mask to accommodate the facial tissues and avoid blood clot formation. While clinical examinations ( Figure 5) showed asymmetry because of excess soft tissue in patient 2, CT showed stability and symmetry of the corrected bones over a 2-year follow-up period ( Figure 6).
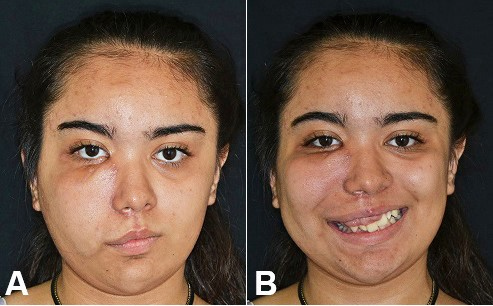
Figure 5
A and B – Gross examination of the patient 2 years after the surgical treatment.
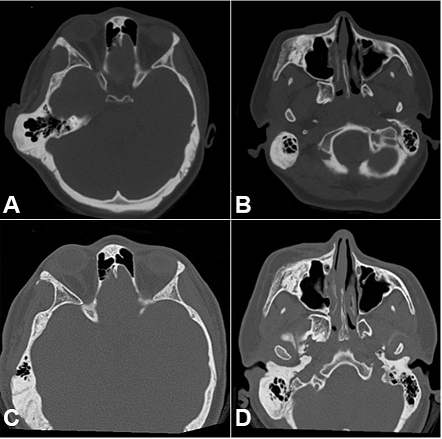
Figure 6
Facial CT. A – Axial plane showing the temporal region before the surgical treatment, B – Axial plane showing the zygomatic-maxillary region before the surgical treatment, C –Axial plane showing the temporal region 2 years after the surgical treatment, D – CT plane showing the zygomatic-maxillary region after the surgical treatment.
Two years after the second surgery, she presented signs and symptoms of new initial bone growth in the right infraorbital region on CT images. The remaining bones subjected to osteoplasty remained unchanged. At the time of writing this report, she was observing a strict follow-up regimen for subsequent osteoplasty procedures. Meanwhile, patient 1 showed stable outcomes for 5 years after surgery.
Virtual planning with the TGH technique
In both cases, presurgical virtual planning was performed with the aim of obtaining a better contour and facial symmetry with the preservation of important structures. The procedure is described as follow.
- 1.
1
Preoperative CT images with a 0.5-mm slice thickness were obtained for each patient (Brilliance CT 64-slice model) and sent to the workstation of the Windows-based navigation system.
- 2.
2
By data superimposition, 3D images were developed using the Digital Imaging and Communications in Medicine protocol and saved as stereolithography (STL) files.
- 3.
3
The median sagittal plane of these images was chosen as the reference plane for manipulation. The healthy side was mirrored on the affected side, with manual changing of positions.
- 4.
4
The mirrored image was superimposed on the pathological image using the Blender system, and the area of resection was generated through image subtraction and manually adjusted. The area of resection was marked in a different color.
- 5.
5
With consideration of the lesion volume planned for resection, perforations were marked on the virtual lesion.
- 6.
6
The Blender system itself creates depth guides, which connect points from the pathological area to the anatomically acceptable surface of the superimposition. The depth of each perforation was virtually adjusted until the maximum depth required for the removal of unwanted bone tissue was reached.
- 7.
7
After the modeling process in the Blender system, the files were sent to the Netfabb program, where they were corrected and manually adjusted for STL printing, with the stitch mesh closing. This model functioned as a transoperative template.
- 8.
8
For printing of the model guides, a reprap type Graber i3 model with ABS filaments was used. For its use in surgery, it requires autoclaving at 125°C or ethylene oxide sterilization, because its melting point is 270 °C. 13 This printer exhibits distortion of only 1mm. 13
- 9.
9
The STL files were printed using slic3r 3D software, which is freely distributed by Creative Commons and Repetier Host.
During surgery, the Weber Ferguson approach was used to correct the deformities in the middle third of the face in both cases. A total of three model guides were printed; the first one contained only the perforations, the second one contained the depth pins for bone wear and transoperative measurements, and the third one indicated the area to be resected. The latter served as a guide for the correction of finer details and confirmation of the surgical plan.
After exposure of the area of interest, the guide containing the perforations was placed over the bone surface and the perforations with the respective depths were created. This was followed by bone resection and wear until the inner end of the perforations was reached; the second template was used for this step. Subsequently, the last template was used for final bone wear. Extensive saline irrigation was used over the operated area ( Figure 7).
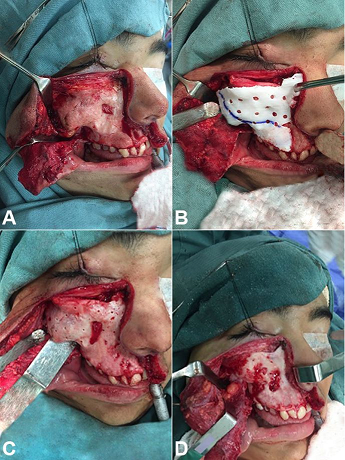
Figure 7
Intraoperative images of the case #2. A – Exhibition of the area of interest; B – Guide placement; C – After realization of the perforations in their respective depths; D – After wear and bone resection.
For patient 1, the mandibular deformities were corrected via a transoral–transcervical approach; the surgical procedures were similar to those described above. The patient did not want to undergo the parietal complementation because he was satisfied with the final esthetic result. In patient 2, the deformities in the mastoid region were corrected via a retroauricular approach. In both surgeries performed for patient 2, the middle third of the face was treated via the same approach.
DISCUSSION
In the present report, we demonstrated that the TGH technique is safe and allows adequate preoperative surgical simulation, reduces the surgical duration, and increases the predictability of the final result in patients with FD.
The management of FD is challenging, particularly when several bones are simultaneously involved. Surgical treatment aims at improvement of esthetics and prevention or correction of functional deformities and complications. 14 Currently used surgical modalities include complete resection and realignment. Complete resection is indicated for isolated lesions or small areas, although it is not well received by patients. In the cases presented in this report, complete resection was not feasible because of the involvement of multiple bones and important structures. 15 Accordingly, we opted for recontouring in order to remove excess fibro-osseous growths and achieve acceptable symmetry while preserving vital structures. 9,10,16 Our technique followed the surgical principles described by Nuss et al. in 1991. 16
At present, new techniques for performing less invasive surgeries are being studied. One example is the real-time image-guided technique, 14 which allows presurgical simulation and transoperative visualization. According to Gui H et al. 14 and Wang Y et al., 17 this technique reduces the surgical duration and risks by identifying the safety margins, preserving vital structures, and achieving reasonable symmetry, even in cases of bilateral dysplasia. However, it requires expensive technology and equipment as well as a steep learning curve in terms of handling specific software and instruments. It also requires time to master the necessary manual skills. All these factors hinder the universal use of this method in addition to making it inaccessible in several cases.
In 2007, Osada et al. 18 used the TGH technique and achieved precise correction of deformities in the right zygomaticomaxillary and naso-orbital regions in a patient with FD. This technique is a predictable and effective method for the management of craniofacial FD, although it does not provide instant feedback about the surgery, unlike the real-time image-guided technique. However, it offers superior reproducibility; moreover, the software used for planning and preparing the templates is cheaper, more accessible, and easy to operate. In addition, it is more direct and straightforward, thus allowing a decrease in the surgical duration. With regard to the precision of the technique, CT can be performed during or after the surgical contouring in order to ensure accuracy as well as safety. The only limitation of the technique is that better soft tissue esthetics may require corrective procedures after the hard tissue correction is complete. In our Patient 2, although the bones affected by FD appeared symmetrical with those on the healthy side ( Figure 6B, D) after surgery, asymmetry due to soft tissue discrepancies was still apparent. The patient will require more surgeries in the future, including dermatoplasty and orthognathic surgery, for improvement of the facial symmetry and function.
CONCLUSION
The findings from this report suggest that the TGH technique for corrective surgeries in patients with facial deformities caused by FD is relatively inexpensive and easy in terms of learning and manipulation; furthermore, it provides good esthetic and functional results.
ACKNOWLEDGEMENTS
We thank the technicians and the “ Centro de Tecnologia da Informação Renato Archer (CTI – Renato Archer)” for preparing us the initials stereolithographic skull models.
REFERENCES
Lichtenstein L. Polyostotic fibrous dysplasia. Arch Surg. 1938;36(5):874-98. http://dx.doi.org/10.1001/archsurg.1938.01190230153012.
Cohen MM Jr, Howell RE. Etilogy of fibrous dysplasia and McCune-Albrights syndrome. Int J Oral Maxillofac Surg. 1999;28(5):366-71. http://dx.doi.org/10.1016/S0901-5027(99)80085-X. PMid:10535539.
Waldron CA. Fibro-osseous lesions of the jaws. J Oral Maxillofac Surg. 1993;51(8):828-35. http://dx.doi.org/10.1016/S0278-2391(10)80097-7. PMid:8336219.
Jeyaraj CP, Srinivas CV. Craniofacial and monostotic variants of fibrous dysplasia affecting the maxillofacial region. J Oral Maxillofac Surg Med Pathol. 2014;26(3):424-31. http://dx.doi.org/10.1016/j.ajoms.2013.05.011.
Ruggieri P, Sim FH, Bond JR, Krishnan Unni K. Malignancies in fibrous dysplasia. Cancer. 1994;73(5):1411-24. http://dx.doi.org/10.1002/1097-0142(19940301)73:5<1411::AID-CNCR2820730516>3.0.CO;2-T. PMid:8111708.
Swain SK, Sahu MC. An extensive fibrous dysplasia of anterior skull base area of a 12-year-old boy – A case report. Pediatr Pol. 2016;91(6):636-9. http://dx.doi.org/10.1016/j.pepo.2016.08.002.
Chapurlat RD, Orcel P. Fibrous dysplasia of bone and McCune-Albright syndrome. Best Pract Res Clin Rheumatol 2008;22(1):55-69.
Chapurlat RD, Meunier PJ. Fibrous dysplasia of bone. Bailliere Clin Rheum 2000;14(2):385-98.
Pinsolle V, Rivel J, Michelet V, Majoufre C, Pinsolle J. Treatment of fibrous dysplasia of the cranio-facial bones. Report of 25 cases. Ann Chir Plast Esthet. 1998;43(3):234-9. PMid:9768065.
Béquignon E, Cardinne C, Lachiver X, et al. Craniofacial fibrous dysplasia surgery: A functional approach. Eur Ann Otorhinolaryngol Head Neck Dis. 2013;130:215-20.
Ricalde P, Horswell BB. Craniofacial fibrous dysplasia of the fronto-orbital region: a case series and literature review. J Oral Maxillofac Surg. 2001;59(2):157-168. http://dx.doi.org/10.1053/joms.2001.20487. PMid:11213984.
Osada Y, Iwasawa M, Tanaka Y. Use of image-guiding template for contouring surgery of midfacial fibrous dysplasia. Ann Plast Surg. 2007;59(4):459-63. http://dx.doi.org/10.1097/01.sap.0000257186.44656.17. PMid:17901743.
Wang T-M, Xi J-T, Jin Y. A model research for prototype warp deformation in the FDM process. Int J Adv Manuf Technol. 2007;33(11-12):1087-96. http://dx.doi.org/10.1007/s00170-006-0556-9.
Gui H, Zhang S, Shen SGF, Wang X, Bautista JS, Voss PJ. Real-time image-guided recontouring in the management of craniofacial fibrous dysplasia. Oral Maxillofac Surg. 2013;116(6):680-5. PMid:24113350.
Valentini V, Cassoni A, Marianetti TM, Terenzi V, Fadda MT, Iannetti G. Craniomaxillofacial fibrous dysplasia: conservative treatment or radical surgery? A retrospective study on 68 patients. Plast Reconstr Surg. 2009;123(2):653-60. http://dx.doi.org/10.1097/PRS.0b013e318196bbbe. PMid:19182626.
Nuss DW, Janecka IP, Sekhar LN, Sen CN. Craniofacial disassembly in the management of skull-base tumors. Otolaryngol Clin North Am. 1991;24(6):1465-97. PMid:1792080.
Wang Y, Sun G, Lu M, Hu Q. Surgical manegemente of maxillofacial fibrous dysplaisa under navigational guidance. Br J Oral Maxillofac Surg. 2015;53(4):336-41. http://dx.doi.org/10.1016/j.bjoms.2015.01.004. PMid:25660597.
Osada Y, Iwasawa M, Tanaka Y. Use of image-guiding template for contouring surgery of midfacial fibrous dysplasia. Annals of Plastic Surgery 2007;59(4):459-63.
Notes
Author notes
Correspondence Matheus Dantas de Araújo Barretto Rua Válson Lopes, 70, Apto 63, torre 1 – Butantã – São Paulo/SP – Brazil CEP: 05360-020 Phone: +55 (84) 99134-5488 matheusdabarretto@gmail.com
Conflict of interest declaration
