Clinical Case Report
Atrial Myxoma – An unusual cause of ischemic stroke in young
Atrial Myxoma – An unusual cause of ischemic stroke in young
Autopsy and Case Reports, vol. 10, no. 4, 2020
Hospital Universitário da Universidade de São Paulo
Received: 25 April 2020
Accepted: 07 May 2020
DOI: 10.4322/acr.2020.178
ABSTRACT: Atrial myxomas are rare primary cardiac tumours with neurological manifestations being reported in 30% of cases. Though a rare cause of ischemic stroke in young patients, considering it as a possibility in absence of any obvious risk factors can help avoid misdiagnosis at early stages. We present a case of left atrial myxoma in a 36-year-old male with no known co-morbidities, showing an unusual clinical presentation of isolated bilateral painless vision loss. With multiple infarcts on Non Contrast Computerised Tomography (NCCT) and a suspicion of Atrial Myxoma on Transesophageal Echocardiography (TEE), patient was successfully managed surgically with confirmation of diagnosis on histopathology.
Keywords - : Myxoma, Stroke, Thrombosis.
INTRODUCTION
Primary cardiac tumors are found in 0.1% of total cases on autopsy,1 represented mostly by the atrial myxoma. Myxoma is considered a “benign tumor” that arises in any of the cardiac chambers and can be uni or bilateral, though 75% occurs in the left atrium.2 They have a spectrum of clinical presentations, with 30% cases presenting neurological signs, ischemic stroke being the most common.3 Atrial myxoma accounts for only <1% cases of ischemic stroke in the young. However, it should be considered a possibility in the absence of any known risk factors to avoid misdiagnosis at early stages.4 Transesophageal echocardiography (TEE) is considered the best diagnostic modality, and surgical excision remains the mainstay of treatment.5
We present an unusual clinical presentation of isolated bilateral loss of vision in a young male with left atrial myxoma, where a diagnosis of left ventricular thrombus was favored over atrial myxoma on cardiac magnetic resonance imaging) (MRI).
CASE REPORT
A 36-year-old previously healthy male presented to our hospital with sudden onset bilateral, painless loss of vision of 12 hours duration along with left-sided continuous headache of moderate to severe intensity and one episode of vomiting. 10 hours prior to the vision loss, he also had one episode of exertional pre-syncope after running a 5-km circuit. There were no other relevant past or family history.
On initial examination, the patient was well oriented, had stable vitals with a regular pulse of 60/min, blood pressure of 128/86 mmHg, and respiratory rate 14/min with SpO2 of 98%. His neurological examination was essentially normal. However, his visual acuity was < 6/60 though bilateral pupils were equally reactive to light with a normal fundus on examination. Cardiovascular examination was normal. ECG revealed ST depression in inferior leads with bradycardia.
Non-contrasted computed tomography (NCCT) of the brain revealed an acute ischemic infarct in left Middle Cerebral Artery (MCA) -Posterior cerebral artery (PCA) watershed areas and distal MCA region along with multiple old lacunar infarcts (Figure 1A). His biochemical parameters remained normal except for a rise in ESR (Erythrocyte Sedimentation Rate, Normal Value ≤ 15 mm/hr) to 30 mm/hr within 3 days. The patient was managed as a case of Stroke in Young. As multiple infarcts on NCCT indicated a thromboembolic phenomenon, a TEE was done to rule out any cardiac cause. On TEE, a left atrial (LA) mass measuring 2.93 cm2, mobile, attached to the interatrial septum (IAS) near the Inferior Vena Cava (IVC) junction was noted with suspicion of atrial myxoma (Figure 1B).
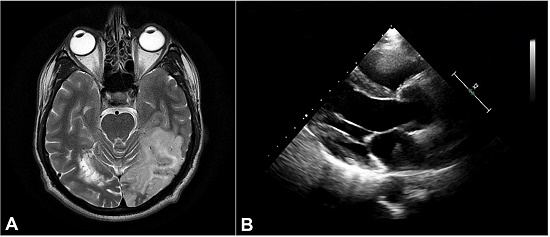
Figure 1
A – NCCT brain showing an acute wedge-shaped infarct in left hemisphere, B – Transesophageal echocardiography (TEE) showing a left atrial mass arising from Interatrial septum.
The ejection fraction was 65%. However, a cardiac MRI done subsequently favored a diagnosis of LA thrombus over myxoma as no stalk was demonstrated. After that, the patient was started on IV Heparin and was taken up for surgery 10 days later. Intraoperatively, a 3cm x 2cm pedunculated LA mass with rough villous surface and a calcified stalk arising from IAS, was noted. He underwent excision of the left atrial mass with autologous pericardial patch closure of the septal defect through the bi-atrial approach. The diagnosis of atrial myxoma was confirmed on histopathologic examination with characteristic features of stellate shaped myxoma cells embedded in a myxoid background (Figure 2). The post-operative period was uneventful.
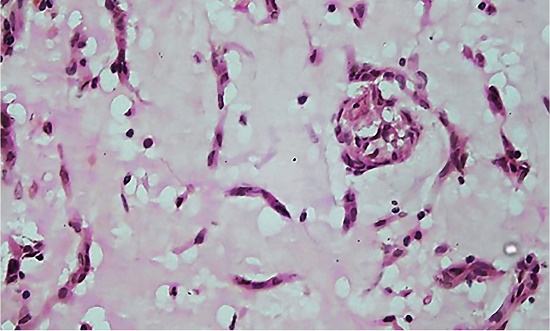
Figure 2
Photomicrograph of the surgical specimen showing the microscopic features of atrial myoma: Stellate shaped myxoma cells embedded in a myxoid stroma.
DISCUSSION
Atrial myxoma is the commonest of the otherwise rare primary cardiac tumors. It is usually seen between the third and sixth decades of life with 2: 1 female preponderance and left atrium being the commonest site accounting for 75% of the cases.1,2,6 Though mostly sporadic, a familial association has been noted in 7% of the cases.3,6Recurrences are reported, and malignant transformation remains a controversial issue.2
Clinical presentation of atrial myxoma often comprises a diagnostic triad of symptoms due to the obstruction of the cardiac outflow or due to embolism or constitutional symptoms.2-4,6Due to a myriad of presentations, diagnosis can be missed in a great number of cases at early stages, increasing the risk of morbidity and mortality. Clinical signs or echocardiography abnormalities may be absent in up to 36% of the cases,6 as was noted in our case. Thus, the presence of an embolic phenomenon, especially in young patients with neurological symptoms, should prompt early neuroimaging and echocardiography.6
The ultrastructural analysis and immunohistochemical investigation suggest that atrial myxoma is more likely derived from a pluripotent mesenchymal stem cell or sub-endothelial cell.7 Angiocardiography was the first diagnostic modality introduced for cardiac tumors, which led to the antemortem diagnosis of atrial myxomas, as prior to which, cases were diagnosed only on autopsy.2 At present, TEE is the best diagnostic modality for diagnosis of atrial myxomas.5 Cardiac MRI is useful in determining the tumor size, attachment, and mobility, which helps the surgical management and planning.6 In cases of ischemic stroke caused by a left atrial myxoma, thrombolytic treatment is considered a safe option of initial management. However, the extent of anticoagulation therapy should be carefully monitored, as hemorrhagic transformation might alter the timing of definitive surgery. In this setting, a delay can increase the overall morbidity and mortality.4
Histopathology is confirmatory for benign atrial myxoma. Few studies,8,9 which attempted classifying myxomas based on myxoma cell population and differentiation, reported that all recurrences were observed in patients with active myxoma with poor differentiation. Recurrences are reported in 1-3% of cases, primarily due to inadequate surgical resection.10 Annual follow up with echocardiography is suggested for a period of 3 to 4 years, especially in sporadic cases.11
CONCLUSION
Albeit rare, considering a differential diagnosis of atrial myxoma in cases of stroke in young, evaluation by early imaging and prompt treatment can help decrease morbidity and mortality resulting from the associated thromboembolic phenomenon.
REFERENCES
Figueroa-Torres Y, Martı’nez-Ojeda JA, Franqui-Rivera H, Martı’nez-Toro J. Benign cardiac neoplasms: The experience at the Cardiovascular Center of Puerto Rico and the Caribbean. P R Health Sci J. 2008;27(4):373-6. PMid:19069370.
Pinede L, Duhaut P, Loire R. Clinical presentation of left atrial cardiac myxoma: A series of 112 consecutive cases. Medicine (Baltimore). 2001;80(3):159-72. http://dx.doi.org/10.1097/00005792-200105000-00002. PMid:11388092.
Ekinci EI, Donnan GA. Neurological manifestations of cardiac myxoma: A review of the literature and report of cases. Intern Med J. 2004;34(5):243-9. http://dx.doi.org/10.1111/j.1444-0903.2004.00563.x. PMid:15151670.
Hatayama S, Ogata T, Okawa M, et al. Ischemic stroke induced by a left atrial myxoma. Brain Nerve. 2012;64(10):1175-9. PMid:23037608.
Centofanti P, Di Rosa E, Deorsola L, et al. Primary cardiac tumors: early and late results of surgical treatment in 91 patients. Ann Thorac Surg. 1999;68(4):1236-41. http://dx.doi.org/10.1016/S0003-4975(99)00700-6. PMid:10543485.
O’Rourke F, Dean N, Mouradian MS, Akhtar N, Shuaib A. Atrial myxoma as a cause of stroke: case report and discussion. CMAJ. 2003;169(10):1049-51. PMid:14609975.
Aiello VD, Campos FP. Cardiac Myxoma. Autops Case Rep. 2016;6(2):5-7. http://dx.doi.org/10.4322/acr.2016.030. PMid:27547737.
Wang JG, Li YJ, Liu H, Li NN, Zhao J, Xing XM. Clinicopathologic analysis of cardiac myxomas: seven years’ experience with 61 patients. J Thorac Dis. 2012;4(3):272-83. http://dx.doi.org/10.3978/j.issn.2072-1439.2012.05.07. PMid:22754666.
Siminelakis S, Kakourou A, Batistatou A, Sismanidis S, Ntoulia A, Tsakiridis K, Syminelaki T, Apostolakis E, Zarogoulidis P, Tsiouda T, Katsikogiannis N, Kougioumtzi I, Dryllis G, Machairiotis N, Mpakas A, Beleveslis T, Zarogoulidis K. Thirteen years follow-up of heart myxoma operated patients: what is the appropriate surgical technique? J Thorac Dis. 2014 Mar;6 (1 Suppl):S32-8. http://dx.doi.org/10.3978/j.issn.2072-1439.2013.10.21. Erratum in: J Thorac Dis. 2014 Jun;6(6):E146.
McCarthy PM, Piehler JM, Schaff HV, et al. The significance of multiple, recurrent and “complex” cardiac myxomas. J Thorac Cardiovasc Surg. 1986;91(3):389-96. http://dx.doi.org/10.1016/S0022-5223(19)36054-4. PMid:3951243.
Semb BK. Surgical considerations in the treatment of cardiac myxoma. J Thorac Cardiovasc Surg. 1984;87(2):251-9. http://dx.doi.org/10.1016/S0022-5223(19)37419-7. PMid:6694416.
Notes
Author notes
Correspondence Khushdeep Kaur Shergill Department of Pathology INHS Asvini, Mumbai, Maharashtra, India. Pin: 400 005 Phone: 7030399112 shergillkhushdeep@gmail.com
Conflict of interest declaration
