Article / Autopsy Case Report
Multiple cardiac rhabdomyomas in tuberous sclerosis complex: case report and review of the literature
Multiple cardiac rhabdomyomas in tuberous sclerosis complex: case report and review of the literature
Autopsy and Case Reports, vol. 9, no. 4, e2019125, 2019
Hospital Universitário da Universidade de São Paulo
Received: 24 July 2019
Accepted: 04 September 2019
ABSTRACT: Cardiac rhabdomyoma is a benign tumor which constitutes the most common cardiovascular feature of the tuberous sclerosis complex, a multisystem genetically determined neurocutaneous disorder. Cardiac rhabdomyomas can be detected in the prenatal ultrasound, are usually asymptomatic and spontaneously regress within the first three years of life. Less often, the tumors’ size, number, and location can produce a mass effect that may lead to blood flow abnormalities or organ dysfunction (heart failure and arrhythmia). In this setting, severe morbidity, and eventually, a lethal outcome despite clinical and surgical treatment may ensue. We describe a fatal case of multiple cardiac rhabdomyomas in a newborn girl. One of the rhabdomyomas was large and unfavorably located, causing significant obstruction of the left ventricular outflow tract. The autopsy identified, in addition to cardiac rhabdomyomas, brain glioneuronal hamartomas (cortical tubers), subependymal nodules and subependymal giant cell tumors, characteristic of the tuberous sclerosis complex. The newborn’s family was investigated for the presence of typical clinical symptoms of the complex and image findings showed significant phenotypical variations and a broad symptom spectrum among the family members. This interesting case underscores the variability of tuberous sclerosis complex and the importance of performing a comprehensive postmortem examination in the identification of the cause of death, especially in the setting of familial disease.
Keywords: Rhabdomyoma, Tuberous Sclerosis, Infant, Newborn, Heart Neoplasms, Neurocutaneous Syndromes.
CASE REPORT
A 7-day-old girl had been born by cesarean delivery at a gestational age of 39 weeks due to prenatal diagnosis of a cardiac tumor in fetal ultrasound. The mother had attended ten prenatal consultations and had negative serologies to HIV, syphilis, and hepatitis B. Relevant family history comprised the presence of dermatological lesions in the patient’s mother, aunt, and grandfather, and both mother and aunt reported having seizures since childhood.
The newborn’s Apgar scores were 7 and 8, and the birth weight was 2745g. Upon initial physical examination, she presented a systolic heart murmur on the left medium sternal border, intensity 3/6. The remaining physical examination was unremarkable. Initially, the patient was hemodynamically stable, without signs of low cardiac output such as hypotension, tachycardia, breathlessness and hypoxemia, alterations in the level of consciousness, cold peripheries, or delayed capillary refill time. Nonetheless, she was kept in the Neonatal Intensive Care Unit (NICU) under rigorous cardiac monitoring. Two echocardiograms were performed and confirmed the presence of multiple tumors in the ventricular walls and interventricular septum. There were two main lesions in the interventricular septum, the largest one located at its basal portion measuring 14X13mm in its longest axis, protruding (8.2mm) into the left ventricular outflow tract, markedly obstructing it. The pressure gradient was approximately 95 mmHg. Rhabdomyomas as the working diagnosis was considered, and surgical intervention was scheduled for the 7th day after birth. The physical examination, at this moment, showed normal intensity and regular peripheral pulses at a rate of 162 per minute. The girl’s blood pressure was 58/33 mmHg, and her room-air oxygen saturation was 97%. Peripheral perfusion was normal, and no respiratory distress (respiratory rate was 37 breaths per minute) was depicted. The cardiovascular examination demonstrated the persistence of the systolic murmur grade 3/6 at the left sternal border. Pre-op laboratory exams, including complete blood count, erythrocyte sedimentation rate, C-reactive protein, serum electrolytes, liver, and renal function tests were all unremarkable. However, during anesthetic induction, the patient developed hemodynamic instability, bradycardia, and cardiac arrest. Unfortunately, she died despite the attempts of resuscitation. An autopsy was performed with the informed consent of the family.
AUTOPSY FINDINGS
The skin examination was normal. The heart weighed 25 g (average heart weight adapted for age and birth weight: 19.1 ± 2.8g) and showed nine cardiac nodules diffusely distributed within the atrial and ventricular chambers, measuring from 0.2 to 1.5 cm. They were firm and white, the biggest one had 1.5 cm, was located in the interventricular septum, protruded into the left ventricular outflow tract, and almost entirely obstructed the aortic valve (Figures 1A, 1B, 1C).
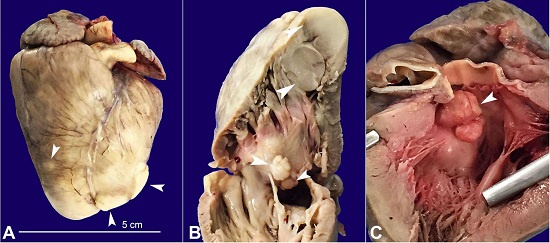
Figure 1
Gross view of the heart. A - Anterior face of the heart showing several whitish nodules (arrowheads); B - Right ventricular outflow tract with some of the nodules represented on the cut surface (arrowheads); C - Left ventricular outflow tract evidencing subaortic obstruction by the largest rhabdomyoma of the interventricular septum (arrowhead).
Histologically, they were composed of vacuolated cells showing almost “empty” cytoplasm with a centrally placed nucleus and cytoplasmic strands extending to the periphery of the cell – consistent with “spider cells” (Figures 2A, 2B).
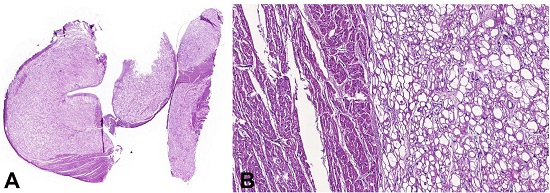
Figure 2
Photomicrograph of the heart. A - Panoramic view of cardiac rhabdomyomas represented by large and clear cell nodules (HE 1x); B - Rhabdomyoma composed of a proliferation of large and vacuolated cells with a centrally placed nucleus and cytoplasmic strands extending to the periphery of the cell – “spider cell” (HE 10X).
The brain weighed 110g (average brain weight adapted for age and birth weight: 337 ± 91g). The cut surface revealed multiple glioneuronal hamartomas (cortical tubers) and subependymal nodules along the walls of the lateral ventricle (Figure 3).
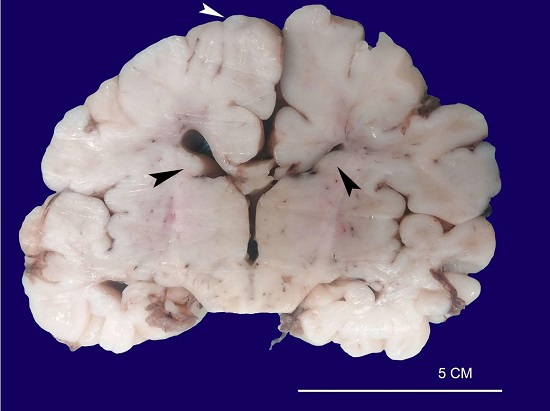
Figure 3
Macroscopic aspect of the brain showing cortical tubers (white arrowhead) and subependymal nodular lesions arising in the walls of the lateral ventricles (black arrowheads).
Histologically, the cortical tubers and subependymal nodules were composed by a proliferation of large and atypical neuronal and glial cells (Figure 4A, 4B). One of the subependymal lesions is represented by a subependymal giant cell tumor (SGCT), comprised of a combination of giant cells with glassy, eosinophilic cytoplasm and round nuclei with conspicuous nucleoli, interspersed with spindled cells and fibrillary elements (Figure 4C, 4D). This type of lesion is characteristic of tuberous sclerosis complex (TSC).
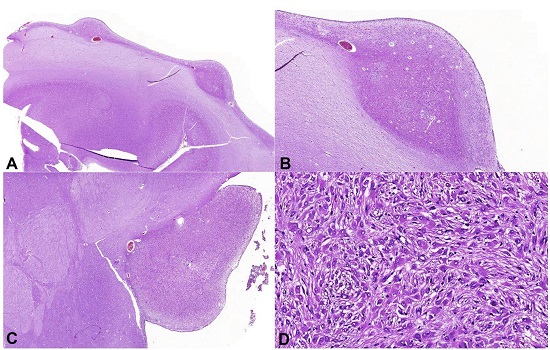
Figure 4
Photomicrographs of the brain. A and B - Subependymal nodules (A, HE 1X; B, HE 3X), comprised by a proliferation of neuronal cells and large and atypical glial cells; C - Large subependymal lesion (HE 1.44X), comprised by the proliferation of large cells of eosinophilic cytoplasm with moderate pleomorphism (D, HE 20X).
No other tumors or hamartomas were identified in the remaining organs.
FAMILY INVESTIGATION
TSC had not been previously diagnosed in the patient’s mother or the family. The autopsy allowed confirmation of the diagnosis and further investigation of the newborn’s family for TSC symptoms. The mother’s dermatological exam found skin lesions such as angiofibroma, ash-leaf spots, shagreen patches, and periungual fibromas. Her head MRI identified multiple cortical tubers, mostly in the frontal and parietal lobes, and white matter heterotopia and subependymal nodules in the lateral ventricles. The chest computed tomography (CT) showed bilateral pulmonary cysts associated to small nodules compatible with lymphangioleiomyomatosis (LAM), while the abdominal CT displayed renal and hepatic hypoattenuating lesions resembling angiomyolipoma, confirmed by kidney biopsy.
DISCUSSION
Tuberous sclerosis complex (TSC) is a multisystem syndrome clinically characterized by dermatologic lesions, neurologic disorders – mostly seizures – and the development of benign tumors in the brain, heart, and kidneys.1 It is genetically determined, first by mutations in either the TSC1 or TSC2 gene – encoding hamartin and tuberin, respectively, which together form a protein complex that participates in cellular hyperplasia – and second by modifications in a tumor suppressor gene in most hamartomas. The high variability in TSC1/TSC2 mutation type and position may partially explain the broad range of clinical symptoms observed in TSC patients. However, timing and location of the second hit in the tumor suppressor gene probably have a higher contribution to the variability of clinical symptoms.2 These mutations are spontaneous in 70% of cases, or they can be inherited in an autosomal dominant manner.3
Cardiac rhabdomyomas are the most common cardiovascular manifestation of TSC, present in more than half of the patients. Although these tumors may appear as an isolated affection, they are associated with TSC in the great majority of cases.4 Despite being pathologically benign and generally asymptomatic, the tumors’ size, number, and location can produce a mass effect and lead to blood flow abnormalities or organ dysfunction (heart failure and arrhythmia). Our case is an example of multiple tumors, one of them with an unfavorable location and big enough to cause significant obstruction of the left ventricular outflow tract. Rhabdomyomas can be detected in 20 to 30 weeks’ prenatal ultrasound, and if present, followed with an electrocardiogram (ECG) to evaluate arrhythmia and an echocardiogram every one to three years until the regression is observed, which generally occurs within the first three years of life.5-7 Unfortunately, tumors that generate hemodynamic instability, as described in this case, can sometimes present poor prognosis and, thereby, require more aggressive intervention.
We reviewed the current literature, which found 31 case reports of cardiac rhabdomyoma and TSC in PubMed (Table 18-36).
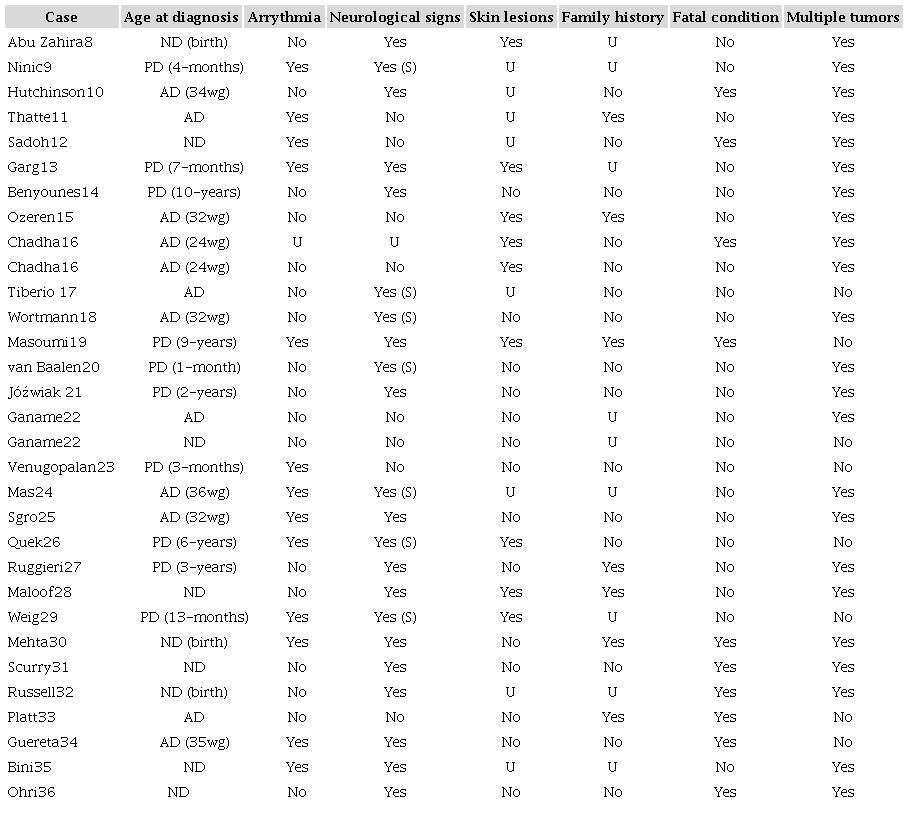
AD = antenatal diagnosis (before birth); ND = neonatal (birth - up to 28 days of life) diagnosis; PD = postneonatal diagnosis; wg = weeks of gestation; S = seizure; U = unknown.
In line with our report, we found that 38.7% of the tumors were diagnosed during gestation, and 32.2% were fatal. Family history of TSC was present in 31.8%, and neurological signs were detected in 73.3% of cases. Multiple tumors were more prevalent than isolated lesions, and arrhythmia was a significant finding in 43.3% of the reported cases.
Among neurological conditions, TSC may present with structural brain abnormalities such as glioneuronal hamartomas – also called cortical tubers and found in at least 80% of TSC patients –, subependymal nodules, seen in about 90% of cases and, with a lower prevalence of 10 to 20%, subependymal giant cell tumors (SGCT).1,37 Although asymptomatic and with a normal appearance in neurological examination, in this case, the newborn’s autopsy identified all of the three brain alterations. The clinical outcome of these tumors, when symptomatic, includes epilepsy or infantile spasms in children younger than 1 year,38 probably related to cortical tubers, and obstructive hydrocephalus due to occlusion of the foramen of Monro caused by SGCT in 10% of cases.39 Intellectual disability, autism spectrum disorders, and behavioral problems can develop in up to 90% of TSC cases, comprising the TSC-associated neuropsychiatric disorders (TAND).40 An association between infantile spasms and the development of TAND has been described.41,42
In this case, not only the newborn’s TSC diagnosis was made by the autopsy, but also the family members were examined for the presence of the disease’s clinical symptoms because of its genetic basis and the possibility of heritability. Another important aspect of the case is TSC’s phenotypical variation among the family. Whereas the newborn presented cardiovascular features, the aunt and grandfather had a history of dermatological lesions, the mother exhibited mostly neurological and dermatological symptoms and had pulmonary, hepatic, and renal image findings. Wide intrafamilial phenotypic variability has been reported,43,44 and family members may sometimes be affected, yet escape the previous diagnosis because of a milder phenotypic presentation.45 At times, the disease can only be diagnosed through genetic testing or image findings, leaving the patient otherwise healthy until adulthood, while in other cases it can present clinical symptoms with a broad spectrum of severity. Regarding cardiac rhabdomyomas, they have been reported to be associated with congestive heart failure, arrhythmias,45,46 or complete cardiovascular collapse and sudden death.46-48
Due to the possibility of fatal consequences, symptomatic tumors must be examined. Regarding the heart, prostaglandins can be used to ensure the patency of the ductus arteriosus and systemic blood flow to stabilize the critically ill newborn while waiting for the involution of the rhabdomyoma.7 Recent reports suggest new clinical treatment modalities with the use of mechanistic target of rapamycin inhibitors, such as sirolimus49 or everolimus.17,50 These drugs may induce accelerated tumor regression, therefore avoiding the need for invasive intervention. However, the minority of cases in which they do not spontaneously regress and become life-threatening, due to either left ventricular outflow tract obstruction, right ventricular outflow tract obstruction, mitral valve insufficiency, or intractable arrhythmias, cardiac surgery might be necessary. Operative management typically involves resection of the intracavitary portion of the tumor without complete excision. As previously stated, the newborn’s main tumor was large and caused significant obstruction of the left ventricular outflow tract during systole. Therefore, the newborn was kept under continuous monitorization in the NICU, and surgical resection was planned as soon as possible, in her first week of life, since the mass began to cause hemodynamic compromise and could result in catastrophic cardiovascular collapse. Other authors have reported cases with similar clinical and imaging presentations, in which aggressive surgical management was successful in treating and preventing hemodynamic compromise with good long-term outcomes.46,51-54 Unfortunately, despite the effort to reverse a life-threatening condition already established, the newborn could not tolerate the additional hypotensive effect of general anesthesia, considering the blood flow compromise caused by the tumor, and did not survive the surgery.
CONCLUSIONS
The reported case demonstrates that, despite histologically benign, large, and problematically placed cardiac rhabdomyomas may present severe morbidity, and, eventually, a lethal outcome. Thus, we highlight the importance of proper diagnosis and timely adequate management of these tumors in order to prevent the potential of catastrophic cardiovascular collapse. Additionally, this case underscores the variability of TSC and the importance of performing a comprehensive postmortem examination in the identification of the cause of death, especially for a familial disease.
References
Randle SC. Tuberous sclerosis complex: a review. Pediatr Ann. 2017;46(4):e166-71. http://dx.doi.org/10.3928/19382359-20170320-01. PMid: 28414398
Rosset C, Vairo F, Bandeira IC, et al. Molecular analysis of TSC1 and TSC2 genes and phenotypic correlations in Brazilian families with tuberous sclerosis. PLoS One. 2017;12(10):e0185713. http://dx.doi.org/10.1371/journal.pone.0185713. PMid: 28968464
Roach ES. Applying the lessons of tuberous sclerosis: the 2015 hower award lecture. Pediatr Neurol. 2016;63:6-22. http://dx.doi.org/10.1016/j.pediatrneurol.2016.07.003. PMid: 27543366
Beghetti M, Gow RM, Haney I, Mawson J, Williams WG, Freedom RM. Pediatric primary benign cardiac tumors: a 15-year review. Am Heart J. 1997;134(6):1107-14. http://dx.doi.org/10.1016/S0002-8703(97)70032-2. PMid: 9424072
Northrup H, Krueger DA. Tuberous sclerosis complex diagnostic criteria update: recommendations of the 2012 Iinternational Tuberous Sclerosis Complex Consensus Conference. Pediatr Neurol. 2013;49(4):243-54. http://dx.doi.org/10.1016/j.pediatrneurol.2013.08.001. PMid: 24053982
Krueger DA, Northrup H, Northrup H, et al. Tuberous sclerosis complex surveillance and management: recommendations of the 2012 International Tuberous Sclerosis Complex Consensus Conference. Pediatr Neurol. 2013;49(4):255-65. http://dx.doi.org/10.1016/j.pediatrneurol.2013.08.002. PMid: 24053983
Hinton RB, Prakash A, Romp RL, Krueger DA, Knilans TK. Cardiovascular manifestations of tuberous sclerosis complex and summary of the revised diagnostic criteria and surveillance and management recommendations from the International Tuberous Sclerosis Consensus Group. J Am Heart Assoc. 2014;3(6):e001493. http://dx.doi.org/10.1161/JAHA.114.001493. PMid: 25424575
Abu Zahira IA, Nir A. Punch-out lesion following regression of a large left ventricular outflow rhabdomyoma. Cardiol Young. 2018;28(1):153-4. http://dx.doi.org/10.1017/S1047951117001512. PMid: 28847327
Ninic S, Kalaba M, Jovicic B, et al. Successful use of sirolimus for refractory atrial ectopic tachycardia in a child with cardiac rhabdomyoma. Ann Noninvasive Electrocardiol. 2017;22(4):e12435. http://dx.doi.org/10.1111/anec.12435. PMid: 28217909
Hutchinson JC, Ashworth MT, Sebire NJ, Arthurs OJ. Multiple cardiac rhabdomyomas visualised using Micro-CT in a case of tuberous sclerosis. Fetal Diagn Ther. 2017;41(2):157-60. http://dx.doi.org/10.1159/000448708. PMid: 27559745
Thatte NM, Guleserian KJ, Veeram Reddy SR. New-onset cardiac rhabdomyoma beyond infancy in a patient with tuberous sclerosis complex. Cardiol Young. 2016;26(2):396-9. http://dx.doi.org/10.1017/S1047951115001183. PMid: 26169191
Sadoh WE, Obaseki DE, Amuabunos EA, et al. Cardiac rhabdomyoma in a neonate with supraventricular tachycardia. World J Pediatr Congenit Heart Surg. 2014;5(1):110-3. http://dx.doi.org/10.1177/2150135113501899. PMid: 24403367
Garg R, Thakkar B, Oswal N. A rare case report of corrected transposition of the great arteries in association with tuberous sclerosis and cardiac rhabdomyomas. Cardiol Young. 2014;24(5):955-7. http://dx.doi.org/10.1017/S1047951113002047. PMid: 24252420
Benyounes N, Fohlen M, Devys JM, Delalande O, Moures JM, Cohen A. Cardiac rhabdomyomas in tuberous sclerosis patients: a case report and review of the literature. Arch Cardiovasc Dis. 2012;105(8-9):442-5. http://dx.doi.org/10.1016/j.acvd.2012.01.009. PMid: 22958887
Ozeren S, Cakiroglu Y, Doger E, Caliskan E. Sonographic diagnosis of fetal cardiac rhabdomyomas in two successive pregnancies in a woman with tuberous sclerosis. J Clin Ultrasound. 2012;40(3):179-82. http://dx.doi.org/10.1002/jcu.21890. PMid: 22287098
Chadha R, Johnson JA, Fruitman D, Cooper SL, Wei XC, Bernier F. A rare case of cardiac rhabdomyomas in a dizygotic twin pair. J Obstet Gynaecol Can. 2011;33(8):854-7. http://dx.doi.org/10.1016/S1701-2163(16)34990-8. PMid: 21846442
Tiberio D, Franz DN, Phillips JR. Regression of a cardiac rhabdomyoma in a patient receiving everolimus. Pediatrics. 2011;127(5):e1335-7. http://dx.doi.org/10.1542/peds.2010-2910. PMid: 21464184
Wortmann SB, Reimer A, Creemers JW, Mullaart RA. Prenatal diagnosis of cerebral lesions in Tuberous sclerosis complex (TSC). Case report and review of the literature. Eur J Paediatr Neurol. 2008;12(2):123-6. http://dx.doi.org/10.1016/j.ejpn.2007.06.006. PMid: 17681840
Masoumi H, Kinney HC, Chadwick AE, Rubio A, Krous HF. Sudden unexpected death in childhood associated with cardiac rhabdomyoma, involuting adrenal ganglioneuroma, and megalencephaly: another expression of tuberous sclerosis? Pediatr Dev Pathol. 2007;10(2):129-33. http://dx.doi.org/10.2350/06-04-0081.1. PMid: 17378684
van Baalen A, Stephani U, Jansen O. Confirmation of tuberous sclerosis complex by high-resolution ultrasonography. J Child Neurol. 2006;21(9):741-2. http://dx.doi.org/10.1177/08830738060210091601. PMid: 16970877
Jóźwiak S, Domańska-Pakieła D, Kwiatkowski DJ, Kotulska K. Multiple cardiac rhabdomyomas as a sole symptom of tuberous sclerosis complex: case report with molecular confirmation. J Child Neurol. 2005;20(12):988-9. http://dx.doi.org/10.1177/08830738050200121101. PMid: 16417848
Ganame J, D’hooge J, Mertens L. Different deformation patterns in intracardiac tumors. Eur J Echocardiogr. 2005;6(6):461-4. http://dx.doi.org/10.1016/j.euje.2005.02.006. PMid: 16293533
Venugopalan P, Babu JS, Al-Bulushi A. Right atrial rhabdomyoma acting as the substrate for Wolff-Parkinson-White syndrome in a 3-month-old infant. Acta Cardiol. 2005;60(5):543-5. http://dx.doi.org/10.2143/AC.60.5.2004977. PMid: 16261787
Mas C, Penny DJ, Menahem S. Pre-excitation syndrome secondary to cardiac rhabdomyomas in tuberous sclerosis. J Paediatr Child Health. 2000;36(1):84-6. http://dx.doi.org/10.1046/j.1440-1754.2000.00443.x. PMid: 10723700
Sgro M, Barozzino T, Toi A, Johnson J, Sermer M, Chitayat D. Prenatal detection of cerebral lesions in a fetus with tuberous sclerosis. Ultrasound Obstet Gynecol. 1999;14(5):356-9. http://dx.doi.org/10.1046/j.1469-0705.1999.14050356.x. PMid: 10623997
Quek SC, Ng KS, Low PS. Cardiac dysrrhthmia in systemic disease. Singapore Med J. 1998;39(9):426-7. PMid: 9885725
Ruggieri M, Carbonara C, Magro G, et al. Tuberous sclerosis complex: neonatal deaths in three of four children of consanguineous, non-expressing parents. J Med Genet. 1997;34(3):256-60. http://dx.doi.org/10.1136/jmg.34.3.256. PMid: 9132502
Maloof J, Sledz K, Hogg JF, Bodensteiner JB, Schwartz T, Schochet SS. Unilateral megalencephaly and tuberous sclerosis: related disorders? J Child Neurol. 1994;9(4):443-6. http://dx.doi.org/10.1177/088307389400900424. PMid: 7822741
Weig SG, Pollack P. Carbamazepine-induced heart block in a child with tuberous sclerosis and cardiac rhabdomyoma: implications for evaluation and follow-up. Ann Neurol. 1993;34(4):617-9. http://dx.doi.org/10.1002/ana.410340417. PMid: 8215250
Mehta AV. Rhabdomyoma and ventricular preexcitation syndrome. A report of two cases and review of literature. Am J Dis Child. 1993;147(6):669-71. http://dx.doi.org/10.1001/archpedi.1993.02160300075027. PMid: 8506838
Scurry J, Watkins A, Acton C, Drew J. Tachyarrhythmia, cardiac rhabdomyomata and fetal hydrops in a premature infant with tuberous sclerosis. J Paediatr Child Health. 1992;28(3):260-2. http://dx.doi.org/10.1111/j.1440-1754.1992.tb02659.x. PMid: 1605981
Russell GA, Dhasmana JP, Berry PJ, Gilbert-Barness EF. Coexistent cardiac tumours and malformations of the heart. Int J Cardiol. 1989;22(1):89-98. http://dx.doi.org/10.1016/0167-5273(89)90140-X. PMid: 2925289
Platt LD, Devore GR, Horenstein J, Pavlova Z, Kovacs B, Falk RE. Prenatal diagnosis of tuberous sclerosis: the use of fetal echocardiography. Prenat Diagn. 1987;7(6):407-11. http://dx.doi.org/10.1002/pd.1970070606. PMid: 3658914
Guereta LG, Burgueros M, Elorza MD, Alix AG, Benito F, Gamallo C. Cardiac rhabdomyoma presenting as fetal hydrops. Pediatr Cardiol. 1986;7(3):171-4. http://dx.doi.org/10.1007/BF02424993. PMid: 3543873
Bini RM, Westaby S, Bargeron LM Jr, Pacifico AD, Kirklin JW. Investigation and management of primary cardiac tumors in infants and children. J Am Coll Cardiol. 1983;2(2):351-7. http://dx.doi.org/10.1016/S0735-1097(83)80174-0. PMid: 6863768
Ohri GL, DeVenecia R, Acs H. Tuberous sclerosis presenting as diaphragmatic hernia in a newborn. Dev Med Child Neurol. 1980;22(4):509-12. http://dx.doi.org/10.1111/j.1469-8749.1980.tb04357.x. PMid: 7409341
Roth J, Roach ES, Bartels U, et al. Subependymal giant cell astrocytoma: diagnosis, screening, and treatment. Recommendations from the International Tuberous Sclerosis Complex Consensus Conference 2012. Pediatr Neurol. 2013;49(6):439-44. http://dx.doi.org/10.1016/j.pediatrneurol.2013.08.017. PMid: 24138953
Pellock JM, Hrachovy R, Shinnar S, et al. Infantile spasms: a U.S. consensus report. Epilepsia. 2010;51(10):2175-89. http://dx.doi.org/10.1111/j.1528-1167.2010.02657.x. PMid: 20608959
Goh S, Butler W, Thiele EA. Subependymal giant cell tumors in tuberous sclerosis complex. Neurology. 2004;63(8):1457-61. http://dx.doi.org/10.1212/01.WNL.0000142039.14522.1A. PMid: 15505165
de Vries PJ, Whittemore VH, Leclezio L, et al. Tuberous sclerosis associated neuropsychiatric disorders (TAND) and the TAND Checklist. Pediatr Neurol. 2015;52(1):25-35. http://dx.doi.org/10.1016/j.pediatrneurol.2014.10.004. PMid: 25532776
Saemundsen E, Ludvigsson P, Rafnsson V. Autism spectrum disorders in children with a history of infantile spasms: a population-based study. J Child Neurol. 2007;22(9):1102-7. http://dx.doi.org/10.1177/0883073807306251. PMid: 17890408
Saemundsen E, Ludvigsson P, Rafnsson V. Risk of autism spectrum disorders after infantile spasms: a population-based study nested in a cohort with seizures in the first year of life. Epilepsia. 2008;49(11):1865-70. http://dx.doi.org/10.1111/j.1528-1167.2008.01688.x. PMid: 18557779
Wang F, Xiong S, Wu L, Chopra M, Hu X, Wu B. A novel TSC2 missense variant associated with a variable phenotype of tuberous sclerosis complex: case report of a Chinese family. BMC Med Genet. 2018;19(1):90. http://dx.doi.org/10.1186/s12881-018-0611-z. PMid: 29843636
Caylor RC, Grote L, Thiffault I, et al. Incidental diagnosis of tuberous sclerosis complex by exome sequencing in three families with subclinical findings. Neurogenetics. 2018;19(3):205-13. http://dx.doi.org/10.1007/s10048-018-0551-y. PMid: 29926239
Northrup H, Koenig MK, Pearson DA, Au KS. Tuberous Sclerosis Complex. In: Adam MP, Ardinger HH, Pagon RA, Wallace SE, Bean LJH, Stephens K, et al., editors. GeneReviews((R)). Seattle: University of Washington; 1993.
Sciacca P, Giacchi V, Mattia C, et al. Rhabdomyomas and tuberous sclerosis complex: our experience in 33 cases. BMC Cardiovasc Disord. 2014;14(1):66. http://dx.doi.org/10.1186/1471-2261-14-66. PMid: 24884933
Gupta A, Narula N, Mahajan R, Rohit M. Sudden death of a young child due to cardiac rhabdomyoma. Pediatr Cardiol. 2010;31(6):894-6. http://dx.doi.org/10.1007/s00246-010-9731-8. PMid: 20505929
Cina SJ, Smialek JE, Burke AP, Virmani R, Hutchins GM. Primary cardiac tumors causing sudden death: a review of the literature. Am J Forensic Med Pathol. 1996;17(4):271-81. http://dx.doi.org/10.1097/00000433-199612000-00001. PMid: 8947350
Breathnach C, Pears J, Franklin O, Webb D, McMahon CJ. Rapid regression of left ventricular outflow tract rhabdomyoma after sirolimus therapy. Pediatrics. 2014;134(4):e1199-202. http://dx.doi.org/10.1542/peds.2013-3293. PMid: 25180276
Aw F, Goyer I, Raboisson MJ, Boutin C, Major P, Dahdah N. Accelerated cardiac rhabdomyoma regression with everolimus in infants with tuberous sclerosis complex. Pediatr Cardiol. 2017;38(2):394-400. http://dx.doi.org/10.1007/s00246-016-1528-y. PMid: 27878332
Foster ED, Spooner EW, Farina MA, Shaher RM, Alley RD. Cardiac rhabdomyoma in the neonate: surgical management. Ann Thorac Surg. 1984;37(3):249-53. http://dx.doi.org/10.1016/S0003-4975(10)60334-7. PMid: 6703807
Jacobs JP, Konstantakos AK, Holland FW 2nd, Herskowitz K, Ferrer PL, Perryman RA. Surgical treatment for cardiac rhabdomyomas in children. Ann Thorac Surg. 1994;58(5):1552-5. http://dx.doi.org/10.1016/0003-4975(94)91963-1. PMid: 7979700
Takach TJ, Reul GJ, Ott DA, Cooley DA. Primary cardiac tumors in infants and children: immediate and long-term operative results. Ann Thorac Surg. 1996;62(2):559-64. http://dx.doi.org/10.1016/0003-4975(96)00349-9. PMid: 8694623
Black MD, Kadletz M, Smallhorn JF, Freedom RM. Cardiac rhabdomyomas and obstructive left heart disease: histologically but not functionally benign. Ann Thorac Surg. 1998;65(5):1388-90. http://dx.doi.org/10.1016/S0003-4975(98)00180-5. PMid: 9594871
Notes
Author notes
Correspondence Paula Frudit Santa Casa de Sao Paulo – School of Medical Sciences Rua Doutor Cesário Mota Júnior, 61 – Vila Buarque – São Paulo/SP – Brazil CEP: 01221-020 Phone: +55 (11) 98432-5194 paulafrudit@gmail.com
Conflict of interest declaration
