Article / Autopsy Case Report
Progressive dyspnea and a right atrial mass in an 80-year-old man
Progressive dyspnea and a right atrial mass in an 80-year-old man
Autopsy and Case Reports, vol. 9, no. 4, e2019135, 2019
Hospital Universitário da Universidade de São Paulo
Received: 16 August 2019
Accepted: 01 October 2019
ABSTRACT: Hepatocellular carcinoma (HCC) is among the five most frequent causes of cancer death worldwide, according to the WHO. The disease is related to alcohol abuse, viral infections, and other causes of cirrhosis, and unfortunately, in some developed countries, the incidence shows an increasing trend. Although the diagnosis of the HCC often relies upon the context of a chronic hepatopathy, some cases may present a silent course, and the initial symptoms ensue when the disease is in an advanced stage with no chance for any therapeutic attempt. The clinical picture of the HCC is varied, and unexpected forms may surprise the clinician. One of the unusual presentations of the HCC is shock by the blockage of the venous return to the right atrium by the inferior vena cava infiltration. Herein we present a case of an old patient who sought medical care complaining of dyspnea. The clinical workup disclosed a right thorax pleural effusion and imaging exams depicted a mass in the right hepatic lobe, invasion of the inferior vena cava (IVC) and the right atrium (RA). During the attempts of clinical investigation, the patient passed away. The autopsy disclosed an HCC involving the right hepatic lobe, with the invasion of the IVC and the RA. The authors highlight the importance of recognizing the bizarre presentation of not so rare diseases.
Keywords: Carcinoma, Hepatocellular, Embolism, Liver Neoplasms, Budd-Chiari Syndrome.
CASE REPORT
An 80-year-old hypertensive and diabetic man was admitted complaining of progressive exertional dyspnea over the last 4 months. The physical examination revealed a well-looking patient with blood pressure of 120/70 mmHg and pulse of 90 bpm. The heart examination was unremarkable but on the lungs auscultation the breath sounds were absent over the inferior 2/3 of the right lung. Abdominal examination demonstrated a small volume ascites and the lower limbs were normal. Initial lab workup showed normal hemogram, mildly decreased renal function and hyperkalemia (potassium of 5.8 mEq/L [reference range (RR); 3.7-5.0 mEq/L]). Liver enzymes and gamma glutamyl transferase were normal. Chest radiography showed right-sided pleural effusion. A needle puncture of the thorax drained 1800ml of bloody effusion. Malignant cells were not seen in either cytology studies or pleural biopsy. The transthoracic echocardiogram revealed normal-sized cardiac chambers, normal left ventricular systolic function and normal valves. An unexpected echo dense mass measuring 3.4 X 2.4 cm filled the right atrium and the intrahepatic portion of the inferior vena cava. Thoracic and abdominal contrast-enhanced computed tomography (CT) confirmed the echocardiographic findings and demonstrated ascites, cystic hypo-attenuated areas in the left kidney as well as a heterogeneous mass on the right hepatic lobe (segment VII), obliterating the right hepatic vein (Figure 1). There was splenomegaly. The patient had no clinical history of previous virus-related hepatic disease or alcoholism. While investigating the possible etiologies for the intracardiac mass, the patient developed cardiogenic shock and died.
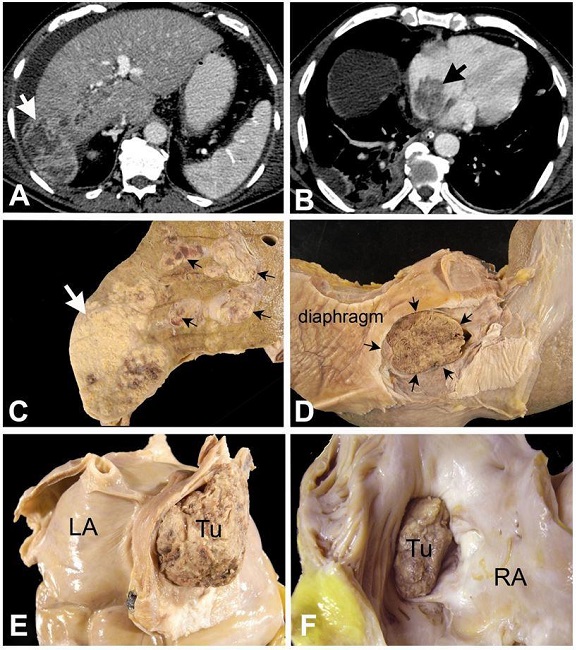
Figure 1
A – Axial contrast enhanced abdominal CT demonstrates a large heterogeneous mass in the periphery of the right hepatic lobe, with exophytic growth (white arrow), associated with tumoral thrombosis of the right portal vein and ascites; B – Axial contrast enhanced CT image obtained at the inferior third of the thorax demonstrates the solid mass into the right atrium (black arrow); C, D, E and F are autopsy images: C – Cross-section of the liver showing the neoplastic mass (white arrow) occupying the right lobe and disseminating via portal and supra-hepatic veins (black arrows); D – Diaphragmatic surface of the liver showing the inferior vena cava completely occluded by tumoral thrombus (black arrows); E – Postero-inferior aspect of the heart showing complete occlusion of the inferior vena cava, at its atrial entrance, by a tumor thrombus (Tu); LA- left atrium; F – Opened right atrium, showing the tumor thrombus invading the right atrial (RA) cavity.
AUTOPSY FINDINGS
The liver weighted 1590 g (RR; 1330-2100 g) and exhibited a poorly-delimited, 7cm-tumour in the right hepatic lobe, with extension to the right hepatic vein and right portal vein branches. Microscopical examination revealed a solid and trabecular neoplasm composed of epithelial cells which resemble hepatocytes with a polygonal shape. Although hepatocellular markers were negative on immunohistochemistry, there was no evidence of non-hepatocellular or cholangiocyte differentiation, supporting the diagnosis of poorly differentiated hepatocellular carcinoma. Both gross and histological evaluation of the liver revealed mild parenchymal fibrosis, mainly portal in location, with few septa, but definitely without cirrhosis. Multiple tumor thrombi invaded the supra-hepatic veins and extended to the inferior vena cava, obliterating it in the form of a massive tumoral thrombus at the right atrial entrance. This tumoral mass protruded into the right atrial cavity (Figure 1). The IVC below the hepatic segment was thrombosed, and generalized visceral edema was present, along with splenomegaly and 2000 ml of ascites. The right lung showed bronchopneumonia, and multiple metastatic nodules of 0,2 to 0,6 cm scattered over both lungs, which on the histologic examination were metastases. The histological analysis of the pleura did not show tumor metastasis. No renal neoplasm was found, but only a cortical and multi-loculated retention cyst measuring 3.5cm in diameter. The heart weight was mildly increased (400g), the pericardium, other chambers, and valves were unremarkable.
Microscopic studies showed a moderately differentiated, predominantly trabecular hepatocarcinoma (Figure 2A). The inferior vena cava thrombus showed neoplastic cells intermingled with fibrin and necrotic debris (Figure 2B). Hematogenous dissemination of HCC was found in both lungs (Figure 2C). Pancreatic steatonecrosis, most likely related to terminal shock (Figure 2D).
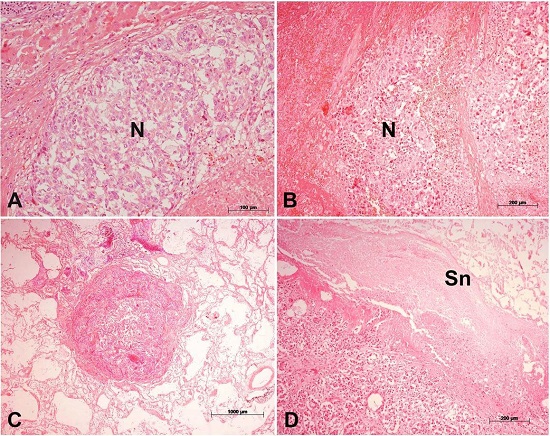
Figure 2
The panel shows some of the microscopic findings. A – Neoplastic nodule (N) in the liver, showing features of hepatocellular carcinoma; B – Tumoral thrombus inside the inferior vena cava. The groups of neoplastic cells (N) are intermingled with necrotic debris and fibrin; C – Small metastatic nodule in the lung parenchyma; D – Acute pancreatitis. There is extensive steatonecrosis (Sn). (H&E; magnification bars: A – 100µm; B – 200 µm; C –1000 µm; D – 200 µm).
DISCUSSION
The hepatocellular carcinoma (HCC) is the fifth most common cancer in men, supposedly because HBV, HCV, and alcohol consumption is more prevalent and possibly more carcinogenic in this population.1-3 Other risk factors associated with HCC include diabetes mellitus (causing nonalcoholic fatty liver disease), tobacco use, hemochromatosis, aflatoxin, pesticides and cirrhosis of any cause.4-6 More than half a million new cases are diagnosed annually worldwide.7 In Brazil the incidence of HCC is varied according to different States of the country, but in São Paulo it is 2.07/100,000 individuals, while in sub-Saharan Africa this rate reaches 20/100,000.1,7
HCC usually occurs in patients with cirrhosis, and therefore, in most of the cases, the presentation is that of chronic liver disease with portal hypertension.8 Symptoms are nonspecific and generally ensue in the setting of an advanced disease including (i) abdominal pain, (ii) abdominal distension, (iii) loss of appetite, (iv) jaundice, (v) ascites, (vi) weight loss, (vii) signs of encephalopathy and (viii) digestive bleeding.7
The affinity of the HCC for the vasculature, exhibiting intravascular growth, was already reported, with the invasion of the portal system. Invasion of the inferior vena cava is reported in as many as 6% of the cases.9-13 Renal carcinoma is the most common neoplasm to invade the IVC, but renal angiomyolipoma, pheochromocytoma, adrenocortical malignant neoplasms are also known to potentially extend to the vena cava and right atrium.14-16 Along with the IVC infiltration, the extensive growth of HCC into the right atrium is even rarer and is generally diagnosed only at autopsy. In recent years, improvement of the diagnostic imaging examination has led to cases similar to ours being reported.12,15,17-23 In 2004, Chang et al.24 published a study of the English-language publications on HCC with invasion of the IVC extending to the RA and found 30 cases, including their 3 cases.
Suspicion of the diagnosis is associated with elevated serum alpha fetoprotein values and imaging examinations. In patients with advanced disease, the echocardiography is useful for detecting IVC and right atrium invasions.10 Transesophageal echocardiography provides more accurate information about the location of the mass in relation to atrial wall, tricuspid valve and superior and IVC.25,26 Right atrial tumor mass may be asymptomatic.19 However, the clinical presentation of involvement of the IVC and RA is usually in the form of the Budd-Chiari syndrome, and cardiogenic shock may result from ball obstruction of the tricuspid valve, right heart failure or complete obstruction of cardiac filling as shown here. Pulmonary emboli and sudden death are also reported.12,27 Differential diagnosis includes a bland thrombus of the IVC, and tumor thrombus is usually continuous with the primary neoplasm with expansion of the vena cava.
Surgical thrombectomy is the main therapeutically option but, radiofrequency ablation, chemoembolization, thalidomide, and sorafenib have also been described as feasible alternatives for advanced hepatocellular carcinoma, despite the high mortality rate within a couple of months whether the patient is treated or not.19,26,28
In our case, one would expect that an octogenarian with diabetes mellitus and hypertension with progressive dyspnea and a large right thorax pleural effusion, would be diagnosed with heart failure of ischemic, hypertensive or both origins, even in the absence of previous chest pain or a history of vascular disease complaints. Our case presents some peculiar clinical features. The HCC reached an advanced stage and a large primary tumor without symptoms and without cirrhosis. As the patient was diabetic and the incidence of non-alcoholic fatty liver disease reached 40% of the population over 70 year, this finding could suggest that the HCC may have arisen in the context of this disease.29
The bloody pleural effusion, responsible for the progressive dyspnea, was interpreted as a result of impaired venous drainage of the right thorax and not due to myocardial disease or by any subpleural pulmonary neoplastic embolism.
The immediate cause of death was shock due to the obstruction of the right atrium by the tumor thrombus.
REFERENCES
Mittal S, El-Serag HB. Epidemiology of HCC: consider the population. J Clin Gastroenterol. 2013;47:S2-6. http://dx.doi.org/10.1097/MCG.0b013e3182872f29. PMid: 23632345
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61(2):69-90. http://dx.doi.org/10.3322/caac.20107. PMid: 21296855
Ferlay J, Parkin DM, Steliarova-Foucher E. Estimates of cancer incidence and mortality in Europe in 2008. Eur J Cancer. 2010;46(4):765-81. http://dx.doi.org/10.1016/j.ejca.2009.12.014. PMid: 20116997
Cabibbo G, Maida M, Genco C, Antonucci M, Cammà C. Causes of and prevention strategies for hepatocellular carcinoma. Semin Oncol. 2012;39(4):374-83. http://dx.doi.org/10.1053/j.seminoncol.2012.05.006. PMid: 22846856
Hamed MA, Ali SA. Non-viral factors contributing to hepatocellular carcinoma. World J Hepatol. 2013;5(6):311-22. http://dx.doi.org/10.4254/wjh.v5.i6.311. PMid: 23805355
Blonski W, Kotlyar DS, Forde KA. Non-viral causes of hepatocellular carcinoma. World J Gastroenterol. 2010;16(29):3603-15. http://dx.doi.org/10.3748/wjg.v16.i29.3603. PMid: 20677332
Gomes MA, Priolli DG, Tralhão JG, Botelho MF. Carcinoma hepatocelular: epidemiologia, biologia, diagnostico e terapias. Rev Assoc Med Bras. 2013;59(5):514-24. http://dx.doi.org/10.1016/j.ramb.2013.03.005. PMid: 24041910
Colombo M, de Franchis R, Del Ninno E, et al. Hepatocellular carcinoma in Italian patients with cirrhosis. N Engl J Med. 1991;325(10):675-80. http://dx.doi.org/10.1056/NEJM199109053251002. PMid: 1651452
Lee IJ, Chung JW, Kim HC, et al. Extrahepatic collateral arterial supply to the tumor thrombi of hepatocellular carcinoma involving inferior vena cava: the prevalence and determinant factors. J Vasc Interv Radiol. 2009;20(1):22-9. http://dx.doi.org/10.1016/j.jvir.2008.09.030. PMid: 19026566
Vallakati A, Chandra PA, Frankel R, Shani J. Intra-atrial tumor thrombi secondary to hepatocellular carcinoma responding to chemotherapy. N Am J Med Sci. 2011;3(9):435-7. http://dx.doi.org/10.4297/najms.2011.3435. PMid: 22362455
Kim SU, Kim YR, Kim DY, et al. Clinical features and treatment outcome of advanced hepatocellular carcinoma with inferior vena caval invasion or atrial tumor thrombus. Korean J Hepatol. 2007;13(3):387-95. http://dx.doi.org/10.3350/kjhep.2007.13.3.387. PMid: 17898555
Pellicelli AM, Barba J, Gomez AJ, Borgia MC. Echocardiographic follow-up of right atrial tumoral invasion by hepatocarcinoma: a case report. Cardiologia. 1992;37(2):151-3. PMid: 1318163
Fleming CJ, Andrews JC, Wiseman GA, Gansen DN, Roberts LR. Hepatic vein tumor thrombus as a risk factor for excessive pulmonary deposition of microspheres during TheraSphere therapy for unresectable hepatocellular carcinoma. J Vasc Interv Radiol. 2009;20(11):1460-3. http://dx.doi.org/10.1016/j.jvir.2009.07.033. PMid: 19875064
Lawindy SM, Kurian T, Kim T, et al. Important surgical considerations in the management of renal cell carcinoma (RCC) with inferior vena cava (IVC) tumour thrombus. BJU Int. 2012;110(7):926-39. http://dx.doi.org/10.1111/j.1464-410X.2012.11174.x. PMid: 22540179
Lazaros G, Samara C, Nikolakopoulou Z, Tassopoulos N. Growth of hepatocellular carcinoma into the right atrium. A case of antemortem diagnosis with magnetic resonance imaging of the heart. Acta Cardiol. 2003;58(6):563-5. http://dx.doi.org/10.2143/AC.58.6.2005323. PMid: 14713184
Molina M, Schiappacasse G, Labra A. Tumors that invade the inferior vena cava: an illustrative review of the main imaging features on computed tomography and magnetic resonance. Rev Chil Radiol. 2016;22:39-46. http://dx.doi.org/10.1016/j.rchira.2016.02.005.
Masahiro N, Masue M, Hirofumi Y, Setsu S. A case of an intra-atrial tumour thrombus from hepatocellular carcinoma (HCC), first indicated by 67 Ga-citrate scintigraphy. Nucl Med (Stuttg). 1995;9:39-42.
Vallakati A, Chandra PA, Frankel R, Shani J. Intra-atrial tumour thrombi secondary to hepatoellular carcinoma responding to chemotherapy. N Am J Med Sci. 2011;3(9):435-7. PMid: 22362455
Pandya H, Shah C, Lakhani J, Patel M. Intra-atrial tumor thrombus secondary to hepatocellular carcinoma. Australas Med J. 2013;6(6):321-4. http://dx.doi.org/10.4066/AMJ.2013.1635. PMid: 23837079
Martínez Baca-López F, Ramírez-Arias E, Rayas-Gómez AL, Bernal-Ruiz EA, Saturno-Chiu G. Hepatocellular carcinoma with invasion into right cardiac cavities: report of a case and literature review. J Am Soc Echocardiogr. 2004;17(2):192-4. http://dx.doi.org/10.1016/j.echo.2003.10.007. PMid: 14752497
Hanada K, Fukui H, Tamura M, Tsujii T. An autopsy case of encapsulated hepatocellular carcinoma with its metastasis into right atrium and pericardium. Kan Tan Sui. 1986;12:471-5.
Chandra P, Shah S, Purandare N, Agrawal A, Rangarajan V. Hepatocellular carcinoma with intra-atrial tumor extension identified on 99mTc-labeled macroaggregated albumin single photon emission computed tomography/computed tomography. Indian J Nucl Med. 2016;31(4):311-2. http://dx.doi.org/10.4103/0972-3919.187449. PMid: 27833324
Demir K, Akyüz F, Poyanli A, Okten A. An asymptomatic huge hepatocellular carcinoma with intra-atrial tumor thrombus in a patient with chronic hepatitis B viral infection. Turk J Gastroenterol. 2006;17(3):203-5. PMid: 16941254
Chang JY, Ka WS, Chao TY, Liu TW, Chuang TR, Chen LT. Hepatocellular carcinoma with intra-atrial tumor thrombi. Oncology. 2004;67(3-4):320-6. http://dx.doi.org/10.1159/000081333. PMid: 15557794
Yoshitomi Y, Kojima S, Sugi T, et al. Echocardiography of a right atrial mass in hepatocellular carcinoma. Heart Vessels. 1998;13(1):45-8. http://dx.doi.org/10.1007/BF02750643. PMid: 9923565
Tse HF, Lau CP, Lau YK, Lai CL. Transesophageal echocardiography in the detection of inferior vena cava and cardiac metastasis in hepatocellular carcinoma. Clin Cardiol. 1996;19(3):211-3. http://dx.doi.org/10.1002/clc.4960190314. PMid: 8674258
Martínez Baca-López F, Ramírez-Arias E, Rayas-Gómez AL, Bernal-Ruiz EA, Saturno-Chiu G. Hepatocellular carcinoma with invasion into right cardiac cavities: report of a case and literature review. J Am Soc Echocardiogr. 2004;17(2):192-4. http://dx.doi.org/10.1016/j.echo.2003.10.007. PMid: 14752497
Sun JH, Zhang YL, Nie CH, et al. Long-term survival after chemoembolization of metastatic right atrial tumor thrombus as a presenting feature of hepatocellular carcinoma: a case study. Oncol Lett. 2012;3(5):975-7. http://dx.doi.org/10.3892/ol.2012.618. PMid: 22783375
Gan L, Chitturi S, Farrell GC. Mechanisms and Implications of Age-Related Changes in the Liver: Nonalcoholic Fatty Liver Disease in the Elderly. Curr Gerontol Geriatr Res. 2011;2011:831536. http://dx.doi.org/10.1155/2011/831536. PMid: 21918648
Notes
Author notes
Correspondence Vera Demarchi Aiello Laboratory of Pathology – Heart Institute (InCor) – Medical School – Universidade de São Paulo (USP) Av. Dr. Enéas C. Aguiar, 44 – São Paulo/SP – Brazil CEP: 05403-000 Phone: + 55 (11) 2661-5252; Fax: +55 (11) 2661-5279 vera.aiello@incor.usp.br
Conflict of interest declaration
