Article / Autopsy Case Report
Gonococcal endocarditis: an ever-present threat
Gonococcal endocarditis: an ever-present threat
Autopsy and Case Reports, vol. 6, no. 2, pp. 19-25, 2016
Hospital Universitário da Universidade de São Paulo
Received: 03 May 2016
Accepted: 02 June 2016
Abstract: The incidence of severe complications of the Neisseria gonorrhoeae infection has presented variations over recent decades since the advent of penicillin. Gonococcal endocarditis (GE) still remains an ever-present threat afflicting the society’s poor and sexually active young population. This entity frequently requires surgical intervention and usually exhibits a poor outcome. The interval between the onset of symptoms and the diagnosis does not usually exceed 4 weeks. One of the characteristics of GE is a proclivity for aortic valve involvement with large vegetation and valve ring abscess formation. The authors report the case of a young man with a 2-week history of fever, malaise, weakness, and progressive heart failure symptoms, who had no previous history of genital complaints or cardiopathy. The physical examination was consistent with acute aortic insufficiency, which was most probably of an infectious origin. The echocardiogram showed thickened aortic cusps and valve insufficiency. After hospital admission, the patient’s clinical status worsened rapidly and he died on the second day. The autopsy findings disclosed aortic valve destruction with vegetation and a ring abscess besides signs of septic shock, such as diffuse alveolar damage, acute tubular necrosis, and zone 3 hepatocellular necrosis. The blood culture isolated N. gonorrhoeae resistant to penicillin and ciprofloxacin. The authors call attention to the pathogen of this particular infectious endocarditis, and the need for early diagnosis and evaluation by a cardiac surgery team.
Keywords: Endocarditis, Bacterial, Neisseria gonorrhoeae, Aortic valve, Autopsy.
CASE REPORT
A 28-year-old male patient sought the emergency facility complaining of fever over the previous 2 weeks accompanied by headache and malaise. Up to this point, he had been treated with an antipyretic. Three days earlier, he began feeling retrosternal pain radiating to the back, which progressed to palpitations, dry cough, nausea, vomiting, progressive dyspnea, orthopnea, and lower limbs edema before he attended the hospital. He denied previous diseases, regular use of medications, alcohol consumption, tobacco smoking, or use of illicit drugs. Retrospectively, we could not obtain any evidence of promiscuous sexual activity or a history of urethral complaint. He worked in a marble factory and was used to heavy-lifting work without physical constraint.
The physical examination revealed an ill-looking patient who was tachypneic, tachycardic, and febrile (38.3°C), with 120/40 mmHg blood pressure and 86% room air oximetry. His skin, mucosae, and conjunctivae examinations were normal. An auscultation of his heart revealed the presence of an intermittent third heart sound, plus an ejection murmur radiating to the carotids, and a diastolic murmur—the latter two at the base of the heart. Pulmonary auscultation revealed decreased sounds in the right base. The abdominal examination revealed a mildly enlarged liver, and the lower limbs exhibited bilateral edema. Laboratory examinations showed a marked leukocytosis 22,800 leukocytes/mm3 (reference value [RV]: 4.4-11.3 × 103/mm3). There were no other abnormalities in the remaining blood cell count and the biochemical work-up. Urinalysis showed sterile leukocyturia (20,000 leukocytes/mm3). HIV and syphilis serology were negative. The electrocardiogram was normal; the plain thoracic X-ray showed a normal cardiac silhouette and an alveolar space-filling opacity in both inferior pulmonary lobes accompanied by a tiny pleural effusion on the right side (Figure 1). The transthoracic echocardiogram revealed normal-sized cardiac chambers as well as normal septum and posterior wall thickness, preserved systolic function, and aortic valve thickening with marked diastolic reflux. The patient was hospitalized and an empiric antibiotic regimen comprising vancomycin, gentamicin, and ceftriaxone was started because of the suspected diagnosis of acute infective endocarditis. The outcome was unfavorable with severe hypotension and persistent respiratory failure requiring vasoactive drugs infusion and non-invasive respiratory support, which meant that the transesophageal echocardiography could not be performed. On the second day of hospitalization, the patient presented cardiac arrest and was unresponsive to the advanced cardio life support maneuvers. The autopsy was performed after informed consent by the family.
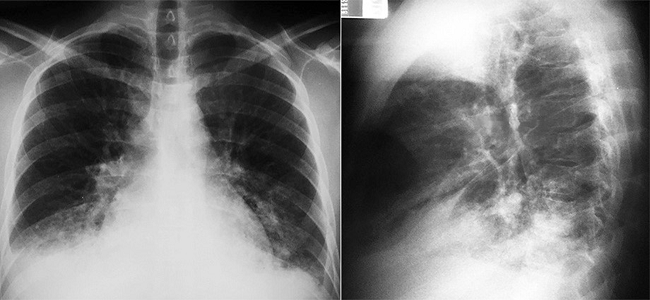
Figure 1
Plain thoracic x-ray showing a normal cardiac silhouette and a heterogeneous opacity in the right inferior pulmonary lobe with a small ipsilateral pleural effusion.
On the day following the patient's death, one out of three blood cultures was positive for Neisseria gonorrhoeae. Bacterial identification was performed using VITEK-2 compact (version 07.01, BioMérrieux, France) and confirmed by manual biochemical tests.1 The isolate was resistant to penicillin and ciprofloxacin, but was susceptible to ceftriaxone. Antibiotic susceptibility was determined by disk diffusion according to Clinical and Laboratory Standards Institute guidelines.2
AUTOPSY FINDINGS
At the opening of the pericardial cavity, moderate yellowish serous pericardial fluid was observed. The heart weighed 392 g (mean RV [mRV]: 327 g), and both ventricles were dilated with mild thickening of the left ventricular wall (Figure 2A). The aortic valve had three cusps and was conspicuously damaged by the formation of vegetation (Figure 2B). The left coronary cusp was perforated and the right coronary cusp was copllapsed. The myocardium and the aortic valve ring's contiguous endocardium showed an extensive area of necrosis, which was identified on gross examination as a whitish area in a valve ring abscess (Figure 2C and Figure 2D).
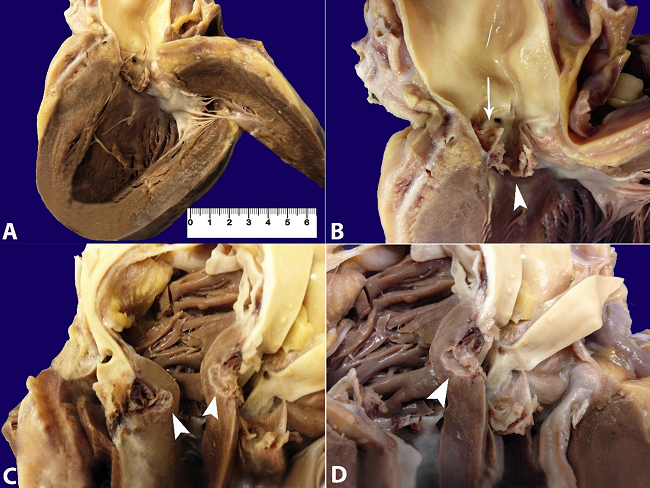
Figure 2
Gross examination of the heart. A - Hypertrophy of the left ventricular wall and collapsed aortic valve cusps; B - Detail of the aortic valve showing one preserved posterior noncoronary cusp and complete destruction of the two coronary cusps. The left coronary cusp is perforated (arrow) and the other showed vegetation (arrowhead); C and D - Valve ring abscess (arrowhead).
Histologically, the valve was replaced by a loose fibrous connective tissue, fibrin, and neutrophils, which were identified on the histological examination (Figure 3A and 3B). The histologic examination of this lesion showed a marked diffuse inflammatory infiltrate predominantly composed of polymorphonuclear neutrophils that extended to the myocardial fibers and the pericardial adipose tissue, which is consistent with a valve ring abscess (Figure 3A, 3B, and 3C). Gram-negative diplococci were found upon Brown-Hopps staining (Figure 3D). The other three valves were enlarged without vegetation or any other lesion.
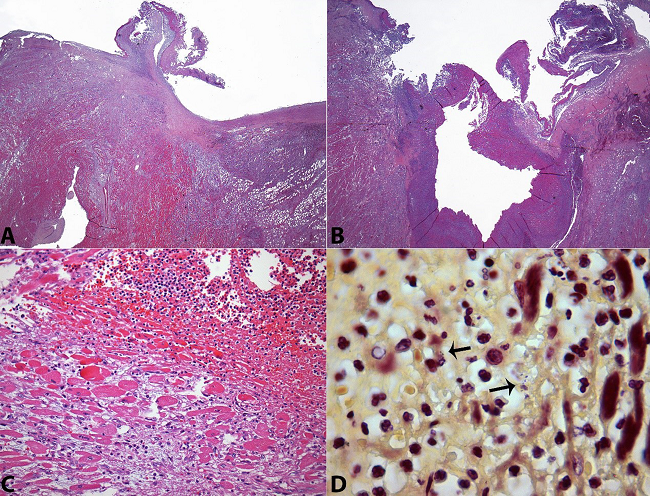
Figure 3
Photomicrography of the valve and ring abscess. A - Panoramic view of the left coronary cusp with a deposition of fibrin and inflammatory exsudate. Note the extensive inflammatory infiltration surrounding the cusps’ implantation (H&E, 12.5X); B - Panoramic view of the valve ring abscess (H&E, 12.5X); C - Acute inflammation of the surrounding myocardium (H&E, 200X); D - Gram-negative diplococci within the abscess (arrows) (Brown-Hopps, 1000X).
Terminal brain congestion and edema (1397 g, mRV: 1365 g) were observed. The lungs (right = 470 g, left = 395 g; mRV: 450 g and 375 g, respectively) showed congestion, edema, and diffuse aggregates of intra-alveolar histiocytes, which sometimes contained hemosiderin or erythrocytes (hemophagocytosis due to sepsis), areas of diffuse alveolar damage with fibrin deposition, and reactive pneumocytes (acute respiratory distress). The liver weighed 1970 g (mRV: 1700 g) and showed diffuse congestion of the sinusoids with pericentral ischemic necrotic foci (shock), and mild portal inflammatory infiltration by lymphocytes and monocytes consistent with reactive hepatitis. The gallbladder, the pancreas, and the gastrointestinal system showed no significant changes.
The lymph nodes and the spleen (180 g, mRV: 112g) were congested with diffusely distributed lymphoid follicles, occasional sinusoidal neutrophils, and focal histiocytes containing erythrocytes (hemophagocytosis related to sepsis). The bone marrow showed preserved maturation of the three hematopoietic series with mild granulocytic reactivity. The kidneys (right = 158 g, left = 149 g; mRV for both: 313 g) showed congestion and acute tubular necrosis (due to shock). Both adrenal glands (right = 5 g, left = 4.5 g; mRV for both: 11.5 g) showed cortical foci of necrosis (adrenal stress due to sepsis and shock). The periadrenal adipose tissue showed a fibrin embolus into a medium caliber artery that was interpreted as due to the sepsis’ intravascular coagulation or may have the origin from the aortic vegetation. The urogenital tract was preserved except for the presence of mild chronic mononuclear inflammatory infiltration in the prostatic urethra and in the bladder.
DISCUSSION
Gonorrhea has remained a global public health problem since ancient times, mostly afflicting society’s poor and sexually active young population and newborns. The incidence is estimated at 600,000 cases annually in the USA.3 In the history of medicine, although it has taken a long time to achieve an efficient treatment for gonorrhea, the control of the disease has been difficult due to the significant number of asymptomatic cases. In 1879, Albert Ludwig Sigesmund Neisser, a German physician and bacteriologist studying patients with urethritis and ophthalmia neonatorum, discovered N. gonorrhoeae, a fastidious Gram-negative diplococcus, which was further proved to be an exclusively human pathogen. The gonococcal infection has a diverse clinical presentation varying from asymptomatic cases, symptomatic infection restricted to the genital tract, pharynx, or rectum, and disseminated infection, which mostly involves the skin and the joints. Although much rarer, other sites may be involved in the disseminated form, such as the heart, meninges, bones, and liver.4,5 According to Albert Newman’s publication of 1933,6 the Ricord’s Treatise on Venereal Disease (1838) was the first publication showing that the gonorrheal infection could disseminate systemically and involve the heart, but the isolation of N. gonorrhoeae from a patient with endocarditis was held by Thayer and Blumer7 in 1895.
In the pre-antibiotic era (i.e. before 1938), gonorrhea was responsible for up to 26% of all bacterial endocarditis8-12 and was almost uniformly fatal.8,13 Since then, the number of endocarditis infections substantially declined, although gonorrhea remained among the four most frequent sexually transmitted diseases worldwide.14 The incidence of gonococcal endocarditis (GE) substantially decreased after the advent of penicillin.8 In parallel, a better outcome was observed, with an 88.5% survival rate (in a report of 26 patients treated exclusively with antibiotics).13 For uncertain reasons many cases occurred during the last three decades of the 20th century, but numbers decreased again in the first decade of the 21st century.13 However, surprisingly, GE currently seems to be showing a resurgence.8
Up to 70 cases of GE were published from 1939 to 2014, 38 of them within the period from 1980 to 2014.13,15 Based on these reviews, GE is characterized by the involvement of a young population, afflicting, in 85% of the cases, patients between 15 and 35 years old, with a slight predominance among males.8,12 GE usually develops in native and intact valves, presenting large vegetation that involves the aortic valve in 50-72% of the cases, accompanied by ring abscesses.8 The primary gonococcal disease or related urinary symptoms occur in 21-27% of the cases, concomitantly or months before the diagnosis of endocarditis.13,15,16
The marked valvular destruction observed in GE explains the high incidence of surgical intervention, which has been reported to be undertaken in 58-72% of the cases; unlike the endocarditis caused by other pathogens.15,17 Although the availability of new-generation antibiotics and refined cardiac surgery techniques, the mortality of GE remains high. The mortality rate was 23% in the review of Ramos et al.15 involving 38 cases diagnosed from 1980 to 2014.
Unfortunately, the clinical picture of GE does not enable its differentiation from other endocarditis. However, some characteristics may call attention to this entity, which include: (i) the time elapsed from the initial symptoms until the diagnosis is intermediate (nearly 4 weeks) between the formerly called sub-acute endocarditis, typically caused by streptococci, and the acute staphylococcal endocarditis9; (ii) young patients; (iii) native and intact valves; (iv) proclivity for aortic valve involvement; and (v) drastic evolution with acute heart failure. Signs and symptoms almost invariably include the presence of fever, cardiac murmur, tachycardia, and signs or symptoms of heart failure. Eventually, arthritis, embolization phenomena, and splenomegaly may be present. A laboratory work-up is not diagnostic, but it may reveal anemia, leukocytosis, and an increase in acute phase proteins. Echocardiography—either transthoracic or transesophageal—almost inevitably renders the presence of vegetation or show valvular thickening and dysfunction. In our case, indeed surprisingly, despite the huge size of the vegetation and the leaflets destruction, the transthoracic echocardiography was of little help for the diagnosis. Blood cultures are positive in more than 90% of cases, which is notably different from other forms of disseminated gonococcal infection where blood cultures show the positivity ranging between 10% and 30%.8
The development of disseminated gonococcal disease occurs mostly in asymptomatic infection and is associated with host susceptibility that includes complement deficiency, systemic lupus erythematosus, HIV infection, and peculiarities of some strains, such as (i) nutritional requirements (arginine-hypoxanthine-uracil growth requirement); (ii) the absence of the surface Protein II complex (generally associated with chromosomal mutation [penA mtr]), which confers more resistance to neutrophil-mediated killing and less intergonococcal adherence; and (iii) the expression of surface protein IA, which confers serum resistance and easier translocation into bilayer membranes.8,18
Our patient fulfilled the epidemiological characteristics of GE and presented a 2-week history of fever. Apparently, symptomatic gonococcal urethritis was not present at the time of the endocarditis diagnosis, since genital complaints were not referred. However, the urinalysis showed a sterile leukocyturia, which could suggest the presence of asymptomatic urethritis. Moreover, although non-specific, the prostatic urethra also showed an inflammatory infiltration, which could raise suspicion of the pathogen’s port of entry. On admission, the diagnosis of infective endocarditis involving the aortic valve was not troublesome. Besides, the apparent recent-onset heart failure symptoms in a previously healthy young patient, the long-term fever, the presence of murmurs at the base of the heart, the wide pulse pressure, the normal cardiac silhouette on thoracic radiography, and the normal electrocardiogram favored the diagnosis of acute aortic valve insufficiency, most probably of infectious origin. Although the transthoracic echocardiogram could not depict aortic vegetation, it confirmed the aortic valve insufficiency with morphological abnormality (thickening). The empirical antibiotic therapy was reasonable for a wide range of pathogens, but, unfortunately, as shown in the literature, the outcome was rapidly fatal. The patient did not have the chance to be evaluated by a cardiac surgery team because of the high risk of removal to a cardiology center and the rapid progression to death. We dare consider that even if this patient had been operated on, the outcome would have likely been the same because of the presence of the large ring abscess depicted at autopsy. Although the etiological diagnosis was done postmortem, the antibiotic regimen choice was appropriate ensuring that it was not the limiting factor for survival. In view of the autopsy findings, our opinion is that the chance of this patient’s survival was lost when the diagnosis could not be concluded during the initial consultations.
Acknowledgements
The authors are thankful to the Microbiology unit of the Central Laboratory of University Hospital of São Paulo for gonococcal isolation and susceptibility tests as well as to Conceição Zanelato and Maria Vaneide de Paiva for assistance in microbiological tests at Adolfo Lutz Institute
REFERENCES
1 Janda WM, Knapp J. Neisseria and Moraxella catarrhalis. In: Murray PR, Baron EJ, Jorgensen JH, Landry ML, Pfaller MA, editors. Manual of clinical microbiology. 8th ed. Washington, DC: ASM Press; 2003. chap. 38.
2 Clinical and Laboratory Standards Institute (CLSI). Performance standards for antimicrobial disk susceptibility tests, approved standard. 12th ed. Wayne, PA: Clinical and Laboratory Standards Institute; 2015. CLSI document M02-A12.
3 Bolan GA, Sparling PF, Wasserheit JN. The emerging threat of untreatable gonococcal infection. N Engl J Med. 2012;366(6):485-7. http://dx.doi.org/10.1056/NEJMp1112456. PMid:22316442
4 Thompson EC, Brantley D. Gonococcal endocarditis. J Natl Med Assoc. 1996;88(6):353-6. PMid:8691495
5 Holmes KK, Counts GW, Beaty HN. Disseminated gonococcal infection. Ann Intern Med. 1971;74(6):979-93. http://dx.doi.org/10.7326/0003-4819-74-6-979. PMid:4996345
6 Newman AB. The prognosis in gonococcal endocaditis: Review of literature and report of case with spontaneous recovery. Am Heart J. 1933;8(6):821-33. http://dx.doi.org/10.1016/S0002-8703(33)90144-1.
7 Thayer WS, Blumer G. Ulcerative endocarditis due to the gonococcus: gonorrheal spticemia. Bull Johns Hopkins Hosp. 1896;7:57-63.
8 Jackman JD Jr, Glamann DB. Gonococcal endocarditis: twenty-five year experience. Am J Med Sci. 1991;301(3):221-30. http://dx.doi.org/10.1097/00000441-199103000-00012. PMid:2000895
9 Thayer WS. Bacterial or infective endocarditis: the Gibson lectures of 1930. Edinburgh Med J. 1931;38:237-65, 297-329.
10 Stone E. Gonorrheal endocarditis. J Urol. 1934;31:869-95.
11 Williams RH. Gonococcic endocarditis. A study of twelve cases with postmorten examinations. Arch Intern Med (Chic). 1938;61(1):26-38. http://dx.doi.org/10.1001/archinte.1938.00180070031003
12 Wall TC, Peyton RB, Corey GR. Gonococcal endocarditis: a new look at an old disease. Medicine (Baltimore). 1989;68(6):375-80. http://dx.doi.org/10.1097/00005792-198911000-00005. PMid:2509857
13 Nie S, Wu Y, Huang L, Pincus D, Tang YW, Lu X. Gonococcal endocarditis: a case report and literature review. Eur J Clin Microbiol Infect Dis. 2014;33(1):23-7. http://dx.doi.org/10.1007/s10096-013-1921-x. PMid:23856883
14 World Health Organization (WHO). Sexual and reproductive health [Internet]. 2016 [cited 2016 Mar 27]. Available from: http://www.who.int/reproductivehealth/news/stis-estimates-2015/en/
15 Ramos A, García-Pavía P, Orden B, et al. Gonococcal endocarditis: a case report ans review of the literature. Infection. 2014;42(2):425-8. http://dx.doi.org/10.1007/s15010-013-0541-9. PMid:24163221
16 Romero Tarín E, González Antuña M, Calzón Díaz S, Riesgo Miranda MA. Gonococcal endocarditis. Rev Esp Cardiol. 1987;40(4):293-6. PMid:3659524
17 Prendergast BD, Tornos P. Surgery for infective endocarditis: who and when? Circulation. 2010;121(9):1141-52. http://dx.doi.org/10.1161/CIRCULATIONAHA.108.773598. PMid:20212293
18 Britigan BE, Cohen MS, Sparling PF. Gonococcal infection: a model of molecular pathogenesis. N Engl J Med. 1985;312(26):1683-94. http://dx.doi.org/10.1056/NEJM198506273122606. PMid:2860565
Notes
Author notes
Correspondence Fernando Peixoto Ferraz de Campos Internal Medicine Division - Hospital Universitário - Universidade de São Paulo (USP) Av. Professor Lineu Prestes, 2565 - Butantã - São Paulo/SP - Brazil CEP: 05508-000 Phone: +55 (11) 3091-9275 E-mail: fpfcampos@gmail.com
Conflict of interest declaration