Article / Autopsy Case Report
Gastrointestinal cytomegalovirus disease in a patient with pemphigus vulgaris treated with corticosteroid and mycophenolate mofetil
Gastrointestinal cytomegalovirus disease in a patient with pemphigus vulgaris treated with corticosteroid and mycophenolate mofetil
Autopsy and Case Reports, vol. 7, no. 1, pp. 23-30, 2017
Hospital Universitário da Universidade de São Paulo
Received: 23 January 2017
Accepted: 01 March 2017
ABSTRACT: Pemphigus vulgaris is an autoimmune disease characterized by the formation of suprabasal intra-epidermal blisters on the skin and mucosal surfaces. Infectious diseases are the main cause of death in patients with pemphigus due to the disrupture of the physiological skin barrier, immune dysregulation, and the use of immunosuppressive medications leaving the patient prone to acquire opportunistic infections. We report the case of a 67-year-old woman diagnosed with pemphigus vulgaris, who was irregularly taking prednisone and mycophenolate mofetil. She was hospitalized because of a 1-month history of watery diarrhea and oral ulcers. Unfortunately, the patient died suddenly on the ward. The autopsy revealed a bilateral saddle pulmonary embolism, Gram-positive cocci bronchopneumonia, and gastrointestinal cytomegalovirus infection, causing extensive gastrointestinal mucosal ulcers.
Keywords: Autopsy, Cytomegalovirus, Opportunistic Infections, Pemphigus.
CASE REPORT
A 67-year-old woman sought the emergency department complaining of a 1-month history of watery diarrhea without mucus or blood, followed by the appearance of lesions in the oral cavity with odynophagia over the last 2 weeks. Despite 10 days of treatment with metronidazole in an outpatient clinic due to the presence of Blastocystis hominis in the stool examination, diarrhea persisted.
The patient had a medical history of pemphigus vulgaris and recurrent episodes of severe major depression. After histopathological confirmation of pemphigus vulgaris, which was initially diagnosed more than 10 years ago, the patient had been followed-up in the dermatologic clinic. Over the last 7 years, she had been hospitalized several times to compensate the pemphigus and the depression. It should be added that she was used to inducing self-scarification of her skin lesions, and had significant problems with treatment compliance (i.e. she did not attend the appointments and refused to properly treat her psychiatric condition). At the time of her last hospitalization, she was irregularly using prednisone 60 mg/day and mycophenolate mofetil (MMF) 3 g/day.
In the emergency room, the physical examination revealed an emaciated patient with erosions covered by hematic crusts on the face, without signs of secondary infection. She also had an ulcer on the dorsal surface of the tongue and some erosions covered with a whitish exudate on the oral mucosa. Due to the diagnostic hypothesis of oral and esophageal candidiasis, the patient was treated with fluconazole with partial relief of the odynophagia. Abnormal laboratory findings included normocytic and normochromic anemia, lymphopenia (530 cells/mm3 (reference value [RV]: 900-3400 cells/mm3), mildly elevated pancreatic enzymes (amylase and lipase), and leukocyturia. Based on the urinalysis result, ciprofloxacin was prescribed; and a urine culture further identified negative-coagulase Staphylococcus sp. The chest x-ray was normal.
On the fourth day of hospitalization, an upper digestive endoscopy evidenced extensive erosive esophagitis, and gastric antral erosions; biopsies were performed. The pathological report was released post mortem. After 6 days of hospitalization, the patient had a sudden cardiac arrest in pulseless electrical activity, and our rapid response team (“blue code”) attempted resuscitation. Unfortunately, the patient did not respond to the standard resuscitation maneuvers. An autopsy was performed with the consent of her relatives.
AUTOPSY FINDINGS
The corpse weighed 42.0 kg and measured 1.60 m (body mass index = 16.4 kg/m2). The ectoscopy revealed an emaciated female corpse. Erosions with fibrin-hematic crusts were observed bilaterally on the malar skin (Figure 1).

Figure 1
Pemphigus vulgaris facial lesions: erosions with fibrin-hematic crusts on the malar region
At the opening of the cavities, no abnormalities were found. There was an organized thrombus in the right atrium extending to the tricuspid valve (Figure 2A). The right lung weighed 744.0 g (mean RV = 450 g) and the left weighed 686.0 g (mRV = 375 g). Both lungs had a massive saddle pulmonary embolism, which extended to the peripheral branches. The cut surface of the lungs showed marked congestion, draining a foamy, pinkish liquid when compressed, with firm consistency in the basal regions (Figure 2B). At microscopy, the lung parenchyma had extensive areas of bronchopneumonia caused by Gram-positive cocci associated with diffuse alveolar damage (congestion, alveolar edema and hemorrhage, and hyaline membranes) and numerous arterial thrombi (Figure 2C, D).
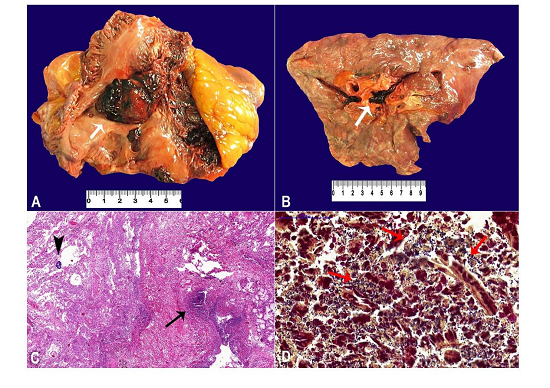
Figure 2
A - Gross examination of the heart showing an organized thrombus in the right atrium (arrow); B - Saddle pulmonary thromboembolism in the right lung (arrow); C - Micrography of the lungs showing suppurative bronchopneumonia (arrow) associated with colonies of cocci (arrowhead) (H&E, 55X); D - Gram-positive cocci in the areas of suppurative pneumonia (arrows) (Brown-Brenn, 400X).
An extensive ulcer, measuring 2.5 cm in diameter, was found on the tongue’s dorsal surface (Figure 3A), which histopathologically corresponded to an ischemic ulcer with ongoing regeneration, neutrophilic inflammatory infiltrate, granulation tissue, and reepithelization (Figure 3B). Within the surrounding areas of the ulcer there were multiple inflammatory, stromal, and endothelial cells presenting cytomegaly and intranuclear eosinophilic inclusion, surrounded by a clear halo, which was consistent with cytomegalovirus infection (Figure 3B).
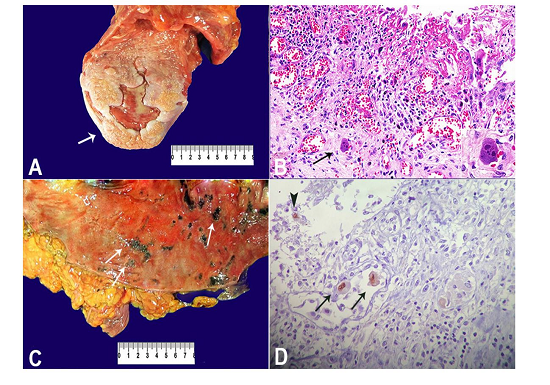
Figure 3
A - Gross examination of the tongue depicting an ulcer on the dorsum (arrow); B - Micrography of the tongue showing mucosal ulcer with endothelial cells exhibiting cytomegalic cytopathic effect (arrow and inset) (H&E, 300X); C - Macroscopic examination of the colonic mucosa exhibiting small ulcers (arrows); D - Micrography of the colon showing a positive immunohistochemistry reaction for cytomegalovirus (CMV) antigens, detecting cytomegalic endothelial (arrow) and inflammatory (arrow head) cells in a colonic ulcer, with nuclear staining (mouse anti-CMV cocktail of DDG9 and CCH2, Diagnostic BioSystems, 400X).
The esophagus presented several small erosions, and the stomach had ulcers with punctuated hemorrhages measuring less than 1.0 cm in diameter. The colonic mucosa was covered by multiple fibrinous plates measuring from 0.7 cm to 2.0 cm in diameter, intermingled with scattered ulcerations measuring on the average of 0.5 cm in diameter, from the cecum to the descendent colon (Figure 3C). The microscopic examination of the gastrointestinal ulcers showed similar findings as the tongue: mucosal ulcers with neutrophilic inflammatory infiltrate, granulation tissue, and endothelial cytomegalic inclusion, stromal, and inflammatory cells. Immunohistochemistry (mouse anti-cytomegalovirus [CMV] cocktail of DDG9 and CCH2, Diagnostic BioSystems, Pleasanton, CA, USA) was performed and all gastrointestinal lesions stained positively for cytomegalovirus in the nuclear inclusions of the cytomegalic cells (Figure 3D).
Additional findings of the autopsy included multiple organ damage due to septic shock, such as discrete lymphomononuclear myocarditis, nutmeg liver, splenitis, and acute tubular necrosis.
DISCUSSION
Taken together, the clinical and autopsy findings described in the present report can be interpreted as a patient with pemphigus vulgaris (the underlying cause of death) and recurrent major depression, under chronic immunosuppression secondary to prednisone and MMF. Tapering off the immunosuppressive therapy was not possible as the patient remained with active pemphigus vulgaris lesions due to treatment non-compliance. As a consequence, she acquired two infections: (i) cytomegalovirus infection reactivation, producing diffuse lesions in the gastrointestinal tract, with prolonged symptomatic diarrhea and odynophagia before her death; and (ii) nosocomial pneumonia due to Gram-positive cocci (an autopsy finding, not clinically suspected). Both infections certainly induced to prothrombotic state, leading to a massive pulmonary embolism, which was the immediate cause of her death.1-3 The malnutrition observed at her physical exam was probably synergistic with the drug-induced immunosuppression, corroborated by the anorexia caused by reduced food intake due to painful oral lesions,4 and lymphopenia, which was evidenced in her first complete blood count at this final hospitalization.5-7
Pemphigus vulgaris is an autoimmune disease characterized by the formation of suprabasal intra-epidermal blisters on the skin and mucosal surfaces, due to the destruction of the epithelial desmosomes by autoantibodies.8 Until the 1950s, the 5-year mortality related to pemphigus vulgaris was 100%.9 Mortality decreased to 25-45% with the use of systemic corticosteroids but adverse effects, such as diabetes, osteoporosis, dyslipidemia, and cataracts, led to the introduction of immunosuppressants as steroid-sparing agents.10,11 Death rates improved to 5-10% with adjuvant immunosuppression, but the risk of opportunistic infections increased.11,12
Infectious diseases are the main cause of death in patients with pemphigus.13-15 Few studies are published on the autopsies of patients with autoimmune bullous disorders concerning the pathology of their death. To the best of our knowledge, in 1984 Aki et al.16 performed the study with the largest number of patients including 18 autopsied cases. Among them, 10 had pemphigus vulgaris and 8 had pemphigus foliaceus. In the group with pemphigus vulgaris, nine deaths were attributed to infectious diseases related to immunosuppressive therapy, such as disseminated strongyloidiasis (two cases), military tuberculosis (two cases), pneumonia (three cases), sepsis related to Gram-positive cocci, and colitis (one case).16 Other isolated autopsy case reports showed bronchiolitis obliterans17 and lymphomatoid granulomatosis18 as causes of death in patients with pemphigus vulgaris.
CMV is a common human pathogen, and a member of the Herpesviridae family. Estimates of the worldwide prevalence of CMV infection range from 40% to 100%, with greater indexes in underdeveloped or developing countries due to higher population density.19 CMV transmission occurs through “close contact”, saliva, urine, sexual contact, and vertical transmission (the latter usually during early childhood). A Brazilian study conducted in Caieiras, a city in the northern area of the São Paulo State, showed that 90% of the infants had maternal antibodies against CMV.20 The immunocompetent hosts most commonly have an asymptomatic infection, and some develop a mononucleosis-like syndrome. The congenital cytomegalovirus infection occurs when a pregnant woman transmits the virus vertically to the fetus. After an acute phase, the CMV can establish a latent infection into the host cells that can reactivate under systemic immunosuppression (AIDS, post-transplantation, post-chemotherapy, and the use of steroids and other immunosuppressive drugs).21 The immunocompromised host with CMV disease can develop lesions in almost all organ systems, but mainly in the gastrointestinal tract (esophagitis, gastritis, colitis, ulcers, and perforations), the lungs (pneumonitis), the liver (hepatitis), the eyes (retinitis) and the nervous system (encephalitis and myelitis). Gastrointestinal CMV disease, with superficial mucosal lesions, can be diagnosed when the CMV nuclear inclusions are only seen in the epithelial cells (owl’s eye aspect) in a tissue sample. The invasive and systemic CMV disease is evidenced when the pathologist detects cytomegalic endothelial cells-with the typical nuclear inclusion-obliterating the vascular lumen, causing ischemic necrosis and ulceration of the gastrointestinal mucosa. Epithelial and stromal cells are also affected.22,23 The clinical manifestations include fever, malaise, weight loss, abdominal pain, diarrhea (watery or bloody), dysphagia, odynophagia, anemia, leucopenia, and viremia.24
CMV infection has been described among patients with pemphigus vulgaris under steroid therapy associated or not with azathioprine or rituximab, causing asymptomatic erythematous gastritis,25 gastric ulcers,22 or skin lesions.23 In a prospective cohort study of patients with pemphigus vulgaris, Leshem et al.12 found an incidence of 8.1% (14/172 patients) of opportunistic infections (OI) within the first year of follow-up. Two patients had fatal CMV disease: one had cutaneous and lung CMV infections, and the other presented systemic CMV. Advanced age and possibly diabetes were associated with an increased risk for OI among patients with pemphigus in this study.12
It should be emphasized that autoimmune bullous disorders can be superinfected or colonized by the herpes simplex virus (HSV).26 Besides HSV, which is the most common virus infection described,26-30 herpes virus 8 (HHV-8) may cause localized lesions of Kaposi sarcoma in areas previously treated with corticosteroid infiltrations,31 multifocal lesions,32 or skin colonization with uncertain clinical significance.33 CMV is described as causing skin superinfection or disseminated disease in some reports.12,23,34 The Herpesviridae virus superinfection must be suspected when there are bullous or ulcerative lesions refractory to immunosuppressive treatment. Diagnosis confirmation relies on histopathological or immunohistochemical analysis of skin samples, and through a polymerase chain reaction to detect CMV-DNA in blood samples (viremia).12,30,35,36 Our case illustrates how an infection can be detrimental to a patient with autoimmune bullous disorder. Despite proper diagnostic and therapeutic measures towards the patient’s complaints and clinical conditions, she had a fatal outcome due to a saddle pulmonary thromboembolism. Epidemiological data provide evidence of the association between acute infections and thromboembolic events, including deep vein thrombosis and pulmonary embolism. The risk of thrombotic events increases more than 2-fold within a period of 2 weeks to 3 months after either community or nosocomial infections of the respiratory and urinary tracts, blood, abdomen, skin, or soft tissues, and Gram-positive bacterial infections.1,2 An autopsy-based study conducted at King’s College Hospital (London, UK) found that among 110 patients with fatal pulmonary embolism associated with well-defined clinical conditions, 26 (23.6%) had infections-predominantly in the respiratory tract-during the 6 weeks prior to death.3 The reasons why an infection predisposes to a prothrombotic state are still not fully understood, but endothelial damage by virus or bacterial infections, dehydration, and white-cell activation have been implicated.37,38 In the current case, CMV gastrointestinal infection and nosocomial pneumonia were the determinants for the patient’s death. The suppurative pneumonia identified at the autopsy was probably not clinically diagnosed because the patient was receiving immunosuppressive drugs, and oral ciprofloxacin for urinary infection and diarrhea, which could mask the signs and symptoms of the respiratory infection.
Patients with bullous autoimmune disorders exhibit a higher infectious risk related to the disrupture of the physiological skin barrier, leading to colonization and infection with bacteria and other microorganisms, immune dysregulation, and the use of immunosuppressive medications.15,39 Infections are one of the main adverse effects of steroids, as they downregulate the innate and cell-mediated adaptive immune responses against intracellular microorganisms.5 MMF inhibits the purine nucleotide synthesis, an essential pathway to lymphocytes proliferation. Therapeutic regimens, including MMF, are associated with an increased likelihood of CMV disease among kidney transplant recipients.6
As a secondary preventive measure to infections, physicians must be aware of the signs and symptoms of infection, especially OI, caused by a myriad of agents (such as CMV), to provide early diagnosis and adequate treatment to the patients under immunosuppressive therapy.
A recent evidence-based guideline on the screening for infectious diseases in patients with autoimmune bullous dermatoses, recommends testing patients for Mycobacterium tuberculosis, and hepatitis B and C viruses, before prescribing rituximab treatment.14 On the other hand, doctors must educate their patients to avoid colonization and infection of their skin (proper hand washing, nail cleaning, and avoiding any physical contact with other patients) and recognize early signs and symptoms of infection to seek health assistance for rapid diagnosis and treatment.36 In addition, patient compliance to the medical prescription and follow-up appointments must be actively persuaded to prevent misuse of medications and consequent prolonged immunosuppression, which can lead to a fatal outcome, such as the one we have described here.
ACKNOWLEDGEMENTS
The authors would like to thank Mrs. Deryn Pompeia and Mr. Reginaldo Silva do Nascimento for their technical assistance in the preparation of this manuscript.
REFERENCES
1 Smeeth L, Cook C, Thomas S, Hall AJ, Hubbard R, Vallance P. Risk of deep vein thrombosis and pulmonary embolism after acute infection in a community setting. Lancet. 2006;367(9516):1075-9. PMid:16581406 http://dx.doi.org/10.1016/S0140-6736(06)68474-2.
2 Schmidt M, Horvath-Puho E, Thomsen RW, Smeeth L, Sørensen HT. Acute infections and venous thromboembolism. J Intern Med. 2012;271(6):608-18. PMid:22026462 http://dx.doi.org/10.1111/j.1365-2796.2011.02473.x.
3 Alikhan R, Peters F, Wilmott R, Cohen AT. Fatal pulmonary embolism in hospitalized patients: a necropsy review. J Clin Pathol. 2004;57(12):1254-7. PMid:15563663 http://dx.doi.org/10.1136/jcp.2003.013581
4 Knudson RM, Kalaaji AN, Bruce AJ. The management of mucous membrane pemphigoid and pemphigus. Dermatol Ther (Heidelb). 2010;23(3):268-80. PMid:20597945 http://dx.doi.org/10.1111/j.1529-8019.2010.01323.x.
5 Migita K, Arai T, Ishizuka N, et al. Rates of serious intracellular infections in autoimmune disease patients receiving initial glucocorticoid therapy. PLoS One. 2013;8(11):e78699. PMid:24260127 http://dx.doi.org/10.1371/journal.pone.0078699
6 Song ATWS, Abdala E, Bonazzi PR, Bacchella T, Machado MC. Does mycophenolate mofetil increase the risk of cytomegalovirus infection in solid organ transplant recipients?: a mini review. Braz J Infect Dis. 2006;10(2):132-8. PMid:16878265 http://dx.doi.org/10.1590/S1413-86702006000200011
7 Santos JI. Nutrition, infection, and immunocompetence. Infect Dis Clin North Am. 1994;8(1):243-67. PMid:8021446
8 Ben Lagha N, Poulesquen V, Roujeau JC, Alantar A, Maman L. Pemphigus vulgaris: a case-based update. J Can Dent Assoc. 2005;71(9):667-72. PMid:16271165
9 Mimouni D, Bar H, Gdalevich M, Katzenelson V, David M. Pemphigus: analysis of epidemiological factors in 155 patients. J Eur Acad Dermatol Venereol. 2008;22(10):1232-5. PMid:18482324 http://dx.doi.org/10.1111/j.1468-3083.2008.02786.x.
10 Bystryn JC, Steinman NM. The adjuvant therapy of pemphigus: an update. Arch Dermatol. 1996;132(2):203-12. PMid:8629830 http://dx.doi.org/10.1001/archderm.1996.03890260105016
11 Stanley JR. Therapy of pemphigus vulgaris. Arch Dermatol. 1999;135(1):76-8. PMid:9923786 http://dx.doi.org/10.1001/archderm.135.1.76
12 Leshem YA, Gdalevich M, Ziv M, David M, Hodak E, Mimouni D. Opportunistic infections in patients with pemphigus. J Am Acad Dermatol. 2014;71(2):284-92. PMid:24815564 http://dx.doi.org/10.1016/j.jaad.2014.03.020
13 Chams-Davatchi C, Valikhani M, Daneshpazhooh M, et al. Pemphigus: analysis of 1209 cases. Int J Dermatol. 2005;44(6):470-6. PMid:15941433 http://dx.doi.org/10.1111/j.1365-4632.2004.02501.x.
14 Keith PJ, Wetter DA, Wilson JW, Lehman JS. Evidence-based guidelines for laboratory screening for infectious diseases before initiation of systemic immunosuppressive agents in patients with autoimmune bullous dermatoses. Br J Dermatol. 2014;171(6):1307-17. PMid:25130049 http://dx.doi.org/10.1111/bjd.13355
15 Lehman JS, Khunger M, Lohse CM. Infection in autoimmune bullous diseases: a retrospective comparative study. J Dermatol. 2013;40(8):613-9. PMid:23772962 http://dx.doi.org/10.1111/1346-8138.12175
16 Aki HE, Sotto MN, Sampaio SA. Post-mortem evaluation in endemic pemphigus foliaceus and pemphigus vulgaris. Med Cutan Ibero Lat Am. 1984;12(2):151-7. PMid:6374324
17 Saito K, Morita M, Enomoto K. Bronchiolitis obliterans with pemphigus vulgaris and Castleman’s disease of hyaline-vascular type: an autopsy case analyzed by computer-aided 3-D reconstruction of the airway lesions. Hum Pathol. 1997;28(11):1310-2. PMid:9385940 http://dx.doi.org/10.1016/S0046-8177(97)90208-2.
18 Jauregui HO. Lymphomatoid granulomatosis after immunosuppression for pemphigus. Arch Dermatol. 1978;114(7):1052-5. PMid:686725 http://dx.doi.org/10.1001/archderm.1978.01640190040014
19 Jong MD, Galasso GJ, Gazzard B, et al. Summary of the II International Symposium on Cytomegalovirus. Antiviral Res. 1998;39(3):141-62. PMid:9833956 http://dx.doi.org/10.1016/S0166-3542(98)00044-8.
20 Almeida LNB, Azevedo RS, Amaku M, Massad E. Cytomegalovirus seroepidemiology in an urban community of São Paulo, Brazil. Rev Saude Publica. 2001;35(2):124-9. PMid:11359197 http://dx.doi.org/10.1590/S0034-89102001000200004
21 Goodgame RW. Gastrointestinal cytomegalovirus disease. Ann Intern Med. 1993;119(9):924-35. PMid:8215005 http://dx.doi.org/10.7326/0003-4819-119-9-199311010-00010
22 Orton DI, Orteu CH, Rustin MHA. Cytomegalovirus-associated gastric ulcer in an immunosuppressed patient with pemphigus vulgaris. Clin Exp Dermatol. 2001;26(2):170-2. PMid:11298108 http://dx.doi.org/10.1046/j.1365-2230.2001.00787.x.
23 Chiu H-Y, Chang C-Y, Hsiao C-H, Wang LF. Concurrent cytomegalovirus and herpes simplex virus infection in pemphigus vulgaris treated with rituximab and prednisolone. Acta Derm Venereol. 2013;93(2):200-1. PMid:22948546 http://dx.doi.org/10.2340/00015555-1429
24 Buckner FS, Pomeroy C. Cytomegalovirus disease of the gastrointestinal tract in patients without AIDS. Clin Infect Dis. 1993;17(4):644-56. PMid:8268345 http://dx.doi.org/10.1093/clinids/17.4.644
25 Hokama A, Taira K, Yamamoto Y, et al. Cytomegalovirus gastritis. World J Gastrointest Endosc. 2010;2(11):379-80. PMid:21173917 http://dx.doi.org/10.4253/wjge.v2.i11.379
26 Oliveira-Baptista D, Janini MER, Fernandes NC, Santos N. Laboratory diagnosis of herpesvirus infections in patients with pemphigus vulgaris lesions. Intervirology. 2013;56(4):231-6. PMid:23735576 http://dx.doi.org/10.1159/000349889
27 Feldmeyer L, Trüeb RM, French LE, Hafner J. Pitfall: pemphigus herpeticatus should not be confounded with resistant pemphigus vulgaris. J Dermatolog Treat. 2010;21(5):311-3. PMid:19895326 http://dx.doi.org/10.3109/09546630903287452
28 Marzano AV, Tourlaki A, Merlo V, Spinelli D, Venegoni L, Crosti C. Herpes simplex virus infection and pemphigus. Int J Immunopathol Pharmacol. 2009;22(3):781-6. PMid:19822095 http://dx.doi.org/10.1177/039463200902200324
29 Nikkels AF, Delvenne P, Herfs M, Pierard GE. Occult herpes simplex virus colonization of bullous dermatitides. Am J Clin Dermatol. 2008;9(3):163-8. PMid:18429645 http://dx.doi.org/10.2165/00128071-200809030-00004
30 Brandão ML, Fernandes NC, Batista DP, Santos N. Refractory pemphigus vulgaris associated with herpes infection: case report and review. Rev Inst Med Trop Sao Paulo. 2011;53(2):113-7. PMid:21537760 http://dx.doi.org/10.1590/S0036-46652011000200010
31 Avalos-Peralta P, Herrera A, Ríos-Martín JJ, Pérez-Bernal AM, Moreno-Ramírez D, Camacho F. Localized Kaposi’s sarcoma in a patient with pemphigus vulgaris. J Eur Acad Dermatol Venereol. 2006;20(1):79-83. PMid:16405614 http://dx.doi.org/10.1111/j.1468-3083.2005.01348.x.
32 Halpern SM, Parslew R, Cerio R, Kirby JT, Sharpe GR. Kaposi’s sarcoma associated with immunosuppression for bullous pemphigoid. Br J Dermatol. 1997;137(1):140-3. PMid:9274643 http://dx.doi.org/10.1111/j.1365-2133.1997.tb03718.x.
33 Meibodi NT, Nahidi Y, Mahmoudi M, et al. Evaluation of coexistence of the human herpesvirus type 8 (HHV-8) infection and pemphigus. Int J Dermatol. 2010;49(7):780-3. PMid:20618497 http://dx.doi.org/10.1111/j.1365-4632.2009.04398.x.
34 Casals DS, Nunes EA, Maruta CW, et al. Disseminated cytomegalovirus disease as a cause of prolonged fever in a bullous pemphigoid patient under systemic steroid therapy. J Dermatol. 2003;30(4):332-6. PMid:12707471 http://dx.doi.org/10.1111/j.1346-8138.2003.tb00396.x.
35 Gee SN, Velez NF, Sepehr A, Burgin S. Two distinct viral infections complicating pemphigus foliaceus. Dermatol Online J. 2012;18(1):3. PMid:22301040
36 Lehman JS, Murrell DF, Camilleri MJ, Kalaaji AN. Infection and infection prevention in patients treated with immunosuppressive medications for autoimmune bullous disorders. Dermatol Clin. 2011;29(4):591-8. PMid:21925003 http://dx.doi.org/10.1016/j.det.2011.06.021
37 Opal SM, Cohen J. Clinical gram-positive sepsis: does it fundamentally differ from gram-negative bacterial sepsis? Crit Care Med. 1999;27(8):1608-16. PMid:10470773 http://dx.doi.org/10.1097/00003246-199908000-00039
38 Visseren FL, Bouwman JJ, Bouter KP, Diepersloot RJ, de Groot PH, Erkelens DW. Procoagulant activity of endothelial cells after infection with respiratory viruses. Thromb Haemost. 2000;84(2):319-24. PMid:10959707
39 Maddur MS, Vani J, Lacroix-Desmazes S, Kaveri S, Bayry J. Autoimmunity as a predisposition for infectious diseases. PLoS Pathog. 2010;6(11):e1001077. PMid:21079687 http://dx.doi.org/10.1371/journal.ppat.1001077
Notes
Author notes
Correspondence Amaro Nunes Duarte-Neto Departamento de Patologia - Faculdade de Medicina - Universidade de São Paulo (USP) Avenida Dr. Arnaldo, 455 - Cerqueira Cesar - São Paulo/SP - Brazil CEP: 01246-903 Phone: +55 (11) 98546-4225 amaro.ndneto@hc.fm.usp.br
Conflict of interest declaration