Article / Clinical Case Report
Small cell neuroendocrine carcinoma of the nasopharynx: a rare case report
Small cell neuroendocrine carcinoma of the nasopharynx: a rare case report
Autopsy and Case Reports, vol. 7, no. 1, pp. 31-35, 2017
Hospital Universitário da Universidade de São Paulo
Received: 24 November 2016
Accepted: 23 February 2017
ABSTRACT: Small cell neuroendocrine carcinoma rarely appears primarily in the head and neck and exhibits aggressive behavior with a poor prognosis. The pathologist has a significant role in the diagnosis, and a consensual treatment still does not exist. The authors report the case of a middle-aged male patient who presented repeated episodes of massive epistaxis. The diagnostic work-up disclosed the diagnosis of small cell neuroendocrine carcinoma of the nasopharynx. The patient was treated with chemotherapy followed by radiotherapy. Imaging examinations performed after the end of treatment showed apparent complete remission of the disease. The patient was kept under active surveillance with no signs of local relapse or distant metastasis after 4 years of follow-up.
Keywords: Carcinoma, Small Cell, Neuroendocrine, Carcinoma, Nasopharynx.
INTRODUCTION
Small cell neuroendocrine carcinoma is a high-grade tumor that usually presents aggressive behavior. The lungs represent the most common primary site, and extra pulmonary cases comprise only 2.5-5% of the cases,1,2 mainly involving the esophagus,3 colon,4 urinary bladder,5 and cervix.6 The involvement of the head and neck structures is even rarer, and in these cases, the larynx is the most frequently involved organ.7 Due to its rarity, the diagnosis and treatment are challenging. We present the case of a nasopharyngeal small cell neuroendocrine carcinoma, which, as far as we know, is the ninth case reported in the literature.8-15
CASE REPORT
A 54-year-old Caucasian male patient sought medical care complaining of recurrent epistaxis and dysphonia for 2 weeks. He had a history of heavy smoking. Physical examination and laboratory tests were normal. The computed tomography (CT) of the paranasal sinuses revealed the presence of an extensive nasopharyngeal lesion measuring 5 × 6 cm predominantly involving the left side, extending to the nasal fossae, parapharyngeal space, and pterygoid muscles. There was neither intracranial extension nor bone involvement.
A nasofibroscopy was performed showing a reddish lesion of the left nasal fossae, which was biopsied. The result was inconclusive (esthesioneuroblastoma vs. small cell carcinoma), but another biopsy favored the diagnosis of small cell neuroendocrine carcinoma, confirmed by the immunohistochemical study, which presented diffuse expression of CAM 5.2 and synaptophysin in rare neoplastic cells, in the absence of neuron-specific enolase, chromogranin, and neurofilaments (Figure 1).
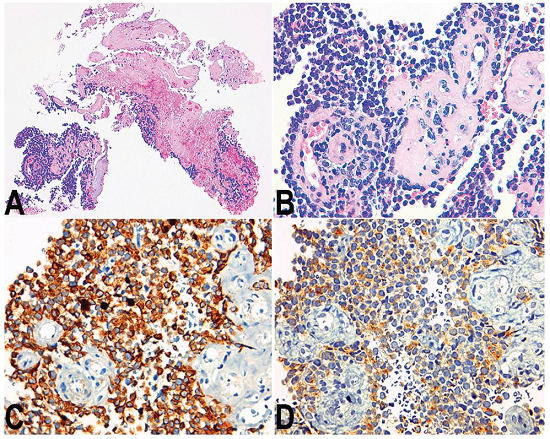
Figure 1
Photomicrography of the biopsy specimen. A - Extensive areas of necrosis (H&E, 100X); B - Sheets of small cells with high nuclear-cytoplasmic ratio surrounding the vessels (H&E, 400X); C - Positivity immunostaining for CAM 5.2 (400X); D - Focal positivity for synaptophysin (400X).
The magnetic resonance imaging (MRI) detected the infiltration of the soft palate and confirmed the presence of a tumor in the nasopharynx measuring 6.5 cm at its longest axis; presenting a similar extension observed with the CT.
Clinical staging was II: cT2N0M0 according to the proposal for the 8th edition of the American Joint Committee on Cancer (AJCC)/Union for International Cancer Control (UICC).16
The patient was treated with primary chemotherapy with carboplatin AUC5 (day 1) and etoposide 100 mg/m2 (days 1, 2, and 3) every 21 days. After the end of the fourth cycle of chemotherapy, the patient underwent a planning CT scan (Figure 2) and started radiotherapy receiving a total dose of 70 Gy to the nasopharynx and 60 Gy to the cervical region.
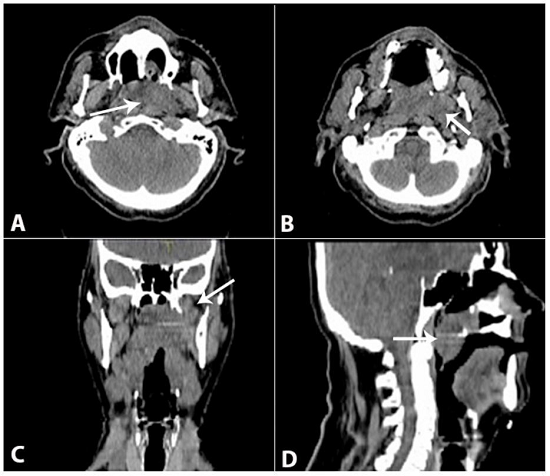
Figure 2
Planning CT scan showing the extensive nasopharyngeal lesion extending to the nasal fossae, parapharyngeal space, and pterygoid muscles, with soft palate invasion. A and B - Axial sections; C - Coronal section; D - Sagittal section.
On the second month of follow-up, another pharyngeal MRI was performed, which demonstrated that the pharynx was free of neoplastic lesions and no cervical lymph nodes were enlarged.
A CT scan of the thorax and abdomen was conducted on the sixth month of follow-up, which showed a spiculated pulmonary nodule measuring 16 mm and another nodular lesion in the pancreatic tail, which were tackled by aspiration biopsy. The cytology was inconclusive for the pulmonary nodule, and negative for malignancy for the pancreatic lesion. The positron emission tomography using 18F-fluorodeoxyglucose (PET-18F-FDG) revealed the radiotracer uptake (SUV max = 6.96) in two lymph nodes of the pulmonary hilum and another in the subcarinal topography, besides a mild enhancement of the radiotracer uptake (SUV max = 2.33) in the middle pulmonary lobe, both suspected of metastasis. No other sites, namely the pancreas and the nasopharynx, showed abnormal radiotracer concentration consistent with a malignant disease. A transbronchial fine-needle aspiration of the hilar lymph nodes was performed by endobronchial ultrasound, and the cytological examination was negative for malignancy. Oncologic surveillance was maintained afterward.
DISCUSSION
The neoplasms with neuroendocrine differentiation involving the head and neck structures, including the nasopharynx, are exceedingly rare. The diagnosis of such an entity is challenging because of the morphological characteristics as well as the biopsy sample size, which does not always allow the distinction among other types of neoplasms. The immunohistochemical study has an indispensable central role to confirm the epithelial and neuroendocrine nature of this kind of tumor, which expresses variably cytokeratins (such as CAM 5.2) and neuroendocrine markers like synaptophysin, neuron-specific enolase, chromogranin and CD56.17 According to the WHO classification, neuroendocrine tumors of the nasal cavity and paranasal sinuses may be divided into typical carcinoid tumors, atypical carcinoid tumors, and small cell neuroendocrine carcinoma.18 Recently, a new classification for neuroendocrine tumors of the head and neck was proposed and is similar to that of the lung and gastrointestinal tract. This classification divides these tumors into neuroendocrine carcinoma grade 1, grade 2, and large or small cell neuroendocrine carcinoma grade 3, which are based on the morphology, the mitotic index, and the presence or absence of necrosis. According to this classification, small cell neuroendocrine carcinomas present tumor cells with small size, scant cytoplasm, nuclei with finely granular chromatin, inconspicuous or absent nucleoli, a high mitotic rate, and frequent necrosis,19 which is in line with the case presented herein. The additional expression of CAM 5.2 and synaptophysin, confirmed by immunohistochemistry, endorsed the diagnosis of small cell neuroendocrine carcinoma. As mentioned above, the expression of cytokeratins and neuroendocrine markers may vary among tumors. In the majority of cases, the analysis of the other eight cases reported in the literature showed the expression of chromogranin,8,11,14,15 synapthophysin,8,11,14 and CD56,11,14,15 with only one expressing neuron-specific enolase.8
To summarize, small cell neuroendocrine carcinomas of the nasopharynx are high-grade tumors, and therefore convey a poor prognosis.
The therapeutic options for these tumors are varied. However, the treatment of choice is still undefined because only eight cases have been reported in the literature to date (Table 1).
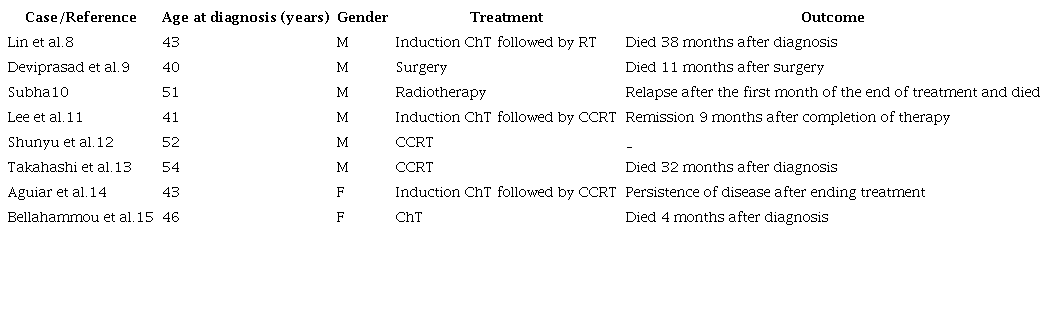
CCRT = concomitant chemoradiotherapy; ChT = chemoradiotherapy; F = female; M = male.
CONCLUSION
Despite the lack of a consensual therapeutic modality, the multimodality approach (chemo and radiotherapy) showed evidence of increment in the survival rate of patients with the diagnosis of sinonasal small cell neuroendocrine carcinoma.20,21 In our case, the option of sequential treatment (chemo followed by radiotherapy) was based on the histology type and the tumor size. Despite the poor prognosis of this entity, we verified no evidence of locoregional relapse or distant metastasis at the end of a 4-year period of follow-up.
REFERENCES
1 Levenson RM Jr, Ihde DC, Matthews MJ, et al. Small cell carcinoma presenting as an extrapulmonary neoplasm: sites of origin and response to chemotherapy. J Natl Cancer Inst. 1981;67(3):607-12. PMid:6268879
2 Remick SC, Ruckdeschel JC. Extrapulmonary and pulmonary small-cell carcinoma: tumor biology, therapy, and outcome. Med Pediatr Oncol. 1992;20(2):89-99. PMid:1310345 http://dx.doi.org/10.1002/mpo.2950200202
3 Haider K, Shahid RK, Finch D, et al. Extrapulmonary small cell cancer: a Canadian province’s experience. Cancer. 2006;107(9):2262-9. PMid:16998932 http://dx.doi.org/10.1002/cncr.22235
4 Brenner B, Shah MA, Gonen M, Klimstra DS, Shia J, Kelsen DP. Small-cell carcinoma of the gastrointestinal tract: a retrospective study of 64 cases. Br J Cancer. 2004;90(9):1720-6. PMid:15150595
5 Shahab N. Extrapulmonary small cell carcinoma of the bladder. Semin Oncol. 2007;34(1):15-21. PMid:17270661 http://dx.doi.org/10.1053/j.seminoncol.2006.10.025
6 Lee SS, Lee JL, Ryu MH, et al. Extrapulmonary small cell carcinoma: single center experience with 61 patients. Acta Oncol. 2007;46(6):846-51. PMid:17653910 http://dx.doi.org/10.1080/02841860601071893
7 Ferlito A, Rinaldo A. Primary and secondary small cell neuroendocrine carcinoma of the larynx: a review. Head Neck. 2008;30(4):518-24. PMid:18302254 http://dx.doi.org/10.1002/hed.20797
8 Lin IH, Hwang CF, Huang HY, Chien CY. Small cell carcinoma of the nasopharynx. Acta Otolaryngol. 2007;127(2):206-8. PMid:17364353 http://dx.doi.org/10.1080/00016480500401027
9 Deviprasad S, Rajeshwari A, Tahir M, Adarsha TV, Gangadhara S. Small cell neuroendocrine carcinoma originating from the lateral nasopharyngeal wall. Ear Nose Throat J. 2008;87(11):E1-3. PMid:19006052
10 Subha S. Neuroendocrine carcinoma of the nasopharynx: a case report. Med Health Sci J. 2011;9(5):101-3. http://dx.doi.org/10.15208/mhsj.2011.185
11 Lee LY, Chang KP, Hsu CL, Chen TC, Kuo TT. Small-cell neuroendocrine carcinoma of the nasopharynx: report of a rare case lacking association with Epstein-Barr virus. Int J Surg Pathol. 2011;19(2):199-202. PMid:18508843 http://dx.doi.org/10.1177/1066896908316904
12 Shunyu N, Lynrah Z, Goyal A, Raphal V. Small-cell neuroendocrine carcinoma of nasopharynx. Clin Rhinol Int J. 2012;5:130-1. http://dx.doi.org/10.5005/jp-journals-10013-1134
13 Takahashi S, Miyashita T, Hoshikawa H, Haba R, Togami T, Shibata T. Accelerated hyperfractionated radiotherapy for small cell carcinoma of the nasopharynx. Head Neck. 2015;37(5):E63-5. PMid:25270910 http://dx.doi.org/10.1002/hed.23888
14 Aguiar A, Jacome M, Azevedo I, Monteiro E, Castro V. Small-Cell neuroendocrine carcinoma originating from the nasopharynx: report of a rare case. Rep Radiother Oncol. 2015;2(3):e3814.
15 Bellahammou K, Lakhdissi A, Akkar O, Kouhen F, Rais F, Dahraoui S, Mrabti H, Errihani H. Small-cell neuroendocrine carcinoma of nasopharynx: a case report. IJSM 2016 Sep 25. [Epub ahead of print].
16 Pan JJ, Ng WT, Zong JF, et al. Proposal for the 8th edition of the AJCC/UICC staging system for nasopharyngeal cancer in the era of intensity-modulated radiotherapy. Cancer. 2016;122(4):546-58. PMid:26588425 http://dx.doi.org/10.1002/cncr.29795
17 Montone KT. The differential diagnosis of sinonasal/nasopharyngeal neuroendocrine/neuroectodermally derived tumors. Arch Pathol Lab Med. 2015;139(12):1498-507. PMid:26619022 http://dx.doi.org/10.5858/arpa.2014-0383-RA.
18 Perez-Ordonez B. Neuroendocrine tumors. In: Barnes L, Eveson JW, Reichart P, Sidransky D, editors. Pathology and genetics of head and neck tumours. Lyon: IARC Press; 2005. p. 26-27. World Health Organization Classification of Tumours, 9.
19 Xu B, Chetty R, Perez-Ordonez B. Neuroendocrine neoplasms of the head and neck: some suggestions for the new WHO classification of head and neck tumors. Head Neck Pathol. 2014;8(1):24-32. PMid:24595420 http://dx.doi.org/10.1007/s12105-014-0531-y
20 Mitchell EH, Diaz A, Yilmaz T, et al. Multimodality treatment for sinonasal neuroendocrine carcinoma. Head Neck. 2012;34(10):1372-6. PMid:22052583 http://dx.doi.org/10.1002/hed.21940
21 van der Laan TP, Bij HP, van Hemel BM, et al. The importance of multimodality therapy in the treatment of sinonasal neuroendocrine carcinoma. Eur Arch Otorhinolaryngol. 2013;270(9):2565-8. PMid:23740425 http://dx.doi.org/10.1007/s00405-013-2554-5
Notes
Author notes
Correspondence Daniela Azevedo Clinical Oncology Department - Centro Hospitalar de São João Urbanização Pé do Cavalo, Rua A, lote 2 - Vila Real - Porto - Portugal CEP: 5000-058 Phone: +35 (19) 6738-6047 dnpadua@hotmail.com
Conflict of interest declaration