Article / Autopsy Case Report
Mycotic aneurysm with aortoduodenal fistula
Mycotic aneurysm with aortoduodenal fistula
Autopsy and Case Reports, vol. 7, no. 2, pp. 27-34, 2017
Hospital Universitário da Universidade de São Paulo
Received: 17 March 2017
Accepted: 08 May 2017
ABSTRACT: Firstly described in the 19th century by Sir William Osler, the mycotic aneurysm (MA) is a rare entity characterized by an abnormal arterial dilation, which is potentially fatal, and is associated with the infection of the vascular wall. Elderly patients are mostly involved, especially when risk factors like chronic diseases, immunosuppression, neoplasia, and arterial manipulation are associated. The authors report the case of a young male patient diagnosed with an aortic aneurysm of infectious origin in the presence of repeated negative blood cultures. The diagnostic hypothesis was raised when the patient was hospitalized for an inguinal hernia surgery. The diagnosis was confirmed based on imaging findings consistent with mycotic aneurism. The patient was treated with an endovascular prosthesis associated with a long-lasting antibiotic therapy. Five months later, the patient attended the emergency unit presenting an upper digestive hemorrhage and shock, from which he died. The autopsy revealed a huge aneurysm of the abdominal aorta with an aortoduodenal fistula. The histological examination of the arterial wall revealed a marked inflammatory process, extensive destruction of the arterial wall, and the presence of Gram-positive bacteria. This case highlights the atypical presentation of a MA associated with an aortoduodenal fistula. Besides the early age of the patient, no primary arterial disease could be found, and no source of infection was detected.
Keywords: Aortic Aneurysm, Fistula, Aneurysm, Infected.
CASE REPORT
A 23-year-old man sought emergency care complaining of a 2-month history of a burning mesogastric pain irradiating to the lumbar region that worsened on the palpation of the right flank. On the tenth postoperative day of an elective right inguinal hernioplasty, an abdominal pulsatile mass was detected. The hernioplasty was indicated before the abdominal symptoms started and preoperative lab work-up did not reveal abnormalities such as leukocytosis. The patient was advised to proceed with further investigation, but he refused.
Due to the worsening of his abdominal pain, he sought medical assistance in a hospital emergency department on the 10th post-operative day. On admission, the physical examination revealed a well-looking patient, slightly pale with normal vital signs, emaciated (the patient mentioned a weight loss of 10 kg over the past 2 months), and was afebrile. The abdominal palpation depicted a painful pulsatile mass in the mesogastrium, and on auscultation there was a bruit in the same region. No signs of peritoneal irritation were found. The patient denied drug addiction and did not recall any infections over recent months. The search for Koch's bacillus in sputum and urine was negative, and the patient's epidemiology was not conducive to tuberculosis. VDRL and ELISA tests were negative and excluded syphilis and HIV infection, respectively.
The aortic angiogram computed tomography (CT) revealed an infrarenal aortic saccular aneurysm measuring 6.7 × 5.2 cm (Figure 1).
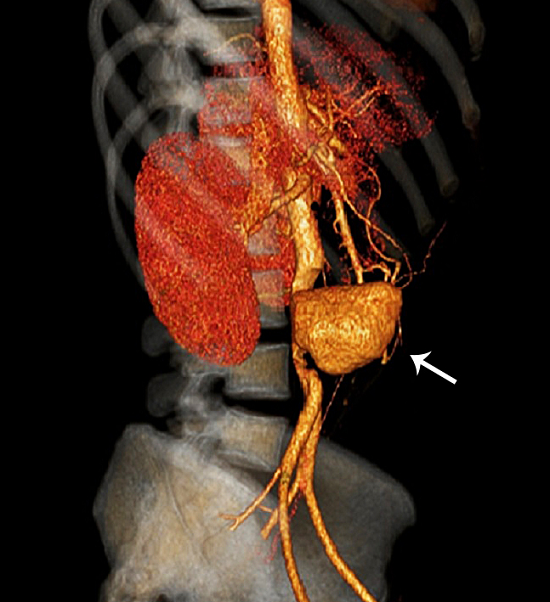
Figure 1
Aortic angiogram CT revealed an infrarenal aortic saccular aneurysm measuring 6.7 × 5.2 cm (arrow).
The aneurysm presented irregular contours and ulcerations, and was surrounded in its entire extension by an irregular dense tissue with fat tissue obliteration (Figure 2A). The aneurysm anteriorly displaced the duodenum and laterally the inferior vena cava and the iliac veins (Figure 2B). The CT findings were consistent with a mycotic aneurysm (MA). The patient was hospitalized and submitted to an endovascular treatment which consisted of the implantation of an aorto-bi-iliac endovascular stent associated with long-lasting empiric antibiotic therapy, which comprised oxacillin and ceftriaxone. A blood culture was sampled before the procedure, but the result was negative. An abdominal CT was undertaken and the patient was discharged clinically well and afebrile, four days after the procedure.
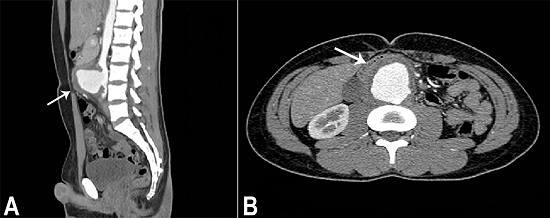
Figure 2
Abdominal CT. A (sagittal plane) - Infrarenal aortic saccular aneurysm presenting irregular contours, ulceration, surrounded by irregular dense tissue (arrow); B (axial plane) - The anterior displacement of the duodenum (arrow).
Over the next 3 months, the patient was hospitalized two more times. At each admission, he was in poor clinical condition, febrile, and presented abdominal pain. He was prescribed vancomycin, meropenem, and micafungin. Blood cultures were repeatedly negative (including anaerobic cultures) and continuing transthoracic and transesophageal echocardiograms were normal.
During his third hospitalization, the patient received 35 days of antimicrobial therapy, ran out of fever and refused to remain longer in the hospital. After 2 months he returned to the emergency facility complaining of severe and diffuse abdominal pain followed by upper digestive hemorrhage and hypovolemic shock. The abdominal CT revealed the presence of thrombi throughout the entire aorta and gaseous foci within the endoprosthesis indicating the presence of an aortoenteric fistula (Figure 3). Unfortunately, the patient died 1 day after hospital admission. An autopsy was performed with an informed consent signed by the family.
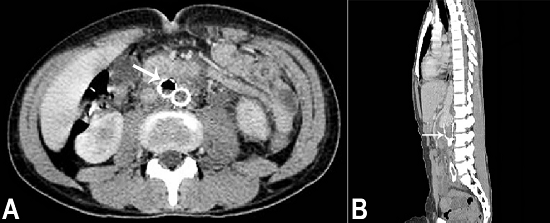
Figure 3
Abdominal CT prior to death. A - (axial plane) Gaseous focus within the endoprosthesis (arrow) indicating aortoenteric fistula; B - (sagittal plane) The presence of thrombi throughout the aortoiliac endoprosthesis (arrow).
AUTOPSY FINDINGS
The autopsy examination revealed the presence of a huge abdominal aortic aneurysm measuring 7.0 cm in extension and 5.0 cm in diameter filled in by an aortoiliac prosthesis, which was extensively thrombosed (Figure 4A). The aortic wall presented a local ulceration that extended towards the third duodenal portion producing an aortoenteric fistula (Figure 4B), which was responsible for the massive intestinal bleeding.

Figure 4
Macroscopic view. A - Segment of abdominal aorta with an aneurysm of 7.0 × 5.0 cm with the lumen filled in by an aortoiliac prosthesis. Note extensive mural thrombosis; B - Third duodenal portion in close relation to the abdominal aneurysm presenting an aorto-enteric fistula covered by blood clots.
The histological examination of this area revealed a marked transmural inflammatory process with an area of destruction of the aorta wall, which was replaced by fibrosis (Figure 5A) and with suppurative areas in the wall of the fistula track and periaortic soft tissues (Figure 5B). The remaining aorta wall had no evidence of inflammatory or degenerative disease (Figure 5C). Verhoeff stain was performed (Figure 5D) as well as an immunohistochemical study to characterize the inflammatory component and exclude the possibility of immunoglobulin (Ig)G4 related disease (CD3, CD20, CD68, CD138, IgG, and IgG4) as well as other aortic vasculitis.
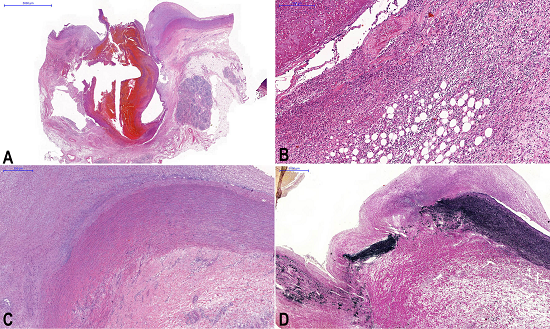
Figure 5
A - Panoramic view of the aorta with the fistula track showing the destruction of arterial wall (H&E 0.45X); B - Periaortic soft tissues infiltrated by inflammatory cells presenting suppurative areas and the fistulous track with hemorrhagic evidence remaining in its light (H&E 9.84X); C - Photomicrography of the aorta showing a segment of the aorta close to the perforation area without pathological alterations (H&E 3.29X); D - Verhoeff staining showing the area of destruction of the aorta wall, which was replaced with fibrosis (Verhoeff 1.61X).
The Brown-Hopps stain revealed the presence of pleomorphic Gram-positive bacilli (Figure 6). Unfortunately, a culture of the aortoenteric fistula’s tissue was not collected during the autopsy examination. The left iliac artery communicated with an inguinal abscess, which was interpreted as a metastatic manifestation of the MA. The heart weighted 340 g, (reference value [RV]: 350 g) and was normal. The cardiac valves were thin and translucent, without any evidence of actual or previous endocarditis. No other organ alteration was observed except for evidence of bronchopneumonia.
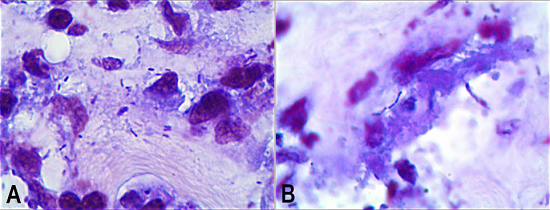
Figure 6
A & B- Photomicrographs of the aneurysm wall showing pleomorphic Gram-positive bacilli (Brown-Hopps, 1000X).
DISCUSSION
In 1885, Sir William Osler first use the term “mycotic aneurysm” describing the autopsy of a 30-year-old man who died after presenting diarrhea, chills, cephalea, cough, and fever. In this autopsy, he found the aortic valve extensively destroyed by a vegetation as well as four aneurysms of the aortic arch with evidence of endocarditis and mycotic endarteritis.1 Currently, the “mycotic” term is used to describe all the infected aneurysms regardless of the etiological agent.2,3
MA is a rare entity, occurring in 0.9% of a series comprising 2,585 patients treated for arterial aneurysms.4 MA has been observed in different age groups including newborns. However, they are much more prevalent in elderly patients with atherosclerosis.2,5 The epidemiology of this entity is changing. In the past, the infectious endocarditis represented the main cause of MA; however, currently, with the availability of the antibiotic therapy, this relationship occurs in the minority of the cases.6-8 The main risk factor for the development of MA are: (i) arterial catheterization; (ii) previous surgeries in distant sites; (iii) immunosuppression due to chronic diseases; (iv) neoplasia; (v) use of corticoids or other cytotoxic agents; (vi) drug addiction; and (vii) hemodialysis.4,5Salmonella spp. and Staphylococcus spp. are the main etiological agents related to MA.6-8
Intriguingly, our patient was young without a known risk factor, and no etiological agent could be isolated. The syphilis, HIV, and TB tests were negative. Autoimmune aortitis (e.g. Takayasu’s arteritis) was excluded since the patient did not present compatible clinical aspects, and the postmortem histological examination did not reveal pathological changes in other sites of the aorta and in other vessels. Also, the patient had no primary vascular disease. Nevertheless, the patient could have had a subclinical and unnoticed bacteremia, which could have been the source for the MA development. Although the patient denied drug addiction, this is information was not entirely credible.
The primary source of infection may be identified in 86% of the patients with abdominal MA.9,10 Upper airway infections, cellulitis, osteomyelitis, pneumonia, urinary tract infections, and non-healed wounds have been described.5
MA may be the result of an infection of the arterial wall, or it may rise as an infection of a previously formed aneurysm.11 The microorganisms generally colonize the vessel by embolization, extension of an adjacent suppurative process, or circulation of viable micro-organisms within the arterial wall during bacteremia. Depending on the virulence of the etiological agent and the blood pressure, the growth of the aneurysm may be fast, reaching centimeters within a month.5 The vessels with congenital abnormalities (aortic coarctation, patent ductus arteriosus) or vessels with atherosclerosis are more prone for the development of MA.2,12,13 In our case, we believe that the MA was formed before the hernioplasty. The fact that the patient’s symptoms started 2 months before the inguinal surgery, and that a pulsatile mass could be palpable before the procedure supports the previous existence of the aneurysm. It is unlikely that the aneurysm was present and became infected after the hernioplasty, since the patient was very young for having an aneurysm, and a primary vascular disease was not found on the autopsy. Moreover, the CT and angiogram images, performed 10 days after the surgery, were consistent with an infectious aneurysm. It was unclear why the aneurysm might not have been detected at the time of the hernioplasty. This elapsed time is insufficient for a MA to be developed. However, the patient’s data and images suggest that he might have had an intermittent bowel obstruction due to intestinal incarceration in his inguinal hernia and at some point, Gram-positive bacteria got into the bloodstream and could have contributed to the development of the MA.
In contrast to the case reported herein, etiology is frequently confirmed. In a study comprising 109 patients with MA,14 101 cases rendered a positive culture. In our case, bacteria identified in the histological examination with the aid of the Brown-Hopps staining were all Gram-positive and pleomorphic, which suggests that the infection was polymicrobial, presumably with multiple types of anaerobic bacilli from the gut.
MA may be found in any artery. However, they are more frequently found in different aortic portions (arch, thoracic, thoracoabdominal, and superior abdominal) and in the great aortic branches, as observed in our patient.4,5,15-17
The symptoms of the MA are mostly non-specific, reflecting the insidious nature of the disease.18,19 However, when present, they are predominantly represented by fever; abdominal, lumbar or thoracic pain; chills; and the presence of a pulsatile mass.20,21 Hence, an imaging exam is crucial for the diagnosis, and CT is the method most often used. The imaging findings that favor the diagnosis of MA are: (i) atypical localization of the aneurysm: (ii) lack of calcium within the vessel wall; (iii) multilobular aspect; (iv) multifocal or saccular configuration; (v) presence of periaortic gas; (vi) adjacent soft tissue reaction; and (vii) simultaneous presence of osteomyelitis.5 The CT findings of our patient favored the diagnosis of MA because of the unusual site of an aneurysm, the early age of onset, the presence of irregular contour, the thickening of the vessel wall, the fat tissue obliteration, and the displacement of the duodenum, inferior vena cava, and iliac veins.
The treatment of choice for the MA is not a consensus and varies according to the anatomic localization and severity of the infection. However, the therapy generally consists of a surgical approach and long-term antibiotic therapy.
In this setting, the surgical approach for patients with the diagnosis of MA presents a high index of mortality, mainly when associated with other comorbidities that substantially increase the surgical risk.13,22,23
Revascularization with aortic exclusion has been a widely performed technique with a survival rate of 61%. However, revascularization with the in situ graft insertion, following the resection and debridement of the involved surrounding infected tissue, presents a survival rate of 75%.22 As MA is a rare disease, experience with its surgical management is not extensive and varies from hospital to hospital.2 Mortality rates can reach 48% regardless of the surgical treatment chosen.2 If, on the one hand, there are controversies about how the surgical intervention should be performed, on the other hand it is mandatory to remove the infected material.2,22
More recently, other studies showed that the endovascular repair technique is an important alternative for MA treatment. It works as damage control and facilitates the subsequent effective open surgery.22 In our case, the endovascular treatment was performed because of the understanding that an aneurysmectomy and an open approach would present an increased risk, and the patient had high chances of dying during the procedure. Furthermore, a femoral vein graft and an aortic ligation with extra-anatomical repair could not withstand the aortic pressure, mainly because they would have to be large and resistant enough. Reconstruction with a conserved corpse aorta from an aorta bank and using a Dacron prosthesis associated with chemotherapy and coverage of biological material are alternatives to MA treatment, but they also represent difficult options.2 Also, the material can be easily infected again, or can be rejected by the patient’s immune system and chemotherapy could worsen the patient’s clinical condition. The poor prognosis of this entity is frequently associated with sepsis and the rupture of the aneurysm.5,24
The aorto-duodenal fistula (ADF) is a rare and potentially fatal complication with the incidence ranging between 0.04% and 0.07%, in autopsies.25-27 It is defined as an abnormal communication between the aorta and the duodenum, and is classified in primary when associated with aneurysms, and secondary when related to surgical procedures—the occurrence of which is 10 times more frequent.28 The third duodenal portion is more often involved since it is localized in the retroperitoneum in close contact with the aorta.29,30 It is most likely that our patient’s case is an example of secondary ADF, since it developed after the endovascular repair and next to the top edge of the graft. Also, the relation between aortic grafting and aortoenteric fistulas is well established.29,30
The clinical features of ADF are: (i) gastrointestinal bleeding (64%); (ii) abdominal pain (32%); and (iii) the presence of a pulsatile mass (25%).31-33 Generally, the patients present self-limited gastrointestinal bleeding (sentinel bleeding) followed by massive bleeding. The principal diagnostic examination is the angiogram CT that presents sensitivity ranging from 40% to 90% and specificity from 33% to 100%.33,34 However, given the frequent sudden onset of the digestive hemorrhage, imaging exams and an endoscopic examination are not always feasible. If there is time for any therapy, the surgical approach is recommended. The mortality rate is 100% without any therapeutic modality due to hypovolemic shock.35,36 Femoral artery pseudoaneurysms (FAP) occur when a blood vessel wall is injured and the blood is contained by the surrounding tissues. FAPs are mostly described in drug addicted or immunosuppressed patients. The main etiologies involve trauma, vessel wall infection, surgeries, and endovascular procedures.37-39 Ultrasound is the most common method to diagnose FAPs.37 In our case, we believe that the infectious process associated with the endovascular intervention were responsible for the FAP. The treatment is controversial, but open surgical repair represents a very high risk option, and arterial ligation tends to be the most indicated procedure, which was the case with our patient.38,39
CONCLUSION
We report an atypical case of MA that evolved with an aortoduodenal fistula—a rare complication. We highlight the early age of the patient, the absence of predisposing factors, the less common site of the aneurysm, and the persistent negative blood cultures. An MA is a rare entity that may exhibit varied presentations, which challenges its diagnosis. Aortoenteric fistulas are rare, but their suspicion should be raised as a possible complication of MA.
REFERENCES
1 Osler W. The gulstonian lectures, on malignant endocarditis. BMJ. 1885;1(1262):467-70. 20751186 http://dx.doi.org/10.1136/bmj.1.1262.467
2 Müller BT Wegener OR, Grabitz K, Pillny M, Thomas L, Sandmann W. Mycotic aneurysms of the thoracic and abdominal aorta and iliac arteries: experience with anatomic and extra-anatomic repair in 33 cases. J Vasc Surg. 2001;33(1):106-13. 11137930 http://dx.doi.org/10.1067/mva.2001.110356
3 Bisdas T, Teebken OE. Mycotic or infected aneurysm? Time to change the term. Eur J Vasc Endovasc Surg. 2011;41(4):570-2. 21232995 http://dx.doi.org/10.1016/j.ejvs.2010.11.036
4 Chan FY, Crawford ES, Coselli JS, Safi HJ, Williams TW Jr. In situ prosthetic graft replacement for mycotic aneurysm of the aorta. Ann Thorac Surg. 1989;47(2):193-203. 2919901 http://dx.doi.org/10.1016/0003-4975(89)90268-3.
5 Cinà CS, Arena GO, Fiture AO, Clase CM, Doobay B. Ruptured mycotic thoracoabdominal aortic aneurysms: a report of three cases and a systematic review. J Vasc Surg. 2001;33(4):861-7. 11296343 http://dx.doi.org/10.1067/mva.2001.111977
6 Johansen K, Devin J. Mycotic aortic aneurysms: a reappraisal. Arch Surg. 1983;118(5):583-8. 6687677 http://dx.doi.org/10.1001/archsurg.1983.01390050059011
7 Gross C, Harringer W, Mair R, Wimmer-Greinecker G, Klima U, Brücke P. Mycotic aneurysms of the thoracic aorta. Eur J Cardiothorac Surg. 1994;8(3):135-8. 8011346 http://dx.doi.org/10.1016/1010-7940(94)90169-4.
8 Malouf JF, Chandrasekaran K, Orszulak TA. Mycotic aneurysms of the thoracic aorta: a diagnostic challenge. Am J Med. 2003;115(6):489-96. 14563506 http://dx.doi.org/10.1016/S0002-9343(03)00394-2.
9 McNamara MF, Finnegan MO, Bakshi KR. Abdominal aortic aneurysms infected by Escherichia coli. Surgery. 1985;98(1):87-92. PMid:3892748
10 McNamara MF, Roberts AB, Bakshi KR. Gram-negative bacterial infection of aortic aneurysms. J Cardiovasc Surg. 1987;28(4):453-5. PMid:3298284
11 Rasoul S, Jaspers RMMJ Jr, van Wijngaarden J. Mycotic aneurysm of the aortic arch. Neth Heart J. 2011;19(9):397-8. 21487749 http://dx.doi.org/10.1007/s12471-011-0109-1
12 Finseth F, Abbott WM. One-stage operative therapy for Salmonella mycotic abdominal aortic aneurysm. Ann Surg. 1974;179(1):8-11. 4817880 http://dx.doi.org/10.1097/00000658-197401000-00002
13 Chan FY, Crawford ES, Coselli JS, Safi HJ, Williams TW Jr. In situ prosthetic graft replacement for mycotic aneurysm of the aorta. Ann Thorac Surg. 1989;47(2):193-203. 2919901 http://dx.doi.org/10.1016/0003-4975(89)90268-3.
14 Lin CH, Hsu RB. Primary infected aortic aneurysm: clinical presentation, pathogen, and outcome. Acta Cardiol Sin. 2014;30(6):514-21. PMid:27122829
15 Parkhurst GF, Dekcer JP. Bacterial aortitis and mycotic aneurysm of the aorta. Am J Pathol. 1955;31(5):821-35. PMid:13248917
16 Brown SL, Busuttil RW, Baker JD, Machleder HI, Moore WS, Barker WF. Bacteriologic and surgical determinants of survival in patients with mycotic aneurysms. J Vasc Surg. 1984;1(4):541-7. 6436514 http://dx.doi.org/10.1016/0741-5214(84)90040-5.
17 Mukherjee JT, Nautiyal A, Labib SB. Mycotic aneurysms of the ascending aorta in the absence of endocarditis. Tex Heart Inst J. 2012;39(5):692-5. PMid:23109770
18 Kuki S, Taniguchi K, Miyagawa S, Takano H. Multiple mycotic arch-thoraco-abdominal aortic aneurysms: a successful case of in situ graft replacement. Eur J Cardiothorac Surg. 2000;17(2):184-6. 10731656 http://dx.doi.org/10.1016/S1010-7940(99)00362-0.
19 Gomes MN, Choyke PL. Infected aortic aneurysms: CT diagnosis. J Cardiovasc Surg. 1992;33(6):684-9. PMid:1287005
20 Garcia SS, Figueiredo EG Teixeira MJ. Aneurismas infecciosos: incidência, diagnóstico e tratamento. Arq Bras Neurocir. 2011;30(3):146-9.
21 Quiñones-Baldrich WJ, Nene SM, Gelabert HA, Moore WS. Rupture of the perivisceral aorta: atherosclerotic versus mycotic aneurysm. Ann Vasc Surg. 1997;11(4):331-41. 9236987 http://dx.doi.org/10.1007/s100169900057
22 Hagendoorn J, de Vries JPPM, Moll FL. Primary infected, ruptured abdominal aortic aneurysms: what we learnedin 10 years. Vasc Endovascular Surg. 2010;44(4):294-7. 20403952 http://dx.doi.org/10.1177/1538574410363746
23 Kan CD, Lee HL, Yang YJ. Outcome after endovascular stent graft treatment for mycotic aortic aneurysm: a systematic review. J Vasc Surg. 2007;46(5):906-12. 17905558 http://dx.doi.org/10.1016/j.jvs.2007.07.025
24 Oderich GS, Panneton JM, Bower TC, et al. Infected aortic aneurysms (aggressive presentation, complicated early outcome, but durable results). J Vasc Surg. 2001;34(5):900-8. 11700493 http://dx.doi.org/10.1067/mva.2001.118084
25 Hirst AE Jr, Affeldt J. Abdominal aortic aneurysm, with rupture into the duodenum: a case report of eight cases. Gastroenterology. 1971;17(4):504-14. PMid:14823205
26 Parry DJ, Waterworth A, Kessel D, Robertson I, Berridge DC, Scott DJ. Endovascular repair of an inflammatory abdominal aortic aneurysm complicated by aortoduodenal fistulation with an unusual presentation. J Vasc Surg. 2001;33(4):874-9. 11296345 http://dx.doi.org/10.1067/mva.2001.112328
27 Romera Barba E, Sánchez Pérez A, Bertelli Puche J, Duque Pérez C, Vazquez Rojas JL. Primary aorto-duodenal fistula: a rare but potentially fatal cause of gastrointestinal bleeding. Cir Esp. 2015;93(2):121-3. PMid:25205236
28 Lemos DW, Raffetto JD, Moore TC, Menzoian JO. Primary aortoduodenal fistula: a case report and review of the literature. J Vasc Surg. 2003;37(3):686-9. 12618713 http://dx.doi.org/10.1067/mva.2003.101
29 Ikonomopoulos DC, Spanos PK, Lazarides DP. Pathogenesis of aortoenteric fistula: an experimental study. Int Angiol. 1986;5(1):33-7. PMid:2942612
30 Bunt TJ. Synthetic vascular graft infections. II graft-enteric erosions and graft-enteric fistulas. Surgery. 1983;94(1):1-9. PMid:6857503
31 Sweeney MS, Gadacz TR. Primary aortoduodenal fistula: manifestations, diagnosis and treatment. Surgery. 1984;96(3):492-7. PMid:6236571
32 Korkut AK, Arpinar E, Yasar T, Guney D. Primary aortoduodenal fistula complicated by abdominal aortic aneurysm. J Cardiovasc Surg. 2000;41(1):113-5. PMid:10836235
33 Morales Ruiz J, Selfa Muñoz A, Salmerón Escobar J. Hypovolemic shock caused by an aortoenteric fistula: an unusual cause of gastrointestinal bleeding. Gastroenterol Hepatol. 2011;34(8):586-7. 21592623 http://dx.doi.org/10.1016/j.gastrohep.2011.03.008
34 Vu QD, Menias CO, Bhalla S, Peterson C, Wang LL, Balfe DM. Aortoenteric fístulas: CT features and potencial mimics. Radiographics. 2009;29(1):197-209. 19168845 http://dx.doi.org/10.1148/rg.291075185
35 Lemos DW, Raffetto JD, Moore TC, Menzoian JO. Primary aortoduodenal fístula: a case report and review of the literature. J Vasc Surg. 2003;37(3):686-9. 12618713 http://dx.doi.org/10.1067/mva.2003.101
36 Ihama Y, Miyazaki T, Fuke C, et al. An autopsy case of a primary aortoenteric fístula: a pitfall of the endoscopic diagnosis. World J Gastroenterol. 2008;14(29):4701-4. 18698688 http://dx.doi.org/10.3748/wjg.14.4701
37 Ahmad F, Turner SA, Torrie P, Gibson M. Iatrogenic femoral artery pseudoaneurysms: a review of current methods of diagnosis and treatment. Clin Radiol. 2008;63(12):1310-6. 18996260 http://dx.doi.org/10.1016/j.crad.2008.07.001
38 Corriere MA, Guzman RJ. True and false aneurysms of the femoral artery. Semin Vasc Surg. 2005;18(4):216-23. 16360579 http://dx.doi.org/10.1053/j.semvascsurg.2005.09.008
39 Qiu J, Zhou W, Zhou W, et al. The treatment of infected femoral artery pseudoaneurysms secondary to drug abuse: 11 years of experience at a single institution. Ann Vasc Surg. 2016;36:35-43. 27427346 http://dx.doi.org/10.1016/j.avsg.2016.03.030
Notes
Author notes
Correspondence Geanete Pozzan Cardiovascular Pathology - Pathological Sciences Department - Faculdade de Ciências Médicas da Santa Casa de São Paulo Rua Riskallah Abib, 292 - São Bernardo do Campo - São Paulo/SP - Brazil CEP: 09771-100 Phone: +55 (11) 4121-8419 pozzange@gmail.com
Conflict of interest declaration