ABSTRACT: Primary pelvic hydatid cysts are a rare entity and are often overlooked as a differential diagnosis of a pelvic-space-occupying lesion particularly in non-endemic regions. Unpreparedness and a hasty decision on the surgical approach may end in life-threatening complications and systemic dissemination of the disease. We report the case of a 55-year-old postmenopausal woman with a history of two previous unsuccessful surgeries to remove pelvic cystic lesions due to dense adhesions between the surrounding gut wall, bladder, and the cyst wall. Clinical and imaging findings failed to diagnose the nature of the cysts, and a laparotomy was contemplated. On the third surgical attempt, the clinical suspicion was considered and by meticulous dissection the cysts were removed thoroughly without undue complications. In the postoperative follow-up period there was no sign of disease recurrence or dissemination.
Keywords:EchinococcosisEchinococcosis,Ovarian CystsOvarian Cysts,ZoonosesZoonoses.
Article / Clinical Case Report
Primary pelvic hydatid cyst in a postmenopausal female: a surgical challenge
Hospital Universitário da Universidade de São Paulo
Received: 10 February 2017
Accepted: 24 April 2017
Hydatid cyst or echinococcosis is a parasitic infection caused by the larval stage of the cestode Echinococcus, especially the genus Echinococcus granulosus. Generally the worm maintains a dog-sheep cycle, and, occasionally, humans become the intermediate host. The disease is mainly prevalent in areas where humans live in close association with dogs and cattle. Although any organ may get infected, primary involvement of the pelvic cavity is very rare. We report an unusual case of hydatid cysts of the pelvis involving the ovary in a postmenopausal woman, which mimicked ovarian neoplasm.
A 55-year-old multiparous woman from a rural area of West Bengal presented to the Gynecology Outpatient Department with a 3-year history of insidious onset of lower abdominal pain and an abdominal mass. She was housewife in a family of low socioeconomic condition and with no history of living in close association with cattle. There was no history of fever, nausea, vomiting, abnormal vaginal bleeding, weight loss, or alteration of bladder and bowel habits. She was a known diabetic patient on regular insulin. She had undergone a hysterectomy for uterine fibroids 15 years ago. She underwent a laparotomy twice in the last 2 years for persistent lower abdominal pain associated with a pelvic mass, which was hindered in both instances due to dense adhesions.
On clinical examination, there was no pallor, icterus, or lymphadenopathy. Abdominal examination revealed a non-tender supra-pubic lump of about 12 cm, which was cystic in consistency, with ill-defined margins and restricted mobility. There was no organomegaly or evidence of ascites. On speculum examination, the vaginal vault was healthy. Bimanual examination confirmed a cystic fullness of around 10 cm with restricted mobility.
Ultrasonography was suggestive of a large unilocular cyst of 9.4 × 11.6 × 7.8 cm in the retrovesical region displacing the vesical wall anteriorly. The contrast enhanced computed tomography (CECT) of the abdomen showed a complex smooth-walled cystic lesion of 112 × 103 × 90 mm in the retrovesical region of the pelvis with few eccentric thin septae in the postero-left-lateral aspect of the cyst (Figure 1A and 1B). No other mural nodule/soft tissue component/calcifications were seen within the lesion. The magnetic resonance imaging (MRI) evidenced a smooth-walled cystic space occupying lesion (SOL) measuring 104 × 99 × 93 mm in the pelvis with a few eccentric intracystic septae without any lymphadenopathy or free fluid in the peritoneal cavity (Figure 1C and 1D). Liver enzymes, other biochemical and hematologic parameters, and tumor markers (i.e. CA125, beta HCG, alpha-fetoprotein) were within normal limits.
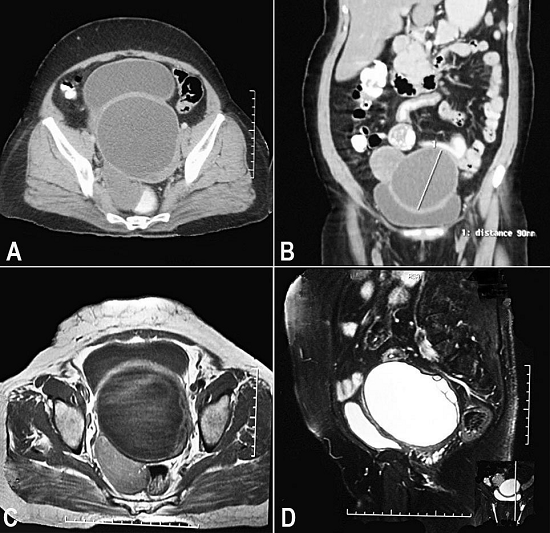
Figure 1
Axial (A) and coronal (B) views of contrast enhanced computed tomography of the abdomen showing a well-defined smooth-walled (maximum wall thickness approximately 5 mm) cystic lesion of 112 × 103 × 90 mm in the retrovesical region of pelvis. A few eccentric thin septae are seen at the postero-left-lateral aspect of the cyst. No abnormal mural nodule/soft tissue component/calcification is seen within the lesion. Axial T2W fat suppressed (C) and sagittal T1W fast spin echo sequences (D) magnetic resonance imaging (plain) of the pelvis showing a well-defined smooth-walled cystic lesion (hypointense in T1W and hyperintense in T2W) in the pelvis just posterior to the urinary bladder. A few thin septae were noted at the postero-left-lateral aspect within the lesion. No features of perilesional inflammation were seen.
Since most of the imaging findings were consistent with an ovarian neoplasm, an exploratory laparotomy was performed. A cystic mass (8 × 8 cm) of pelvic origin was found buried under the loops of intestine and adherent to the sigmoid colon postero-laterally and to the bladder wall anteriorly. By sharp dissection, the bladder and gut walls were separated from the cyst wall. During the surgical procedure, an accidental injury broke up the cyst wall allowing the drainage of some cystic fluid, revealing many scolices and yielding the macroscopic diagnosis of hydatid cyst. After sealing the puncture site with hemostatic clamps, the whole mass was carefully enucleated (Figure 2A). Underneath the sigmoid colon, another small palpable lump was dissected and it appeared to be the right ovary with cystic changes. Although left ovary appeared normal it was also removed. Thorough peritoneal lavage was performed with saline followed by external abdominal drainage. The surgical specimen showed gross features of an echinococcal cyst (Figure 2B), and microscopic examination revealed the ectocyst, the pericyst, and the hydatid scolices (Figure 3). Her postoperative period was uneventful and she was prescribed albendazole (400 mg) tablets twice daily for 1 month. At regular follow-up for 6 months, no further recurrence was found clinically or on imaging.
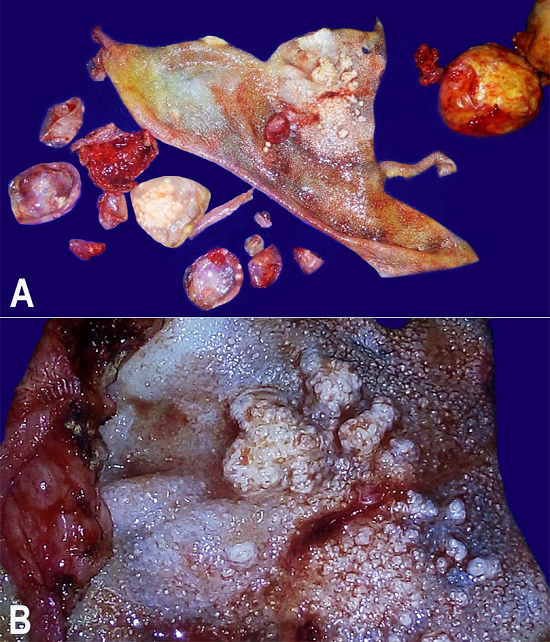
Figure 2
Gross examination of the surgical specimen. A - Panoramic cut view of the large “mother cyst” with a white, thick, cyst wall and multiple “daughter cysts”; B - Magnified view of the inner cyst wall (germinal layer) with numerous embedded scolices.
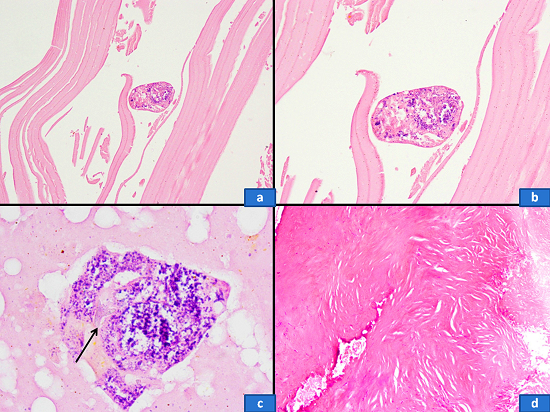
Figure 3
Photomicrography of the cystic lesion. a - The lamellate membrane forming the ectocyst. An occasional hydatid scolex was also noted (H&E, 200X); b - The scolex contained hydatid micronuclei (H&E, 400X); c - Occasional scolex showed a hydatid hooklet as a refractile structure (H&E, 1000X); d - The pericyst showed thick, focally calcified, fibrocollagenous tissue (H&E, 200X).
Echinococcus granulosus is a 5 mm long worm that completes its life cycle in dogs as a definitive host and sheep, goats, cattle, or pigs as intermediate hosts. Humans are accidentally infected following ingestion of the eggs, which are excreted through the feces of the definitive hosts. The eggs, in the form of oncospheres, penetrate through the intestinal wall and disseminate via blood and lymphatic circulation to different organs where they grow to form hydatid cysts. On average, it takes 5-20 years for the cyst to enlarge to the point of causing symptoms.
Hydatid cysts are a disease that has worldwide prevalence, with the highest being in the Middle East, Australia, Argentina, New Zealand, Chile, Africa, Eastern Europe, and the Mediterranean region (particularly Lebanon and Greece).1 Although it is thought that the worldwide prevalence and incidence of cystic Echinococcosis has decreased considerably over the past few decades, certain areas with a previously low level of infection have still shown an emergence of the disease.2 Some parts of India—particularly north and central India (e.g. Kashmir, Andhra Pradesh, Tamil Nadu)—also show a high prevalence. The annual incidence ranges from ≤1-200/100,000 population.3 However, detailed epidemiological data from India are scarce. A hospital-based retrospective study from Kolkata (Eastern India), revealed that the median age of patients is 33 years with female preponderance.4 Worldwide, the increased female prevalence is considered to be mainly due to their participation in activities predisposing them to infections (e.g. farming and animal breeding). Low public awareness about the fact that ingestion of contaminated food spreads the disease, is one of the most important factors causing the transmission of infection in endemic areas of India.5 There are several other risk factors, such as rural area, poor economic condition, and low education level—all of which were present in our case. There was no history of farming or close association with dogs and cattle. Therefore, cystic echinococcosis was not initially considered as a possible differential diagnosis.
Incidental diagnosis is quite frequent in asymptomatic patients while imaging for other purposes. However, symptoms are mainly due to (i) pressure; (ii) rupture into the pleural or peritoneal cavity; (iii) secondary infection; or (iv) allergic reaction.6 Echinococcal cysts are mostly found in the liver (60-70%), followed by the lungs (10-25%), the spleen, kidney, brain, bones, heart, and ovary.7 The incidence of hydatid cysts in female reproductive organs is very low (0.5%),8 where it is usually found in the pouch of Douglas and within the uterine cavity. According to our literature search, only four cases of ovarian hydatid cysts have been reported in English in the last 10 years. Peritoneal hydatid cysts, secondary to rupture from other organs, occurs in 13% of cases;1 however, primary peritoneal hydatid cysts are extremely rare.9 Common gynecological symptoms due to peritoneal or ovarian hydatid cysts are abdominal pain, abdominal lump, menstrual irregularities, infertility, pressure symptoms over adjacent organs (i.e. bladder and rectum), and obstructed labor.10 Pelvic echinococcosis may resemble malignancy and a mimic multicystic ovarian neoplasm.11 Our patient presented with a large abdomino-pelvic mass and a dull, aching, lower abdominal pain. Her advanced age, post-hysterectomy status, and repeated imaging findings of a large complex, cystic mass with intracystic septation raised the possibility of an ovarian neoplasm. The diagnosis of hydatid disease was not firstly considered because of its rarity and the absence of consistent epidemiology (i.e. close association with dogs and raising sheep, goats, cattle, or pigs).
Different imaging modalities play an important role in the diagnosis of hydatid disease. Plain abdominal x-rays may show calcifications of the cyst wall.12 Ultrasonography (USG) is cost effective and useful in identifying cyst membrane, septa, and hydatid sand within the cyst, presenting the sensitivity ranging between 93% and 98%.13 Hydatid cysts are classified by the USG into six category types: (i) type 1: univesicular and <50 mm in diameter; (ii) type 2: univesicular with a prominent laminated layer and a tendency to have positive serological testing for echinococcus; (iii) type 3: cysts with prominent laminations and daughter cysts, and a highly positive serological test; (iv) type 4: appears as solid mass; (v) type 5: degenerated and calcified cysts; (vi) type 6: multiple cysts with daughter cysts involving one or more organs.13,14 CT yields better delineation of the cyst and the disease extension showing a sensitivity of 90-100%.15 Serological tests are also important and usually involve a screening test using ELISA or an indirect hemagglutination test followed by confirmation using an immunoblot or gel diffusion test (sensitivity 60-90%).1 False positive results may occur in the case of cysticercosis. However, in our case, the imaging failed to identify the nature of the cyst, and serological tests were not carried out due to the lack of clinical suspicion.
An echinococcus cyst consists of an outer acellular laminated membrane white in color (like a hard-boiled egg), which is called an ectocyst, and an inner germinal layer called an endocyst. The germinal layer is cellular, consists of nuclei embedded in a protoplasmic mass, produces brood capsules with scolices, and secretes hydatid fluid and forms the outer layer. When ruptured, the draining fluid can give rise to an anaphylactic shock due to high antigenicity. Hosts tissue reaction produces a thick fibrous layer outside the ectocyst, which is known as a pericyst.
According to the World Health Organization, surgery is the treatment of choice for all symptomatic hydatid cysts.16 Total cystectomy, whenever possible, is the gold standard. However, for peritoneal cysts that are densely adhered to the surrounding viscera, uncovering and draining can be performed as an alternate therapeutic approach. Nevertheless, the abdominal cavity should be isolated with gauzes soaked in 20% saline to prevent secondary hydatosis and any allergic reaction.17 The most common intra- and postoperative complications are due to accidental cyst rupture and secondary infection, which can be markedly reduced by preoperative medical treatment with albendazole.1,11 In our case, although there were adhesions and an accidental rupture of a cyst wall, all the contents of the cysts including its wall was removed and the abdominal cavity thoroughly washed with normal saline. Another alternative treatment is PAIR (puncture, aspiration, injection, re-aspiration) therapy with concomitant anti-helminthic drugs. In PAIR therapy, initially USG-guided percutaneous aspiration of cyst is carried out followed by the injection of protoscolicidal substances (20% NaCL solutions, 95% ethanol or betadine solution). The solution is re-aspirated after a contact period of 15 min. Indications are large multiple cysts of liver, spleen, kidney, or bones; inoperable cases; and relapse after surgery. The efficacy of exclusive medical treatment is limited to a success rate of 40%.
The risk of recurrence following surgery ranges between 8% and 22%, and cases usually occur within 2 years of the operation.18 In our case, the patient was prescribed tablets of albendazole 800 mg daily for 1 month, and there was no evidence of recurrence at the sixth month of follow-up.
Although the incidence of hydatid cysts in the female reproductive system is almost less than 0.5%,9 an increasing number of cases are being reported even from the non-endemic areas, presenting a challenging diagnosis, as in our case. Hence, primary pelvic and ovarian hydatid cysts should be kept in mind while evaluating a case of pelvic cystic SOL. Preoperative suspicion, proper intra-operative dissection, and postoperative follow-up may eliminate the morbidity and mortality associated with the condition.
Correspondence Sayanti Paul Department of Obstetrics and Gynecology - IPGMER & SSKM Hospital Bhowanipore, Kolkata, West Bengal - India 700020 Phone: +91 983799167 ichchedana.sayanti@gmail.com

Figure 1
Axial (A) and coronal (B) views of contrast enhanced computed tomography of the abdomen showing a well-defined smooth-walled (maximum wall thickness approximately 5 mm) cystic lesion of 112 × 103 × 90 mm in the retrovesical region of pelvis. A few eccentric thin septae are seen at the postero-left-lateral aspect of the cyst. No abnormal mural nodule/soft tissue component/calcification is seen within the lesion. Axial T2W fat suppressed (C) and sagittal T1W fast spin echo sequences (D) magnetic resonance imaging (plain) of the pelvis showing a well-defined smooth-walled cystic lesion (hypointense in T1W and hyperintense in T2W) in the pelvis just posterior to the urinary bladder. A few thin septae were noted at the postero-left-lateral aspect within the lesion. No features of perilesional inflammation were seen.

Figure 2
Gross examination of the surgical specimen. A - Panoramic cut view of the large “mother cyst” with a white, thick, cyst wall and multiple “daughter cysts”; B - Magnified view of the inner cyst wall (germinal layer) with numerous embedded scolices.

Figure 3
Photomicrography of the cystic lesion. a - The lamellate membrane forming the ectocyst. An occasional hydatid scolex was also noted (H&E, 200X); b - The scolex contained hydatid micronuclei (H&E, 400X); c - Occasional scolex showed a hydatid hooklet as a refractile structure (H&E, 1000X); d - The pericyst showed thick, focally calcified, fibrocollagenous tissue (H&E, 200X).
