Article / Autopsy Case Report
Pancreatic hamartoma in a premature Trisomy 18 female
Pancreatic hamartoma in a premature Trisomy 18 female
Autopsy and Case Reports, vol. 7, no. 4, pp. 26-29, 2017
Hospital Universitário da Universidade de São Paulo
Received: 14 May 2017
Accepted: 12 November 2017
ABSTRACT: Pancreatic hamartomas are extremely rare tumors in adults and even more so in children. They are lesions characterized by acinar, islet and ductal components found in varying proportions and in a disorganized pattern. We report a case of a premature female with trisomy 18 diagnosed by amniocentesis. The newborn was delivered by cesarean section at thirty-three weeks of gestation and expired within one hour of birth. Postmortem examination exhibited numerous features associated with Trisomy 18 including lanugo on the torso and arms, micrognathia, microstomia, left low-set ear with small flat pinna, closed ear canal, clenched fists with overlapping fingers, rocker-bottom feet, narrow pelvis, large right diaphragmatic hernia and left pulmonary hypoplasia. Microscopic examination of the pancreas revealed an area, 1.2 cm in greatest dimension, with branching ducts and cysts lined by cuboidal epithelium intermingled within primitive mesenchymal proliferation and exocrine glands. The cysts measured up to 0.2 cm and were surrounded by a collarette of proliferating spindle cells as highlighted by Masson’s trichrome stain. A diagnosis of pancreatic hamartoma was rendered. A total of thirty-four cases of pancreatic hamartomas have been reported in the literature including twenty-seven in adults, five in children and two in newborns. Our case may be the third pancreatic hamartoma reported in association with Trisomy 18. We recommend that careful examination of the pancreas be performed in individuals with Trisomy 18 to further characterize this lesion as one of the possible abnormal findings associated with this syndrome.
Keywords: Edwards Syndrome, hamartoma, pancreas, pancreatic neoplasm, Trisomy 18.
CASE REPORT
The premature female was born at 33 weeks of gestation to a 25 years old mother, gravida 3 para 2, with a history of pre-eclampsia in the previous pregnancy. In this pregnancy, the fetus was found to have Trisomy 18 karyotype by amniocentesis. Fetal ultrasound at 21 weeks gestational age showed left displacement of the heart and absent nasal bone. The mother presented to at 33 weeks of gestation for premature rupture of membranes and contractions. Fetal ultrasound performed at that time showed normal amniotic fluid volume, intrauterine growth restriction with estimated fetal weight of 1519 grams (less than the 10th percentile for gestational age), right side congenital diaphragmatic hernia with liver herniating into the thorax, left mediastinal shift of the heart, hypoplastic left lung, and clenched hands with limited movement of upper extremities suggestive of arthrogryposis. Doppler interrogation of umbilical artery was normal. The baby was delivered by cesarean section.
At the time of birth, the infant was floppy and apneic for approximately 8 minutes requiring stimulation and intermittent positive pressure ventilation. APGAR score was 1, 2 and 4 at 1, 5 and 10 minutes, respectively. Intubation and resuscitation were not performed given the poor prognosis, and she expired at less than one hour of life.
On postmortem examination, the newborn exhibited numerous features associated with Trisomy 18 which included the following: lanugo on the torso and arms, micrognathia, microstomia, left low set ear with small flat pinna and closed ear canal, bilateral clenched fists with index and fifth fingers overlapping the third and fourth fingers, rocker-bottom feet, narrow pelvis, large right diaphragmatic hernia, and left pulmonary hypoplasia. No cardiovascular and genitourinary defects were identified. The placenta was small for the gestational age weighing 250 grams (normal is 342 grams for 33 weeks of gestation).
Although no abnormalities were noted on gross examination of the pancreas, microscopic examination revealed an area measuring 1.2 cm in greatest dimension characterized by branching duct-like structures and cysts lined by cuboidal epithelium that measured up to 0.2 cm. Primitive mesenchymal proliferation and exocrine gland formation were found in between (Figure 1A, 1B and 1C). A proliferation of spindle cells forming a collarette around the cysts were highlighted by Masson’s trichrome stain (Figure 1D). A diagnosis of pancreatic hamartoma was rendered. The uninvolved pancreatic parenchyma showed normal exocrine and endocrine components, congestion and extramedullary hematopoiesis.
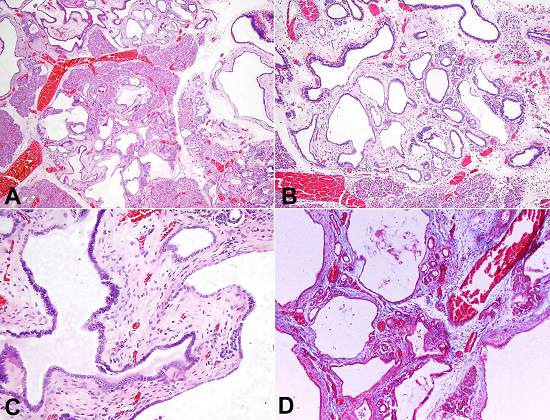
Figure 1
Photomicrography of the pancreatic hamartoma, showing in A, B and C - cysts and ducts lined by cuboidal epithelium, primitive mesenchymal proliferation and exocrine gland formation (H&E, 10X, 20X and 40X respectively); in D - mesenchymal proliferation by Masson’s trichrome stain (20X).
DISCUSSION
Pancreatic hamartomas are characterized as lesions with acinar, islet and ductal cellular components present in varying proportions and in a disorganized pattern. The exocrine and endocrine pancreatic tissue is well differentiated, and the ductal elements are irregularly branching or cystically dilated. The endocrine component may be seen as single cells or small group of cells interspersed within the exocrine component. Fibrous tissue, fat and primitive mesenchyme is often found in between these elements.1-6
Pancreatic hamartomas are extremely rare tumors in adults but are even more so in children. Only a total of thirty-four cases of pancreatic hamartomas have been reported in the literature including twenty-seven in adults, five in children and two in newborns. Two of the five reported in children had Trisomy 18 and one adult had SAPHO syndrome, a chronic inflammatory disorder.1-23
In 1960, Smith et al.23 originally described a pancreatic lesion in a two-month old girl with Trisomy 18 at the time of autopsy. The lesion was located at the tail of the gland, and was characterized by acinar and islet pancreatic tissue within a dense fibrous network and dilated ducts. In 1964, Rohde22 and colleagues reported a second pancreatic lesion in a Trisomy 18 child. In his case, a two-and-a-half-month girl was found to have an unusual pancreatic lesion described as having prominent immature connective tissue with sparse or absent acini, and distorted, often dilated ducts. These areas were surrounded by normal pancreatic tissue. The histologic description of these lesions is very similar to those seen in our case, and, despite the fact that these lesions were not labeled as hamartomas, their microscopic description seem to indicate that they may be considered hamartomas as well. Our case then, may be the third pancreatic hamartoma reported in association with Trisomy 18 Syndrome. Ectopic pancreas has also been reported in association with Trisomy 18.22-24 It is, therefore, recommended that careful examination of the pancreas be performed in individuals with Trisomy 18 to further characterize these lesions as one of the possible abnormal findings associated with Trisomy 18 Syndrome.
REFERENCES
1 Burt TB, Condon VR, Matlak ME. Fetal pancreatic hamartoma. Pediatr Radiol. 1983;13(5):287-9. 6622090 http://dx.doi.org/10.1007/BF00973350
2 Flaherty MJ, Benjamin DR. Multicystic pancreatic hamartoma: a distinctive lesion with immunohistochemical and ultrastructural study. Hum Pathol. 1992;23(11):1309-12. 1427759 http://dx.doi.org/10.1016/0046-8177(92)90301-I.
3 Izbicki JR, Knoefel WT, Müller-Höcker J, Mandelkow HK. Pancreatic hamartoma: a benign tumor of the pancreas. Am J Gastroenterol. 1994;89(8):1261-2. PMid:8053450
4 McFaul CD, Vitone LJ, Campbell F, et al. Pancreatic hamartoma. Pancreatology. 2004;4(6):533-7, discussion 537-8. 15340246 http://dx.doi.org/10.1159/000080528
5 Pauser U, Kosmahl M, Kruslin B, Klimstra DS, Klöppel G. Pancreatic solid and cystic hamartoma in adults: characterization of a new tumorous lesion. Am J Surg Pathol. 2005;29(6):797-800. 15897746 http://dx.doi.org/10.1097/01.pas.0000157748.18591.d7
6 Nagata S, Yamaguchi K, Inoue T, et al. Solid pancreatic hamartoma. Pathol Int. 2007;57(5):276-80. 17493175 http://dx.doi.org/10.1111/j.1440-1827.2007.02090.x.
7 Anthony PP, Faber RG, Russell RC. Pseudotumours of the pancreas. BMJ. 1977;1(6064):814. 851741 http://dx.doi.org/10.1136/bmj.1.6064.814
8 Noltenius H, Colmant HJ. Excessive hyperplasia of the exocrine pancreatic tissue and Wernicke’s encephalopathy (author’s transl). Med Klin. 1977;72(50):2155-8. PMid:593198
9 Wu SS, Vargas HI, French SW. Pancreatic hamartoma with Langerhans cell histiocytosis in a draining lymph node. Histopathology. 1998;33(5):485-7. 9839177 http://dx.doi.org/10.1046/j.1365-2559.1998.0491c.x.
10 Sepulveda W, Carstens E, Sanchez J, Gutierrez J. Prenatal diagnosis of Congenital Pancreatic Cyst: Case Report and Review for the Literature. J Ultrasound Med. 2000;19(5):349-52. 10811411 http://dx.doi.org/10.7863/ultra.19.5.349
11 Pauser U, da Silva MT, Placke J, Klimstra DS, Klöppel G. Cellular hamartoma resembling gastrointestinal stromal tumor: a solid tumor of the pancreas expressing c-kit (CD117). Mod Pathol. 2005;18(9):1211-6. 15803185 http://dx.doi.org/10.1038/modpathol.3800406
12 Thrall M, Jessurun J, Stelow EB, et al. Multicystic adenomatoid hamartoma of the pancreas: a hitherto undescribed pancreatic tumor occurring in a 3-year-old boy. Pediatr Dev Pathol. 2008;11(4):314-20. 17990924 http://dx.doi.org/10.2350/07-04-0260.1
13 Sampelean D, Adam M, Muntean V, Hanescu B, Domsa I. Pancreatic hamartoma and SAPHO syndrome: a case report. J Gastrointestin Liver Dis. 2009;18(4):483-6. PMid:20076824
14 Kawakami F, Shimizu M, Yamaguchi H, et al. Multiple pancreatic hamartomas: A case report and reviews of the literature. World J Gastrointest Oncol. 2012;4(9):202-6. 23293730 http://dx.doi.org/10.4251/wjgo.v4.i9.202
15 Durczynski A, Wiszniewski M, Olejniczak W, Polkowski M, Sporny S, Strzelczyk J. Asymptomatic solid pancreatic hamartoma. Arch Med Sci. 2011;7(6):1082-4. 22328895 http://dx.doi.org/10.5114/aoms.2011.26624
16 Kim HH, Cho CK, Hur YH, et al. Pancreatic hamartoma diagnosed after surgical resection. J Korean Surg Soc. 2012;83(5):330-4. 23166894 http://dx.doi.org/10.4174/jkss.2012.83.5.330
17 Yamaguchi H, Aishima S, Oda Y, et al. Distinctive histopathologic findings of pancreatic hamartomas suggesting their “hamartomatous” nature: a study of 9 cases. Am J Surg Pathol. 2013;37(7):1006-13. 23715157 http://dx.doi.org/10.1097/PAS.0b013e318283ce4c
18 Sueyoshi R, Okazaki T, Lane GJ, Arakawa A, Yao T, Yamataka A. Multicystic adenomatoid pancreatic hamartoma in a child: Case report and literature review. Int J Surg Case Rep. 2013;4(1):98-100. 23143293 http://dx.doi.org/10.1016/j.ijscr.2012.10.001
19 Addeo P, Tudor G, Oussoultzoglou E, Averous G, Bachellier P. Pancreatic hamartoma. Surgery. 2014;156(5):1284-5. 23652195 http://dx.doi.org/10.1016/j.surg.2013.02.021
20 Inoue H, Tamed M, Yamada R, et al. Pancreatic hamartoma: a rare cause of obstructive jaundice. Endoscopy 2014;46(Suppl 1):E157-8. http://dx.doi.org/10.1055/s-0034-1364953
21 Matsushita D, Kurahara H, Mataki Y, et al. Pancreatic hamartoma: a case report and literature review. BMC Gastroenterol. 2016;16(1):3. 26762320 http://dx.doi.org/10.1186/s12876-016-0419-2
22 Rohde RA, Hodgman JE, Cleland RS. Multiple congenital anomalies in the E1-Trisomy (Group 16-18) Syndrome. Pediatrics. 1964;33:258-70. PMid:14117382
23 Smith DW, Patau K, Therman E, Inhorn S. A new autosomal trisomy syndrome: multiple congenital anomalies caused by an extra chromosome. J Pediatr. 1960;57(3):338-45. 13831938 http://dx.doi.org/10.1016/S0022-3476(60)80241-7.
24 Lewis AJ. The pathology of 18 trisomy. J Pediatr. 1964;65(1):92-101. 14178832 http://dx.doi.org/10.1016/S0022-3476(64)80557-6.
Notes
Author notes
Correspondence Patricia Isabel Delgado, MD University of Miami School of Medicine Department of Pathology and Laboratory Medicine Jackson Memorial Hospital, East Tower 1611 NW 12th Ave, Suite 2147 Miami, FL, USA 33136 Phone: 1(305) 585-6637 patricia.delgado@jhsmiami.org
Conflict of interest declaration