Letter to the Editor
Fas-ligand and caspase-3 positivity in three cases of histiocytic sarcoma: a different etiopathogenic pathway?
Fas-ligand and caspase-3 positivity in three cases of histiocytic sarcoma: a different etiopathogenic pathway?
Autopsy and Case Reports, vol. 8, no. 1, e2018001, 2018
Hospital Universitário da Universidade de São Paulo
Received: 09 September 2017
Accepted: 01 December 2017
Abstract: Histiocytic sarcoma (HS) is a rare malignant neoplasia of hematopoietic origin and unknown etiology. We studied three patients with histiocytic sarcoma reviewing the morphological and immunohistochemical aspects. We evaluated in particular, if apoptosis may be unbalanced in this disease. All cases have morphological and immunohistochemical features consistent with the diagnosis of histiocytic sarcoma. The markers CD163, CD68, vimentin, lysozyme, and S-100 were positive in all cases. Similarly, the three samples were positive for Fas-ligand and Caspase-3. It is well-known that neoplasms may induce increased levels of Fas-ligand with the blockade of the apoptosis process. In the context of HS, the increased Fas-ligand expression represents a new area for research. Indeed, it is linked to proinflammatory stimulus and, maybe with the association of an infection.
Keywords: Histiocytic sarcoma, Diagnosis, Immunohistochemistry.
Dear editors,
Histiocytic sarcoma (HS) is a rare hematopoietic malignancy of unknown etiology.1,2 The diagnosis is made after ruling out other neoplasms such as melanoma, undifferentiated carcinomas, and anaplastic lymphomas.2,3 The use of immunohistochemical markers such as CD163, CD68, and lysozyme is mandatory for a definitive diagnosis.2 The histiocytic marker CD163 has been proposed as the most specific.2,3
We present the immunohistochemical behavior of apoptotic marker pathways in three cases of HS under our service. We believe that the apoptosis' mechanisms might contribute to understanding the etiology of this disease.
The first case is a 30-year-old man with painful cutaneous lesions on the left thorax, intermittent fever, unmeasured weight loss and bilateral cervical/axillary adenomegaly. The second case is a 66-year-old woman with four months of non-painful left cervical adenomegaly, without B-symptoms. She had a melanoma 23 years ago. And, the third patient is a 42-year-old woman with abdominal pain and a palpable mass in the small intestine.
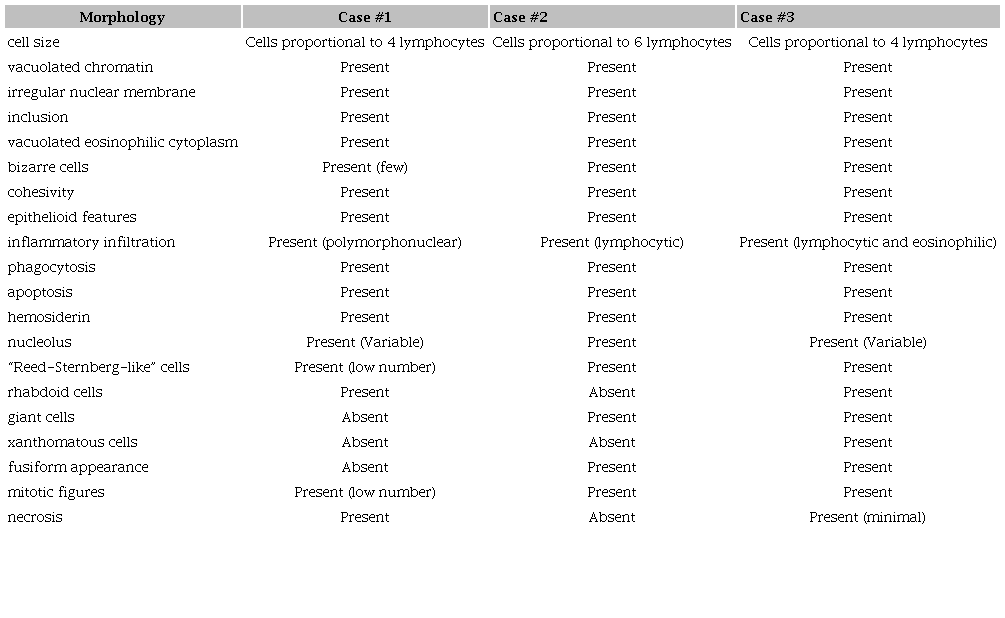
We reviewed the morphological aspects, using the criteria supported by Hornick et al.4 These histopathological aspects were (i) cell size; (ii) chromatin pattern; (iii) evident nucleolus; (iv) nuclear inclusion; (v) cytoplasm aspect; (vi) presence of bizarre types of cell; (vii) the presence of giant cells; and (viii) the presence of Reed-Sternberg-like cells or rhabdoid cells or xanthomized cells. Other relevant morphological aspects, also cited by literature, are cohesivity, epithelioid features, fusiform features, inflammatory infiltrate, phagocytosis, apoptosis, mitosis, necrosis, and hemosiderin.2,4
We also performed the immunohistochemistry study with cytokeratin (AE1/AE3), EMA, Vimentin, S100 protein, HMB-45, Melan-A, Myeloperoxidase (MPO), CD21, CD1a, CD35, CD45, CD15, CD30, CD20, CD3, CD68, Lysozyme, and CD163. Additionally, we used Fas Ligand, Bax and Caspase-3 to study apoptosis. For immunohistochemical characterization, the antibody-staining pattern was considered and classified as either diffuse or focal.
We confirmed the diagnosis of HS in all three cases. Twelve relevant morphological criteria were simultaneously present in all the HS cases2-4 with minimal variability. There were: medium to large cell size, vacuolated chromatin, irregular cell membrane, intranuclear inclusion, vacuolar eosinophilic cytoplasm, the presence of bizarre cells, cohesivity, epithelioid features, phagocytosis, inflammatory background, apoptosis and hemosiderin (Table 1).
The final immunohistochemical characterization of all the HS cases is summarized in Table 2. CD163, CD68, vimentin, and lysozyme were positive in all patients. Only S-100 protein was not uniform, and its positivity was focal or irregular. Concerning apoptosis, two markers showed positive results in all three cases: Fas-ligand and caspase-3. The Bax marker was negative in all of the cases.Figure 1 and 2 show some images of the results.
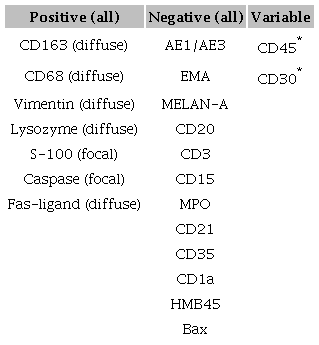
* A cutaneous lesion was present only in case 01.
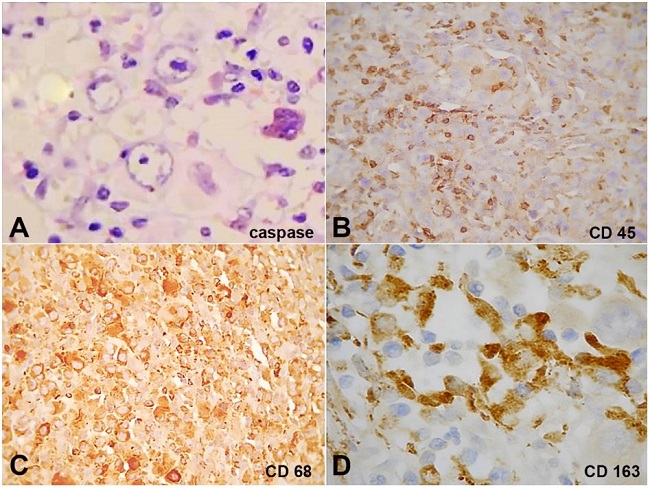
Figure 1
Immunohistochemical markers. A - Caspase 3, focal, 200x; B - CD45, in histological section of skin, 200x; C - CD68, diffuse, 400x; D - CD163, diffuse, 400x.
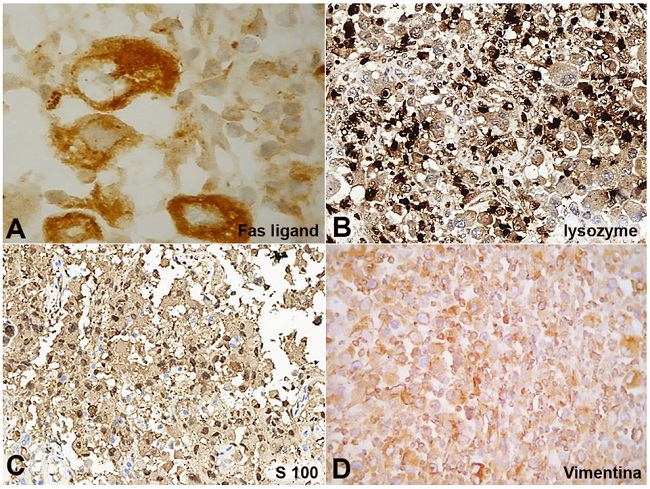
Figure 2
Immunohistochemical markers. A - Fas-ligand, diffuse, 1000x; B - Lysozym, diffuse, 200x; C - S-100, focal, 200x; and D - Vimentin, diffuse, 400x.
Fas and Fas-ligand are transmembrane glycoproteins, whose connections can start apoptosis.5,6 Fas and Fas-ligand are also named CD95 and CD95L and are part of the tumor necrosis factor receptor family. Natural ligand (CD95L or Fas-ligand) or agonistic antibodies may stimulate the receptor.7 Fibroblasts, epithelial cells, tumor cells and hematopoietic cells have Fas receptors. Inflammatory, necrosis and bacterial infections may activate the Fas-ligand. In microbial infections, there is a marked proinflammatory scenario with activation of Fas-ligand and therefore Fas-ligand induced apoptosis.5
Fas/FasL has an important function in killing cancer cells. Cytotoxic lymphocytes recognize the tumor cells in an antigen-specific mechanism and these cells are attacked by direct systems: perforin/granzyme or the CD95/CD95L engagement. Other pathways activated an upregulation of TNFα and INFγ, which cause more expression of Fas receptors, inducting cell death. However, the Fas/Fas ligand system plays many roles in cancer. Indirect roles are the suppression of the immune response in the cancer micro-environment by either tumor-generated CD95L or by CD95L expressed by endothelial cells. The main direct effect is the promotion of tumor growth and invasiveness. Another point is the low baseline level of CD95/CD95L signaling in cancer cells which is important for their survival.7
Cancers such as hepatocellular and esophageal carcinoma, lymphoma, melanoma and gastric adenocarcinoma express Fas-ligand, inducing apoptosis by the T-cell, which presents the Fas receptor. Fas-ligand was primarily recognized as associated with the remodeling tissue and deletion of potentially autoreactive cell in thymus, maintaining immune tolerance. Thus, the apoptosis process occurs, again, in a context of inflammation, in this case, due to a neoplasm.5,6
The relationship of apoptosis reduction and lymphoma was first described in the follicular lymphoma due to positive Bcl-2, an anti-apoptotic protein, followed by other neoplasms with imbalances of pro-apoptotic and anti-apoptotic factors.6 In some types of leukemia the Fas-ligand is expressed on the plasmatic membrane of the neoplastic cells, which bind to T-cell with Fas expression. Due to this coupling, apoptosis ensues, and the immune action against the tumor is blocked.5,6
Here, we present an unusual finding of HS. Apoptosis and neoplasms have been studied for many years, but, in the context of HS, we could not find any report on this observation in humans. HS is an aggressive and a controversial disease in the literature. Therefore, supplementary knowledge about its etiology is needed.
These three cases are not conclusive due to the small series’ size. However, they represent another possibility in understanding HS. In addition to the potential site for future therapies, the inquiry remains on finding out what factors may stimulate Fas-ligand. Infection and the inflammatory response as well as the microenvironment alterations are possibilities that may contribute to neoplasm growth.
REFERENCES
1 Swerdelow SH, Campo E, Harris ES, et al. WHO Classification of tumours of haematopoietic and Lymphoid Tissues. 5th ed. Lyon: International Agency for research on Cancer; 2017.
2 Emile JF, Abla O, Fraitag S, et al. Revised classification of histiocytoses and neoplasms of the macrophage-dendritic cell lineages. Blood. 2016;127(22):2672-81. 26966089 http://dx.doi.org/10.1182/blood-2016-01-690636
3 Swerdlow SH, Campo E, Pileri SA, et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood. 2016;127(20):2375-90. 26980727 http://dx.doi.org/10.1182/blood-2016-01-643569
4 Hornick JL, Jaffe ES, Fletcher CDM. Extranodal histiocytic sarcoma: clinicopathologic analysis of 14 cases of a rare epithelioid malignancy. Am J Surg Pathol. 2004;28(9):1133-44. 15316312 http://dx.doi.org/10.1097/01.pas.0000131541.95394.23
5 Caulfield AJ, Lathem WW. Disruption of Fas-Fas ligand signaling, apoptosis and innate immunity by bacterial pathogens. PLoS Pathog. 2014;10(8):e1004252. 25101900 http://dx.doi.org/10.1371/journal.ppat.1004252
6 Bergantini APF, Castro FA, Souza AM, Fett-Conte AC. Chronic myeloid leukemia and the Fas-FasL system. Rev Bras Hematol Hemoter. 2005;27(2):120-5.
7 Peter ME, Hadji A, Murmann AE, et al. The role of Cd95 and CD95 ligand in cancer. Cell Death Differ. 2015;22(4):549-59. 25656654 http://dx.doi.org/10.1038/cdd.2015.3
Notes
Author notes
Correspondence Cristiano Claudino Oliveira Department of Pathology - Botucatu School of Medicine (FMB) - São Paulo State University (UNESP) Distrito de Rubião Junior, s/n - Botucatu/SP - Brazil CEP: 18618-000 Phone: +55 (14) 3811-6238 cristiano_c_oliveira@hotmail.com
Conflict of interest declaration