Article / Autopsy Case Report
Disseminated toxoplasmosis in a patient with advanced acquired immunodeficiency syndrome
Disseminated toxoplasmosis in a patient with advanced acquired immunodeficiency syndrome
Autopsy and Case Reports, vol. 8, no. 1, e2018012, 2018
Hospital Universitário da Universidade de São Paulo
Received: 01 December 2017
Accepted: 24 January 2018
Abstract: Extracerebral toxoplasmosis, with pulmonary involvement and shock, is a rare form of toxoplasmosis in patients with advanced AIDS. It can mimic pneumocystosis, histoplasmosis, and disseminated tuberculosis, and should be considered in the differential diagnosis of causes of respiratory failure and fulminant disease in this group of individuals, especially in areas where the Toxoplasma gondii infection is highly prevalent and in those without proper use of antimicrobial prophylaxis. We report the case of a 46-year-old male patient who presented to the emergency department with uremia, requiring urgent dialysis. During the laboratorial investigation, the patient had confirmed HIV infection, with a low CD4+ peripheral T-cell count (74 cells/µL). During hospitalization, the patient presented drug-induced hepatitis due to trimethoprim/sulfamethoxazole in a prophylactic dose, requiring interruption of this medication. On the 55th day of hospitalization, the patient developed refractory shock and died. At the autopsy, disseminated toxoplasmosis with encephalitis and severe necrotizing pneumonia were diagnosed, with numerous tachyzoites in the areas of pulmonary necrosis.
Keywords: AIDS, Toxoplasma gondii, Autopsy.
CASE REPORT
A 46-year-old male with a history of drug, alcohol and tobacco abuse was admitted to the emergency department with altered level of consciousness. At admission, no close companion was present to provide a detailed medical history. A complete clinical examination and various laboratory tests were performed, and uremia was diagnosed. The lab tests showed a serum creatinine level of 17.2 mg/dL (reference range [RR] = 0.7-1.2 mg/dL); serum urea 318 mg/dL (RR = 10-50 mg/dL); hemoglobin 4.8 g/dL (RR: 13-18g/dL) with intense poikilocytosis; peripheral white blood cell count 5,220 cells/mm3 (RR = 4,000-11,000 cells/mm3); total peripheral lymphocyte count 400 cells/mm3 (RR = 900-3,400 cells/mm3); arterial pH 7.208 (RR: 7.37-7.44) and serum bicarbonate 6.1 mmol/L (RR: 23-29 mmol/L). A head computed tomography (CT) and a lumbar puncture were also carried out, both of which showed normal results. Dialytic treatment and adjuvant measures were then initiated, and the patient’s mental and general condition improved progressively over the next few days. The serology panel requested at admission showed two ELISA positives for HIV antibodies, non-immunization for HBV, a positive Toxoplasma immunoglobulin (Ig)G (by electrochemiluminescence, with 650.0 IU/mL [RR ≥ 3 IU/mL]) and a positive HVA IgG. Other serologic tests, including anti-hepatitis C virus (HCV) and anti-T. cruzi antibodies were negative. The HIV infection was then confirmed by Western blotting. However, the patient was not aware of his HIV status. The TCD4+ peripheral count was 74 cells/µL, TCD8+ peripheral count was, 171 cells/ µL (TCD4+/TCD8+ ratio = 0.43) and the HIV-1 RNA viral load in the blood was 66,309 copies/mL (branched DNA) or 4.82 Log. The blood cultures were negative.
On the 28th day of hospitalization, the patient manifested acute hepatitis, with significant elevation of serum canalicular enzymes (direct bilirrubin (DB) = 2.63 mg/dL [RR < 0.3 mg/dL], alkaline phosphatase (AP) = 1,248 U/L [RR: 40-129 U/L]) and liver enzymes (AST = 172 U/L [RR < 37 U/L], ALT = 447 U/L [RR < 41 U/L]) attributed to trimethoprim/sulfamethoxazole (TMP/SMX) prescribed to prevent opportunistic infections. The detection of HCV RNA by the real-time polymerase chain reaction assay was negative in the blood. These antibiotics were discontinued for 2 weeks, when the canalicular enzyme levels returned to the reference range. Combined antiretroviral therapy (ART) was not prescribed during hospitalization due to the patient’s liver dysfunction. On the 55th day of hospitalization, the patient developed refractory shock attributed to nosocomial pneumonia as he presented cough, fever, and thoracic rales (see chest x-ray and chest CT in Figure 1). Bronchoalveolar lavage (BAL) was not performed due to the poor clinical condition of the patient.
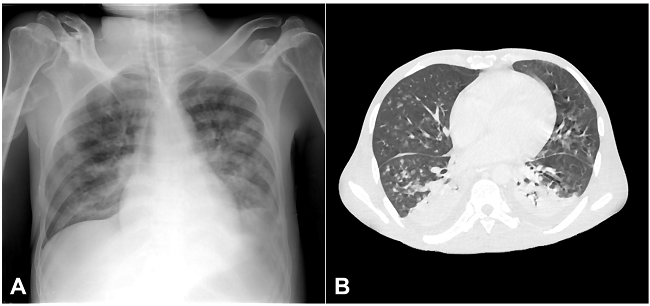
Figure 1
Radiological findings of pulmonary toxoplasmosis: A - supine chest x-ray showing diffuse bilateral pulmonary opacities and obliteration of left inferior hemithorax. Also note the tracheal tube and venous catheters; B - Axial computed tomography (lung window) showing bilateral centrilobular ground-glass opacities, consolidation of posterior portions of the lungs, and small bilateral pleural effusions.
Broad-spectrum antibiotics and vasoactive drugs were prescribed associated with dialysis and mechanical ventilation, but the patient did not respond favorably and died. The blood cultures were negative, and the microscopic analysis and culture of the sputum were negative. The autopsy was requested and was performed with the agreement of the patient’s relatives.
AUTOPSY FINDINGS
The corpse weighed 65 kg and measured 177 cm (body mass index 20.74). The external examination was unremarkable. The lungs were heavy (the right lung weighed 1,126 g and the left lung 936.0 g [RR = 400-800 g]), congested, and showed signs of anthracosis. The parenchymal cut surface was violaceous, friable, with diffuse consolidation (lung hepatization), associated with abundant purulent secretions draining from the airways. Microscopic examination revealed significant congestion and pneumonia characterized by scattered foci of necrosis, a variable number of mononuclear and polymorphonuclear inflammatory cells (Figure 2) associated with granular eosinophilic cysts and free arc-shaped structures with eccentric nuclei (measuring 7 µm) compatible with Toxoplasma gondii cysts and tachyzoites, respectively (Figure 3).
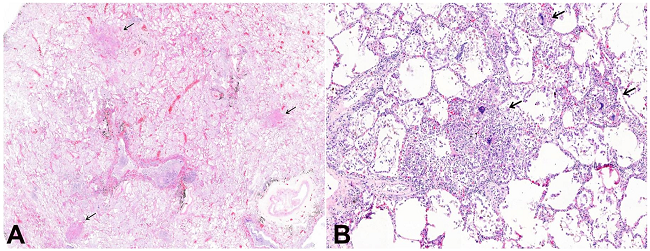
Figure 2
Micrograph of pulmonary toxoplasmosis. A - Septal congestion, alveolar edema, foci of necrosis scattered in the lung parenchyma (arrows). Anthracosis is observed (H&E, 10X); B - Interstitial pneumonia and areas of intra alveolar exudate and lytic necrosis, T. gondii forms, not seeing at this magnification (H&E, 70X).
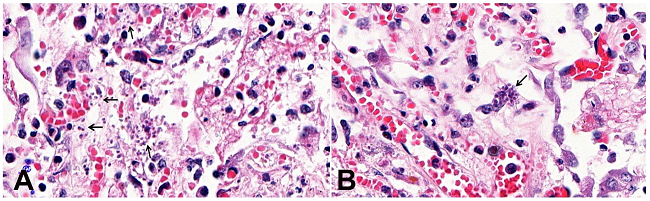
Figure 3
Micrograph of pulmonary toxoplasmosis. A - Areas of exudative pneumonia with free T. gondii tachyzoites (arrows) in the alveolar space. The inflammatory tissue reaction is weak, with few alveolar macrophages, lymphocytes, and neutrophils (H&E 630X); B - Rupture of a T. gondii cyst in a lung parenchymal cell (H&E 630X).
The cysts were periodic acid Schiff (PAS) positive and the immunohistochemistry (IHC) for the T. gondii antigen (Rabbit Polyclonal Antibody, Cell Marque™) was positive in both cysts and tachyzoites (Figure 4).
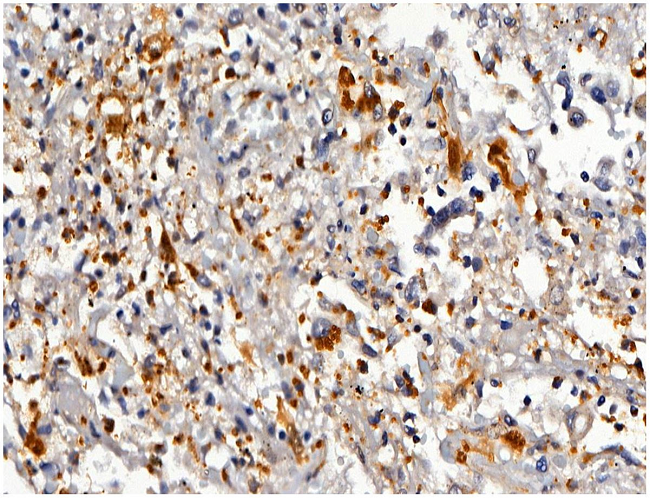
Figure 4
Micrograph of pulmonary toxoplasmosis. The immunohistochemistry revealed numerous intracellular and extracellular forms of T. gondii (Peroxidase, Rabbit Polyclonal Antibody, Cell Marque™ 400X).
The liver weighed 1,570 g (RR: 1,400-1,600 g), was extremely congested, and had a finely granular capsule. The histology demonstrated cirrhosis with dense mononuclear inflammatory infiltrates in the portal tracts, focal steatosis, centrilobular congestion, and necrosis of hepatocytes. The spleen weighed 268.0 g (RR; 100.0 -150.0 g), and the red pulp was friable. Microscopic analysis showed that the red pulp exhibited splenitis and hemorrhagic foci, and that the white pulp had an intense lymphocytic depletion. The kidneys were both atrophic, with a granular external surface. Microscopic examination revealed diffuse glomerular hyalinization, some glomeruli with collapse of the glomerular tuft (collapsing glomerulopathy), cystic dilatation of the renal tubules, chronic interstitial nephritis associated with interstitial fibrosis, and tubular atrophy (Figure 5).
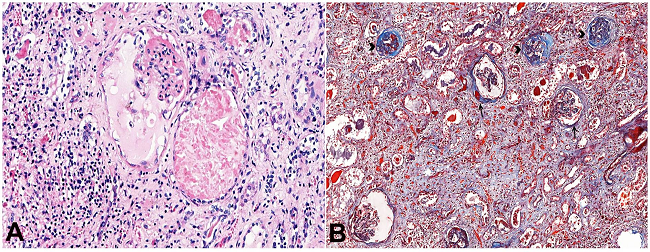
Figure 5
Micrograph showing some histopathological aspects of AIDS-associated nephropathy. A - Collapse of glomerular capillaries, dilatation of the Bowman capsule, podocytes hypertrophy, tubular cystic dilatation, and interstitial chronic nephritis with interstitial fibrosis (H&E, 200X); B - The Masson’s Trichrome stain shows collagen deposition in the glomeruli, some with thick Bowman capsule (arrows) and others with glomerular sclerosis (arrowheads). On the bottom, interstitial fibrosis is evident (Masson’s trichrome, 90X).
The bone marrow was hypercellular due to increased granulocytic-lineage cells, and numerous plasmacytes and micromegakaryocytes. The brain weighed 1,302 g (RR: 1,200-1,600 g) and was congested and edematous. Microscopy showed sparse areas of necrosis associated with congestion, edema, and discrete interstitial and perivascular lymphohistiocytic infiltrate. The T. gondii antigen was detected by immunohistochemistry in the cytoplasm of inflammatory cells, confirming central nervous system involvement (Figure 6). The heart weighed 512 g (RR: 200-350 g) and had concentric left ventricular hypertrophy, with no signs of T. gondii infection at the microscopic level. The eyes were not examined, and other organs had no significant histological features. The Grocott’s, Gram, and Ziehl-Neelsen stains were negative in the lungs, brain, heart, and bone marrow. Tissues were not collected for postmortem microbiological analysis.
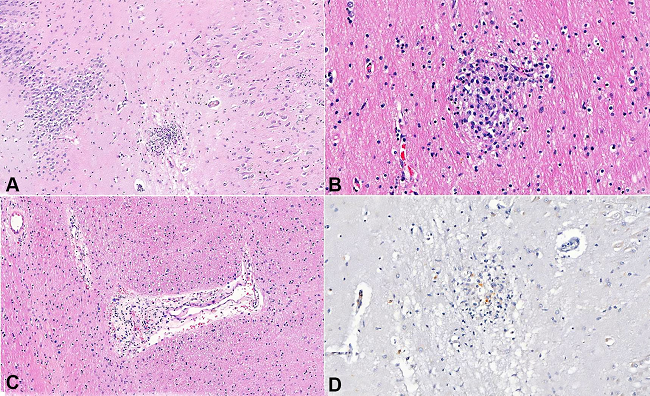
Figure 6
Micrograph of diffuse T. gondii encephalitis. A - Glial nodule in the hippocampus associated with neuropil edema (H&E 100X); B - Glial nodule in the subcortical white matter. T. gondii forms were not seeing in the inflammatory reaction composed of histiocytes and lymphocytes (H&E 300X); C - Perivascular inflammatory infiltrate with discrete hemorrhage and edema (H&E 200X); D - The T. gondii antigen was detected by IHC in the cytoplasm of rare inflammatory cells (Peroxidase, Rabbit Polyclonal Antibody, Cell Marque™ 200X).
DISCUSSION
The case reported herein depicts a male patient who was unaware of his HIV status and arrived in the emergency facility with uremia due to a late-stage of AIDS-associated nephropathy, requiring urgent dialytic treatment. The patient was also in the late stage of AIDS, according to the low peripheral TCD4+ lymphocyte count. He evolved to refractory shock after 50 days of hospitalization and died. The autopsy showed an unsuspected pre-mortem disseminated toxoplasmosis (class I of discrepancy by Goldman criteria),1 affecting the brain and the lungs. Death was a result of severe disseminated toxoplasmosis causing diffuse necrotizing pneumonia, encephalitis, and refractory septic shock. Morphological features of alcoholic liver disease in the cirrhotic stage were also found. The liver disease decompensated whilst the patient was taking prophylactic TMP/SMX, leading to drug discontinuation, which certainly influenced the reactivation and spread of T. gondii
The Brazilian public health system is internationally recognized as a model for the treatment of HIV/AIDS infection. However, cases of late-stage diagnosed HIV infection (or late entry, defined as CD4+ T cells <350 cells/μL, the presence of AIDS-defining diseases, or death attributed to HIV at diagnosis) still represent a problem in the care of people living with HIV/AIDS (PLHA) in the country.2
Toxoplasmosis has a worldwide distribution and is caused by the intracellular protozoan parasite T. gondii3 The Brazilian seroprevalence of T. gondii infection is considered one of the highest in the world; it is estimated at 36-92% among pregnant women and 56% among young military men.4 Most of the newly infected immunocompetent people tend to be asymptomatic and a latent T. gondii infection can persist for a lifetime, and can reactivate in the case of immune system impairment (such as AIDS). A few people with primary infection may have prolonged lymphadenopathy and, rarely, pneumonitis and hepatitis.3-5
AIDS patients with toxoplasmosis commonly experience T. gondii reactivation, with focal encephalitis, producing mass effect as the main clinical feature.6 The extracerebral disease may also occur, with lungs and eyes being the most common affected sites, although any organ or system can eventually be involved when the infection is disseminated.3,5,7 Even though the real incidence of cerebral toxoplasmosis—as an AIDS-defining disease in Brazil—is not well known, this incidence is declining since the introduction of ART, corresponding to 10% of all reported cases of AIDS in the São Paulo State between 2001 and 2005.8 The odds of reactivated toxoplasmosis can be up to 30% amid PLHA with low TCD4+ peripheral count (less than 100 cells/µL), with positive serum anti-T. gondii IgG, who don’t receive proper prophylaxis or ART.9 Discontinuation of TMP/SMX increases the risk of T. gondii encephalitis10 as we observed in the clinical course of this current case report.
Toxoplasma pneumonitis accounts for up to 4% of all HIV-related pulmonary diseases11 and can be associated with respiratory insufficiency, metabolic acidosis, disseminated intravascular coagulation, refractory septic shock, encephalitis, and myocarditis.3,12,13 The clinical and radiological features of T. gondii pneumonitis can mimic Pneumocystis jirovecii pneumonia.5,14,15 The most common symptoms are cough associated with pulmonary rales, fever, dyspnea, and respiratory failure. Rabaud et al.5 described 64 cases of AIDS-associated pulmonary toxoplasmosis in France, with isolated lung disease in 61%, lung disease associated with cerebral toxoplasmosis in 25%, ocular lesion in 8%, fatal septic shock in 15.6%, chest roentgenographic abnormalities in 51.5%, clinical response to treatment in 47%, recurrence in 23%, and death due to toxoplasmosis in 37% of cases. Although the diagnosis of cerebral toxoplasmosis is preemptive in most cases, the diagnosis of pulmonary toxoplasmosis should be confirmed by methods that demonstrate the presence of T. gondii in the pulmonary parenchyma, since the differential diagnosis of the most common causes of pulmonary disease in PLHA (such as pneumocystosis, mycobacteriosis, and cryptococcosis) must be ruled out.5,13-16 The diagnosis of pulmonary toxoplasmosis can be made by (i) visualizing tachyzoites through the microscopic examination of bronchoalveolar lavage (stained by Giemsa, indirect immunofluorescence or IHC); (ii) cell or laboratory animal inoculation of BAL, tissues or other fluids; (iii) detecting the T. gondii-B1 gene; and (iv) histopathologic exam of tissue samples.5,15-17 Most cases of pulmonary toxoplasmosis have high serum titers of anti-T. gondii IgG, but serology is not a well-validated method for the diagnosis of extracerebral toxoplasmosis.5 High serum levels of lactic dehydrogenase can be detected in pulmonary toxoplasmosis.14
The chest x-ray in pulmonary toxoplasmosis may be normal or exhibit bilateral and diffuse interstitial infiltrates (most common finding), bilateral interstitial infiltrates with nodular densities, solitary nodular lesions, nodular densities with pleural effusion, isolated pleural effusion, bilateral white lung, and pneumothorax.5,15,18,19 The tomographic findings in the pulmonary toxoplasmosis include bilateral septal and peribronchovascular thickening, ground-glass opacities, atelectasis, random nodules, lymph node enlargement, and pleural effusion.20
The histopathology of pulmonary toxoplasmosis is described in a few case reports and autopsy studies.3,19,21,22 The lungs are usually heavy, often having a combined weight of more than 2,000 g. The cut surface may exhibit congestion, consolidation, cavitation, petechial hemorrhages, disseminated miliary necrotic nodules (measuring 2-6 mm in diameter) or foci of necrotizing bronchopneumonia. The microscopic patterns of pulmonary toxoplasmosis include interstitial pneumonitis, diffuse alveolar damage, and focal or diffuse necrotizing pneumonia. The inflammatory reaction may be minimal or discrete lymphohistiocytic. Neutrophils and eosinophils are variable, and hyperplasia of type II pneumocytes can be seen. The parasite is found in the areas of coagulative necrosis as free tachyzoites or as cysts of bradyzoites. The tachyzoites are the rapidly dividing infective form of T. gondii, measuring less than 10 µm and can be mistaken for necrotic debris using the hematoxylin-eosin (H&E) stain. The Giemsa stain highlights the tachyzoites in the tissues. The bradyzoites can form cysts inside the cytoplasm of histiocytes and alveolar macrophages, the endothelial cells, alveolar lining cells, and smooth muscle cells lining the airways and blood vessels.
Cysts containing bradyzoites can be seen by H&E and are highlighted by the PAS stain.3,16,19,21,22 Some authors propose the histopathology of pulmonary toxoplasmosis has evolutionary morphological aspects: the early stage being interstitial pneumonitis with diffuse alveolar damage and few parasites in the cytoplasm of cells. With the progression of the disease, cellular lysis, tissue necrosis, and the release of tachyzoites into the extracellular medium occur.16,22 It is important to emphasize that, in autopsies of patients with advanced AIDS, multiple opportunistic infections and other pathologies can be detected during the histopathological examination.19,22
Another important finding in the present autopsy was HIV-associated nephropathy. The patient had no previous history of systemic arterial hypertension, and an earlier echocardiogram showed normal left ventricular function, with slight hypertrophy of the posterior wall of the left ventricle and septum (both 1.2 cm thick). HIV-associated nephropathy (HIVAN) is a rapidly progressive disease (from weeks to a few months), conferring high morbidity.23 The prevalence of HIVAN is variable, being more common in Afro-descendants, depending on the geographical area (more common in Africa). Also, it is more common in individuals with low CD4+ T cell count and high viral load, which was the case with our patient. In Brazil, the prevalence of HIVAN is estimated to be 5.6%.24
CONCLUSION
The present autopsy shows the importance of the postmortem exam in patients with advanced AIDS (as well as with other immunocompromising conditions), revealing pathologies not clinically diagnosed. Two major diagnoses were made in this autopsy: (i) disseminated toxoplasmosis with pulmonary and cerebral disease; and (ii) HIV-associated nephropathy. Pulmonary toxoplasmosis must be considered in the differential diagnosis of interstitial pneumonia and respiratory failure in PLHA who are not taking prophylactic and antiretroviral treatment.
The unfortunate outcome of the case reported herein highlights the importance of general preventive measures in the care of PLHA: regular testing and early diagnosis of HIV infection; regular medical follow-up; screening for previous infectious and common diseases including toxoplasmosis, hypertension, metabolic and renal disease; the introduction of ART; and prophylaxis against opportunistic diseases, as well as their early diagnosis and treatment.25,26
ETHICAL APPROVAL
The manuscript is in accordance with our institution’s ethics committee (HCFMUSP-CAPPesq 108/17).
ACKNOWLEDGEMENTS
The authors wish to thank Mrs. Deryn Pompeia for her technical assistance in the preparation of this manuscript.
REFERENCES
1 Goldman L, Sayson R, Robbins S, Cohn LH, Bettmann M, Weisberg M. The value of the autopsy in three medical eras. N Engl J Med. 1983;308:1000-5.
2 Grangeiro A, Escuder MM, Pereira JC. Late entry into HIV care: lessons from Brazil, 2003 to 2006. BMC Infect Dis. 2012;12:99.
3 Eza DE, Lucas SB. Fulminant toxoplasmosis causing fatal pneumonitis and myocarditis. HIV Med. 2006;7(6):415-20.
4 Dubey JP, Lago EG, Gennari SM, Su C, Jones JL. Toxoplasmosis in humans and animals in Brazil: high prevalence, high burden of disease, and epidemiology. Parasitology. 2012;139(11):1375-424.
5 Rabaud C, May T, Lucet JC, Leport C, Ambroise-Thomas P, Canton P. Pulmonary toxoplasmosis in patients infected with human immunodeficiency virus: a French National Survey. Clin Infect Dis. 1996;23(6):1249-54.
6 Luft BJ, Remington JS. Toxoplasmic encephalitis. J Infect Dis. 1988;157:1-6.
7 Barbosa CJ, Molina RJ, de Souza MB, et al. Disseminated toxoplasmosis presenting as sepsis in two AIDS patients. Rev Inst Med Trop São Paulo. 2007;49(2):113-6.
8 Vidal JE, Oliveira AC. AIDS-related cerebral toxoplasmosis in São Paulo State, Brazil: marked improvements in the highly active antiretroviral therapy-era but the challenges continue. Braz J Infect Dis. 2013;17(3):379-80.
9 Grant IH, Gold JW, Rosenblum M, et al. Toxoplasma gondii serology in HIV-infected patients: the development of central nervous system toxoplasmosis in AIDS. AIDS. 1990;4(6):519-21.
10 Abgrall S, Rabaud C, Costagliola D. Incidence and risk factors for toxoplasmic encephalitis in human immunodeficiency virus-infected patients before and during the highly active antiretroviral therapy era. Clin Infect Dis. 2001;33(10):1747-55.
11 Derouin F, Sarfati C, Beauvais B, Garin YJF, Larivière M. Prevalence of pulmonary toxoplasmosis in HIV-infected patients. AIDS. 1990;4:1036.
12 Lucet JC, Bailly MP, Bedos JP, Wolff M, Gachot B, Vachon F. Septic shock due to toxoplasmosis in patients infected with the human immunodeficiency virus. Chest. 1993;104:1054-8.
13 Okenshendler E, Cadranel J, Sarfati C, et al. Toxoplasma gondii pneumonia in patients with the acquired immunodeficiency syndrome. Am J Med. 1990;88:18N-21N.
14 Pugin J, Vanhems P, Hirschel B, Chave JP, Flepp M. Extreme elevations of serum lactic dehydrogenase differentiating pulmonary toxoplasmosis from Pneumocystis pneumonia. N Engl J Med. 1992;326(18):1226.
15 Velásquez JN, Ledesma BA, Nigro MG, et al. Pulmonary toxoplasmosis in human immunodeficiency virus-infected patients in the era of antiretroviral therapy. Lung India. 2016;33(1):88-91.
16 Pomeroy C, Filice GA. Pulmonary toxoplasmosis: a review. Clin Infect Dis. 1992;14(4):863-70.
17 Lavrard I, Chouaid C, Roux P, et al. Pulmonary toxoplasmosis in HIV-infected patients: usefulness of polymerase chain reaction and cell culture. Eur Respir J. 1995;8:697-700.
18 Goodman PC, Schnapp LM. Pulmonary toxoplasmosis in AIDS. Radiology. 1992;184(3):791-3.
19 Hofman P, Bernard E, Michiels JF, Thyss A, Le Fichoux Y, Loubière R. Extracerebral toxoplasmosis in the acquired immunodeficiency syndrome (AIDS). Pathol Res Pract. 1993;189(8):894-901.
20 de Souza Giassi K, Costa AN, Apanavicius A, et al. Tomographic findings of acute pulmonary toxoplasmosis in immunocompetent patients. BMC Pulm Med. 2014;14:185.
21 Jautzke G, Sell M, Thalmann U, et al. Extracerebral toxoplasmosis in AIDS. Histological and immunohistological findings based on 80 autopsy cases. Pathol Res Pract. 1993;189(4):428-36.
22 Nash G, Kerschmann RL, Herndier B, Dubey JP. The pathological manifestations of pulmonary toxoplasmosis in the acquired immunodeficiency syndrome. Hum Pathol. 1994;25(7):652-8.
23 Rosenberg AZ, Naicker S, Winkler CA, Kopp JB. HIV-associated nephropathies: epidemiology, pathology, mechanisms and treatment. Nat Rev Nephrol. 2015;11(3):150-60.
24 Cavalcante MA, Coelho SN, Lacerda HR. Prevalence of persistent proteinuria in stable HIV/AIDS patients and its association with HIV nephropathy. Braz J Infect Dis. 2007;11(5):456-61.
25 Palella FJ Jr, Delaney KM, Moorman AC, et al. Declining morbidity and mortality among patients with advanced human immunodeficiency virus infection. HIV Outpatient Study Investigators. N Engl J Med. 1998;338(13):853-60.
26 Gallant JE, Moore RD, Chaisson RE. Prophylaxis for opportunistic infections in patients with HIV infection. Ann Intern Med. 1994;120(11):932-44.
Notes
Author notes
Correspondence Amaro Nunes Duarte-Neto Departamento de Patologia - Faculdade de Medicina - Universidade de São Paulo (USP) Avenida Dr. Arnaldo, 455 - Cerqueira Cesar - São Paulo/SP - Brasil CEP: 01246-903 Phone: +55 (11) 98546-4225 amaro.ndneto@hc.fm.usp.br
Conflict of interest declaration