Article / Clinical Case Report
Traumatic injury to the internal carotid artery by the hyoid bone: a rare cause of ischemic stroke
Traumatic injury to the internal carotid artery by the hyoid bone: a rare cause of ischemic stroke
Autopsy and Case Reports, vol. 8, no. 1, e2018010, 2018
Hospital Universitário da Universidade de São Paulo
Received: 21 December 2017
Accepted: 19 January 2018
Abstract: Central nervous system (CNS) ischemic events, besides being a common and devastating disease, are accompanied by severe disability and other morbidities. The cause of such events is not always that simple to diagnose, and among the young, a broad spectrum of possibilities should be considered. We present the case of a young man who presented two episodes of CNS ischemia with a 1 year gap between them, which occurred in the same situation while he was walking and carrying a heavy backpack. The second event first presented as a transient ischemic attack followed by a stroke the day after. The diagnostic work-up showed an indentation of the greater cornu of the hyoid bone over the internal carotid artery, which injured the media and intimal layers. At the arterial injury site, a micro thrombus was found, which explained the source of the embolic event to the CNS. The patient was operated on, and the procedure included the resection of the posterior horn of the hyoid bone, the resection of the injured segment of the internal carotid artery followed by carotid-carotid bypass with the great saphenous vein. The postoperative period and the recovery were uneventful as was the 5-month follow-up. We call attention to this unusual cause of stroke and present other cases reported in the literature.
Keywords: Stroke, Brain Ischemia, Hyoid Bone, Carotid Artery diseases, Surgical procedures, Operative.
INTRODUCTION
Ischemic central nervous system (CNS) injury of atherosclerotic origin is not a common entity among the young. In this age group, disregarding the hematological diseases, the external or surgical traumas and post-extensive radiation of the neck play an essential role in the carotid artery lesion with subsequent embolization source to the CNS.1,2 Additionally, and regardless of age, rare cases have been reported on the particular vascular insult resulting from the close contact between the hyoid bone and the internal carotid artery (ICA).3-5
In the face of the available literature evidence, we should consider the possibility of carotid artery injury caused by the hyoid bone in cases of ischemic cerebrovascular events of unknown origin, especially in the young population. We also call attention to the critical clue of repetitive events under the same situation, such as sports playing movements, carrying heavy weights on the back, shoulder or neck, which can bring the contact between the ICA and the hyoid bone even closer.
Case Report
A 29-year-old man sought the emergency room (ER) complaining of a short-term paralysis of the left upper limb that occurred 2 days prior, when he was hiking along a walking trail with a backpack; the paralysis completely subsided later that day. On the next day, a left facial paralysis ensued accompanied by dysphagia and recurrence of the left upper limb weakness. Once again, the latter partially subsided within a couple of hours. At the ER, the patient still presented with facial paralysis and left upper limb paresis, plus slight difficulty in walking, and slurred speech.
His past medical history was unremarkable except for a similar complaint 1 year before, when he underwent a thorough laboratory and CNS imaging work-up without a conclusive diagnosis. With the working diagnosis hypothesis of multiple sclerosis, the patient was hospitalized. On physical examination, he was conscious and alert with normal vital signs. The neurological examination disclosed (i) left central facial nerve paralysis; (ii) dysarthric speech; (iii) hyposensitivity in the inferior two-thirds of the left hemiface; (iv) deviation of the tongue to the right side; and (v) Grade 2 motor strength of the left upper limb and Grade 4 of the left leg with a hemiplegic gait requiring a four-point stick to walk. No spasticity or fasciculation was present. The remaining physical examination was unremarkable.
The laboratory work-up, which included renal function tests, electrolytes, lipid profile, hepatic function tests, peripheral blood count, erythrocyte sedimentation rate, hemoglobin electrophoresis, C-reactive protein, and thyroid function analysis, was within the normal limits. The serology for HIV, hepatitis B, and C, Chagas’ disease, syphilis, antinuclear antibody, and rheumatoid factor was negative. The research for anti-cardiolipin antibodies, lupus anticoagulant, and factor V Leiden were negative. The cerebrospinal spinal fluid analysis included biochemical, cytological, microbiological, and protein electrophoresis, which were normal. The brain computed tomography (CT) depicted a hypoattenuating area in the cortico-subcortical boundary of the right parietal lobe, which did not show enhancement after the contrast medium injection. The brain magnetic resonance imaging showed an extensive zone of altered signal in the cortical and subcortical areas of the right frontal and parietal lobes, as well as the insula, which presented diffusion restriction, T2-weighted hyper signal, and mild expansive effect. The aspect was compatible with an acute ischemic event in the territory of the right middle cerebral artery. Additional small foci of altered signal and diffusion restriction coexist in sparse regions of the parietal lobe and on the right periventricular white matter, which is consistent with ischemic events of undetermined chronology (Figure 1).
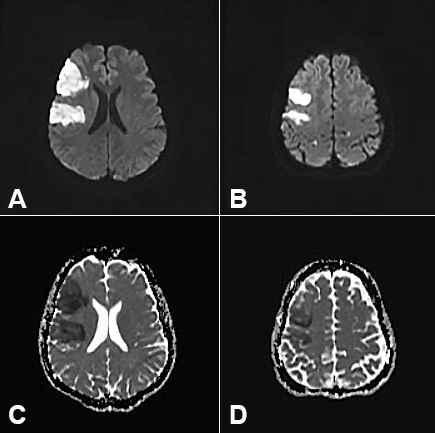
Figure 1
Brain magnetic resonance imaging. Diffusion-weighted images (A and C) and apparent diffusion coefficient map (B and D) showing areas of water-free-motion restriction in the right frontal lobe, compatible with acute ischemic stroke.
The Holter electrocardiogram (ECG), transthoracic Doppler echocardiography, and the carotid duplex scan were normal. However, the CT angiographic analysis of the carotid arteries depicted an enlarged greater horn of the hyoid bone, which had close contact with the ICA, where a hypoattenuating irregularity on the parietal wall was found, accompanied by a tiny flap of the endothelium consistent with a thrombus (Figures 2 and 3).
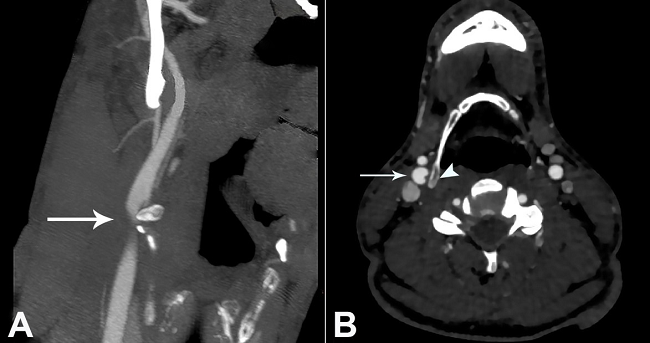
Figure 2
Computed tomography of the neck after intravenous contrast injection. A - Impingement of the internal carotid artery (ICA) by the greater horn of the hyoid bone (arrow); B - The close contact between the hyoid bone (arrowhead) and the ICA (arrow). Note the irregularity on the arterial wall, which leans against the hyoid bone.
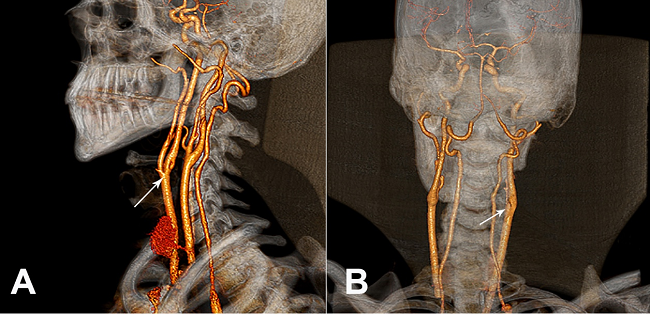
Figure 3
3D reformation of the neck computed tomography showing the indentation imprint in the internal carotid artery (arrows). A - Anterior left oblique view; B - Posterior view.
The patient was prescribed physiotherapy and 300 mg daily of salicylate, with substantial improvement of the gait and the dysarthria, with Grade 4 motor strength in the left upper limb and Grade 5 in the left lower limb on the day of hospital discharge.
Based on the clinical features and the imaging study, the working diagnosis considered the external compression of the right ICA by the hyoid bone. Therefore, a right cervical surgical exploration was performed with dissection of the common, internal, and external carotid arteries. During the surgical procedure, the external compression of the ICA by the posterior horn of the hyoid bone was confirmed. There was no evidence of arterial perforation nor any presence of a pseudoaneurysm—only close contact between both structures. The surgical procedure included the resection of the posterior horn of the hyoid bone, the resection of the injured segment of the ICA, followed by carotid-carotid bypass with the great saphenous vein. At the opening of the arterial segment, evident atherosclerosis and a small intimal thrombus adjacent to the compression site were seen (Figure 4).
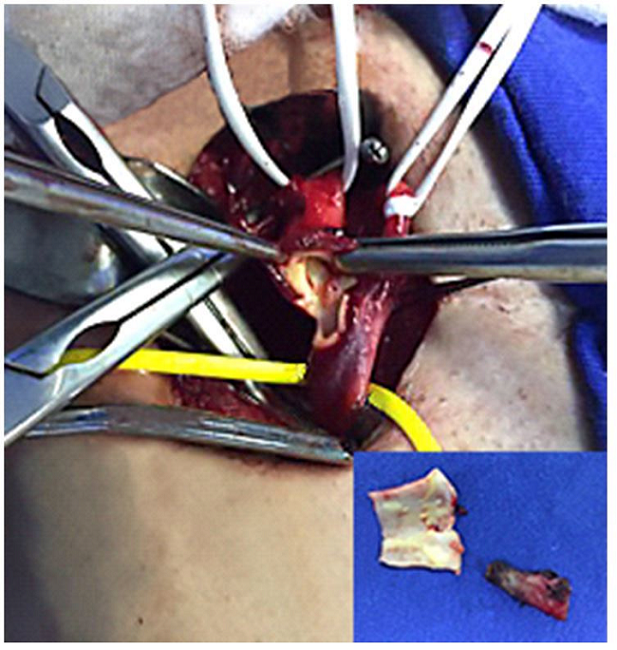
Figure 4
Intraoperative exposure of the internal carotid artery, which was resected (inset). Note the tiny plaques of atherosclerosis.
The microscopic examination showed focal intimal thickening with fibrosis, and an accumulation of foam cells—typical of uncomplicated atherosclerotic plaques. Also, there was an organizing intimal thrombus (Figure 5).
![Photomicrographs of the carotid artery showing a focal organizing thrombus (asterisk in A) and atherosclerotic plaques characterized by intimal fibrosis and accumulations of foam cells (arrows in B) (H&E 10X [A] and 20X [B]).](../2236-1960-acrep-08-01-e2018010-gf05.jpg)
Figure 5
Photomicrographs of the carotid artery showing a focal organizing thrombus (asterisk in A) and atherosclerotic plaques characterized by intimal fibrosis and accumulations of foam cells (arrows in B) (H&E 10X [A] and 20X [B]).
At the external half of the medial layer a focal replacement of the smooth muscle cells by fibrosis and a scarcity of elastic fibers were detected, which were not accompanied by inflammatory infiltration (Figure 6).
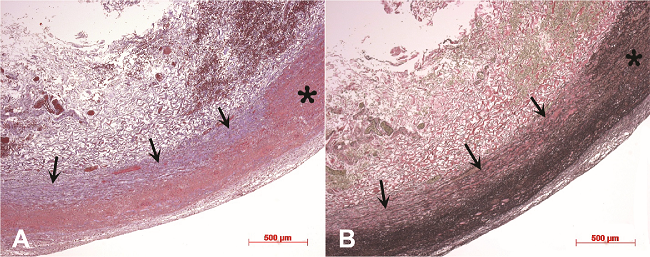
Figure 6
Photomicrographs of a segment of the carotid arterial wall showing, at the external half of the medial layer, a focal replacement of the smooth muscle cells with fibrosis (arrows in A) and a scarcity of elastic fibers in the same region (arrows in B). Compare with the normal-appearing arterial wall at the right extremities of the panels (asterisks). Masson’s trichrome stain 5X (A), and Verhoeff’s elastic stain 5X (B).
The patient outcome was uneventful and he was discharged on day 3 of the postoperative period. The 5-month follow-up did not show new events or any other complaint.
DISCUSSION
Ischemic cerebrovascular events in young adults present a different etiologic distribution when compared with older patients.6 According to the Helsinki Young Stroke Registry, the most common identified cause of stroke in the young is cardioembolic (18.7%) followed by arterial dissection (15.5%), small vessel disease (13.9%), large artery atherosclerosis (8.4%), and a considerable amount of undetermined etiology (33.1%).7 Presumably, among the latter, the traumatic injury to the carotid artery caused by the hyoid bone is included but underdiagnosed. In this setting, the anatomical redundant variant of the of the greater horn of the hyoid bone may entrap, hook, or protrude into the carotid artery causing structural injuries with subsequent dissection, thrombosis, or turbulent blood flow velocities, and shear stress speeding up early atherogenesis.8-10 In 1998, Abdelaziz et al.3 published a case of transient ischemic attack (TIA) in which the hyoid bone caused 90% of stenosis at the site of the artery injury caused by the greater horn. Since then, it has been observed that arterial intramural mechanical stress, endothelial damage, muscular hyperplasia, and increased connective tissue formation result in a rough, damaged, vascular surface and thrombi formation. In subsequent years, other cases involving vascular injury by the hyoid bone and ischemic cerebrovascular events were published. Schneider and Kortmann11 retrieved three cases of pseudoaneurysm of the carotid artery caused by the mechanical trauma of the hyoid bone. One was diagnosed at autopsy after massive cervical bleeding; one presented as a cervical pulsatile mass and the other as an ischemic stroke. In their case report, Schneider and Kortmann11 proposed an occupational relationship for the carotid injury, since their patient was a bricklayer and worked carrying heavy weights on his shoulder, which, in the authors’ interpretation, compressed the tissue of the neck against the hyoid bone. Kölbel et al.12 reported the case of a TIA caused by the interposition of the greater horn of the hyoid bone between the right carotid arteries, causing its entrapment. In this case, during the surgical procedure, even though the ICA was apparently normal in its structure and palpation it seemed ectactic. The authors speculated that repetitive traumatic injury to the arterial wall with subsequent intimal thrombus formation was responsible for the TIA. Pearlman et al.13 showed the association of carotid wall bruising and intimal damage at the site of contact with the hyoid bone. Hong et al.14 described a case of repetitive symptomatic brain ischemic lesions associated with the movements of a golf player, which subsided after partial resection of the hyoid bone. The authors interpreted that the repetitive motion of the sternocleidomastoid muscle (during the head rotation) compressed the ICA against the greater horn of the hyoid bone, and was considered the culprit physiopathological process for this vasculopathy. Similarly, Tokunaga et al.15 published a case in which the ICA moved inward and outward, depending on the head rotation and swallowing, because the origin of the ICA was hooked by the greater cornu of the hyoid bone. In this case, the surgical procedure revealed that the ICA was thickened at the site of the hyoid bone contact, and the histology demonstrated medial necrosis, inflammatory changes, and a mural organized thrombus, without atherosclerosis. Carotid artery dissection is another injury caused by an elongated hyoid bone associated with neck rotation, demonstrating constitutional damage to the artery layers by direct compression on the artery.16 Exceptionally, the elongated lesser horn of the hyoid bone also may be responsible for the compression or irritation of the carotid artery as reported by Janczack et al.17 in a case of recurrent syncope in a middle-aged man. Due to the scarcity of reported cases, or even the real paucity of cases, this entity is unknown to many doctors and is therefore not taken into account in the differential diagnosis of the cause of a stroke.
The case reported herein calls attention to the initial presentation as TIA followed by a stroke. The recurrence of a similar episode presented 1 year before, which happened in the same situation with the same patient when he was hiking and carrying a heavy backpack, was indeed intriguing. In the first episode, the diagnostic work-up failed to demonstrate any diagnosis and the neurological symptoms wholly subsided, most probably because the lesion was not detectable by the imaging study. The diagnostic approach in this second episode—although extensive, which included a carotid duplex—did not provide any abnormality that could explain the etiology of the stroke. However, a thorough and meticulous interpretation of the carotid angiogram, with the aid of the 3D reformation, identified the hyoid bone indentation on the ICA.
We interpreted that the elongated greater cornu of the hyoid bone associated with carrying the backpack may have enhanced the compression of the carotid artery by the hyoid bone, which is why the events occurred a second time in the same situation. The weight of the backpack pulling the patient’s trunk backward forced his neck forward (to maintain the center of gravity) and facilitated the impingement of the ICA by the greater cornu of the hyoid bone.
Atherosclerotic plaques were found on the gross and microscopic examinations of the resected arterial specimen, which were well-formed but not large enough to cause stenosis or obstruction. The patient had no risk factors for atherosclerosis, nor any familial history of early atherosclerosis. We considered the presence of these plaques as arising from the turbulent flow and the shear stress, and the elastic layer injury was caused by the continuous compression on the arterial wall by the hyoid bone. The damage to these arterial layers may have caused local thrombosis and embolization to the CNS. The option for the carotid resection and saphenous bypass, besides the hyoid corn removal, was based on the surgeon's concern at the very moment during surgery. At that point, it was not possible to assure, based on the gross view of the artery injury, how extensive the mechanical damage to the vessel was. The surgeon's decision took into account the possibility that an injured vessel wall could, in the future, be a site prone to the development of a pseudoaneurysm or dissection, as demonstrated in the literature review.
The awareness of this harmful and disabling entity, which can be surgically treated, is of paramount importance to good medical practice.
References
1 McIntyre WB, Ballard JL. Cervicothoracic vascular injuries. Semin Vasc Surg. 1998;11(4):232-42. PMid:9876030
2 Ramadan F, Rutledge R, Oller D, Howell P, Baker C, Keagy B. Carotid artery trauma: a review of contemporary trauma center experiences. J Vasc Surg. 1995;21(1):46-56. 7823361 http://dx.doi.org/10.1016/S0741-5214(95)70243-1.
3 Abdelaziz OS, Ogilvy CS, Lev M. Is there a potential role for hyoid bone compression in pathogenesis of carotid artery stenosis? Surg Neurol. 1999;51(6):650-3. 10369234 http://dx.doi.org/10.1016/S0090-3019(99)00022-1.
4 Metz H, Murray-Leslie RM, Bannister RG, Bull JW, Marshall J. Kinking of the internal carotid artery. Lancet. 1961;1(7174):424-6. 13769898 http://dx.doi.org/10.1016/S0140-6736(61)90004-6.
5 Quattlebaum JK Jr, Wade JS, Whiddon CM. Stroke associated with elongation and kinking of the carotid artery: long-term follow-up. Ann Surg. 1973;177(5):572-9. 4704042 http://dx.doi.org/10.1097/00000658-197305000-00010
6 Stack CA, Cole JW. A diagnostic approach to stroke in young adults. Curr Treat Options Cardiovasc Med. 2017;19(11):84. 28948451 http://dx.doi.org/10.1007/s11936-017-0587-6
7 Putaala J, Metso AJ, Metso TM, et al. Analysis of 1008 consecutive patients aged 15 to 49 with first ever ischemic stroke. The Helsinki young stroke registry. Stroke. 2009;40(4):1195-203. 19246709 http://dx.doi.org/10.1161/STROKEAHA.108.529883
8 Wells DR, Archie JP Jr, Kleinstreuer C. Effects of carotid artery geometry on the magnitude and distribution of wall shear stress gradients. J Vasc Surg. 1996;23(4):667-78. 8627904 http://dx.doi.org/10.1016/S0741-5214(96)80048-6.
9 Salzar RS, Thubrikar MJ, Eppink RT. Pressure-induced mechanical stress in the carotid artery bifurcation: a possible correlation to atherosclerosis. J Biomech. 1995;28(11):1333-40. 8522546 http://dx.doi.org/10.1016/0021-9290(95)00005-3.
10 Renard D, Rougier M, Aichoun I, Labauge P. Hyoid bone-related focal carotid vasculopathy. J Neurol. 2011;258(8):1540-1. 21327849 http://dx.doi.org/10.1007/s00415-011-5940-1
11 Schneider CG, Kortmann H. Pseudoaneurysm of the common carotid artery due to ongoing trauma from the hyoid bone. J Vasc Surg. 2007;45(1):186-7. 17210407 http://dx.doi.org/10.1016/j.jvs.2006.08.075
12 Kölbel T, Holst J, Lindh M, Mätzsch T. Carotid artery entrapment by the hyoid bone. J Vasc Surg. 2008;48(4):1022-4. 18992420 http://dx.doi.org/10.1016/j.jvs.2008.04.062
13 Pearlman RC, Naficy MA, Koby MB, Nyanzu M. Carotid artery compression by the hyoid bone. Vasc Endovascular Surg. 2012;46(8):686-7. 22989429 http://dx.doi.org/10.1177/1538574412460101
14 Hong JM, Kim TJ, Lee JS, Lee JS. Repetitive internal carotid artery compression of the hyoid: a new mechanism of golfer’s stroke. J Neurol Neurosurg Psychiatry. 2011;82(2):233-4. 20587482 http://dx.doi.org/10.1136/jnnp.2010.213777
15 Tokunaga K, Uehara T, Kanamaru H, et al. Repetitive artery-to-artery embolism caused by dynamic movement of the internal carotid artery and mechanical stimulation by the hyoid bone. Circulation. 2015;132(3):217-9. 26195490 http://dx.doi.org/10.1161/CIRCULATIONAHA.114.015144
16 Yukawa S, Yamamoto S, Hara H. Carotid artery dissection associated with an elongated hyoid bone. J Stroke Cerebrovasc Dis. 2014;23(8):e411-2. 25126696 http://dx.doi.org/10.1016/j.jstrokecerebrovasdis.2014.04.006
17 Janczak D, Skora J, Rucinski A, Szuba A. Recurrent syncope caused by compression of internal carotid artery by anomalous hyoid bone. Vasa. 2012;41(3):221-4. 22565624 http://dx.doi.org/10.1024/0301-1526/a000189
Notes
Author notes
Correpondence Erasmo Simão Silva Vascular and Endovascular Division - Department of Surgery - School of Medicine - Hospital das Clínicas - University of São Paulo (USP) Av. Dr. Eneas de Carvalho Aguiar, nº 255 - Cerqueira Cesar - São Paulo/SP - Brazil Phone: +55 (11) 3061-8718 CEP: 05403-000 ersimao@usp.br
Conflict of interest declaration