Abstract: Mesothelial/monocytic incidental cardiac excrescences (MICE) are unusual findings during the histological analysis of material from the pericardium, mediastinum, or other tissues collected in open-heart surgery. Despite their somewhat worrisome histological appearance, they show a benign clinical course, and further treatment is virtually never necessary. Hence, the importance of recognizing the entity relays in its differential diagnosis, as an unaware medical pathologist may misinterpret it for a malignant neoplasm. Other mesothelial and histiocytic proliferative lesions, sharing very close histological morphology and immunohistochemistry features with MICE, have been described in sites other than the heart or the mediastinum. This similarity has led to the proposal of the common denomination “histiocytosis with raisinoid nuclei.” We report three cases from the pathology archives of the Heart Institute of São Paulo University (Incor/HC-FMUSP), diagnosed as “mesothelial/monocytic incidental cardiac excrescence,” with immunohistochemical documentation, and provide a literature review of this entity.
Keywords:Heart Diseases Incidental Findings Epithelium HistiocytesHeart Diseases Incidental Findings Epithelium Histiocytes.
Article / Clinical Case Report
Occurrence of mesothelial/monocytic incidental cardiac excrescences in material from open-heart procedures: case reports and literature review
Hospital Universitário da Universidade de São Paulo
Received: 31 August 2017
Accepted: 02 October 2017
Mesothelial/monocytic incidental cardiac excrescences (MICE) are benign lesions usually found incidentally in the pericardium. The pathologist has to be aware of such entities, as they may morphologically mimic a malignant lesion, such as metastatic carcinoma. Also, in the past, histiocytic proliferations have been described in many sites of the human body, usually mesothelial-layered surfaces. Recent studies have proposed uniting these entities under the common category of “histiocytosis with raisinoid nuclei.” The aim of this paper is to report three cases diagnosed in a tertiary heart center and to provide a review of the ongoing changes in the pathological classification of these lesions.
We searched the pathology archives of the Heart Institute of São Paulo University (Incor/HC-FMUSP) for cases diagnosed as “mesothelial/monocytic incidental cardiac excrescence,” retrieving a total of three cases with immunohistochemical documentation. All three cases are detailed further in this paper.
The authors also performed a literature review. In the PubMed database, a search with the descriptor “histiocytosis with raisinoid nuclei” raised two results, and another one with the descriptor “mesothelial/monocytic incidental cardiac excrescence” offered 14 results. Both searches were performed in April 2017.
A 79-year-old male patient with a long history of Parkinson disease and heart failure due to double aortic dysfunction (severe stenosis and moderate insufficiency) returned to follow-up with signs of clinical deterioration. For the past 4 years, he’d already had indications for surgical treatment but refused it. In the presence of a worse clinical condition, he consented to surgery. An echocardiogram was performed, which showed the double aortic valve dysfunction and measured a left ventricular ejection fraction of 29%. After proper pre-operatory stabilization, the aortic valve was replaced with a biological prosthesis.
The patient’s original aortic valve was sent to pathological analysis, confirming chronic calcified valvulopathy. An incidental finding at histological examination was an agglomerate of polyhedral cells and histiocytes admixed with fibrin and erythrocytes, and focally forming strips or tubular arrays that were not attached to the valve, which appeared to correspond macroscopically to a thrombus (Figures 1A and 1B). Immunohistochemistry was positive for calretinin in some of the cells and for CD68 in others, allowing the identification of a biphasic lesion, with mesothelial cells and macrophages, respectively (Figures 1C and 1D), and the diagnosis of a mesothelial/monocytic incidental cardiac excrescence.
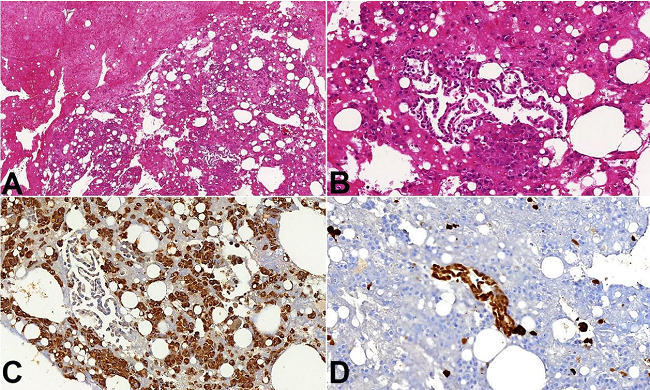
Figure 1
Photomicrographies showing the main histological findings. Images A - (case 1; 10X) and B - (case 1; 20X) show cells that are interspersed within agglomerates of fibrin and present as loosely arranged clusters or glandular-like structures. Occasionally, the glandular structures show columnar cells. Image C - Histiocytes show strong cytoplasmic immunostaining for CD68 (case 1; 20X). Image D - Mesothelial cells show strong immunohistochemical positivity for calretinin. (case 1; 20X). Objective magnifications are in parenthesis after the case number.
A 26-year-old patient with the previous diagnosis of epileptic seizures, cognitive deficit, chronic lymphedema of the lower limbs, bicuspid aortic valve and total atrioventricular block, who had an implanted internal pacemaker, was admitted for surgical replacement of the aortic valve by a biological prosthesis. Pathological examination of the native valve was consistent with chronic endocarditis, but in the histological analysis isolated sheets and blocks of epithelioid cells within a fibrin matrix were also found, which were associated with macrophagic reaction (Figure 2A), an incidental finding, without attachment to the valve. Immunohistochemistry was positive for pan-keratin (AE1/AE3) and WT1 in the epithelioid cells and CD68 in the macrophages. Such findings are consistent with mesothelial/monocytic incidental cardiac excrescence.
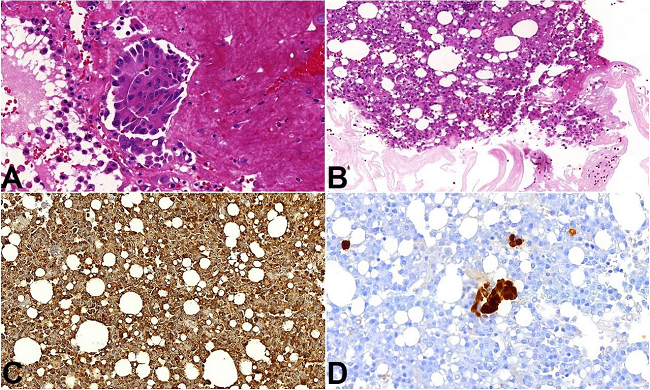
Figure 2
Photomicrographs from cases 2 and 3. Image A - Hematoxylin and eosin (H&E) staining displays sheets and blocks of epithelioid cells within a fibrin matrix, associated with macrophages (case 2; 20X). Image B - Agglomerate of polyhedral cells and histiocytes embedded in fibrin (case 3; 20X). Image C - CD68 immunostaining shows cytoplasmic positivity in the solid cells clusters demonstrating the histiocytic nature of these cells (case 3; 20X). Image D - Calretinin immunostaining shows mesothelial cells in middle of histiocytic cells (case 3; 20X). Objective magnifications are in parenthesis after the case number.
Our third patient was a 45-year-old male patient with precedent rheumatic fever, followed by mitral valve stenosis, which evolved with left atrial enlargement and atrial fibrillation. The patient underwent cardiac catheterization, which identified a 50% occlusive lesion of the second left marginal artery. He had heart failure and complained of dyspnea at usual efforts. A mitral valve replacement was performed.
During surgery, in the mediastinum, the surgeon described an area that grossly resembled fibrosis, which was excised and submitted for pathological study. Microscopically it was composed of fibrotic tissue with foci of calcifications and thrombi. Additionally, and apart from the fibrotic area, an agglomerate of polyhedral cells and histiocytes were found embedded in fibrin (Figure 2B), compatible with mesothelial/monocytic incidental cardiac excrescence (Figures 2C and 2D).
Given the histological findings and immunohistochemistry profiles, all the previously reported cases were diagnosed as MICE. These are non-neoplastic collections of mesothelial cells intermingled with foamy macrophages interspersed in fibrin. Although they are benign lesions with little, if any, clinical repercussion, pathologists should be aware of their existence, as the finding may induce a misdiagnosis of primary local malignancy or even metastatic carcinoma.1
MICE was initially considered to be a type of histiocytoid hemangioma by Rosai et al.2 who, in 1979, proposed the term for a proliferative process of endothelial cells that has a distinctive histiocytoid appearance and manifests itself in a variety of sites. Using immunohistochemistry and electron microscopy, Veinot et al.3 proposed the name MICE for the lesion by demonstrating in 1994 that MICE were mainly composed of mesothelial cells and histiocytes. Histologically, there are compact clusters of histiocytes, fat globules, and mesothelial cells, without intervening stroma.4
MICE are usually incidental findings in specimens from patients who underwent cardiac surgery for other reasons, such as valve replacement5 or even endomyocardial biopsy.3 They have been described as nodules in the pericardium, or as a loose material in the pericardial fluid, and occasionally some cases were submitted to the pathology laboratory as a “thrombus.” In the examples here described, these fragments were not distinguished macroscopically and were sent for histological analysis admixed in the excised material.
Although its physiopathology isn’t fully elucidated, some theories have been proposed. Two of the most discussed are the artifactual and reactive hypotheses. According to the former, MICE are aggregates of mesothelial cells and other debris that were not drawn into the bypass pump and were transported by the suction catheter to the intravascular space.6,7 The latter states that in the scenario of heart surgical procedures, mesothelial cells would gain access to the bloodstream and couple with histiocytes and fibrin through an unknown mechanism.7
These are very reasonable theories for patients who underwent previous thoracic manipulation, but they can’t explain all the cases of MICE, such as the ones diagnosed in endomyocardial biopsies from patients without precedent surgeries. Again, in the context of the reactive theory, not only mechanical, but other forms of stimuli, such as inflammation, or even neoplasms, may be the trigger. For example, Argani et al.8 reported a patient with no previous cardiac instrumentation who had adenocarcinoma of the right lung. During the operation, a fragment of tissue was found in the pericardial cavity that was diagnosed as MICE. Ton et al.9 described a case in which the patient with both antiphospholipid syndrome and prior cardiac catheterization developed a mass in the ventricular surfaces of the left coronary and right coronary cusps of the aortic valve. It was expected to be a thrombus, but pathological examination diagnosed it as MICE.
Few cases have been reported so far, which makes it difficult to appraise the incidence and prevalence of MICE in the general population. Courtice et al.6 published a study where they found material histologically compatible with MICE in 82% of the contents from extracorporeal bypass pump filters and in 13% of the ones from mediastinal drains. Hence, considering the large number of patients that are submitted to cardiac and mediastinal procedures nowadays, it could be hypothesized that this entity may be actuality more prevalent than previously thought.
At gross examination, these excrescences are described as small fragments of tissue (measuring from <1 mm up to 3 cm), which vary in color from gray-white to brown or dark red.10 Microscopic analysis shows that MICE are composed of two cell populations. The first population is represented by plump histiocytoid cells with pale pink cytoplasm and prominent nuclear grooves. Immunohistochemistry studies show they are positive for CD68 and lysozyme. The second are the mesothelial cells, which have a cuboidal appearance, with less cytoplasm, and small nuclei, and show diverse arrangements (ranging from sheets, tubules, or even papillary patterns). They show positivity for keratins (which can be demonstrated with pan-keratin tests, e.g. AE1/AE3), CK 5/6, WT1, and calretinin. It is important to emphasize that MICE collections have no supporting stroma. Both cell types are found to be embedded in fibrin.5,10
As an entity, MICE has been known for a few decades; however, over time benign histiocytic proliferations have been described in other sites of the human body, such as intralymphatic histiocytosis and nodular mesothelial/histiocytic hyperplasia. The resemblance between some of them have already been recognized, but they have remained as separate entities.
In 2014, Jiao et al.11 reported a case of MICE and described its striking similarities with nodular histiocytic/mesothelial hyperplasia (NHMH), which is also predominantly composed of histiocytes and striped mesothelium, and may well be a reactive lesion resulting from trauma, tumor, or inflammation. They proposed it belonged to the same disease spectrum as MICE and that the term “NHMH” could refer to both lesions.11
However, it was only in 2016 that some authors tried to unite all of the histiocytic proliferations into one single category, with the proposed name of “histiocytosis with raisinoid nuclei.”12,13 Along with MICE and NHMH, there are also reactive eosinophilic pleuritis, histioeosinophilic granuloma (HEG) of the thymus, and intralymphatic histiocytosis.
Reactive eosinophilic pleuritis (REP) is an inflammatory reaction composed by an aggregate of histiocytes, mesothelial cells, and eosinophils in the visceral lung pleura.14,15 It is commonly observed in association with pneumothorax and ruptured pleural blebs.16 HEG of the thymus has similar findings to REP and is also associated with a reaction to air entering the thoracic/mediastinal cavity. This is a very likely explanation for a significant parcel of the reported cases that occurred in patients with myasthenia gravis who underwent thymectomy.17,18 According to the 2016 study by Michal et al.,12 these eosinophils might be one of the reasons for the delay in the recognition that REP and HEG took longer to be linked to other histiocytic proliferations.
Intralymphatic histiocytosis (ILH) consists of benign histiocytes in dilated lymph vessels. It is another reactive lesion and has been reported to follow chronic inflammation, prior surgery, systemic disease (e.g. rheumatoid arthritis), orthopedic implants, and even neoplastic lesions.19,20 Most cases show cutaneous manifestations, but at least one visceral case has already been described.21
All these entities have very similar histopathological findings: histiocytes with eosinophilic cytoplasm, and irregular nuclei with delicate grooves, which are described as “raisinoid.” These histiocytes are embedded in fibrin and admixed with variated populations of other inflammatory and mesothelial cells.12
The general and surgical pathologist should be aware of this entity when examining material excised from several sites of the human body, and should be able to make the differential diagnosis with other inflammatory and neoplastic lesions.
Correspondence Vitor Gabriel Ribeiro Grossi Anatomic Pathology Department - Hospital das Clinicas Rua Dr. Ovídio Pires de Campos, nº 255, 10º andar - Cerqueira Cesar - São Paulo/SP - Brazil CEP: 01246-100 Phone: +55 (11) 2137-4700 vitor.grossi@hc.fm.usp.br

Figure 1
Photomicrographies showing the main histological findings. Images A - (case 1; 10X) and B - (case 1; 20X) show cells that are interspersed within agglomerates of fibrin and present as loosely arranged clusters or glandular-like structures. Occasionally, the glandular structures show columnar cells. Image C - Histiocytes show strong cytoplasmic immunostaining for CD68 (case 1; 20X). Image D - Mesothelial cells show strong immunohistochemical positivity for calretinin. (case 1; 20X). Objective magnifications are in parenthesis after the case number.

Figure 2
Photomicrographs from cases 2 and 3. Image A - Hematoxylin and eosin (H&E) staining displays sheets and blocks of epithelioid cells within a fibrin matrix, associated with macrophages (case 2; 20X). Image B - Agglomerate of polyhedral cells and histiocytes embedded in fibrin (case 3; 20X). Image C - CD68 immunostaining shows cytoplasmic positivity in the solid cells clusters demonstrating the histiocytic nature of these cells (case 3; 20X). Image D - Calretinin immunostaining shows mesothelial cells in middle of histiocytic cells (case 3; 20X). Objective magnifications are in parenthesis after the case number.
