Article / Clinical Case Report
Bullous systemic lupus erythematosus - a case report
Bullous systemic lupus erythematosus - a case report
Autopsy and Case Reports, vol. 9, no. 1, e2018069, 2019
Hospital Universitário da Universidade de São Paulo
Received: 29 November 2018
Accepted: 08 December 2018
ABSTRACT: Skin involvement in systemic lupus erythematosus (SLE) occurs in more than 75% of patients with this condition. Vesicles and blisters in lupus erythematosus (LE) may be present in SLE secondary to interface vacuolar changes in the epidermis, in discoid LE also secondary to vacuolar epidermal changes, and in bullous LE secondary to antibodies anti-collagen VII deposits with neutrophilic aggregates. In addition, blisters can occur due to the association of SLE with other autoimmune blistering diseases (e.g. bullous pemphigoid). BSLE is a rare blistering disease that mainly occurs in females (30-40 years old), and less frequently in children and adolescents. The most common presentation is rapid and widespread development of tense vesicles and bullae over erythematous macules or plaques. Preferential sites are: superior trunk, proximal superior limbs, and face (lips) with symmetrical distribution. Mucosal involvement is common on perioral, pharyngeal, laryngeal, and genital areas. The involvement of sun-exposed areas is not mandatory. The lesions usually progress with no scarring, but hypo or hyperchromia may be present. We report an 18-year-old female patient with blistering lesions at admission, who was diagnosed with BSLE. She was initially treated with systemic prednisone and hydroxychloroquine. Her condition evolved with relapsing lesions, which required the introduction of Dapsone. The authors emphasize the relevance of recognizing BSLE—a rare presentation of SLE—which may evolve with marked clinical presentation.
Keywords: Lupus Erythematosus, Systemic, Blister, Dapsone, Diagnosis, Therapeutics.
INTRODUCTION
Skin involvement in systemic lupus erythematosus (SLE) occurs in more than 75% of patients with this condition. Vesicles and blisters in SLE may be detected in three different conditions: (1) due to an interface vacuolar dermatitis; (2) due to the association of SLE with other autoimmune blistering diseases (e.g. bullous pemphigoid); and (3) due to an autoimmune blistering disease related to antibodies anti-collagen VII. This last condition refers to bullous systemic lupus erythematous (BSLE).
BSLE is a rare blistering disease that mainly occurs in females (3-40 years old), and less frequently in children and adolescents.1-2 The involvement of sun-exposed areas is not mandatory, and is marked by the rapid and widespread development of tense vesicles and bullae over erythematous macules or plaques. Preferential sites are: superior trunk, proximal superior limbs, and face (lips)3 with symmetrical distribution. Mucosal involvement is common on perioral, pharyngeal, laryngeal, and genital areas. The lesions usually progress with no scarring, but hypo or hyperchromia may be present.
CASE REPORT
An 18-year-old female was hospitalized presenting slightly painful and pruritic vesicles and bullae on the face, oral mucosa, abdomen, thighs, and dorsum over the last 2 weeks. She also reported weight loss, migratory polyarthralgia with morning stiffness longer than 30 minutes, myalgia, fatigue, fever, alopecia, and malar rash associated with photosensitivity over the last 2 months. She was taking medroxyprogesterone acetate every 3 months as a contraceptive method. Her past medical history included a pregnancy at the age of 17, with low titers in the Venereal Disease Research Laboratory test, and a negative treponemal test.
The admission clinical examination revealed a prostrated and pale patient, with normal vital signs. Skin findings showed the presence of a malar rash, and multiple tense vesicles and bullae varying from 1 mm to 6 cm, over erythematous macules and plaques (Figure 1). These lesions were present on the face, including eyelids, arms, abdomen, dorsum, and thighs. Involvement of the vermilion border of the lips and oral mucosa was present. No lymphadenopathy and signs of arthritis were detected; the remaining physical examination revealed no alterations.
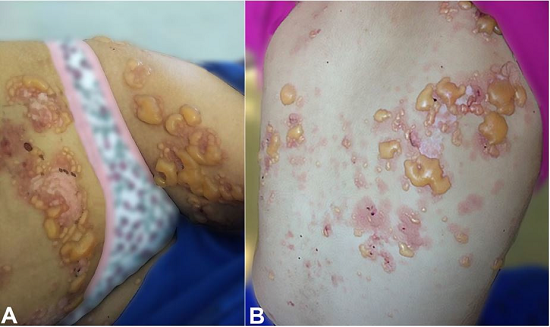
Figure 1
BSLE. Arciform and linear tense bullae of varying sizes over erythematous macules and plaques on abdomen, thighs (A) and dorsum (B). BSLE = bullous systemic lupus erythematous.
BSLE was considered as the first hypothesis, followed by the differential diagnosis of other conditions, such as: epidermolysis bullosa acquisita, bullous pemphigoid, dermatitis herpetiformis, linear immunoglobulin (Ig)A bullous dermatosis, and drug eruption.
General laboratory evaluation showed a complete blood cell count with hemoglobin levels of 11.0 g/dL (reference range [RR]: 12.3-15.3 g/dL), leukocytes of 2880 per mm3 (RR: 4.4-11.3 × 103/mm3) and platelet count of 105,000 per mm3 (RR: 150-400 × 103 /mm3). The direct Coombs assay was positive, but lactate dehydrogenase and bilirubin were at normal levels. Urinalysis revealed the presence of proteinuria, hematuria, and leukocyturia, with no casts. The spot urine protein/creatinine ratio was 856 mg/g (RR: < 300 mg/g). Renal function was preserved (creatinine of 0.63 mg/dL [RR: 0.4-1.3 mg/dL] and urea of 15 mg/dL [RR: 5-25 mg/dL]). Complement was at low levels, C3 < 21 mg/dL (RR: 67-149 mg/dL) and C4 = 7 mg/dL (RR: 10-38mg/dL); anti-nuclear antibody was positive (1/640) with a homogeneous nuclear pattern, anti-double-stranded DNA and anti-histone were positive.
Skin histopathology revealed subepidermal cleavage with a neutrophilic inflammatory infiltrate (Figure 2). Direct immunofluorescence (DIF) showed positivity for IgG at the basement membrane zone (BMZ) in a linear pattern (Figure 3A). Salt-split skin indirect immunofluorescence showed the presence of IgG deposits at the dermal side of cleavage (negative for IgA and IgM) (Figure 3B). ELISA was positive for antibodies against type VII collagen.
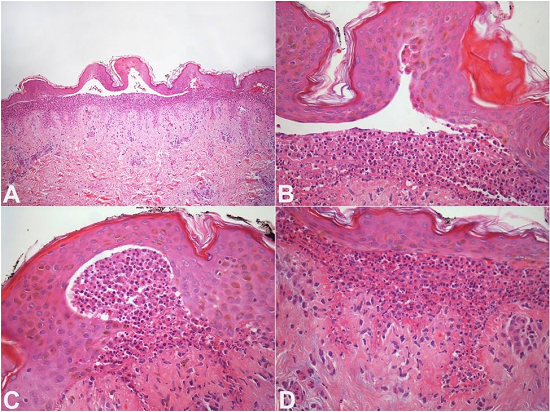
Figure 2
BSLE. Skin histopathology. A - Subepidermal cleavage with neutrophils. The inflammatory cells are disposed along the papillary dermis; B - Subepidermal cleavage partially filled with neutrophils; C - Subepidermal blister with neutrophils and inflammatory cells at the periphery of the bulla; D - Neutrophils are present in the superficial dermis. Note the thickening of the basement membrane. (H&E, A:100X; B, C, and D: 400X). BSLE = bullous systemic lupus erythematous.
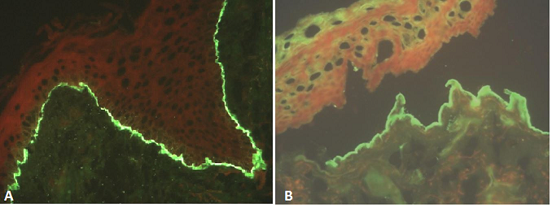
Figure 3
A - Direct immunofluorescence shows homogenous IgG deposit at the basement membrane zone (400X); B - Indirect immunofluorescence (salt-split skin), human foreskin substrate: IgG deposits at the dermal side of the split (400X).
The clinical and laboratory findings confirmed the diagnosis of BSLE.
The patient was treated with oral prednisone 1 mg/kg/d along with hydroxychloroquine 400 mg/d 5 days per week. Skin lesions and other SLE symptoms markedly improved after 1 week of treatment and the patient was discharged. However, the skin lesions on the vermilion border of the lips, eyelids, forearms, dorsum, and thighs relapsed soon after, despite adherence to treatment. Hospitalization was necessary due to the extent of skin involvement and systemic symptoms. Dapsone 200 mg/d was added to the therapeutic regimen, with subsequent improvement of the clinical features. Hydroxychloroquine was withdrawn due to hemolysis.
DISCUSSION
BSLE is a rare autoimmune blistering disorder that usually presents as tense bullae and/or vesicles in patients with a previously known or concurrent diagnosis of SLE. Due to the rarity of the disease, epidemiologic data are limited. A French study4 showed an incidence of bullous skin disorders (excluding bullous pemphigoid) of 3.4 cases per million per year. The occurrence of BSLE among children and adolescents with SLE is around 1%.2 Data on BSLE are scarce; therefore, the knowledge about the disease is mostly based on case reports or case series.
BSLE patients usually present with clinical features of SLE. The blisters predominantly occur on the face, neck, upper extremities, oral mucosa, and vermilion border. Development of bullae is triggered by sun exposure. Bullous lesions are tense and heal with no scarring.5-13 Mucosal involvement features include either blisters or painless ulcers. Esophagitis dissecans was reported.14 Differential diagnoses usually include other subepidermal bullous diseases such as bullous pemphigoid (BP), epidermolysis bullosa acquisita (EBA), linear IgA bullous dermatosis, and dermatitis herpetiformis (DH).15
The histopathology of BSLE usually shows a subepidermal bullae with neutrophil-predominant inflammatory infiltrate below the bullae formation. The presence of micro abscesses in the dermal papillae is also frequent, which makes the histopathology features of BSLE very similar to DH.16 In BSLE, neutrophils tend to be in a dispersed arrangement (Figure 2C) and may extend into the papillary dermal collagen (Figure 2D). Deposits of mucin are often seen in the deep dermis.17 In counterpart, the neutrophilic infiltrate in DH is focal and localized at the papillary dermis.
The immunopathologic findings in BSLE include: direct immunofluorescence with linear IgG deposits at BMZ, and occasional granular IgA, IgM, and C3 deposits.17 Salt-split skin indirect immunofluorescence reveals IgG deposition at the dermal side of cleavage, and is a helpful tool for BSLE.13 The presence of autoantibodies against type VII collagen may be detected in patients with BSLE.18,19
Camisa and Sharma20 were the first authors to establish diagnostic criteria for BSLE. In 1988, with the use of immunofluorescence, these diagnostic criteria were reviewed and are shown in Table 121,22

BMZ = basement membrane zone; BSLE = bullous systemic lupus erythematosus; DH = dermatitis herpetiformis; DIF = direct immunofluorescence; IF = indirect immunofluorescence; Ig = immunoglobulin; SLE = systemic lupus erythematosus.
The current therapeutic regimens for BSLE are based on the pathogenesis of the disease, case-reports, and case-series. Dapsone is used as first-line therapy6,11,23-25 along with glucocorticoids and immunosuppressants. Other therapeutic options include mycophenolate mofetil5,7,13,26 methotrexate,27 rituximab,28 hydroxychloroquine and intravenous immunoglobulin.8
REFERENCES
Watanabe T, Tsuchida T. Classification of lupus erythematosus based upon cutaneous manifestations. Dermatological, systemic and laboratory findings in 191 patients. Dermatology. 1995;190(4):277-83. http://dx.doi.org/10.1159/000246716. PMid:7655105
Lourenço DM, Gomes RC, Aikawa NE, Campos LM, Romiti R, Silva CA. Childhood-onset bullous systemic lupus erythematosus. Lupus. 2014;23(13):1422-5. http://dx.doi.org/10.1177/0961203314544187. PMid:25074872
Nico MMS, Lourenço SV. Multiple Blisters Along the Lip Vermilion are a Clue to Bullous Lupus Erythematosus. Acta Derm Venereol. 2012;92(4):404-5. http://dx.doi.org/10.2340/00015555-1276. PMid:22293956
Bernard P, Vaillant L, Labeille B, et al. Incidence and distribution of subepidermal autoimmune bullous skin diseases in three French regions. Arch Dermatol. 1995;131(1):48-52. http://dx.doi.org/10.1001/archderm.1995.01690130050009. PMid:7826096
Li AW, Weed JG, Stamey CR, Subtil A, Tomayko MM, Antaya RJ. Bullous systemic lupus erythematosus in a 6‐year‐old boy. Pediatr Dermatol. 2017;34(6):e313-6. http://dx.doi.org/10.1111/pde.13258. PMid:28851077
Maggio MC, Corsello G, Prinzi E, Cimaz R. Systemic lupus erythematosus and bullous pemphigoid with dramatic response to dapsone. Am J Case Rep. 2017;18:317-9. http://dx.doi.org/10.12659/AJCR.902351. PMid:28352068
Mahesh E, Madhyastha PR, Varma V, Gurudev KC, Gireesh MS, Bande SR. Unusual presentation of systemic lupus erythematosus. Saudi J Kidney Dis Transpl. 2017;28(3):653-6. http://dx.doi.org/10.4103/1319-2442.206468. PMid:28540909
Juhász M, Rogge M, Chen M, Czernik A, Kim SJ, Geller L. Case of pediatric bullous systemic lupus erythematosus treated with intravenous immunoglobulin. Pediatr Dermatol. 2017;34(1):e54-6. http://dx.doi.org/10.1111/pde.13015. PMid:27778386
Grabell DA, Matthews LA, Yancey KB, Chong BF. Detection of type VII collagen autoantibodies before the onset of bullous systemic lupus erythematosus. JAMA Dermatol. 2015;151(5):539-43. http://dx.doi.org/10.1001/jamadermatol.2014.4409. PMid:25671758
Christodoulou G, Powell M, Nguyen V, Watters K, Jafarian F. An atypical case of bullous systemic lupus erythematosus in a 16‐year‐old boy. Pediatr Dermatol. 2014;31(6):e164-6. http://dx.doi.org/10.1111/pde.12389. PMid:25040539
Liu KL, Shen J, Yang C, Chen YJ. Bullous systemic lupus erythematosus in a child responding to Dapsone. Pediatr Dermatol. 2014;31(4):e104-6. http://dx.doi.org/10.1111/pde.12340. PMid:24846529
Burke KR, Green BP, Meyerle J. Bullous lupus in an 18‐year‐old. Pediatr Dermatol. 2011;28(4):483. http://dx.doi.org/10.1111/j.1525-1470.2011.01502.x. PMid:21793895
Tincopa M, Puttgen KB, Sule S, Cohen BA, Gerstenblith MR. Bullous lupus: an unusual initial presentation of systemic lupus erythematosus in an adolescent girl. Pediatr Dermatol. 2010;27(4):373-6. http://dx.doi.org/10.1111/j.1525-1470.2010.01179.x. PMid:20653856
Yogarajah M, Sivasambu B, Jaffe EA. Bullous systemic lupus erythematosus associated with esophagitis dissecans superficialis. Case Rep Rheumatol. 2015;2015:930683. http://dx.doi.org/10.1155/2015/930683. PMid:25821624
Contestable JJ, Edhegard KD, Meyerle JH. Bullous systemic lupus erythematosus: a review and update to diagnosis and treatment. Am J Clin Dermatol. 2014;15(6):517-24. http://dx.doi.org/10.1007/s40257-014-0098-0. PMid:25358414
Burrows NP, Bhogal BS, Black MM, et al. Bullous eruption of systemic lupus erythematosus: a clinicopathological study of four cases. Br J Dermatol. 1993;128(3):332-8. http://dx.doi.org/10.1111/j.1365-2133.1993.tb00180.x. PMid:8471519
Vassileva S. Bullous systemic lupus erythematosus. Clin Dermatol. 2004;22(2):129-38. http://dx.doi.org/10.1016/j.clindermatol.2003.12.020. PMid:15234014
Shirahama S, Furukawa F, Yagi H, Tanaka T, Hashimoto T, Takigawa M. Bullous systemic lupus erythematosus: detection of antibodies against noncollagenous domain of type VII collagen. J Am Acad Dermatol. 1998;38(5 Suppl):844-8. http://dx.doi.org/10.1016/S0190-9622(98)70472-4. PMid:9591800
Bain EE, Grover RK, Plunkett RW, Beutner EH. Detection of collagen VII autoantibodies to NC1 and NC2 domains of collagen VII by ELISA in suspected epidermolysis bullosa acquisita and bullous lupus erythematosus patients. J Dermatol Sci. 2012;65(2):155-6. http://dx.doi.org/10.1016/j.jdermsci.2011.12.004. PMid:22225828
Camisa C, Sharma HM. Vesiculobullous systemic lupus erythematosus: report of two cases and a review of the literature. J Am Acad Dermatol. 1983;9(6):924-33. http://dx.doi.org/10.1016/S0190-9622(83)70210-0. PMid:6358284
Camisa C, Grimwood RE. Indirect immunofluorescence in vesiculobullous eruption of systemic lupus erythematosus. J Invest Dermatol. 1986;86(5):606. http://dx.doi.org/10.1111/1523-1747.ep12355583. PMid:3528314
Camisa C. Vesiculobullous systemic lupus erythematosus: a report of four cases. J Am Acad Dermatol. 1988;18(1 Pt 1):93-100. http://dx.doi.org/10.1016/S0190-9622(88)70014-6. PMid:3279084
Piette EW, Werth VP. Dapsone in the management of the autoimmune bullous diseases. Dermatol Clin. 2011;29(4):561-4. http://dx.doi.org/10.1016/j.det.2011.06.018. PMid:21924998
Hall RP, Lawley TJ, Smith HR, Katz SI. Bullous eruption of systemic lupus erythematosus. Dramatic response to dapsone therapy. Ann Intern Med. 1982;97(2):165-70. http://dx.doi.org/10.7326/0003-4819-97-2-165. PMid:7049027
Hans-Bittner NR, Bittner GC, Hans G Fo, Takita LC. Bullous systemic lupus erythematosus in a 10-year-old child. An Bras Dermatol. 2017;92(5 Suppl 1):37-9. http://dx.doi.org/10.1590/abd1806-4841.20176074. PMid:29267441
Pehr K. Mycophenolate mofetil and erythromycin for bullous lupus erythematosus of childhood. J Cutan Med Surg. 2012;16(1):50-3. http://dx.doi.org/10.1177/120347541201600110. PMid:22417996
Malcangi G, Brandozzi G, Giangiacomi M, Zampetti M, Danieli MG. Bullous SLE: response to methotrexate and relationship with disease activity. Lupus. 2003;12(1):63-6. http://dx.doi.org/10.1191/0961203303lu241cr. PMid:12587829
Alsanafi S, Kovarik C, Mermelstein AL, Werth VP. Rituximab in the treatment of bullous systemic lupus erythematosus. J Clin Rheumatol. 2011;17(3):142-4. http://dx.doi.org/10.1097/RHU.0b013e318214f30c. PMid:21441817
Notes
Author notes
Correspondence Eduardo Messias Hirano Padrão Rua Leonardo Motta, 100, Apto 92 - Vila Indiana - São Paulo/SP - Brazil CEP: 05586-090 Phone: +55 (11) 97547-3987 padraoedu@gmail.com
Conflict of interest declaration
