Article / Autopsy Case Report
Late stillbirth due to listeriosis
Late stillbirth due to listeriosis
Autopsy and Case Reports, vol. 8, no. 4, e2018051, 2018
Hospital Universitário da Universidade de São Paulo
Received: 06 August 2018
Accepted: 31 August 2018
Abstract: Listeriosis is a sporadic infectious disease, which affects high-risk populations, such as the elderly, pregnant women, newborns, and immunocompromised patients. During pregnancy, listeriosis usually presents like a mild non-specific infection, but it may be responsible for fetal loss, preterm labor, early onset neonatal sepsis, and neonatal death. We report the case of a late stillbirth secondary to maternal chorioamnionitis. Listeria monocytogenes was isolated from the amniotic fluid and the fetal pleural fluid. The fetal autopsy revealed a disseminated inflammatory response with multi-organ involvement. This case illustrates the importance of the prevention and the diagnosis of listeriosis during gestation and may help us to understand the physiopathology of fetal loss due to listeriosis.
Keywords: Listeria Monocytogenes, Stillbirth, Infant, Newborn, Chorioamnionitis, Autopsy.
INTRODUCTION
Listeria monocytogenes is a Gram-positive bacillus that causes listeriosis, a rare (incidence of 0.7/100.000; 12/100.000 in pregnant women) but potentially fatal infection.1 An increased prevalence of listeriosis has been reported in the last two decades in several European countries, including Spain (with an incidence of 0.75/1000 pregnancies between 2005 and 2010).2,3
Sporadic and outbreak-related cases are linked to the consumption of food contaminated with Listeria spp.4 During pregnancy, Listeria infection occurs more frequently in the third trimester (66%), and in most cases, the maternal sickness is usually mild with non-specific symptoms (fever, flu-like syndrome, plus abdominal and back pain) or even asymptomatic.5 The fetal and neonatal complication becomes less frequent as the gestational age increases. However, the complication may be represented by preterm birth (≈ 65%), early onset neonatal sepsis (≈ 60%), and fetal loss (≈ 25%).6
Regarding the late stillbirth (after 28 weeks of gestation), the ethiopathogenic role of the maternal listeriosis is not fully understood due to the scarcity of published articles on this issue.7 It is hypothesized that after ingestion of the contaminated food, the Listeria enters into the bloodstream through bacterial translocation, and then disseminates, having a particular predilection for the fetoplacental unit, causing fetal infection.8
The intracellular growth of L. monocytogenes, the administration of antibiotics during the delivery, and the parental position opposing to the fetal autopsy performance and microbiological investigation hamper the study of the role of listeriosis as the cause of stillbirth.
In this work, we report an antepartum stillbirth at 29 weeks of gestation secondary to maternal infection by L. monocytogenes. The thorough clinical and anatomopathological description of our case may contribute to the improvement of the knowledge of this entity.
CASE REPORT
A 37-year-old pregnant woman (29 weeks of gestation) was attended in the Emergency Department because of a premature labor. She complained of abdominal pain, fever, and loss of dark amniotic fluid over the last hour. The day before, she attended the physician complaining of a decrease in fetal movements, but fetal distress was ruled out with the aid of the cardiotocography and fetal ultrasonography.
However, on admission, a new cardiotocography was performed and revealed prolonged fetal bradycardia (70 bpm). Therefore, a cesarean section was promptly performed. The neonate was born extremely hypotonic with intense mucocutaneous pallor without respiratory movements. The heart auscultation revealed asystole. Unsuccessful advanced resuscitation maneuvers were performed for 10 minutes. The Apgar score at 10 minutes was zero, and cardiac electric activity remained asystole. The maneuvers were withdrawn, and the newborn was considered dead.
Neonatal blood analysis showed severe acidosis (pH= 6.8), anemia with a hemoglobin of 7 g/dL (reference range [RR]: 12-16 g/dL), and hematocrit 23% (RR: 35-47%). A complete blood count and biochemistry analysis of the mother showed anemia with hemoglobin of 6.7 g/dL (RR: 12-16 g/dL), leukocytosis of 16.3 × 103/mm3 (RR: 4-11 × 103/mm3), and serum C-reactive protein of 59 mg/L (RR: 0-5 mg/L). Antibiotic regimen with cefazolin was started. The Kleihauer-Betke test did not detect fetal hemoglobin in the maternal bloodstream.
AUTOPSY FINDINGS
The ectoscopic examination revealed a well-formed male newborn weighing 1500 g. After the thoracic cavity overture, multiple pulmonary hemorrhagic foci were found and bilateral mild pleural effusion was drained. Cerebral examination showed a massive bilateral intraventricular hemorrhage. Microscopic study of the lungs revealed collapsed alveoli, with multiple hemorrhagic foci and disseminated microabscesses formed by a necrotic material, cellular debris, and inflammatory cells. Similar lesions were seen in the esophagus, spleen, liver, and suprarenal glands (Figure 1). Microscopic study of the placenta and umbilical cord showed the deposit of fibrin and leucocyte aggregates, which were consistent with an acute inflammatory process. L. monocytogenes was isolated in the amniotic and pleural fluids, and in the blood culture of the mother. After the microbiological results, the antibiotic regimen was changed to ampicillin. Further blood cultures resulted negative. Post-partum anamnesis revealed that the mother consumed cheese from a rural area some days before the delivery.
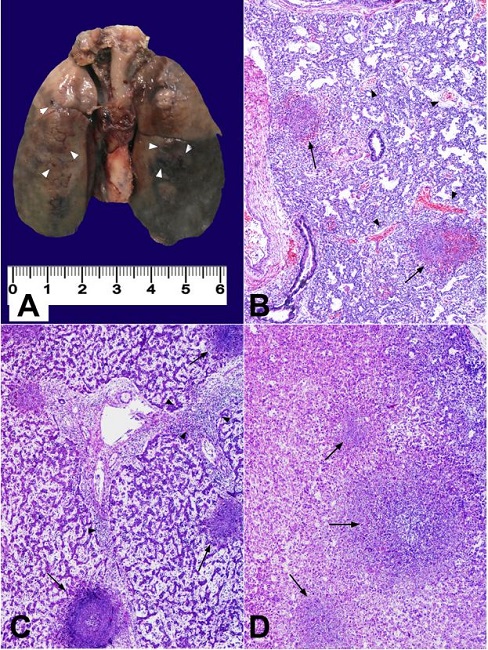
Figure 1
Findings in the fetus necropsy. A - Macroscopic appearance of lungs. Brown coloured areas and dark spotting (hemorrhagic and necrotic foci) are seen (marked with white arrow heads); B - Cresyl violeta staining of lung parenchyma showing collapsed alveolar spaces, with hemorrhagic areas (arrow heads) and abscesses (black arrows); C - Cresyl violeta staining of liver parenchyma, showing a portal space with active inflammatory process (arrow heads), surrounded by three abscesses (black arrows); D - Cresyl violeta staining of adrenal gland showing a central abscess with other similar satellite lesions (black arrows).
DISCUSSION
Stillbirth is defined as no sign of life in a neonate at delivery.9 It is considered late stillbirth when it occurs after the 28th week of gestation. The rate of stillbirths is 3-5 per 1000 births in high-income countries, where maternal or fetal infection accounts for 10-25% of them.7
Usually, maternal infections lead to stillbirth when they occur during the second and early third trimester of gestation. The most frequent microorganisms are summarized in Table 1

* Likely most common viral etiologic agent.** Associated Not likely causative.*** Probably the most common cause. CR = case reports; HIV = human immunodeficiency virus; IDS = immunodeficiency syndrome; U = uncommon. Modified from McClure and Goldenberg7
Infection may cause stillbirth through various mechanisms, including severe maternal illness, direct fetal infection, and placental injury. Ascending gestational infections secondary to bacteria, such as Escherichia coli, group B Streptococcus, or Ureaplasma urealyticum, are well-recognized causes of stillbirth. Nevertheless, it is likely that intracellular bacteria have an underestimated rate of miscarriage and stillbirth,8 specifically L. monocytogenes, which lacks substantial evidence of its physiopathogenic role due to the scarcity of information. Mylonakis et al.10 reported a series of cases of 11 maternal listeriosis with 2 stillbirths secondary to the infection acquired in the second trimester of gestation. Monari et al.11 reviewed 109 late stillbirths and reported 20 cases to be secondary to maternal infections; 2 of them were caused by L. monocytogenes. The cause of the fetal death secondary to late maternal infection is not well known due to the scarcity of published articles. Barikbin et al.12 reported 16 cases of perinatal listeriosis, with a high incidence of neonatal disease (14/16) with 2 cases of fatal outcome (granulomatosis infantiseptica in one patient and septic shock in the other).
The importance of the early diagnosis of maternal listeriosis lies in the reduction of adverse fetal outcomes when prompt antibiotic treatment is started.10,13 An important aspect to consider in the medical history is the previous ingestion of potentially contaminated food, which can be the clue for the diagnosis of maternal listeriosis. This information was present in up to 20% of cases of listeriosis in pregnancy.6 In the present case, the mother ate soft cheese from a rural area a few days before the delivery, but this fact was overlooked during her first visits to the Emergency Department. Another critical fact to make an early diagnosis is the microbiologic assessment. The highest concentration of bacteria are found in the fetal lung and gut, suggesting that the infection is acquired in utero via the inhalation of infected amniotic fluid.8 However, the isolation of Listeria in amniotic fluid is rare (≈10%).10 In the present case, Listeria was isolated in the stillborn pleural fluid and amniotic fluid, but not in the maternal blood, which may be due to previous antibiotic administration. We believe it is better to use new molecular diagnostic techniques to detect listeriosis in this high-risk population. The real-time polymerase chain reaction has shown a high sensitivity for early (less than 24 hours) detection of Listeria, and it can detect the bacteria despite the previous administration of antibiotic treatment.14,15
This case is the paradigm of a preventable and treatable maternal and fetal disease with a fatal outcome. It is essential for the health care providers of pregnant women to be aware of the non-specific symptoms of listeriosis and to make a targeted anamnesis focusing on the consumption of potentially contaminated food (e.g. unpasteurized soft cheese, hot dogs, refrigerated meat spreads, unwashed raw fruits and vegetables). An early diagnosis can be achieved by detecting this potential source of infection, and prompt maternal antibiotic treatment will allow a better neonatal outcome.
REFERENCES
1 Sapuan S, Kortsalioudaki C, Anthony M, et al. Neonatal listeriosis in the UK 2004 e 2014. J Infect. 2017;74(3):236-42. http://dx.doi.org/10.1016/j.jinf.2016.11.007. PMid:27867063
2 Allerberger F, Wagner M. Listeriosis: a resurgent foodborne infection. Clin Microbiol Infect. 2010;16(1):16-23. PMid:20002687
3 Sisó C, Goncé A, Bosch J, Salvia MD, Hernández S, Figueras F. Listeriosis in pregnancy: a secular trend in a tertiary referral hospital in Barcelona. Eur J Clin Microbiol Infect Dis. 2012;31(9):2125-32. http://dx.doi.org/10.1007/s10096-012-1545-6. PMid:22327372
4 The American College of Obstetricians and Gynecologists. Committee opinion nº 614: management of pregnant women with presumptive exposure to Listeria monocytogenes. Obstet Gynecol. 2014;124(6):1241-4. http://dx.doi.org/10.1097/01.AOG.0000457501.73326.6c. PMid:25411758
5 Janakiraman V. Listeriosis in pregnancy: diagnosis, treatment, and prevention. Rev Obstet Gynecol. 2008;1(4):179-85. PMid:19173022
6 Wadhwa Desai R, Smith MA. Pregnancy-related listeriosis. Birth Defects Res. 2017;109(5):324-35. http://dx.doi.org/10.1002/bdr2.1012. PMid:28398675
7 McClure EM, Goldenberg RL. Seminars in fetal & neonatal medicine infection and stillbirth. Semin Fetal Neonatal Med. 2009;14(4):182-9. http://dx.doi.org/10.1016/j.siny.2009.02.003. PMid:19285457
8 Baud D, Greub G. Intracellular bacteria and adverse pregnancy outcomes. Clin Microbiol Infect. 2011;17(9):1312-22. http://dx.doi.org/10.1111/j.1469-0691.2011.03604.x. PMid:21884294
9 Silva FT, Gonik B, McMillan M, et al. Stillbirth: case definition and guidelines for data collection, analysis, and presentation of maternal immunization safety data. Vaccine. 2016;34(49):6057-68. http://dx.doi.org/10.1016/j.vaccine.2016.03.044. PMid:27431422
10 Mylonakis E, Paliou M, Hohmann EL, Calderwood SB, Wing EJ. Listeriosis during pregnancy: a case series and review of 222 cases. Medicine. 2002;81(4):260-9. http://dx.doi.org/10.1097/00005792-200207000-00002. PMid:12169881
11 Monari F, Gabrielli L, Gargano G, et al. Fetal bacterial infections in antepartum stillbirth: a case series. Early Hum Dev. 2013;89(12):1049-54. http://dx.doi.org/10.1016/j.earlhumdev.2013.08.010. PMid:24041816
12 Barikbin P, Sallmon H, Huseman D, et al. Clinical, laboratory, and placental findings in perinatal Listeriosis. Fetal Pediatr Pathol. 2016;35(5):307-14. http://dx.doi.org/10.1080/15513815.2016.1179822. PMid:27212240
13 Lamont RF, Sobel J, Mazaki-Tovi S, et al. Listeriosis in human pregnancy: a systematic review. J Perinat Med. 2011;39(3):227-36. http://dx.doi.org/10.1515/jpm.2011.035. PMid:21517700
14 Kaur S, Malik SVS, Vaidya VM, Barbuddhe SB. Listeria monocytogenes in spontaneous abortions in humans and its detection by multiplex PCR. J Appl Microbiol. 2007;103(5):1889-96. http://dx.doi.org/10.1111/j.1365-2672.2007.03414.x. PMid:17953598
15 Le Monnier A, Abachin E, Beretti JL, Berche P, Kayal S. Diagnosis of Listeria monocytogenes meningoencephalitis by real-time PCR for the hly gene. J Clin Microbiol. 2011;49(11):3917-23. http://dx.doi.org/10.1128/JCM.01072-11. PMid:21918022
Notes
Author notes
Correspondence Antonio Segado-Arenas Division of Neonatology - Hospital Universitario Puerta del Mar Avenida Ana de Viya 21 (3rd floor), Cádiz CP 11009 - Spain Phone: +34 956 002 314 a.segadoarenas@gmail.com
Conflict of interest declaration
