Article / Clinical Case Report
Ameloblastoma with distinctive granular cell pattern: an 8 case study
Ameloblastoma with distinctive granular cell pattern: an 8 case study
Autopsy and Case Reports, vol. 8, no. 4, e2018052, 2018
Hospital Universitário da Universidade de São Paulo
Received: 11 July 2018
Accepted: 10 September 2018
Abstract: Ameloblastoma with granular cell pattern (AGC) is classified as an unusual histological subtype of solid/multicystic ameloblastoma, characterized by granular changes in stellate-like cells located within the inner portion of the epithelial follicles. Studies have revealed that lysosomal overload causes cytoplasmic granularity; however, the mechanism involved remains poorly understood. Here we report on eight cases of granular cell ameloblastoma, in the posterior region of the mandible. The age of the patients included in this case series ranged from 35 -64 years old and 87.5% of cases occurred in non-Caucasians, with a slight gender predilection for men (62.5%). There was no evidence of recurrence, and the majority of the cases were treated with surgical resection (87.5%). All tumors displayed histopathological features consistent with the diagnosis of ameloblastoma with granular cell pattern.
Keywords: Ameloblastoma, Odontogenic Tumors, Neoplasms.
INTRODUCTION
Odontogenic tumors comprise a complex group of lesions of diverse histopathological subtypes and clinical behavior, which originate from tissue remnants of tooth formation. Ameloblastoma is a true neoplasm with a particular tendency to rise from enamel organ type tissue and presents a slow growth pattern as well as the potential to be infiltrative and recurrent. Ameloblastoma is the second most common odontogenic tumor (11%).1,2 This lesion is found almost exclusively in the lower jaw, near the angle of the mandible, and occurs most often between the third and fifth decades of life.1,3
According to clinical and radiographic features, ameloblastomas are classified into conventional solid or multicystic, extraosseous or peripheral, desmoplastic or unicystic types.4 They can display distinctive histological findings, mainly characterized by the presence of cystic areas composed of peripheral columnar cells in a palisaded basal cell layer that surrounds hyperchromatic granular cells. There are six histological variants, follicular, plexiform, acanthomatous, desmoplastic, granular and basal cell, with follicular and plexiform being the most common subtype.3,4
Despite current nomenclature using the term “granular cell ameloblastoma”, this subtype is not classified as such by the latest update of the World Health Organization Classification of Head and Neck Tumours5. This study will therefore describe and discuss all findings under the term Ameloblastoma with granular cell pattern (AGC). AGC is a rare type of ameloblastoma that represents only 3-5% of all histological subtypes. Said tumor mostly presents as a combination of different histological patterns, particularly the follicular variant.1,6 This tumor was first observed and described by Krompecher in 1918 at which time it was called pseudoxanthomatous cells.1,2 It appears to be aggressive, with a marked propensity for recurrence and can progress to metastasis.2,7
Histologically, AGC is characterized by the presence of granular cells, which are usually small structures of around 1μm (compared to rare giant granular cells that are up to 30μm). These cells typically appear within the central area of ameloblastic tumor follicles, progressively replacing the stellate reticulum, which may also extend to the peripheral columnar or cuboidal cells.2 The granular cell can vary from the most common round shape to cuboidal, columnar or angular, in which the nuclei are pyknotic and hyperchromatic, and the cytoplasm is filled with acidophilic granules, identified as lysosomal aggregates.2,8 The presence of granular cells represents either a transitional or matured stage in the life cycle of ameloblastomas. They start as normal stellate reticulum-like cells, followed by the production of cytoplasmic granules and finally tissue degeneration and formation of cystic areas is seen.6
The current treatment for AGC is similar to other subtypes of ameloblastomas, which consists of accurate preoperative diagnosis and radical surgical methods. Regular follow-up management is recommended as conservative treatments, such as enucleation or curettage, exhibit a high rate of recurrence.2 Previous studies suggest that AGC represents more aggressive lesions, whereas others propose that AGC is just a transitional phase during the pathological course of ameloblastomas, therefore not requiring particular attention.1,2
The prognosis of AGC is considered favorable when treated early; however, a recurrence rate of over 30% has been reported, implying a potentially unknown, aggressive behavior on the part of the AGC9. A proper regular follow-up is therefore recommended, as recurrence has been shown to happen up to eight years after initial treatment.1,2 The uncertain clinical behavior reported for this type of ameloblastoma emphasizes the necessity of better understanding its pathogenesis and clinical impact. As a consequence, this article aims to report on the clinical and histological features of a retrospective case series of eight mandibular AGC.
CASE SERIES
Given that AGC represents a rare pathological entity and that there are only a few reports in the literature comparing their clinical and histopathological characteristics, a retrospective study of eight patients was performed. Clinical, demographic and histopathological data for all AGC diagnosed in AC Camargo Hospital Cancer Center in the Department of Pathological Anatomy, SP Brazil, from 1953 to 2001, were evaluated in accordance with the approval given by the Faculty Research Ethics Committee of the School of Dentistry, University of São Paulo - USP (Protocol number 171/08; FR-216880).
All slides were stained with hematoxylin-eosin (HE) and were reviewed using an optical microscope (Olympus Cover); tumors were classified according to WHO criteria5. Two independent evaluators analyzed the slides, and in cases where there was disagreement a third evaluator was requested, and the result was established in consensus.
RESULTS
The clinical features of all patients were obtained from their medical records, which included age, ethnicity, gender, the location of the lesion, treatment, recurrence, radiographic and histopathological findings, shown in Table 1

C = Caucasian; nC = non-Caucasian; nd = not detected; SR = surgical resection; M = Men; F = Female.
This retrospective analysis of an AGC case series included eight patients ranging from 35 to 64 years old (average age of 46.8 years) with a slight predilection for men (62.5%) and for non-Caucasians (87.5%). The majority of the cases were treated with surgical resection (87.5%) and presented no evidence of recurrence during a follow-up of more than 3 years. All AGC included in this study were found in the posterior region of the mandible particularly near the angle, with a multilocular radiographic pattern.
Paraffin-embedded sections of all eight cases were stained with hematoxylin and eosin to further morphological analysis as shown in Figure 1, 2 to 3. All cases presented a tumor area composed of granular cells with small and hyperchromatic nuclei (high magnification on Figure 1A, arrow) surrounded by ameloblastic epithelium arranged in palisaded basal cell layer (Figures 1A and 1B, arrowheads).
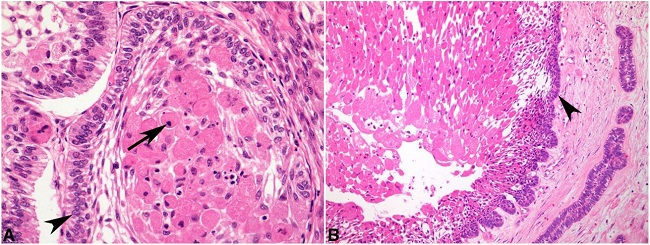
Figure 1
Photomicrograph of the tumors. A - Case 4 highlights classical granular cell content with varied cell shape, pyknotic nuclei (arrow) and palisaded epithelial basal cell layer surrounding granular cell clusters (arrowhead) (H&E, 400X); B - Case 2 presents localized granular cell cluster surrounded by typical palisaded epithelium (arrowhead) (H&E,100X).

Figure 2
Photomicrograph of the tumors. A - Case 7 presents extensive granular cell content in between epithelial and inflammatory tissue (asterisk) (H&E, 200X); B - Case 3 shows widespread typical ameloblastic epithelium interlaced within areas of granular cells (H&E, 200X); C - Case 6 displays extensive granular cell cluster with partially absent epithelial periphery (H&E, 200X); D - Case 8 illustrates the epithelial boundary of a granular cell cluster within mature connective tissue (H&E, 200X).
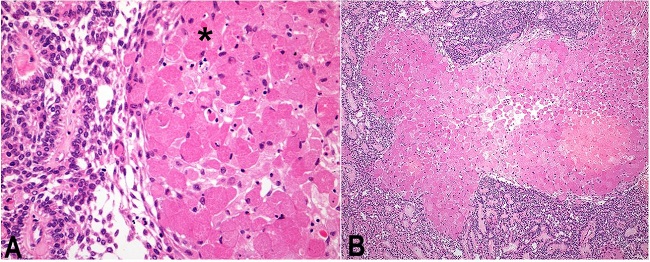
Figure 3
Photomicrograph of the tumors. A - Case 1 shows granular cell nests (asterisk) surrounded by highly dense and proliferative ameloblastic epithelium (H&E, 400X); B - Case 5 shows extensive granular cell cluster within the ameloblastic epithelium (H&E, 100X).
The granular cells were usually packed and distributed in one or multiple clusters throughout the epithelium, forming islands of tumor mass, sometimes also presenting a great number of inflammatory cells (Figure 2A, asterisk). There was variation in both size and shape of these granular cells within the clusters, which were mostly round and presenting large cytoplasm. The connective tissue wrapping the tumor area was well-developed and prominently poorly vascularized (Figures 2B, 2C, and 2D). Cell pleomorphism and mitosis were absent.
This eight-case series of AGC showed the presence of multiple typical granular cells in the center of the neoplastic nests, where the peripheral palisaded columnar or cuboidal cells were entirely or partially limiting the tumor areas. The differential diagnosis included granular cell myoblastoma and granular cell ameloblastic fibroma; however, the histopathological findings associated with the clinical and multilocular radiographic features resulted in the final diagnosis of AGC for all cases.
DISCUSSION
Ameloblastoma with granular cell pattern is rare, representing less than 5%, and unusual histological variant of the odontogenic tumor ameloblastoma. Reichart et al.9 have reported via an extensive review study from 1960 to 1993 only 56 cases of AGC out of a total of 1593 cases of ameloblastomas (3.5%). Likewise, Kameyama et al.10 in a clinicopathological study observed only one AGC out of 77 ameloblastomas included in said study.
A case series published by Hartman et al.11 evaluated all radiograph and clinical aspects of 20 patients from the United States of America diagnosed with AGC, where they reported a slight predilection for men of 66%, similar to our findings (62.5%). Both studies revealed that all cases of AGC were found in the posterior region of the mandible which aligns with reports found in the literature. They also described that AGC was predominant in Caucasians (60%) in contrast to our study which showed a prevalence of cases in non-Caucasians (87.5%). Regarding average age, Hartman et al. found an average age of 40.7 compared to our finding of 46.8.
Histopathologically, AGC showed significant tumor cell transformation in the cytoplasm caused by the presence of lysosomes, which corroborates with our results. These cells are usually stellate reticulum-like and present very coarse, granular and eosinophilic cytoplasm. The “granular change” is reported to happen due to a dysfunctional status of neoplastic cells, and the pathogenesis of this tumor seems to be age-related.6,8,9. However, whether the granular cell change in ameloblastoma is a degenerative process or a harbinger of a more aggressive course remains unclear.12
The differential diagnosis of AGC includes other oral lesions with a similar morphology of granular cell accumulation, such as granular cell myoblastoma, congenital epulis and granular cell ameloblastic fibromas. Although the morphology of all granular cells is similar, the tissue of origin is particular to each condition. Also, importantly, these lesions present different biological pathways and should be discriminated from AGC.3,6,12
There has been considerable interest regarding the nature of granular cells in ameloblastoma ever since it was first described. Studies have reported that these granular cells are derived from the odontogenic epithelium, rather than the connective tissue, as they highly express cytokeratins.13,14 The transformation of neoplastic cells into granular cells is currently attributed to “a degenerative process in a long-standing lesion or a dysfunctional status of neoplastic cells” or a “metabolic change in response to an aging phenomenon or an indicator of a more aggressive course”.2,10 Ultrastructural analyses have previously shown the presence of lysosomal aggregates in the cytoplasm of granular cells, and apoptotic cell fragments were detected between these cells within the neoplastic clusters, corroborating this hypothesis.14
The basement membrane represents the barrier between the adjacent tissues, which provides mechanical support and plays an important role in the development of the disease by regulating the cell migration, adhesion, and proliferation. Proteins from the basement membrane have been shown to be expressed in several types of odontogenic tumors and can define differences between their biological behavior.15 An immunohistochemical study of a single case of AGC has shown moderate to strong protein expression of laminin and fibronectin within the granular cells, whereas collagen IV was utterly absent.16 This corroborates similar findings in follicular ameloblastomas and supports the hypothesis of age-related granular transformation, although further studies of AGC are required.
The biological behavior of AGC does not seem to differ from the other histologic subtypes of ameloblastoma, as it has been reported to be locally aggressive and recurrent. Reichart et al.9 reported a 33.3% recurrence rate for this lesion, which was higher than the more common follicular, plexiform and acanthomatous subtypes. In Hartman’s study11, 11 of 15 patients (73%) developed recurrent AGC lesions. This was different from our case series, where no recurrence was detected among the 8 cases evaluated over more than three years of follow-up; therefore, no correlation of AGC with recurrence could be confirmed. This difference in recurrence rate is likely due to the surgical procedure performed in our sample, as the majority of the cases received less conservative treatment (surgical resection 87.5%).
Metastatic AGC is rare, and its etiopathogenesis remains unknown. None of the AGC cases reported here were metastatic; however, a few cases have been previously reported. When spreading occurred it mostly moved to the cervical lymph nodes, the lungs, the thorax or to the neck.17-20 These cases presented a long duration from the time of diagnosis to the end of treatment, ranging from 18 to 45 years. The exception to this being the latest case report which involved only two years of progression.21 Most of them were recurrent with aggressive behavior, however this unfavorable prognosis cannot be strictly linked to AGC as almost all the aforementioned reports may not reflect current treatments and have not shown any strong molecular correlation to their pathogenesis. Early diagnosis is therefore essential and is currently the best available measure to prevent distinct recurrences and potential malignancy.
CONCLUSION
This study reported on eight retrospective cases of AGC with no history of recurrence or aggressive behavior. AGC is a rare condition with particular histopathological findings of granular cell transformation, whose clinical significance is still under investigation. The treatment and prognosis of AGC are similar to that of other subtypes of ameloblastoma; however, case reports highlighted the potential risk of developing recurrences and metastasis if early diagnosis and proper treatment are not provided. A better understanding of the molecular pathogenesis of AGC would, therefore, improve diagnosis and therapeutic procedures.
REFERENCES
1 Dave A, Arora M, Shetty VP, Saluja P. Granular cells in ameloblastoma: an enigma in diagnosis. Indian J Dent. 2015;6(4):211-4. http://dx.doi.org/10.4103/0975-962X.165048. PMid:26752883
2 Wu Y-H, Chang JY-F, Wang Y-P, Chiang C-P. Langerhans cells in granular cell ameloblastoma. J Formos Med Assoc. 2017;114:781-2. http://dx.doi.org/10.1016/j.jfma.2017.02.007. PMid:28284724
3 Hunasgi S, Koneru A, Chauhan DS, Guruprasad Y. Rare giant granular cell ameloblastoma: a case report and an immunohistochemical study. Case Rep Dent. 2013;2013:372781. http://dx.doi.org/10.1155/2013/372781. PMid:23533826
4 Nikitakis NG, Tzerbos F, Triantafyllou K, Papadimas C, Sklavounou A. Granular cell ameloblastoma: an unusual histological subtype report and review of literature. J Oral Maxillofac Res. 2011;1(4):e3. PMid:24421980
5 Wright JM, Vered M. Update from the 4th Edition of the World Health Organization Classification of Head and Neck Tumours: odontogenic and maxillofacial bone tumors. Head Neck Pathol. 2017;11(1):68-77. http://dx.doi.org/10.1007/s12105-017-0794-1. PMid:28247226
6 Etebarian A, Asareh F, Motahhary P. Granular cell type of a unicystic ameloblastoma: an unusual case and review of the literature. J Oral Maxillofac Pathol. 2014;18(2):331. http://dx.doi.org/10.4103/0973-029X.140927. PMid:25328325
7 Jansari TR, Samanta S, Trivedi P, Shah MJ. Granular cell ameloblastoma of mandible. Indian J Pathol Microbiol. 2014;57(2):305-7. http://dx.doi.org/10.4103/0377-4929.134725. PMid:24943773
8 Yamunadevi A, Madhushankari GS, Selvamani M, Basandi PS, Yoithapprabhunath TR, Ganapathy N. Granularity in granular cell ameloblastoma. J Pharm Bioallied Sci. 2014;6(5, Suppl 1):S16-20. http://dx.doi.org/10.4103/0975-7406.137253. PMid:25210361
9 Reichart PA, Philipsen HP, Sonner S. Ameloblastoma: biological profile of 3677 cases. Eur J Cancer B Oral Oncol. 1995;31B(2):86-99. http://dx.doi.org/10.1016/0964-1955(94)00037-5. PMid:7633291
10 Kameyama Y, Takehana S, Mizohata M, et al. A clinicopathological study of ameloblastomas. Int J Oral Maxillofac Surg. 1987;16(6):706-12. http://dx.doi.org/10.1016/S0901-5027(87)80057-7. PMid:3125270
11 Hartman KS. Granular-cell ameloblastoma. Oral Surg Oral Med Oral Pathol. 1974;38(2):241-53. http://dx.doi.org/10.1016/0030-4220(74)90063-2. PMid:4528580
12 Thillaikarasi R, Balaji J, Gupta B, et al. Cystic granular cell ameloblastoma. J Maxillofac Oral Surg. 2010;9(3):310-3. http://dx.doi.org/10.1007/s12663-010-0083-y. PMid:22190813
13 Dina R, Marchetti C, Vallania G, Corinaldesi G, Eusebi V. Granular cell ameloblastoma: an immunocytochemical study. Pathol Res Pract. 1996;192(6):541-6. http://dx.doi.org/10.1016/S0344-0338(96)80103-8. PMid:8857640
14 Kumamoto H, Ooya K. Immunohistochemical and ultrastructural investigation of apoptotic cell death in granular cell ameloblastoma. J Oral Pathol Med. 2001;30(4):245-50. http://dx.doi.org/10.1034/j.1600-0714.2001.300409.x. PMid:11302245
15 Henriques ÁCG, Vasconcelos MG, Galvão HC, de Souza LB, de Almeida Freitas R. Comparative analysis of the immunohistochemical expression of collagen IV, MMP-9, and TIMP-2 in odontogenic cysts and tumors. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2011;112(4):468-75. http://dx.doi.org/10.1016/j.tripleo.2011.05.033. PMid:21911204
16 Lapthanasupkul P, Poomsawat S, Chindasombatjaroen J. Investigation of basement membrane proteins in a case of granular cell ameloblastoma. Int J Oral Sci. 2012;4(1):45-9. PMid:22361945
17 Cranin AN, Bennett J, Solomon M, Quarcoo S. Massive granular cell ameloblastoma with metastasis: report of a case. J Oral Maxillofac Surg. 1987;45(9):800-4. http://dx.doi.org/10.1016/0278-2391(87)90207-2. PMid:3476703
18 Hoke HF Jr, Harrelson AB. Granular cell ameloblastoma with metastasis to the cervical vertebrae: observations on the origin of the granular cells. Cancer. 1967;20(6):991-9. http://dx.doi.org/10.1002/1097-0142(196706)20:6<991::AID-CNCR2820200609>3.0.CO;2-V. PMid:6026472
19 Tsukada Y, Delapava S, Pickren JW. Granular-cell ameloblastoma with metastasis to the lungs: report of a case and review of the literature. Cancer. 1965;18(7):916-25. http://dx.doi.org/10.1002/1097-0142(196507)18:7<916::AID-CNCR2820180722>3.0.CO;2-T. PMid:14308241
20 Takahashi K, Kitajima T, Lee M, et al. Granular cell ameloblastoma of the mandible with metastasis to the third thoracic vertebra: a case report. Clin Orthop Relat Res. 1985;(197):171-80. PMid:4017332
21 Bansal A, Bhatnagar A, Saxena S. Metastasizing granular cell ameloblastoma. J Oral Maxillofac Pathol. 2012;16(1):122-4. PMid:22434948
Notes
Author notes
Correspondence Ana Maria Hoyos Cadavid Department of Stomatology - School of Dentistry - Universidade de São Paulo (USP) Avenida Professor Lineu Prestes, 2227 - Butantã - São Paulo/SP - Brazil CEP: 05508-000 Phone: +55 (11) 98283-3520 ana_hoyosc@hotmail.com
Conflict of interest declaration
