Article / Autopsy Case Report
Fatal necrotizing Candida esophagitis in a patient with leukocytoclastic cutaneous vasculitis and ankylosing spondylitis
Fatal necrotizing Candida esophagitis in a patient with leukocytoclastic cutaneous vasculitis and ankylosing spondylitis
Autopsy and Case Reports, vol. 9, no. 2, e2018070, 2019
Hospital Universitário da Universidade de São Paulo
Received: 19 June 2018
Accepted: 24 December 2018
Abstract: Esophageal infection by Candida spp. is a common opportunistic entity in immunocompromised hosts; however, systemic fungal dissemination due to perforation or transmural necrosis, also known as necrotizing Candida esophagitis (NCE), is rare. We report the case of a 61-year-old male patient with diagnosed ankylosing spondylitis, severe arteriosclerosis, and vasculitis under immunosuppressive therapy who presented NCE with fungal and bacterial septicemia diagnosed at autopsy. Necrotizing esophagitis is a rare manifestation of Candida infection, which may be a final complication in severely ill patients. Unfortunately, it may be underdiagnosed, and we call attention to this devastating complication in patients with leukocytoclastic cutaneous vasculitis and ankylosing spondylitis.
Keywords: Necrosis, Esophagitis, Candidiasis, Invasive, Mycoses.
CASE REPORT
A 61-year-old man was referred to the emergency care facility because of symptomatic hypotension diagnosed by the primary care physician. His past medical history included the diagnosis of ankylosing spondylitis over the last 10 years, bilateral cataracts, and a major depressive disorder with progressive and marked weight loss. Over the last 5 months, he complained of pain with necrosis in the extremities (hands and feet). Since then, he had been empirically prescribed prednisone and pentoxifylline by his primary care physician, and started wearing gloves. He lived alone and showed signs of depression since the loss of his parents, and had become progressively anorectic. Despite this empiric therapeutic attempt, the clinical picture worsened over the following months, with generalized weakness, asthenia, and exertional dyspnea. He had been submitted to amputation of the distal phalanx of the right ring finger and was prescribed ciprofloxacin and clindamycin a couple of days before the hospital admission. He was a smoker (50 pack-years) but denied alcoholism and illicit drugs usage. On admission, the physical examination revealed an emaciated and pale patient with blood pressure of 100/70 mmHg, rhythmic pulse of 80 beats per minute. He was afebrile, eupneic, and had room air oximetry of 93%, capillary glucose of 94 mg/dL, and a body mass index of 17 kg/m2. The pulmonary examination revealed bilateral diminished breath sounds. The cardiac examination was unremarkable and the abdomen presented signs of moderate ascites. On examination of his back, a grade-4 sacral bedsore was found. Multiple necrotic ulcers in the fingers, feet, pretibial, and around the left knee regions were evident (Figure 1). The laboratory work-up revealed: pancytopenia with hemoglobin of 4.3 g/dL (reference value [RV]; 12.3-15.3 g/dL), leukocytes of 2170/mm3 (RR: 4.4-11.3 × 103/mm3, and platelets of 15.000/mm3 (RV: 150-400 × 103/mm3); an abnormal prothrombin time with the International Normalized Ratio (INR) of 1.59 (RV:1), hypoalbuminemia, and elevated C-reactive protein. The electrolytes, renal function tests, hepatic enzymes, creatine phosphokinase and lactic dehydrogenase, thyroid hormones, iron profile, folic acid dosage were within normal limits. Vitamin B12 was 173 pg/mL (RV: 211-911 pg/mL). Urinalysis revealed proteinuria, leukocyturia, hematuria, but no casts, and the culture was negative, but it was sampled on antibiotics. Serology was negative for HIV, HCV, and syphilis. However, hepatitis B surface antigen and HBe were positive and so was anti-HBc and anti-HBe (consistent with chronic hepatitis B in replication phase). Rheumatoid factor, ANA, complement, cryoglobulin, and cold agglutinin were negative. The echocardiogram revealed inferior and lateral left ventricular hypokinesis and signs of pulmonary hypertension. The abdominal ultrasound showed chronic calculous cholecystitis, ascites, and bilateral pleural effusion. Two sets of blood culture were sampled and were negative.
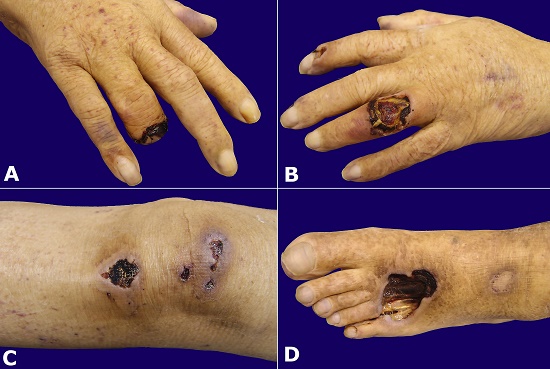
Figure 1
Gross examination of the distal right ring-finger after amputation (A) and necrotic skin ulcers; B – Left ring finger; C – Left knee; D – left foot.
Perilesional skin biopsy showed a mild perivascular lymphomononuclear inflammatory infiltrate with capillary fibrinoid necrosis, which was interpreted as small vessel vasculitis (Figure 2). The Grocott special stain was negative for fungi.
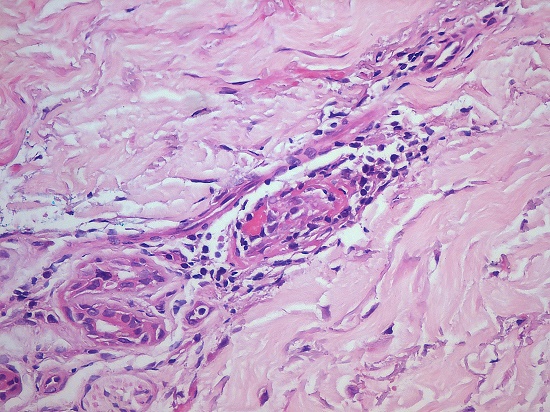
Figure 2
Photomicrograph of the skin biopsy showing mild lymphomononuclear pericapillary infiltrate with fibrinoid necrosis close to skin adnexa (H&E, 400X).
With the working diagnosis of small vessel vasculitis of unknown cause, malnutrition, and major depression, the patient was promptly treated with packed red blood cell transfusion, intravenous crystalloids, 60 mg/daily of prednisone (after oral ivermectin) parenteral vitamin B12 supplementation, and the antibiotic regimen was maintained for 10 days. The outcome was unfavorable; the patient worsened his mood disturbance by refusing to eat (but not because of dyspeptic complaints) and to talk to health professionals. After 15 days of hospitalization, there was a marked worsening of the respiratory symptoms with cough and oxygen desaturation. The antibiotic regimen was changed to vancomycin and meropenem, but the patient died soon after.
AUTOPSY FINDINGS
After receiving the required consent from the patient’s family member, an autopsy was performed. On external examination, the corpse showed signs of undernourishment with bedsores in the sacral regional, right calcaneus, left lower back and multiple perimalleolar lesions, bilaterally.
The right distal ring-finger phalanx had been surgically removed with no apparent signs of infection. The histological examination of the skin lesions showed the presence of chronic ulcerated dermatitis with a vascular pattern and areas of acute inflammatory process, necrosis, and adjacent phlebitis. Special stains for fungi were negative in the skin lesions samples at autopsy. These findings were compatible with a partially treated cutaneous vasculitis.
We also observed bilateral crystalline opacities and bone deformity in a dorsal spine with hyperextension stiffness. Histological examination of the lumbar joints showed a chronic inflammatory process, signs of old hemorrhage; fibrosis, and adjacent vertebral bone marrow with reactional lymphoplasmacytic infiltrate, which were consistent with the clinical diagnosis of ankylosing spondylitis.
There were large and bilateral pleural effusions, and the lungs presented areas of subpleural emphysema and airways filled with mucus. The histological sections taken from the lungs indicated diffuse alveolar damage and some organized foci of aspiration bronchopneumonia, with pseudohyphae and yeasts consistent with Candida spp. (Figure 3). The epicardial surface showed fibrin exudate, and the aorta and coronary arteries exhibited moderate atherosclerosis.
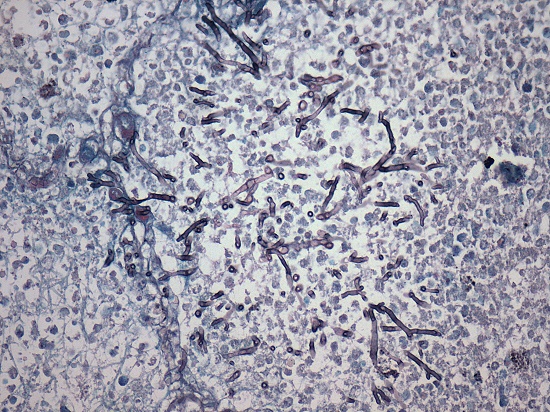
Figure 3
Photomicrograph of an area of aspiration pneumonia in the lung showing pseudohyphae of Candida spp. (Grocott, 400X).
The esophagus presented whitish and eroded mucosa with floppy and grayish pseudomembrane on the middle third where the esophageal wall was thickened (Figure 4).
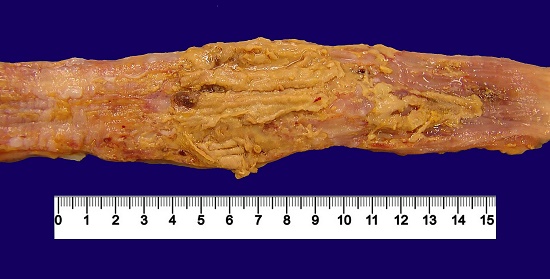
Figure 4
Gross view of the esophagus showing mucosa with loosely adherent grayish pseudomembranes.
The abdomen showed a small amount of peritoneal effusion with fibrin deposits on the parietal peritoneal surface of the large bowel (Figure 5). On histological examination, the esophagus showed full thickness necrosis, and diffuse inflammatory infiltrate rich in neutrophils (necrotizing esophagitis) (Figure 6). Bacterial colonies, pseudohyphae, and yeasts consistent with Candida spp were detected on special stains in the esophagus, with invasion extended to the adventitia (Figure 7). Cultures or molecular tests for specific Candida species were not available for autopsy samples.
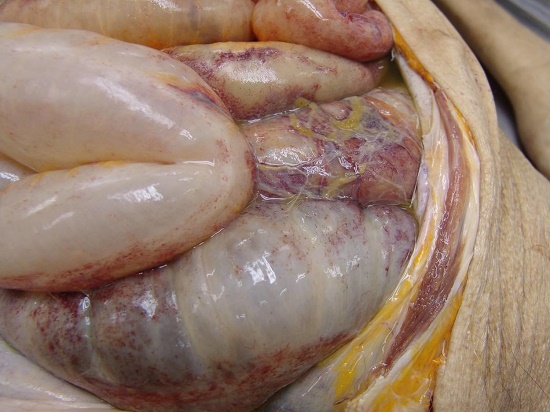
Figure 5
Gross aspect of abdominal cavity, showing peritoneal fibrinous exudate on the colonic surface.
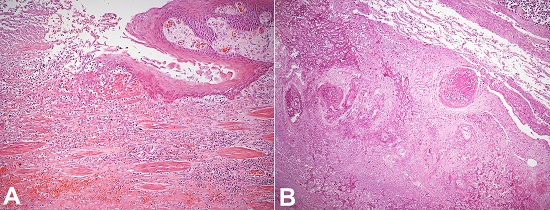
Figure 6
A – Photomicrograph of the esophagus showing an active ulcer with inflammatory exudate (H&E, 100X); B – Coagulative necrosis of esophageal submucosa and vessels (H&E, 100X).
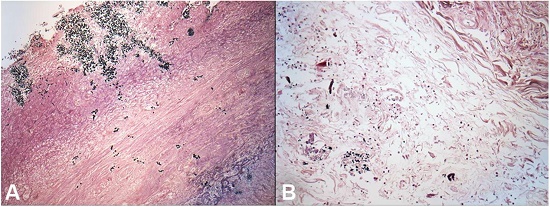
Figure 7
Photomicrograph of the esophagus showing invasive Candida spp. from mucosal ulcer and submucosa in A (Grocott; 100X) through adventitia; in B (yeasts in left bottom) (Grocott; 200X).
The sections of peritoneum showed acute exudative inflammation with bacterial colonies, pseudohyphae, and yeasts consistent with Candida spp. (Figure 8).
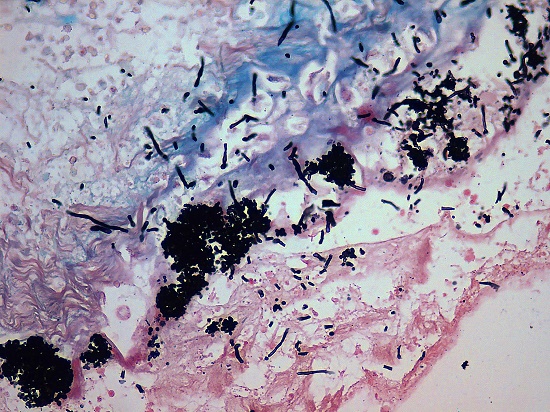
Figure 8
Photomicrograph of the peritoneum with exudative inflammation, pseudohyphae, and spores of Candida spp. (Grocott, 400x).
The liver and spleen were grossly congested. Upon histology, the liver showed reactive findings related to sepsis, with mild portal infiltration, mild cholangitis, and centrilobular necrosis, while the pancreas showed mild chronic and acute ischemic pancreatitis. An 8.0 mm urinary calculus was found within the right ureter.
According to the autopsy findings, we interpreted the immediate cause of death as being the fungal septicemia due to necrotizing Candida esophagitis (NCE) related to general malnourishment, long-term use of corticotherapy and broad-spectrum antibiotics. The necrotic skin lesions were interpreted as vasculitis of uncertain etiology, possibly related to ankylosing spondylitis.
DISCUSSION
Severe infections of the esophagus are uncommon in the general population and usually occur in immunocompromised patients. In this setting, the main etiological agents comprise viruses and fungi, such as cytomegalovirus (CMV), herpes virus (HSV) and Candida spp.1
Candida spp. may be present in 20% to 25% of normal esophagus mucosae and the colonization is the primary way of opportunist dissemination.2-5 In addition to the fungal pathogenic factors, the spread is related to the breakdown of the mucosal immune barrier and T-cell response, which are fundamental to the protective mechanism.3,6 Among the risk factors for NCE and other esophageal opportunistic infections are the immunodeficiency status (primary or secondary), functional or mechanical gastric obstruction, endocrine disorders, and malnutrition.4,7 In the present case, we considered that the corticotherapy, the recent surgical intervention followed by the broad-spectrum antibiotic usage and starvation, were the main reasons for the emergence of the fungal infection.
Most cases of esophageal candidiasis occur in the form of superficial esophagitis. Transmural infection, such as NCE is rare, and our case is one of the few reports found in the literature.3,5,8
Clinical features include dysphagia, odynophagia, chest pain, hypotension, and blackened vomiting.4,7 Complications of NCE, such as perforation, mediastinitis, pneumonia, and tracheoesophageal fistula, may be the initial presentation. However, the cases accompanied by perforation are related to severe neutropenia.5 More rarely, NCE presents initially with sepsis, which hampers an accurate diagnosis, since the esophagus is not usually investigated as a primary site of infection, and some cases can course asymptomatic, similar to our case. The fungal infection was not detected before death in this patient. Candida yeasts and pseudohyphae were demonstrated in the esophagus, peritoneum, and lungs (aspiration) at autopsy. No other systemic dissemination sites, including skin lesions, were detected. This insidious presentation in an overall very ill patient certainly has prevented a specific diagnosis and treatment.
The diagnosis of NCE is made on endoscopic examination. The differential diagnosis, especially on biopsy samples, with other necrotizing esophagitis such as the black esophagus syndrome, also can be made. A biopsy is usually not required for endoscopic diagnosis but either a biopsy or a brushing may be necessary in guiding therapy.3,4,9,10 When a biopsy is performed, the sections show necrotic debris, transmural coagulative necrosis without epithelium, and frequent submucosal involvement, in addition to yeasts with pseudohyphae.3,9,11,12 However, quite often, the diagnosis is performed at autopsy.3
Standard antifungal therapy of NCE is fluconazole and itraconazole with flucytosine or amphotericin B. The latter is usually prescribed in fluconazole-resistant Candida. Aggressive intervention, such as esophagectomy, should be considered in cases of extensive involvement and in signs of complication.5
Candida infection in this case was associated with immunosuppression caused by autoimmune diseases (arthropathy and vasculitis), corticoid therapy, and malnutrition.
Moreover, the association between vasculitis and ankylosing spondylitis is a rare underlying cause of death. Apparently, the literature supports this clinical picture in three distinct ways. First is the association between symptomatic aortic valve disease and aortitis large vessel vasculitis with spondyloarthropathies.13 Second is vasculitis related to treatment with biologic agents, particularly anti-tumor necrosis factor drugs. In these cases, cutaneous manifestation predominates in nearly 75% of the patients, especially in the form of purpura; however, ulcers, nodules, and visceral manifestations do occur. The treatment is based in the withdrawal of the drug with improvement in over 90% of cases.14 Finally, there are some case reports of different types of vasculitis in ankylosing spondylitis, including cutaneous, immunoglobulin-A-related retinal and cerebral vasculitis.13,15-19 These rare, sporadic, and localized forms of vasculitis seem to illustrate the third clinical picture in which ankylosing spondylitis could be the underlying cause of leukocytoclastic vasculitis.
In the present case, we did not find signs of large vessel vasculitis or thromboangiitis obliterans (Buerger disease) at autopsy. Also, there was no history of any drugs that could explain these findings.
CONCLUSION
We interpreted the case as a rare and severe manifestation of cutaneous leukocytoclastic vasculitis in a setting of ankylosing spondylitis, which was complicated with fatal sepsis due to undiagnosed NCE. Medical staff should keep in mind the investigation of esophageal candidiasis in very ill patients despite the lack of local specific clinical symptoms.
REFERENCES
Baehr PH, McDonald GB. Esophageal infections: risk factors, presentation, diagnosis, and treatment. Gastroenterology. 1994;106(2):509-32. http://dx.doi.org/10.1016/0016-5085(94)90613-0. PMid:7980741
Al-Shawwa B, D’Andrea L, Quintero D. Candida esophageal perforation and esophagopleural fistula: a case report. J Med Case Rep. 2008;2(1):209. http://dx.doi.org/10.1186/1752-1947-2-209. PMid:18559100
Gock M, Schäfer M, Perren A, Demartines N, Clavien PA. Fatal esophageal perforation caused by invasive candidiasis. Ann Thorac Surg. 2005;80(3):1120-2. http://dx.doi.org/10.1016/j.athoracsur.2004.02.147. PMid:16122508
Underwood JA, Williams JW, Keate RF. Clinical findings and risk factors for Candida esophagitis in outpatients. Dis Esophagus. 2003;16(2):66-9. http://dx.doi.org/10.1046/j.1442-2050.2003.00305.x. PMid:12823199
Gaissert HA, Breuer CK, Weissburg A, Mermel L. Surgical management of necrotizing Candida esophagitis. Ann Thorac Surg. 1999;67(1):231-3. http://dx.doi.org/10.1016/S0003-4975(98)01144-8. PMid:10086557
Gaissert HA, Roper CL, Patterson GA, Grillo HC. Infectious necrotizing esophagitis: outcome after medical and surgical intervention. Ann Thorac Surg. 2003;75(2):342-7. http://dx.doi.org/10.1016/S0003-4975(02)04343-6. PMid:12607636
Jones JM. Necrotizing Candida esophagitis: failure of symptoms and roentgenographic findings to reflect severity. JAMA. 1980;244(19):2190-1. http://dx.doi.org/10.1001/jama.1980.03310190042020. PMid:7420723
Jones JM, Glass NR, Belzer FO. Fatal Candida esophagitis in two diabetics after renal transplantation. Arch Surg. 1982;117(4):499-501. http://dx.doi.org/10.1001/archsurg.1982.01380280079016. PMid:7039554
Gurvits GE, Shapsis A, Lau N, Gualtieri N, Robilotti JG. Acute esophageal necrosis: a rare syndrome. J Gastroenterol. 2007;42(1):29-38. http://dx.doi.org/10.1007/s00535-006-1974-z. PMid:17322991
Gurvits GE. Black esophagus: acute esophageal necrosis syndrome. World J Gastroenterol. 2010;16(26):3219-25. http://dx.doi.org/10.3748/wjg.v16.i26.3219. PMid:20614476
Guarner J, Brandt ME. Histopathologic diagnosis of fungal infections in the 21st century. Clin Microbiol Rev. 2011;24(2):247-80. http://dx.doi.org/10.1128/CMR.00053-10. PMid:21482725
Kim YH, Choi SY. Black esophagus with concomitant candidiasis developed after diabetic ketoacidosis. World J Gastroenterol. 2007;13(42):5662-3. http://dx.doi.org/10.3748/wjg.v13.i42.5662. PMid:17948944
Ernst D, Baerlecken NT, Schmidt RE, Witte T. Large vessel vasculitis and spondyloarthritis: coincidence or associated diseases? Scand J Rheumatol. 2014;43(3):246-8. http://dx.doi.org/10.3109/03009742.2013.850737. PMid:24438209
Ramos-Casals M, Brito-Zerón P, Muñoz S, et al. Autoimmune diseases induced by TNF-targeted therapies: analysis of 233 cases. Medicine. 2007;86(4):242-51. http://dx.doi.org/10.1097/MD.0b013e3181441a68. PMid:17632266
Gillott TJ, Struthers GR. Cutaneous necrotizing vasculitis, erythema nodosum and ankylosing spondylitis. Rheumatology. 1999;38(4):377-8. http://dx.doi.org/10.1093/rheumatology/38.4.377. PMid:10378721
Kobak S, Yilmaz H, Karaarslan A, Yalcin M. Leukocytoclastic vasculitis in a patient with ankylosing spondylitis. Case Rep Rheumatol. 2014;2014:653837. http://dx.doi.org/10.1155/2014/653837. PMid:24653851
Machet L, Jan V, Ouakil H, Vaillant L, Estève E, Lorette G. Cutaneous leukocytoclastic vasculitis in a case of ankylosing spondylitis. Acta Derm Venereol. 1997;77(4):324. PMid:9228232
Taylan A, Yildiz Y, Sari I, Ozkok G. Vasculitis and long standing ankylosing spondylitis in a patient with familial Mediterranean fever. J Res Med Sci. 2014;19(10):1009-11. PMid:25538787
Beauvais C, Kaplan G, Mougenot B, Michel C, Marinho E. Cutaneous vasculitis and IgA glomerulonephritis in ankylosing spondylitis. Ann Rheum Dis. 1993;52(1):61-2. http://dx.doi.org/10.1136/ard.52.1.61. PMid:8427517
Notes
Author notes
Correspondence Mario Luiz Marques Piubelli Department of Pathology - Faculty of Medicine - Universidade de São Paulo (USP) Avenida Dr. Eneas Carvalho de Aguiar, 155 – Cerqueira Cesar – São Paulo/SP – Brazil CEP: 05403-000 Phone: +55 (11) 94141-6930 marquemario@gmail.com
Conflict of interest declaration