Article / Clinical Case Report
Cholesterol crystal embolism to the gastrointestinal tract: a catastrophic case
Cholesterol crystal embolism to the gastrointestinal tract: a catastrophic case
Autopsy and Case Reports, vol. 9, no. 2, e2018082, 2019
Hospital Universitário da Universidade de São Paulo
Received: 01 February 2019
Accepted: 11 March 2019
Abstract: Cholesterol crystal embolism is a rare and easily overlooked cause of colonic ischemia. The gastrointestinal tract is the third most common organ system affected by cholesterol emboli, second only to kidney and skin. Here we present a catastrophic case of gastrointestinal cholesterol crystal embolism leading to extensive post-operative bowel infarction and ultimately death. For a practicing pathologist, careful attention to the vessels of any ischemic bowel and recognition of the subtle but distinct angular imprint of cholesterol crystals facilitates prompt identification of the atheroemboli. In some cases, early identification may help mitigate further tissue damage. In more acute and severe cases, identification of the cholesterol crystal emboli may be important primarily for documentation of procedural complications.
Keywords: Cholesterol, Embolism, Gastrointestinal Tract, Ischemia.
CASE REPORT
A 62-year-old man underwent bilateral common femoral endarterectomy and aortobifemoral bypass for severe claudication secondary to extensive atherosclerosis. On postoperative day 1, he developed melena and hematochezia. Emergency colonoscopy demonstrated severe ischemia of the proximal colon and severe circumferential ischemic proctitis. Due to severe hypoxia, hypotension, and lactic acidosis, exploratory laparotomy was performed on postoperative day 2. Necrosis of the distal ileum and cecum was identified, and these segments were subsequently resected. Dusky areas in the remainder of the small bowel and colon were left intact, pending future operative evaluation. On the following day, the patient developed multi-organ failure, and a second-look exploratory laparotomy was performed. Diffuse small bowel and ascending colon necrosis was identified, and these segments were resected. Intraoperatively, the patient became acidotic and hyperkalemic, and upon transport to the intensive care unit, the patient expired.
PATHOLOGIC RESULTS
Gross examination of the small bowel and colon resections revealed dusky serosa and dark red to grey mucosa with attenuated folds (Figure 1).
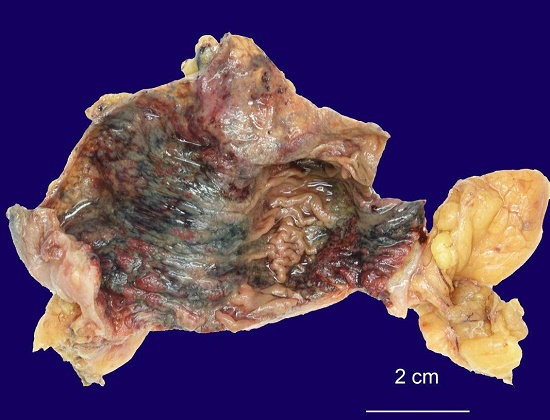
Figure 1
Ascending colon showing dusky mucosa with necrosis, hemorrhage, and attenuated folding (formalin-fixed specimen).
On the histologic sections, extensive mucosal and focal submucosal necrosis was present throughout the bowel (Figure 2). In the submucosa, numerous thin, needle-shaped clefts were noted in congested arterioles. Fibrin thrombi were also seen in some of the arterioles (Figure 2A. Additionally, focal areas of viable mucosa were noted on histologic sections. These areas were notable for absence of the aforementioned vascular changes.
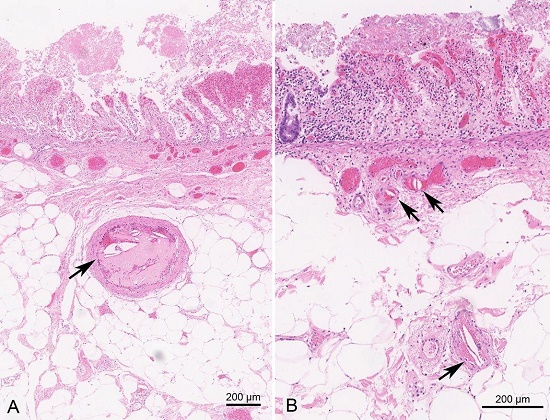
Figure 2
Photomicrograph of the ascending colon. A – Cholesterol crystal clefts (arrow) in an arteriole located in the submucosa of the cecum. Note the fibrin thrombus associated with the cholesterol crystal cleft. Mucosal necrosis is present; B – Cholesterol crystal clefts (arrows) in arterioles of varying sizes within the superficial and mid layers of the submucosa. (All images digitally scanned at 40X).
DISCUSSION
Needle-shaped clefts are the characteristic imprint of cholesterol crystals. The cholesterol itself is no longer present, as it dissolves during tissue processing (due to serial incubations in ethanol and xylene). When the needle-shaped clefts are present in small blood vessels in association with histologic features of tissue ischemia, the findings are diagnostic of cholesterol crystal embolism (CCE).1-3 Fibrin thrombi develop secondarily as a result of obstructed blood flow.1,3
CCE also referred to as atheroembolism or cholesterol embolization syndrome is a rare manifestation of the atherosclerotic disease.4 It occurs when an atherosclerotic plaque in the aorta or other major artery ruptures and releases cholesterol crystals and atheroma debris into the bloodstream. The crystals embolize to small and medium-sized arteries and arterioles, resulting in end-organ damage.1 The location of the disrupted atherosclerotic plaque determines the pattern of end-organ damage and thus its clinical manifestations. CCE can occur upon spontaneous rupture of an atherosclerotic plaque, after iatrogenic mechanical trauma (e.g., vascular surgery, angiography, or angioplasty), or as a side effect of medications targeting the coagulation system (e.g., anticoagulants, or thrombolytics).5
The incidence of CCE is estimated to be less than 0.5% based on studies of unselected autopsy populations.6 However, it has been reported to be 1.4% after coronary catheterization.7 The risk factors for cholesterol crystal formation are primarily those of atherosclerosis (e.g., smoking, hypercholesterolemia, hypertension, and obesity), while the risk factors for cholesterol crystal embolization include atherosclerosis, vascular manipulation, anticoagulation, and thrombolytic therapy.5,8
The gastrointestinal tract is the third most frequent organ system affected (13.4%), following kidney (31.5%) and skin (15.5%).6 Within the alimentary tract, the colon is the most common site of involvement (42.3%), followed by small intestine (33%), stomach (12.3%), rectum (9.2%), and esophagus (1.5%).9 CCE may also involve the pancreas, liver, and gallbladder.10,11 CCE involving the digestive tract often presents as abdominal pain, diarrhea, and gastrointestinal bleeding.5,8,12 Because the clinical presentation is not specific, the disease can masquerade as other conditions (e.g., infection, tumor, inflammatory bowel disease). CCE can present as either a chronic indolent disease that resolves over time or an acute catastrophic multi-organ disorder with poor prognosis.5 Although histologic evaluation is the gold standard for diagnosis, the co-occurrence of 3 clinical findings: (1) history of known risk factors for CCE (e.g., vascular surgery, anticoagulation), (2) acute onset renal failure with creatinine elevation of greater than 50% of baseline, and (3) signs of cutaneous vascular compromise (e.g., livedo reticularis, purples toes) or funduscopic evidence of retinal atheroemboli, has proved to be relatively specific for disseminated CCE.13 Thus, non-invasive procedures such as funduscopic examination can be useful. No specific treatment for CCE exists; however, early and aggressive supportive therapy (e.g., use of blood pressure lowering agents - in the context of cardiac failure, hemodialysis, nutritional support, discontinuation of inciting medications) can improve prognosis.13 Nonetheless, overall mortality rates are still as high as 80%.14
In the present case, the patient had multiple atherosclerotic plaques (identified by imaging) along the entire abdominal aorta, placing him at risk for multi-organ CCE. Indeed, besides the extensive gastrointestinal tract involvement, acute renal and liver failure emerged after aortobifemoral bypass. Although an autopsy was not performed, the widespread aortic atherosclerosis, diffuse bowel infarction, and acute renal and liver dysfunction point to multi-organ CCE as the ultimate cause of his demise. Nonetheless, since no post-surgical angiography studies were performed, we cannot entirely exclude the possibility that vasoconstriction of large arteries contributed to the vascular compromise, as has been described previously.15 It is also worth noting that the patient’s abdominal aorta clamp time (35 minutes) and blood loss (1.5 L) would be considered moderate16 and would not be expected to be the primary cause of the patient's massive vascular compromise.
From a histologic standpoint, the differential diagnosis of colonic ischemia includes infection (e.g., cytomegalovirus, E. coli O157:H7), vasculitis, mesenteric ischemia due to prolonged aortic clamping, mesenteric thrombosis, radiation-induced vasculopathy, mesenteric myointimal hyperplasia,17 and enterocolic lymphocytic phlebitis,18 none of which were observed in this case. Additionally, a thrombus overlying an atheromatous plaque can become loose and occlude downstream large caliber arteries (thromboembolism).3,4,19 Thromboembolism is distinguished from CCE in that it usually involves a single target organ and the emboli are predominantly composed of fibrin, whereas CCE is characterized by multiple cholesterol crystal emboli in small arterioles. Of note, some studies report the presence of CCE in small vessels mostly less than 200 µm in diameter,2,20 whereas in the current case the size of the affected arterioles ranged from 50 to 950 µm in diameter, similar to the findings of Flory.1
CONCLUSION
CCE is a type of vasculopathy that occurs when an atherosclerotic plaque ruptures and releases cholesterol crystals and atheroma debris into downstream arterioles. Although the digestive tract is a commonly affected site, CCE is a relatively uncommon cause of colonic ischemia overall and as such must be kept in mind in the differential diagnosis of ischemia, particularly in the setting of a recent vascular procedure and/or certain patient-specific risk factors (atherosclerosis, smoking, hypercholesterolemia, hypertension, obesity). Unfortunately, due to the relatively non-specific symptoms, a high degree of clinical suspicion is necessary for establishing a timely diagnosis. For the pathologist, attention to the character and content of the vessels is critical for identification. If not viewed carefully, cholesterol clefts may be overlooked as artefactual fractioning of vessel lumina rather than an indication of a potentially widespread and morbid pathologic process.
REFERENCES
Flory CM. Arterial occlusions produced by emboli from eroded aortic atheromatous plaques. Am J Pathol. 1945;21(3):549-65. PMid:19970827
Kassirer JP. Atheroembolic renal disease. N Engl J Med. 1969;280(15):812-8. http://dx.doi.org/10.1056/NEJM196904102801506. PMid:4887250
Eliot RS, Kanjuh VI, Edwards JE. Atheromatous embolism. Circulation. 1964;30(4):611-8. http://dx.doi.org/10.1161/01.CIR.30.4.611. PMid:14211824
Saric M, Kronzon I. Aortic atherosclerosis and embolic events. Curr Cardiol Rep. 2012;14(3):342-9. http://dx.doi.org/10.1007/s11886-012-0261-2. PMid:22437371
Ben-Horin S, Bardan E, Barshack I, Zaks N, Livneh A. Cholesterol crystal embolization to the digestive system: characterization of a common, yet overlooked presentation of atheroembolism. Am J Gastroenterol. 2003;98(7):1471-9. http://dx.doi.org/10.1111/j.1572-0241.2003.07532.x. PMid:12873565
Moolenaar W, Lamers CB. Cholesterol crystal embolization in the Netherlands. Arch Intern Med. 1996;156(6):653-7. http://dx.doi.org/10.1001/archinte.1996.00440060081009. PMid:8629877
Fukumoto Y, Tsutsui H, Tsuchihashi M, Masumoto A, Takeshita A. The incidence and risk factors of cholesterol embolization syndrome, a complication of cardiac catheterization: a prospective study. J Am Coll Cardiol. 2003;42(2):211-6. http://dx.doi.org/10.1016/S0735-1097(03)00579-5. PMid:12875753
Fries C, Roos M, Gaspert A, et al. Atheroembolic disease: a frequently missed diagnosis: results of a 12-year matched-pair autopsy study. Medicine. 2010;89(2):126-32. http://dx.doi.org/10.1097/MD.0b013e3181d5eb39. PMid:20517183
Moolenaar W, Lamers CB. Cholesterol crystal embolisation to the alimentary tract. Gut. 1996;38(2):196-200. http://dx.doi.org/10.1136/gut.38.2.196. PMid:8801196
Konstantinidis IT, Warshaw AL, Deshpande V, et al. Cholesterol crystal embolization presenting as either solid or cystic pancreatic lesion. J Surg Oncol. 2010;102(6):706-8. http://dx.doi.org/10.1002/jso.21521. PMid:20976733
Moolenaar W, Lamers CB. Cholesterol crystal embolization to liver, gallbladder, and pancreas. Dig Dis Sci. 1996;41(9):1819-22. http://dx.doi.org/10.1007/BF02088752. PMid:8794801
Moolenaar W, Lamers CB. Cholesterol crystal embolization and the digestive system. Scand J Gastroenterol Suppl. 1991;188(Suppl 188):69-72. http://dx.doi.org/10.3109/00365529109111232. PMid:1775943
Belenfant X, Meyrier A, Jacquot C. Supportive treatment improves survival in multivisceral cholesterol crystal embolism. Am J Kidney Dis. 1999;33(5):840-50. http://dx.doi.org/10.1016/S0272-6386(99)70415-4. PMid:10213638
Ghanem F, Vodnala D, K Kalavakunta J, et al. Cholesterol crystal embolization following plaque rupture: a systemic disease with unusual features. J Biomed Res. 2017;31(2):82-94. PMid:28808190
Imanaka K, Kyo S, Ban S. Possible close relationship between non-occlusive mesenteric ischemia and cholesterol crystal embolism after cardiovascular surgery. Eur J Cardiothorac Surg. 2002;22(6):1032-4. http://dx.doi.org/10.1016/S1010-7940(02)00590-0. PMid:12467838
Bruls S, Quaniers J, Tromme P, Lavigne JP, Van Damme H, Defraigne JO. Comparison of laparoscopic and open aortobifemoral bypass in the treatment of aortoiliac disease: results of a contemporary series (2003-2009). Acta Chir Belg. 2012;112(1):51-8. http://dx.doi.org/10.1080/00015458.2012.11680795. PMid:22442910
Yantiss RK, Cui I, Panarelli NC, Jessurun J. Idiopathic myointimal hyperplasia of mesenteric veins: an uncommon cause of ischemic colitis with distinct mucosal features. Am J Surg Pathol. 2017;41(12):1657-65. http://dx.doi.org/10.1097/PAS.0000000000000905. PMid:28817406
Louie CY, DiMaio MA, Charville GW, Berry GJ, Longacre TA. Gastrointestinal Tract Vasculopathy: Clinicopathology and Description of a Possible “New Entity” With Protean Features. Am J Surg Pathol. 2018;42(7):866-76. http://dx.doi.org/10.1097/PAS.0000000000001060. PMid:29624512
Tunick PA, Kronzon I. Atheromas of the thoracic aorta: clinical and therapeutic update. J Am Coll Cardiol. 2000;35(3):545-54. http://dx.doi.org/10.1016/S0735-1097(99)00604-X. PMid:10716454
Quinones A, Saric M. The cholesterol emboli syndrome in atherosclerosis. Curr Atheroscler Rep. 2013;15(4):315. http://dx.doi.org/10.1007/s11883-013-0315-y. PMid:23423524
Notes
Author notes
Correspondence Karen E Matsukuma Department of Pathology and Laboratory Medicine - Davis Medical Center- University of California 4400 V Street – Sacramento/CA – USA PO Box: 9581795817 Phone: +1 (91) 6734-2529 kmatsukuma@ucdavis.edu
Conflict of interest declaration
