Original Research Papers
Cropping duration and non-rhizomorphic mycelial phenotype of Pleurotus djamor woody1 co-segregate in the hybrid progenies
Cropping duration and non-rhizomorphic mycelial phenotype of Pleurotus djamor woody1 co-segregate in the hybrid progenies
Journal of Horticultural Sciences, vol. 17, núm. 1, pp. 220-226, 2022
Society for Promotion of Horticulture

Esta obra está bajo una Licencia Creative Commons Atribución-NoComercial-CompartirIgual 4.0 Internacional.
Recepción: 17 Septiembre 2021
Aprobación: 15 Marzo 2022
Abstract: Crop duration of the cultivated Pleurotus spp. is 45 to 50 days. P. djamor isolate woody-1 was collected as natural selection and was found to be short cropping duration variety with total cropping duration of 30 days but it is less palatable. It produced very thin, loose and non-rhizomorphic mycelia appearing light white color. Whereas, other commercial Pleurotus varieties such as P. florida and P. djamor MDU1 are long crop duration varieties and palatable producing thick, compact and rhizomorphic mycelia with bright white color. Co-segregation of non-rhizomorphic mycelial phenotype and short cropping duration trait of P. djamor woody- 1 in hybrid progenies was evaluated. Hybrid strains viz., H2W12 and H2W14 have thin, loose and non-rhizomorphic mycelium and they produced primordia in 9-10 days after spawning with total cropping duration of 29-32 days. Whereas, hybrid strain namely Pf1W2 has thick, compact and rhizomorphic mycelial phenotype and it produced primordia in 20 days after spawning with the total cropping duration of 47 days. This study indicated that genes governing short cropping duration and non-rhizomorphic mycelial pattern were tightly linked and co-segregated in the progenies. Thus, non-rhizomorphic mycelial phenotype of P. djamor woody1 can be used as a phenotypic marker for selection of hybrid cultivar having short cropping duration with other desired agronomic traits in future breeding strategy.
Keywords: Basidiocarp, hybridization, mycelium and Pleurotus.
INTRODUCTION
Mushrooms have been identified as an excellent food source to alleviate malnutrition in developing countries. Oyster mushroom has proteins, carbohydrates, vitamins, calcium, and iron (Hilal et al., 2012). It is a good dietary supplements which can lower cholesterol (Khatun et al., 2007). Globally, Pleurotus is the second largest cultivated mushroom after Shiitake (Royse et al., 2017). Pleurotus species are popular and widely cultivated throughout the world especially in Asia, America and Europe because of their simple, low-cost production technology and high biological efficiency. Pleurotus species are efficient lignin degraders which can grow on a wide variety of agricultural wastes and acclimatize a wide range of temperatures. The productivity and quality of cultivated edible mushrooms mainly depend on the genetic makeup of the strain (Kaur and Sodhi 2012). The improvement of Pleurotus mushroom production primarily utilizes natural selection. The productivity of the mushroom strain can be improved to some extent by manipulating the environmental and physiological conditions during cultivation. However, genetic manipulation of the ruling mushroom variety can enhance the productivity and quality of the mushroom. Genetic manipulation of mushroom can be done by hybridization, protoplast fusion and genetic engineering for strain improvement. Selection and mating of genetically diverse parents is an important approach to exploit heterosis through hybridization. The objectives of mushroom breeding are to obtain Pleurotus strain with desirable agronomic traits such as high yield, wider substrate utilisation, spore lessness, wide temperature tolerance, good palatability, good texture of the fruiting body and resistance to pest and diseases.
Recently, P. djamor isolate woody1 was collected as natural selection process and it has short crop duration (30 days) and high biological efficiency. However, it is leathery in sensory while eating, contains less plectenchymatous tissue in the pileus. Thus, it is less palatable (Praveen et al., 2017). But several ruling Pleurotus spp. including P. florida and P. djamor MDU1 are long crop duration varieties with good palatability. In order to transfer the short cropping duration trait into the commercially ruling mushroom cultivar, P. djamor woody1 need to be crossed with any of the ruling oyster mushroom and the suitable hybrid possessing short cropping duration along with desired agronomic traits such as good palatability and high yielding potential has to be selected. There are some sequential steps followed for carrying out successful breeding process (hybridization) between two Pleurotus spp. starting with collection and culturing of basidiospores; crossing monokaryotic mycelia and evaluation of successful crosses and finally analysis of the hybrid strain for desired agronomic traits in comparison with parental strains (Barh et al., 2019). Pleurotus has tetrapolar / bifactorial mating system and requires two compatible mating type for dikaryotic mycelial formation and basidiocarp initiation and need to carry out several crosses to get the dikaryons for obtaining hybrid with desired agronomic traits (Raper and Raper, 1966; Casselton and Olesnicky, 1998).
Several crosses need to be made to find a hybrid having short crop duration trait with desired agronomic trait or a hybrid with several desirable traits. Thus, it would be wise to have additional phenotypic marker that could co-segregate with any one of the desirable trait for screening the hybrid having other desired traits. In our previous studies conducted during 2018 and 2020 on breeding between P. djamor woody1 and P. djamor MDU1 or P. florida resulted in several hybrids having both short cropping duration and long cropping duration (Reihana et al., 2018; Samundeeswari, 2020). We speculated that short cropping duration and non-rhizomorphic mycelial phenotypes could co-segregate in the hybrid progenies. Keeping these points in mind, cropping duration and mycelial phenotypic characters of three selected hybrid progenies (H2W12, H2W14 and Pf1W2) of P. djamor woody1 were analysed upon crossing with P. florida. In this study it was found that non-rhizomorphic phenotypic character co-segregate with the short cropping duration in hybrid progenies.
MATERIAL AND METHODS
Pleurotus culture and growth medium
Dikaryotic mycelia isolated from the basidiocarps of different Pleurotus spp. viz., P. djamorwoody1, P. florida, P. djamor MDU1 and hybrids strains viz., H2W12, H2W14 and Pf1W2 (obtained upon crossing between P. djamor woody1 and P. florida) were used in this study. Mycelial cultures were cultured on PDA medium. Spawn production was carried out on sorghum/paddy grains. Mushroom cultivation for analysing the primordial formation and cropping duration was carried out on paddy straw.
Somatic hybridization of different Pleurotus spp.
Collection of basidiospores was carried out by placing healthy pileus in sterilized Petri plate in such a way that gills were facing down the bottom plate for an hour to allow shedding of basidiospore from the pileus. Then, the basidiospores were collected by adding 10 ml of sterilized water and counted using haemocytometer. The basidiospore stock suspension was serially diluted to the concentration of 300 spores /ml and about 30 to 100 basidiospores were spread plated using sterilized glass L rod and incubated at 28! for 4 to 6 days or until individual small white mycelial colonies appear with the diameter of 3-5 mm. Markedly fast growing monokaryotic colonies with typical radial growth were identified and sub cultured on fresh PDA plate in a grid form at equi-distance.
Somatic hybridization was carried out between P. florida and P. djamor isolate woody 1. Dual culture technique was employed for pairing monokaryons of two parental strains at possible combinations. Small discs of monokaryons from two parental strains were cut and inoculated at two centimeters apart from each other on PDA medium at the center of the plate and incubated at 28OC The plates were incubated until two monokaryotic mycelia grow towards each other and the hyphae of two monokaryons were interwoven or fused with each other. A compatible mating consisted of formation of fluffy and vigorous mycelia (with thick and bright white color) at the confrontation zone/merger region of anastomosis/ junction point of two monokaryotic mycelia. From this junction point, the fluffy putative dikaryotic mycelium was taken and sub-cultured onto the new PDA plate and incubated for five days. Dikaryotic mycelia (crossed/hybrid/ paired mycelia) were further confirmed by the presence of clamp connection under light microscope.
Assessment of mycelial growth of different Pleurotus spp.
To assess the radial growth of mycelium and mycelial growth pattern, the dikaryotic mycelia of P. djamor woody1, P. florida, P. djamor MDU1 and hybrid strains viz., H2W12, H2W14 and Pf1W2 were inoculated onto the PDA medium. The cultures were incubated at 28°C. Three replications were maintained for each culture. The radial growth of the mycelium was recorded when anyone of the isolates completely covered the Petri plate. Mycelial growth patterns such as fluffiness, color and rhizomorphic pattern were noted.
Spawn preparation
Paddy straw was used as a substrate for the bed preparation. The substrate was cut into 5 cm long bits, soaked in cold water for 4 hours and pasteurized in hot water for 30 min at 80°C. The transparent polythene bags (30 x 60 cm length and 80-gauge thickness) were used for the cultivation of oyster mushroom. Initially, hands were thoroughly washed with alcohol. The bottom end of the bag was tied with a thread and the bag was turned inside out. Then, the dried straw was mixed thoroughly to get a uniform moisture level in all areas. The well-grown bed spawn was taken out, squeezed thoroughly and divided into two halves. (Two beds are prepared from the single spawn bag). Bits of chopped straw (5 cm long) were placed at the bottom of polythene bag to make a layer (10 cm height) on which 40 g of spawn was sprinkled. Second layer of straw to a height of 10 cm was placed and 40 g of spawn was sprinkled on top of the second layer. In the same way, five layers of straw and four layers of spawn were kept in the polythene bag and finally the bag was tied at the top. Six ventilation holes of one-cm diameter were made at random in the polythene bag. Then, these beds were kept in spawn running room where the temperature was maintained at 28oC. The fully spawn run beds were taken to the cropping room in which the temperature was maintained at 25±2°C and RH- > 80% for initiation of basidiocarp (Krishnamoorthy et al., 2005).
The spawn preparation was carried out as described by Krishnamoorthy et al. (2005). The paddy or sorghum grains were washed in water thoroughly to remove chaffy and damaged grains. The grains were cooked in vessel for 30 minutes just to soften them. Then, the excess water from the cooked grains was drained off and grains were spread evenly over a hessian cloth on a platform to remove the excess water. At 50% moisture level, calcium carbonate (CaCO3) was applied on the grains (dried grains) @ 40 g /kg of grains. Then, the grains were filled in polythene bags up to 3/4th height (approximately 300-330 g / bag), PVC ring was inserted, edges were folded down and the mouth of the bag was plugged tightly with non-absorbent cotton. After plugging with cotton plug, the bag was covered with a piece of paper and tied tightly around the neck with a jute thread or a rubber band. The bags were arranged inside an autoclave and sterilized at 20 lbs for 2 hours. Then, the bags were taken after cooling and kept inside the laminar air flow chamber for inoculation.
The mycelial culture (10 mm diameter disc) of Pleurotus spp. was cut and transferred to a bag. The inoculated bags were incubated in a clean room under room temperature (28±2°C). The spawn running period was recorded.
Bed preparation
Paddy straw was used as a substrate for the bed preparation. The substrate was cut into 5 cm long bits, soaked in cold water for 4 hours and pasteurized in hot water for 30 min at 80°C. The transparent polythene bags (30 x 60 cm length and 80-gauge thickness) were used for the cultivation of oyster mushroom. Initially, hands were thoroughly washed with alcohol. The bottom end of the bag was tied with a thread and the bag was turned inside out. Then, the dried straw was mixed thoroughly to get a uniform moisture level in all areas. The well-grown bed spawn was taken out, squeezed thoroughly and divided into two halves. (Two beds are prepared from the single spawn bag). Bits of chopped straw (5 cm long) were placed at the bottom of polythene bag to make a layer (10 cm height) on which 40 g of spawn was sprinkled. Second layer of straw to a height of 10 cm was placed and 40 g of spawn was sprinkled on top of the second layer. In the same way, five layers of straw and four layers of spawn were kept in the polythene bag and finally the bag was tied at the top. Six ventilation holes of one-cm diameter were made at random in the polythene bag. Then, these beds were kept in spawn running room where the temperature was maintained at 28oC. The fully spawn run beds were taken to the cropping room in which the temperature was maintained at 25±2°C and RH- > 80% for initiation of basidiocarp (Krishnamoorthy et al., 2005).
Primordia formation and cropping duration assessment
The days required for the primordia formation were recorded after spawning and the days required for the harvest of the first, second and third flushes and total cropping duration of each variety were recorded. The yield and biological efficiency were recorded.
RESULTS AND DISCUSSION
Assessment of mycelial growth pattern of different
Pleurotus spp.
To assess which Pleurotus spp. grow actively on the culture media, six isolates of Pleurotus spp. viz., P. djamor woody1, P. florida, P. djamor MDU1 and hybrid strains viz., H2W12, H2W14 and Pf1W2 were cultured on PDA medium. Among the various Pleurotus spp. tested, P. djamor isolate woody1 grew to the maximum level of 88.67 mm followed by the hybrid strains Pf1W2 (86.33), H2W12 (85.67 mm), H2W14 (85.33 mm) and P. djamor isolate MDU1 (77.67 mm). Whereas, the minimum mycelial growth was recorded by P. florida on PDA medium (75 mm) (Fig. 1 and Table 1).
| Pleurotusstrains | Radial mycelial growth (mm)* | Mycelial growth pattern |
| P. djamor woody1 | 88.67a | Thin, loose and non-rhizomorphic growth; dull white in color |
| Hybrid - H2W12 | 85.67c | Thin, loose and non-rhizomorphic growth; dull white in color |
| Hybrid - H2W14 | 85.33c | Thin, loose and non-rhizomorphic growth; dull white in color |
| Hybrid Pf1W2 | 86.33b | Thick, compact and rhizomorphic growth; bright white in color |
| P. florida | 75.00d | Thick, compact and rhizomorphic growth; bright white in color |
| P. djamor MDU1 | 77.67e | Thick, compact and rhizomorphic growth; bright white in color |
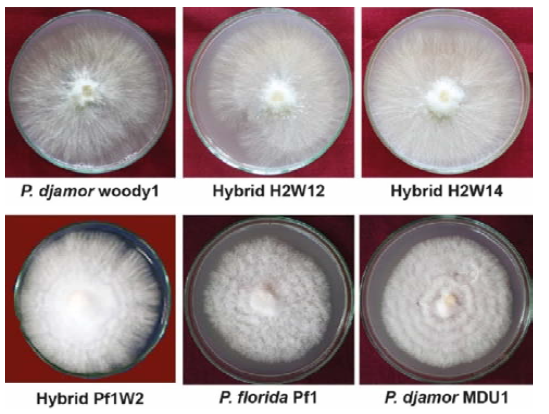
Fig. 1
Colony characters of different Pleurotus spp.
Mycelia of P. djamor woody1 (one of the parental strains used for hybridization) appeared as thin, loose and non-rhizomorphic filament and light white in color. Similarly, the hybrid strains such as H2W12 and H2W14 also produced thin, loose and non- rhizomorphic filament and light white in color. Whereas, hybrid strain namely Pf1W2 produced thick, compact and rhizomorphic mycelium and appeared bright white in color as that of other parental strain P. florida (Fig. 1 and Table 1). Mostly, the mycelial growth phenotype of Pleurotus appears as rhizomorphic like radial growth with thick and white in color. But, mycelial growth characters of P. djamor woody1 and some of its hybrid progeny appeared as loose, thin and non-rhizomorphic type. This is the important phenotypic and distinguishing character for the identification of this isolate during culturing time. Similar type of varied mycelial phenotypic characters in different Pleurotus spp. was noticed in different Pleurotus spp. Mycelia of P. sajor-caju and P. djamor woody1 and hybrid strains such as H2W12 and H2W14 appeared as thin, loose and non- rhizomorphic filament and light white in color on spawn substrates (sorghum/ paddy grains) as observed on PDA medium. Whereas, hybrid strain namely Pf1W2 and other parental strain namely. florida produced thick, compact and rhizomorphic mycelium and appeared bright white in color on spawn substrates as grown on PDA medium (Table 2). In other studies, it was reported that P. eous covered the spawn within 7 to 20 days on different grains used as spawn substrate (Sahu et al., 2014). Blue oyster mushroom took spawn 18.5 days for the spawn production when paddy grain was used as a substrate (Sumi and Geetha, 2017).
platypus were compact. Whereas, mycelia of P. citrinopileatus were highly fluffy. Similarly, mycelial pattern in P. fossulatus, P. flabellatus, P. sapidus andP. ostreatus was slightly fluffy (Mishra et al., 2015).
Spawn running period and mycelial pattern
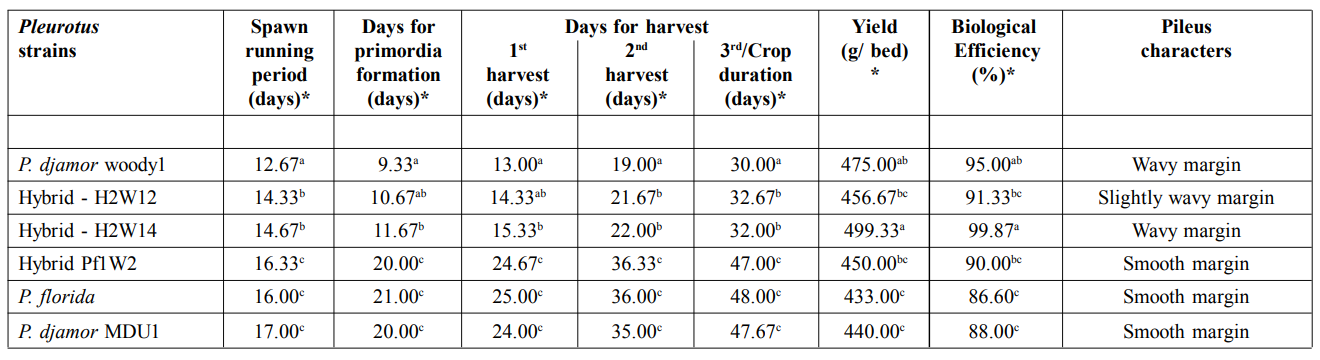
Days required for spawn development was analysed for different Pleurotus cultivars such as viz., P. djamor woody1, P. florida, P. djamor MDU1 and hybrid strains viz., H2W12, H2W14 and Pf1W2. Days required for spawn development for P. djamor woody1 and hybrid strains viz., H2W12 and H2W14 were ranged from12 to 15 days and that for hybrid strain namely Pf1W2 and P. florida were 16 to 17 days. Mycelial growth pattern of P.
Days required for spawn development was analysed for different Pleurotus cultivars such as viz., P. djamor woody1, P. florida, P. djamor MDU1 and hybrid strains viz., H2W12, H2W14 and Pf1W2. Days required for spawn development for P. djamor woody1 and hybrid strains viz., H2W12 and H2W14 were ranged from12 to 15 days and that for hybrid strain namely Pf1W2 and P. florida were 16 to 17 days. Mycelial growth pattern of P.
Mycelia of P. djamor woody1 (one of the parental strains used for hybridization) appeared as thin, loose and non-rhizomorphic filament and light white in color. Similarly, the hybrid strains such as H2W12 and H2W14 also produced thin, loose and non- rhizomorphic filament and light white in color. Whereas, hybrid strain namely Pf1W2 produced thick, compact and rhizomorphic mycelium and appeared bright white in color as that of other parental strain P. florida (Fig. 1 and Table 1). Mostly, the mycelial growth phenotype of Pleurotus appears as rhizomorphic like radial growth with thick and white in color. But, mycelial growth characters of P. djamor woody1 and some of its hybrid progeny appeared as loose, thin and non-rhizomorphic type. This is the important phenotypic and distinguishing character for the identification of this isolate during culturing time. Similar type of varied mycelial phenotypic characters in different Pleurotus spp. was noticed in different Pleurotus spp. Mycelia of P. sajor-caju and P. djamor woody1 and hybrid strains such as H2W12 and H2W14 appeared as thin, loose and non- rhizomorphic filament and light white in color on spawn substrates (sorghum/ paddy grains) as observed on PDA medium. Whereas, hybrid strain namely Pf1W2 and other parental strain namely. florida produced thick, compact and rhizomorphic mycelium and appeared bright white in color on spawn substrates as grown on PDA medium (Table 2). In other studies, it was reported that P. eous covered the spawn within 7 to 20 days on different grains used as spawn substrate (Sahu et al., 2014). Blue oyster mushroom took spawn 18.5 days for the spawn production when paddy grain was used as a substrate (Sumi and Geetha, 2017).
Days required for primordia formation
Days required for primordia formation was analysed for different Pleurotus cultivars such as P. djamor woody1, P. florida, P. djamor MDU1 and hybrid strains viz., H2W12, H2W14 and Pf1W2. Days required for primordia formation for P. djamor woody1 and that for hybrid strains viz., H2W12 and H2W14 were ranged between 9 to 12 days and that for hybrid strain Pf1W2, parental strain P. florida were 20 to 21 days. In general, primordia (pin head formation) formation of Pleurotus spp. occurs at 20 days after spawning (Table 2). Ahmed (1998) reported that pinhead formation of oyster mushroom occurred between 23 and 27 days from spawning in different substrates. Fan et al. (2000) observed that pinhead formation took place between 20-23 days. Patra and Pani (1995) also recorded 20-24 days taken for the pinhead formation on paddy straw substrate.
Agronimic traits for the different Pleurotus supp.
* *Mean of three replicationsIn the column, mean values followed by a common letter are not significantly different (pd”0.05, DMRT analysis).
Days required for basidiocarp production and total crop duration
Days required for the first flush basidiocarp production for P. djamor woody1 and hybrid strains viz., H2W12 and H2W14 were between 13 to 15 days and that for hybrid strain Pf1W2 and parental strain P. florida was 24 to 25 days after spawning. Similarly, days required for the second flush basidiocarp production for P. djamor woody1 and hybrid strains viz., H2W12 and H2W14 ranged between 19 to 23 days and that for hybrid strain Pf1W2, parental strains viz., P. florida and P. djamor MDU1 was 35 to 36 days. Total cropping duration for the P. djamor woody 1 and hybrid strains viz., H2W12 and H2W14 ranged between 30 to 33 days and that for P. florida and P. djamor MDU1 was 47 to 48 days. Margin and outer surface of basidiocarps of P. djamor woody1 and hybrid H2W14 appeared wavy and the hybrid H2W12 appeared slightly wavy. Whereas, margin and outer surface of basidiocarps of hybrid Pf1W2, P. florida and P. djamor MDU1 appeared smooth. The hybrid H2W14 gave the highest yield of 499.33 g with biological efficiency of 99.87 % followed b P. djamor isolate woody 1 (475.00 g and 95 %), hybrids H2W12 (456.67 g and 91.33 %), Pf1W2 (450.00g and 90.00 %) and P. djamor var MDU 1 (440.0 g and 88.00 %) and P. florida (433.00 g and 86.60 %). (Fig. 2; Table 2). Baral et al. (2017) developed an intraspecific hybrid of P. flabellatus showing better nutritional quality, earliness in production and higher yield compared to their parental strains. Interspecific hybrids viz., P1xC9 and P 3xC8, obtained by crossing between P. citrinopileatus and P. pulmonarius , showed desirable traits such as higher productivity and biological efficiency and less offensive aroma compared to their parental strains (Rosnina et al., 2016).
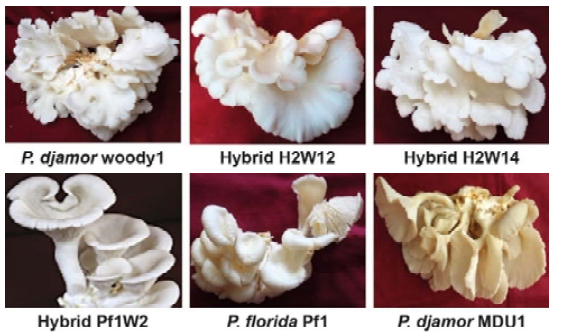
Fig. 2
Basidiocarp phenotypic characters of different Pleurotus spp
Thus, this study clearly showed that hybrid strains viz., H2W12 and H2W14 are short crop duration varieties with non-rhizomorphic mycelial type. Whereas, hybrid strain Pf1W2 is long cropping duration variety with rhizomorphic mycelial type. The present study clearly showed that short crop duration phenotype and non-rhizomorphic phenotype co-segregate together in the hybrid strains. Thus, from this study, it was concluded that non-rhizomorphic mycelium character can be used as a phenotypic marker to screen and select the short duration hybrid strains with additional desirable agronomic traits in the breeding program. Pleurotus breeding program involves five steps such as 1. collection of basidiospores 2. culturing of individual basidiospore to form monokaryotic mycelium. 3. crossing/ mating between monokaryotic mycelia of two Pleurotus spp. 4. evaluation of successful cross/dikaryon by checking clamp connection and 5. analysis of the hybrid strain for desired agronomic traits by mushroom cultivation (Petersen and Ridley, 1996). This study showed that only putative dikaryotic crosses/ hybrids having non-rhizomorphic phenotype at the fourth step of breeding program could be further evaluated in the fifth step for screeding/analysis of the hybrid progenies having short crop duration with other desired agronomic traits. Thus, co- segregating phenotypic marker saves the time and speed up the screening process of development of hybrid strain having short cropping duration with desired agronomic traits such as good palatability and high yielding potential. Hence, having co- segregating phenotypic marker (non-rhizomorphic phenotype) with short cropping duration traits of P. djamor woody1 would facilitate in speeding up breeding program with other commercially cultivated ruling Pleurotus cultivar.
Acknowledgments
The authors are thankful to Tamil Nadu Agriculture University for giving facility and technical help rendered by R. Reihana is highly appreciated.
REFERENCES
Ahmad, I., Fuad, I. and Khan, Z. K. 2015. Mycelia Growth of Pink Oyster (Pleurotus djamor) Mushroom in Different Culture Media & Environmental Factors. Agriculture and Food Sciences Research, .(1): 6-11.
Barh, A., Sharma, V.P., Annepu, S.K., Kamal, S., Sharma, S. and P Bhatt. 2019. Genetic improvement in Pleurotus (oyster mushroom): a review. 3 Biotech, . (9):322.
Baral, D., Roy, A. and Thapa, S. 2018. Strain improvement in oyster mushroom (Pleurotus sp.) through hybridization. The Pharma Innovation Journa,l, .(4): 286-289
Casselton, L. A. and Olesnicky, N. S. 1998. Molecular genetics of mating recognition in basidiomycete fungi. Microbiology and molecular biology reviews, 62(1): 55-70.
Fan, L., Pandey, A., Mohan, R. and Soccol, C. 2000. Use of various coffee industry residues for the cultivation of Pleurotus ostreatus in solid state fermentation. Engineering in Life Sciences, 20(1), 41-52.
Hilal, A., Dunder, A. and Yildiz, A. 2012. Effect of using different lignocellulosic wastes for cultivation of Pleurotus ostreatus (Jacq.) P. Kmm. on mushroom yield, chemical composition and nutritional value. Afr. J. Biotechnol., .(4): 662-666.
Kaur, J, and Sodhi, H. S. 2012. Molecular characterization of mutant strains of Calocybe indica using RAPD-PCR. CME .:4.
Khatun, K., Mahtab, H., Khanam, P., Sayeed, M. and Khan, K. 2007. Oyster mushroom reduced blood glucose and cholesterol in diabetic subjects. Mymensingh medical journal: MMJ, 16(1): 94-99.
Krishnamoorthy, A. S., Marimuthu, T. and Nakkeeran, S. 2005. Mushroom biotechnology. Department of Plant Pathology, Tamil Nadu Agriculture University, Coimbatore, p.82.
Patra, A. and Pani, B. 1995. Yield response of different species of oyster mushroom to paddy straw. Curr Agric Res, .(Suppl): 11-14.
Petersen, R.H. and Ridley, G.S. 1996. A New Zealand Pleurotus with multiple-species sexual compatibility. Mycologia, 88 (2):198-207.
Praveen, T., Reihana, R., Parthiban, V.K. and Ramamoorthy, V. 2018. “Molecular characterization and phenotypic study of new Pleurotus djamor isolate KKM.” Int. J. Curr. Microbiol. Appl. Sci. . (8):3574-3582.
Raper, C. A. and Raper, J. R. 1966. Mutations modifying sexual morphogenesis in Schizophyllum. Genetics, 54(5): 1151-1168.
Reihana, R., Praveen, T., Parthiban, V.K. and Ramamoorthy, V. 2018. Studies on yield attributes of new Pleurotus djamor isolate KKM 1. Mushroom Res. 27 (2): 145-150,2018.
Rosnina, A.G., Tan, Y.S., Abdullah, N. and Vikineswary, S. 2016. Morphological andmolecular characterization of yellow oyster mushroom, Pleurotus citrinopileatus, hybrids obtained by interspecies mating. World J. Microbiol. Biotechnol. 32 (2):18.
Royse, D.J., Baars, J. and Tan, Q. 2017. “Current overview of mushroom production in the world.” In: Edible and medicinal mushrooms: technology and applications (Eds.) Diego, C.D., Pardo-Giménez, A., Willey publications, p.:5-13.
Sahu, S.K., Singh, D.P., Patel, R., Awadhiya, G.K. and Vishwavidhyalaya, I.G.K. 2014. Screening of suitable grains substrates for spawn development, growth and yield of Pleurotus eous. American International Journal of Research in Formal, Applied and Natural Sciences . (1):86-89.
Samundeeswari, S. 2020. Studies on development of elite Pleurotus spp. for cultivation. M. Sc thesis. Tamil Nadu Agricultural University, Coimbatore.
Sumi, I. and D Geetha. 2017. Comparative performance of common oyster and blue oyster mushroom. Trends in Biosciences 10 (39):8149-8158.