CULTIVATION OF EDIBLE ECTOMYCORRHIZAL MUSHROOMS IN JAPAN
CULTIVO DE SETAS ECTOMICORRIZAS COMESTIBLES EN JAPÓN
CULTIVATION OF EDIBLE ECTOMYCORRHIZAL MUSHROOMS IN JAPAN
Revista Fitotecnia Mexicana, vol. 40, no. 4, pp. 379-389, 2017
Sociedad Mexicana de Fitogenética, A.C.
Abstract: Japan is a mycophilic country where hundreds of species of wild edible fungi are consumed, including ectomycorrhizal species. Worldwide, the cultivation of ectomycorrhizal edible fungi represents a challenge of great economic, ecological and cultural relevance. This paper presents a review of the advances in the cultivation of edible ectomycorrhizal fungi developed in Japan. The greatest achievements have been carried out in some species of shimeji (Lyophyllum spp.), matsutake (Tricholoma matsutake) and the hypogeous fungus called shoro (Rhizopogon roseolus). In addition, some advances have been made in species of the genera Astraeus, Cantharellus, Sarcodon, Suillus and Tuber. Some of these latter species have received only interest in Japan, because they have culinary value exclusively in Eastern Asia. The cultivation of L. shimeji, a mushroom highly valued in the Japanese culture, has been achieved in the absence of host plants. The cultivation of matsutake, an iconic fungus of Japanese culture, has received great attention due to its dramatic decline of natural production in Pinus densiflora forests from the 1940s to the present, in Japan. The studies have been developed in laboratory, greenhouse and field conditions and have focused on the application of forest management techniques, the establishment of mycorrhizal tree plantations and the inoculation of trees with mycelium and spores. The key to the successful cultivation of these fungi requires a better understanding of the physiological, ecological, biochemical and genetic mechanisms involved in the mycorrhizal symbiotic relationship between trees and fungi, as well as in the understanding of the biotic and abiotic factors involved in the formation of basidiomata in field conditions. The methodological advances in these areas may allow the successful establishment of commercial plantations inoculated with fungi native to Japan. One of the main challenges is the enormous native diversity of species of ectomycorrhizal fungi that exist naturally in Japan, which represents a competitive factor for the success of plantations aimed to produce specific fungal species. However, given the great interest that exists in the country, this area of research looks very promising in the following decades.
Keywords: Lyophyllum shimeji, Tricholoma matsutake, Rhizopogon roseolus, mycophilia, Japan, cultivation of edible ectomycorrhizal fungi.
Resumen: Japón es un país micofílico en donde se consumen centenares de especies de hongos comestibles silvestres, incluyendo especies ectomicorrízicas. A nivel mundial, el cultivo de los hongos comestibles ectomicorrízicos representa un reto de gran relevancia económica, ecológica y cultural. En el presente trabajo se presenta una revisión de los avances del cultivo de los hongos comestibles ectomicorrízicos desarrollados en Japón. Los mayores logros se han tenido en algunas especies de shimeji (Lyophyllum spp.), del matsutake (Tricholoma matsutake) y del hongo hipogeo denominado shoro (Rhizopogon roseolus). Además se han logrado algunos avances en especies de los géneros Astraeus, Cantharellus, Sarcodon, Suillus y Tuber. Algunas de estas últimas especies han recibido solo interés en Japón, porque poseen valor culinario exclusivamente en Asia oriental. El cultivo de L. shimeji, un hongo altamente valorado en la cultura japonesa ha sido posible en ausencia de plantas hospederas. El cultivo de matsutake, un hongo icónico de la cultura japonesa, ha recibido una gran atención debido a la dramática declinación de su producción natural en los bosques de Pinus densiflora a partir de la década de 1940 a la fecha en Japón. Los estudios han sido desarrollados tanto en condiciones de laboratorio, invernadero y campo y se han centrado en la aplicación de técnicas de manejo forestal, del establecimiento de plantaciones de árboles micorrizados y de la inoculación de árboles con micelio y con esporas. La clave para el cultivo exitoso de dichos hongos requiere de una mejor comprensión de los mecanismos fisiológicos, ecológicos, bioquímicos y genéticos involucrados en la relación simbiótica entre árboles y hongos, así como en el entendimiento de los factores bióticos y abióticos involucrados en la formación de basidiomas en condiciones de campo. Los avances metodológicos en estas áreas podrán permitir el establecimiento exitoso de plantaciones comerciales inoculadas con hongos nativos de Japón. Uno de los principales retos es la enorme diversidad nativa de especies de hongos ectomicorrízicos que existen de manera natural en dicho país, lo cual representa un factor de competencia para el éxito de plantaciones destinadas a producir hongos específicos. Sin embargo, dado el gran interés que existe en el país, esta área de investigación luce prometedora en las próximas décadas.
Palabras clave: Lyophyllum shimeji, Tricholoma matsutake, Rhizopogon roseolus, micofilia, Japón, cultivo de hongos comestibles ectomicorrízicos.
INTRODUCTION
The Japanese love collecting and eating wild forest mushrooms (Bertelsen, 2013; Imazeki, 1988; Neda, 2003; Ogawa, 1978; Ogawa, 1983; Tsing, 2015), and their ancient mycophilic culture embraces several hundred of such ectomycorrhizal mushroom species (Imazeki and Hongo, 1987, 1989; Yamada, 2002). This interest has led many studies of these edible mushrooms. Examples include Lyophyllum shimeji (Kawam.) Hongo, Tricholoma matsutake (S. Ito & S. Imai) Singer, T. bakamatsutake Hongo, T. portentosum (Fr.) Quél., Rhizopogon roseolus (Corda) Th. Fr., Suillus grevillei (Klotzsch) Singer, Boletus edulis Bull., Amanita caesareoides Lj. N. Vassiljeva, Entoloma sepium (Noulet & Dass.) Richon & Roze, Cantharellus cibarius Fr., and Tuber indicum Cooke & Massee. Documents dating back a century describe the deep appreciation that people in western Japan have for collecting two particular mushrooms: L. shimeji (called “shimeji” or “hon”-shimeji, which means “true” shimeji) and T. matsutake (simply called “matsutake”) (Yamada et al., 2012). A modern Japanese proverb translates as: “Matsutake smells best, but shimeji tastes best.”
L. shimeji is one of the few ectomycorrhizal mushrooms that have been commercially cultivated without its host plant. Advanced cultivation techniques allow them to be sold in Japanese markets year-around. By contrast, the highly-valued T. matsutake is not as available because efforts to cultivate it, with or without its mycorrhizal host, have not yet been successful (Suzuki, 2005; Yamada, 2015).
The special aroma and firm texture of matsutake fascinates people who love the traditional Japanese dietary culture of “Washoku” which is celebrated at New Year. Matsutake is served at various ceremonial occasions and enjoyed as a seasonal food harvested from nature (http://www.unesco.org/culture/ich/en/lists; Figure 1). Therefore, many Japanese also are eager to see matsutake commercially cultivated to increase supplies and reduce costs.

Figure 1.
Matsutake-zukushi (“full-of-matsutake dishes”). The matsutake dishes illustrated here were served in atraditional Japanese hotel which is in the pine forests of Gifu Prefecture where they were harvested.
In the early 1990s, Japan’s economy was a global leader that some called an “Economic Animal.” Since then, Japan has shifted to a more stable state economy, and its agricultural and forestry sectors increasingly embrace the need for sustainable use of biological resources and emphasize nature conservation. Additionally, the Fukushima nuclear accident in March 2011 affected a wide geographic area of natural forests in the eastern part of Honshu Island. Forest mushroom researchers were initially disheartened by the disaster. However, the economic slowdown, changing forest management priorities, and nuclear contamination of some mushroom habitat soon motivated them to shift their research foci to new and better techniques for inoculating, transplanting and establishing mycorrhized tree seedlings more widely, especially planting them in areas with safe levels of radionuclides. The hope is that these techniques will provide new sources of these valued mushrooms.
In this paper, we review recent studies in the cultivation of the edible ectomycorrhizal mushrooms, especially L. shimeji, T. matsutake and R. roseolus. Then we propose future research topics and activities.
Cultivation of Lyophyllum Shimeji and related species
Growing L. shimeji on artificial nutrient substrates, without host plants, has been studied since the 1970s. A basic cultivation system was developed in the early 1990s (Ohta, 1994a, 1994b; Watanabe et al., 1994; Figure 2). A starch-based nutrient substrate enables the selection of dikaryotic fungal strains that would fruit within several months of inoculation onto the experimental medium. Although most ectomycorrhizal mushroom species show poor growth on nutrient media without glucose, L. shimeji readily catabolizes starch and can use it as a sole carbon source. Because starch, unlike glucose, does not increase osmotic pressure when it is present at high concentrations in the nutrient medium, starch-based solid media provide a sufficient and constant supply of carbon for the prolonged growth period of L. shimeji mycelium. As a result, such media can support, in pot cultivation, both the growth and fruiting of L. shimeji (Kawai, 1998; Ohta, 1998a).
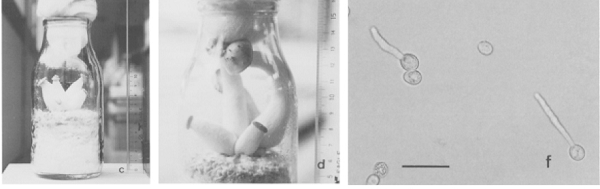
Figure 2.
In vitro fruiting of L. shimeji without a host. These cultivated mushrooms of L. shimeji produced basidiospores (right) that show germination tubes. The images are reproduced from Ohta (1994b).
This cultivation system was technically refined for large scale commercial production and has supplied markets since 2004 (Figure 3). Although several mushroom companies now grow the mushroom commercially, only one company has recently produced large commercial quantities (https://agribio.takara-bio.co.jp/kinoko/kinoko.html). The market price of shimeji is about 200-300 yen/100 g (8-12 USD/lb), which is one to three times higher than the price of other cultivated mushrooms such as shiitake (Lentinula edodes (Berk.) Pegler), enokitake (Flammulina velutipes (Curtis) Singer), oyster mushrooms (Pleurotus ostreatus (Jacq.) P. Kumm.), or button mushrooms (Agaricus bisporus (J. E. Lange) Imbach).

Figure 3.
Commercially cultivated L. shimeji fruit bodies packaged for sale. The image is downloaded from: https://agribio.takara-bio.co.jp/kinoko/product/daikoku/index.html.
Since the mid-1980s, researchers also tried to inoculate trees in the forest with L. shimeji mycelium, but although some fruiting occurred, mycorrhizae formation and growth of the mycelium were inadequately recorded. By the mid-1990s, techniques for soil inoculation were improved and ectomycorrhizal colonization was confirmed by Kawai (1997). The method entailed co-culture of mycelium and non-mycorrhizal pine trees in pot culture in a greenhouse. The soil was enhanced with starch as a carbon source and the mycelium was layered with the roots. The L. shimeji mycelium colonized the pine roots, produced ectomycorrhizae, and fruited from the pots (Figure 4).
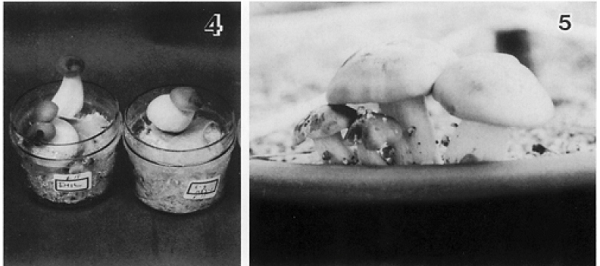
Figure 4.
Fruiting of L. shimeji with pine host under greenhouse condition. Left photograph: saprotrophic fruiting. Right photograph: symbiotic fruiting with pine host in the clay pot (Kawai, 1997).
This inoculation technique was then applied in natural forests with pine and oak trees (Hasegawa and Kawai, 2000; Kawai, 1999). Inoculated L. shimeji mycelia became established and annual fruiting started one to two years later. Subsequently, this inoculation technique has been widely applied in Japan and several provinces have reported success with establishing fruiting in new locations (e.g.Mizutani, 2005; Todo, 2012). Because this soil inoculation technique is easy to apply and works with both pines and oaks, L. shimeji cultivation in such forest sites (Figure 5) has become quite valuable. This approach might also be applicable to L. fumosum, a related ectomycorrhizal species, because culture characteristics for growing this mushroom on nutrient media are quite like those of L. shimeji.

Figure 5.
Fruiting of L. shimeji after field inoculation and establishment of a mycorrhizal association with forest oak trees.This soil inoculation technique was applied by local foresters in Nagano Prefecture, Japan.
Fortunately, mating tests on cultivated L. shimeji basidiomata can be used to refine breeding procedures (Kawai, 2000; Maeta et al., 2008a; Ohta, 1998a). For instance, artificial mass production methods might be improved by breeding for higher starch catabolic activity. Selection might also be used to discover more effective or more productive strains for the field inoculation of forest sites.
However, mating trials have shown that L. shimeji consists of at least two incompatible groups, that is, two different biological species within the Japanese population (Murakami et al., 2006). Because the sexually incompatible groups can be clearly distinguished with DNA markers (Maeta et al., 2008b), taxonomic revisions are needed for this morphological species (Larsson and Sundberg, 2011; Visnovsky et al., 2014; Yamada et al., 2012).
CULTIVATION OF TRICHOLOMA MATSUTAKE
Unlike L. shimeji, T. matsutake has not yet been successfully cultivated. Natural matsutake production in Japan occurs exclusively in Pinus densiflora forests. Such forests, in various regions of the country, have been managed for the sustainable harvest of T. matsutake for over a century (Fujiwara, 2011; Hamada, 1953, 1964, 1974; Mimura, 1909; Masui, 1927; Ogawa, 1978). However, during the 1940 - 1950s, drastic social change altered the condition of pine forests in ways that were unfavorable for T. matsutake production. As the forests aged, litter accumulated on the forest floor, resulting in eutrophication and higher soil humidity. Additionally, pine wilt disease killed pines in much of the western part of Honshu Island where T. matsutake was originally quite productive (Suzuki, 2005; Yamada and Kobayashi, 2008; Yamada, 2015). As a result, T. matsuake production declined sharply in the 1960 - 1970s, a trend that has continued, for Japan, until the present.
Nagano prefecture appears to be an exception to this decline. The prefecture is in the central region of Honshu Island, in a high elevation area that is surrounded by alpine mountains. T. matsutake populations persisted in pine forest growing on the steep mountains in the region, and they occupy most of the suitable forest habitat (Figure 6). For example, several local foresters still gain all their income from the harvest of T. matsutake (Fujiwara 2011). One experimental forest site has accumulated 36 years of T. matsutake harvests data (Figure 7). These data allow analyses of environmental factors affecting fruit-body productivity.
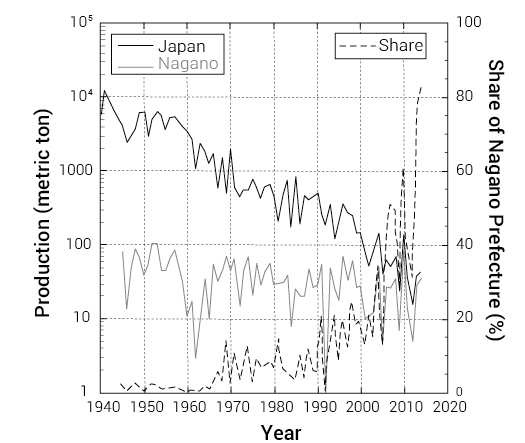
Figure 6.
Production of T. matsuake in Japan and Nagano Prefecture. The original data is derived from the databases of the Ministry of Agriculture, Forestryand Fisheries (MAFF) and Nagano Prefectural Forestry Center. The share of total Japanese production of T.matsuake that is harvested in Nagano Prefecture is illustrated by the dotted line.
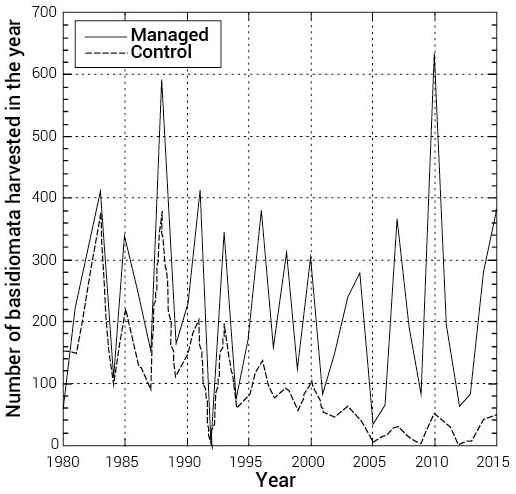
Figure 7.
Harvest of T. matsuake at the Toyooka Experimental Forest site in Nagano Prefecture. The solid line indicates matsutake harvest levels sampled in theforest plots (about 0.5 ha) located in forests managed for enhanced matsutake production by removing shrubs and reducing the litter layer every few years. The dotted line shows harvest levels from neighboring plots that were not managed. The graph was created from the original data of Furukawa et al. (2016).
Wang et al. (1997) inspired Japanese research groups to reconsider previous concepts of T. matsutake ecology and appropriate management of T. matsutake - P. densiflora associations. The paper sparked new research which contributed to a better understanding of the physiological, morphological and ultrastructural nature of T. matsutake ectomycorrhizae and the uniquely dense extramatrical mycelium or “shiro” that the fungus produces (Gill et al., 2000; Guerin-Laguette et al., 2000, 2004; Vaario et al., 2000, 2002; Yamada et al., 1999a, 1999b).
Considering the distinctive ectomycorrhizae that T. matsutake forms and applying that understanding to further improvements of in vitro inoculation techniques, Japanese researchers have succeeded in propagating pine seedlings with many clustered T. matsutake mycorrhizae imbedded in a large mass of “shiro”-like mycelium in 1 L pots. This combination of the dense masses of ectomycorrhizae and mycelia are like what the fungus produces in vivo (Kobayashi et al., 2007; Yamada et al., 2006). A recently started national T. matsuake cultivation project (2015 - 2019) aims to further improve the efficiency and reliability of in vitro mycorrhization techniques, so that high numbers of T. matsutake seedlings can be transplanted into the field to establish the first cultivation trials with this species (Figure 8; Yamada, 2015; Yamanaka, 2016).

Figure 8.
In vitroT. matsutake shiros formed on host pine seedlings in four liters of potting soil. This shows the largest T. matsutake – P. densiflora “shiros” (a dense mass of ectomycorrhizae and extramatrical mycelia) that have been produced under laboratory and greenhouse conditions.
In a manner similar to the successful efforts with L. shimeji, researchers would like to develop an in vitro system to study the fruiting of matsutake, with and without a host plant. Only one example has been reported (Ogawa and Hamada, 1964, 1975), where T. matsutake developed primordia on a nutrient substrate in the absence of a host. There are no reports, so far, of basidioma maturation without a host plant. Clémençon (2004) and Moore et al. (2011) provide current interpretations of Agaric primordia which might pertain to these previous efforts. Although the matsutake genome has been described (JGI web site and others), we still lack the means to interpret this information as it applies to the life-cycle, symbiotic relations, nutritional needs, and reproductive biology of the organism in situ. If we develop a greater understanding of the relevant factors in the matsutake life cycle, we might yet be able to induce fruiting under controlled conditions.
CULTIVATION OF RHIZOPOGON ROSEOLUS
The Japanese call the truffle Rhizopogon roseolus (= R. rubescens) “shou-ro,” which means “raindrops from pine leaves”. The name describes its habitat, namely, pine stands that develop on coastal sand dunes. P. thunbergii (Japanese black pine) and P. densiflora (Japanese red pine) are the host pine species. The pretty shape, unique texture, and mild fragrance of shou-ro is appreciated in the simple Japanese soup called “Osuimono” (Neda, 2003; Ogawa, 1983). Supplies of the truffle have become limited, however, because most of its coastal habitat has been converted to other human uses, especially in urban areas. The problem of decreased supplies can be somewhat ameliorated through habitat management such as the removal of shrubs and the litter layer. Amending the soil with charcoal also improves the growth of R. roseolus and subsequent fruiting (Ogawa, 1992). In combination with the conservation of pine stands in urban areas, these management practices have been applied in various locations (Ogawa, 2007). The recovery of R. roseolus production is occasionally reported in scientific papers and in newspapers.
Intending to develop propagation protocols, Yamada et al. (2001) reported that R. roseolus fruited in a small plot within a year of inoculating a P. densiflora seedling in a laboratory (Figure 9). With the goal of mass producing mycorrhized seedlings for plantation establishment, it also might be possible to scale-up the process with P. thinbergii. Kawai et al. (2008) reported that R. roseolus has a bipolar incompatibility mating system. Therefore, breeding trials similar to those with L. shimeji would improve the efficacy of strain selection. More recently, Shimomura (2013) and Shimomura et al. (2012) reported using spores and cultured mycelia to prepare effective inoculant for use in combination with elegant techniques of inducing mycorrhizal syntheses in field trials (Figure 10).
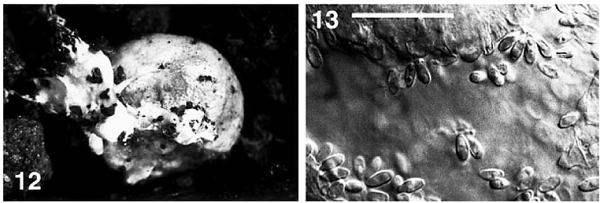
Figure 9.
Fruiting of R. roseolus on the open pot soil under laboratory condition. Photographs were reproduced from Yamada et al. (2001).

Figure 10.
Fruiting of R. roseolus under field condition. Photographs were reproduced from Shimomura (2013).
CULTIVATION TRIALS WITH OTHER MUSHROOMS
Japanese researchers have also conducted research on the cultivation of a variety of other edible ectomycorrhizal mushroom species (Table 1). Some of these species, such as T. bakamatsutake, E. sepium, Suillus spp., Astraeus hygrometricus (Pers.) Morgan, and Sarcodon aspratus (Berk.) S. Ito have never been targeted for cultivation efforts in western countries because their use is unique to Japanese, and in some cases other Asian cuisines (Fangfuk et al., 2010a; Kobayashi, 2005; Masuno et al., 2016; Murakami, 2004; Obana and Yamada, 2012; Ogawa, 1992; Yamanaka et al., 2014). For example, S. aspratus is also quite popular in Korea and China (Marcotullio, 2011) and Astraeus spp. in Thailand (Fangfuk et al., 2010a; Phosri et al., 2013).
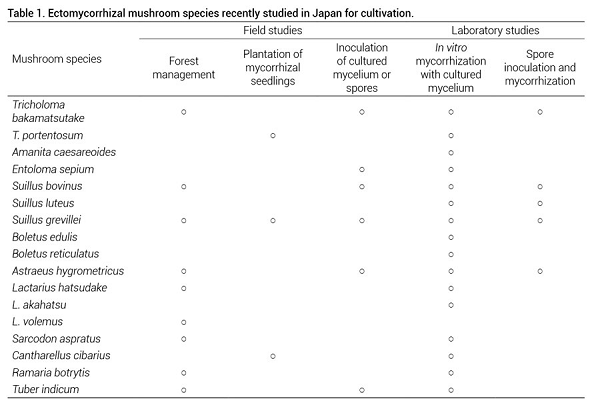
By contrast, A. caesareoides (the Asian relative of A. caesarea (Scop.) Pers.), B. edulis, C. cibarius and T. indicum (the Asian relatives of T. melanosporum Vittad.) have recently become the subject of cultivation trials (Endo et al., 2013, 2014; Ogawa et al., 2014, Yamada and Endo, 2011; Yamanaka, 2016) because data are available from research conducted on closely related species in western countries. So far, Japanese researchers have only succeeded with C. cibarius, producing fruit-bodies using in vitro mycorrhiza synthesis and greenhouse pot trials (Figure 11). Given the large global market for chanterelles, field trials with inoculated seedlings are expected soon.

Figure 11.
Japanese C. cibarius fruiting in pot culture with pine hosts under laboratory conditions. Shown here three (4 L) pots where C. cibarius fruited from inoculated pine seedlings.
FUTURE PERSPECTIVES
For decades, Japan has been a global leader in the development of superior strains of wood-decay mushrooms for cultivation. Species include: shiitake, enokitake, nameko (Pholiota microspora (Berk.) Sacc.), maitake (Grifola frondosa (Dicks.) Gray) and buna-shimeji (Hypsizygus marmoreus (Peck) H. E. Bigelow). This research foundation contributed to the successful commercial cultivation of the ectomycorrhizal fungus L. shimeji. These advances were achieved because the Japanese research community has developed innovative ideas for the controlled fruiting of mycorrhizal mushrooms, regardless of their autecology in nature. These ideas derive from knowledge about the physiology and genetics of the fungi, as well as the biochemistry of their mycelial grown on various nutrient media (Fukuda et al., 2003; Hasebe et al., 1998, Kawagishi et al., 2004; Kikuchi et al., 2009; Miyazaki et al., 2005; Ohta, 1998b;Yamanaka et al., 2000).
A better understanding of the genetic determinants of primordia and basidioma morphogenesis, such as the “Hox gene system” in animals or “ABC system” in flowering plants, likely will be central to further advancements in the cultivation of ectomycorrhizal mushroom species. Even if host-free cultivation methods are developed, chances are good that scientists specializing in mycorrhizal symbioses will remain essential to the application of such knowledge in a cost-effective manner. Several additional considerations apply when the intent is to establish mycorrhized seedlings in plantations or forest settings to sustainably produce edible mushrooms in more natural settings.
First, to obtain reliable fruiting a better understanding of mushroom synecology will be needed when such seedlings are introduced into native or managed forests. This will be a complex undertaking because Japan is a global hot spot for biodiversity and thousands of mycorrhizal fungi grow in the country’s diverse forest ecosystems (Imazeki and Hongo, 1987; 1989; Miyamoto et al., 2015; Tedersoo and Nara, 2010). Because of the high level of species diversity among the native ectomycorrhizal fungi, extensive competition among them is very probable.
By extrapolation, introduced fungal species or strains likely also will encounter substantial competition from already established native fungi. Advanced establishment techniques will be needed to sustain desired species or strains of introduced fungi in this context. This situation contrasts with the elegant and successful case of Lactarius deliciosus cultivation in New Zealand (Guerin-Laguette et al., 2014), where competing ectomycorrhizal fungal populations are comparatively sparse. In addition, as biodiversity correlates with climatic and geological diversity in Japan, in situ cultivation efforts may need to be specifically tailored to local forest communities.
Second, as with L. shimeji, some of the species selected for cultivation might consist of several cryptic species, each with their own ecological adaptations. For instance, Japanese A. hygrometiricus, B. reticulatus, C. cibarius and T. indicum are defined as the taxa sensu lato, and might very well need more detailed taxonomic analyses, including new species descriptions as required (Endo et al., 2014; Fangfuk et al., 2010b; Kinoshita et al., 2011; Ogawa et al., 2014; Phosri et al., 2013). Valid species identification is not only fundamental to effective research, but is also important for marketing the selected mushrooms as safe or superior foods.
Third, it is possible that native species diversity could be undesirably altered by the spread of non-native fungi growing in nearby plantations or by the introduction of selected fungal cultivars in native forests.
So, to compete among international efforts to cultivate edible ectomycorrhizal mushroom species, Japanese mycorrhizologists will need to build on their previous investigations and focus on research topics that are unique, scientifically intriguing, and commercially valuable. For instance, because T. matsutake is such a quintessentially Japanese mushroom, it would be a great honor for our scientists to be the first to develop reliable techniques for its artificial cultivation; in vitro, in situ or both. Advances in growing ectomycorrhizal mushrooms without their host plants will improve our understanding of their nutritional needs and might have enormous potential to broaden commercially-viable means of inoculation and production.
If advances such as those made with L. shimeji, R. roseolus, C. cibarius and T. matsutake are also applied to the other species in Table 1, the future looks bright for providing the large Japanese and global markets for edible mushrooms with increased supply, extended seasons of production, and a greater variety of culinary opportunities.
BIBLIOGRAPHY
Bertelsen C. D. (2013) Mushroom: A Global History. Reaktion Books. London, UK. 160 p.
Clémençon H. (2004) Cytology and Plectology of the Hymenomycetes. J. Cramer. Stuttgart, Berlin. 488 p.
Endo N., S. Gisusi, M. Fukuda and A. Yamada (2013)In vitro mycorrhization and acclimatization of Amanita caesareoides and its relatives on Pinus densiflora. Mycorrhiza 23:303-315.
Endo N., F. Kawamura, R. Kitahara, D. Sakuma, M. Fukuda and A. Yamada (2014) Synthesis of Japanese Boletus edulis ectomycorrhizae with Japanese red pine. Mycoscience 55:405-416.
Fangfuk W., K. Okada, R. Petchang, C. To-anun, M. Fukuda and A. Yamada (2010a)In vitro mycorrhization of edible Astraeus mushrooms and their morphological characterization. Mycoscience 51:234-241.
Fangfuk W., R. Petchang, C. To-anun, M. Fukuda and A. Yamada (2010b) Identification of Japanese Astraeus, based on morphological and phylogenetic analyses. Mycoscience 51:291-299.
Fujiwara G. (2011) How to Manage Forest for Matsutake. Zenrinkyo. Tokyo. (In Japanese).
Fukuda M., S. Ohno and M. Kato (2003) Genetic variation in cultivated strains of Agaricus blazei. Mycoscience 44:431-436.
Furukawa H., K. Masuno and Y. Takeuchi (2016) Forest management of matsutake productive sites for the optimization to global warming. Annual Reports of the Nagano Prefecture Forestry Research Center 30:87-100. (In Japanese).
Gill W. M., A. Guerin-Laguette, F. Lapeyrie and K. Suzuki (2000) Matsutake – morphological evidence of ectomycorrhiza formation between Tricholoma matsutake and host roots in a pure Pinus densiflora forest stand. New Phytologist 147:381-388.
Guerin-Laguette A., L. M. Vaario, W. M. Gill, F. Lapeyrie, N. Matsushita and K. Suzuki (2000) Rapid in vitro ectomycorrhizal infection on Pinus densiflora roots by Tricholoma matsutake. Mycoscience 41:389-393.
Guerin-Laguette A., K. Shindo, N. Matsushita, K. Suzuki and F. Lapeyrie (2004) The mycorrhizal fungus Tricholoma matsutake stimulates Pinus densiflora seedling growth in vitro. Mycorrhiza 14:397-400.
Guerin-Laguette A., N. Cummings, R. C. Butler, A. Willows, N. Hesom-Williams, S. Li and Y. Wang (2014)Lactarius deliciosus and Pinus radiata in New Zealand: towards the development of innovative gourmet mushroom orchards. Mycorrhiza 24:511-523.
Hamada M. (1953) Matsutake (in Japanese). Shizen 8:56-64. (In Japanese).
Hamada M. (1964) General Introduction to Tricholoma matsutake. In: Matsutake (Tricholoma matsutake Singer): its Fundamental Studies and Economic Production of the Fruit-body. The Matsutake Research Association. Kyoto, Japan. p 6. (In Japanese)
Hamada M. (1974) Matsutake Nikki (diaries of matsutake). Kyoto University. Kyoto. (In Japanese).
Hasebe K., I. Ohira and I. Arita (1998) Genetic relationship between high-, medium-, and low-temperature-type fruiting of Lentinula edodes in wood log culture. Reports of The Tottori Mycological Institute 36:21-28.
Hasegawa M. and M. Kawai (2000) Ectomycorrhization between Lyophyllum shimeji and Quercus phillyraeoides. Annual Reports of Nara Forest Research Institute 30:21-26. (In Japanese).
Imazeki R. (1988) Forest Life Science. Tojusha. Tokyo. (In Japanese).
Imazeki R. and T. Hongo (1987) Colored Illustrations of Mushrooms of Japan I. Hoikusha. Osaka. (In Japanese).
Imazeki R. and T. Hongo (1989) Colored Illustrations of Mushrooms of Japan II. Hoikusha. Osaka. (In Japanese).
Kawagishi H., K. Hamajima, R. Takanami, T. Nakamura, Y. Sato, Y. Akiyama, M. Sano and O. Tanaka (2004) Growth promotion of mycelia of the matsutake mushroom Tricholoma matsutake by D-isoleucine. Bioscience, Biotechnology, and Biochemistry 68:2405-2407.
Kawai M. (1997) Artificial ectomycorrhizal formation on roots of air-layered Pinus densiflora saplings by inoculation with Lyophyllum shimeji. Mycologia 89:228-232.
Kawai M. (1998) Mass-production of fruit-bodies of Lyophyllum shimeji without host plant. Japanese Journal of Mycology 39:117-120. (In Japanese).
Kawai M. (1999) Mycorrhizal colonization and Fruiting of L. shimeji by the inoculation of cultured mycelium in the soil under forest conditions. Annual Reports of Nara Forest Research Institute 29:1-27. (In Japanese).
Kawai M. (2000) On the monokaryotic mycelial state and its mating type of Lyophyllum shimeji. Annual Reports of Nara Forest Research Institute 30:17-20. (In Japanese).
Kawai M., M. Yamahara and A. Ohta (2008) Bipolar incompatibility system of an ectomycorrhizal basidiomycete, Rhizopogon rubescens. Mycorrhiza 18:205-210.
Kikuchi K., N. Matsushita and K. Suzuki (2009) Fruit body formation of Tylopilus castaneiceps in pure culture. Mycoscience 50:313-316.
Kinoshita A., H. Sasaki and K. Nara (2011) Phylogeny and diversity of Japanese truffles (Tuber spp.) inferred from sequences of four nuclear loci. Mycologia 103:779-794.
Kobayashi H., T. Watahiki, M. Kuramochi, S. Onose and A. Yamada (2007) Production of pine seedlings with shiro-like structure of the matsutake mushroom (Tricholoma matsutake (S. Ito et Imai) Sing.) in a large culture bottle. Mushroom Science and Technology 15:151-155.
Kobayashi H. (2005) Study on the Japanese “Harushimeji” (Entoloma spp.): its mycorrhizal morphology, ecology, and utilization. PhD Thesis of the University of Tsukuba. (In Japanese).
Larsson E. and H. Sundberg (2011) Lyophyllum shimeji, a species associated with lichen pine forest in northern Fennoscandia. Mycoscience 52:289-295.
Maeta K., A. Koike, H. Mukaiyama, T. Ochi, T. Terashita, Y. Kitamoto and T. Aimi (2008a) Preparation and crossing of mating-capable monokaryons via protoplasting of the dikaryotic mycelia of a mycorrhiza mushroom, Lyophyllum shimeji. Journal of Wood Science 54:337-340.
Maeta K., A. Koike, T. Ochi, T. Mukaiyama, T. Terashita, Y. Kitamoto and T. Aimi (2008b) Biological species of Lyophyllum shimeji. Mushroom Science and Technology 16:105-108.
Marcotullio M. C. (2011) Sarcodon mushrooms: biologically active metabolites. In: Phytochemicals: Bioactivities and Impact on Health. I. Rasooli (ed.). InTech Europe. Rijeka, Croatia. pp:77-94.
Masui K. (1927) A study of the ectotrophic mycorrhizas of woody plants. In: Memoirs of the College of Science. University of Kyoto. Series B. Vol. 3. pp:152-279.
Masuno K., M. Fukuda, A. Yamada, M. Ichikawa, H. Furukawa and K. Katagiri (2016) Developments of mushroom cultivation technique and promotion system for better use of forest environment, by the used of locally accumulated forest plant biomass. Annual Reports of the Nagano Prefecture Forestry Research Center 30:47-86. (In Japanese).
Mimura K. (1909) Matsutake jinkou hanshoku shiken (Some experiments on the cultivation of Tricholoma matsutake). Bulletin of Forestry and Forest Products Research Institute 7:93-108.
Miyamoto Y., A. Sakai, M. Hattori and K. Nara (2015) Strong effect of climate on ectomycorrhizal fungal composition: evidence from range overlap between two mountains. The ISME Journal 9:1870-1879.
Miyazaki Y., M. Nakamura and K. Babasaki (2005) Molecular cloning of developmentally specific genes by representational difference analysis during the fruiting body formation in the basidiomycete Lentinula edodes. Fungal Genetics and Biology 42:493-505.
Mizutani K. (2005) Fruit body occurrence during 5 years by inoculation of Lyophyllum shimeji mycelial culture in Pinus densiflora forest. Bulletin of the Gifu Prefectural Forest Science Research Institute 34:1-6. (In Japanese).
Moore D., G. D. Robson and A. P. J. Trinci (2011) 21st Century Guidebook to Fungi. Cambridge University Press. New York, USA. 627 p.
Murakami S., E. Nagasawa and K. Matsumoto (2006) Taxonomic study in the biological species of Lyophyllum shimeji. In: Abstracts of the 10th Meeting of Japanese Society of Mushroom Science and Biotechnology. p:48. (In Japanese).
Murakami Y. (2004) Increase of fruiting of mycorrhizal fungi as a result of environmental modification in a pine forest. In: Abstracts of Papers Presented at the 48th Annual Meeting of the Mycological Society of Japan. A2904. (In Japanese).
Neda H. (2003) Kinoko Hakubutsukan (Mushroom museum). Yasaka Shobo. Tokyo. (In Japanese).
Obana R. and A. Yamada (2012) Fungal isolation of edible Thelephoraceae mushrooms. In: Abstracts of Papers Presented at the 56th Annual Meeting of the Mycological Society of Japan. B6. (In Japanese).
Ogawa M. (1978) The Biology of Matsutake Mushroom. Tsukiji Shokan Publishing Co. Ltd. Tokyo. (In Japanese).
Ogawa M. (1983) Natural History of Mushrooms. Tsukiji Shokan Publishing Co. Ltd. Tokyo. (In Japanese).
Ogawa M. (1992) How to Cultivate Wild Mushrooms. Zenkoku Ringyo Fukyu Kyokai. Tokyo. (In Japanese).
Ogawa M. (2007) Reviving Pine Trees by Charcoal Treatment and Mycorrhizal Developmet. Tsukiji Shokan Publishing Co. Ltd. Tokyo. (In Japanese).
Ogawa M. and M. Hamada (1964) Method for growing the fruit-body primordium of matsutake on the artificial medium (article for patent application). In: Matsutake (Tricholoma matsutake Singer): its Fundamental Studies and Economic Production of the Fruit-body. Matsutake Research Association (ed.). Kyoto, Japan. pp:115-117. (In Japanese).
Ogawa M. and M. Hamada (1975) Primordia formation of Tricholoma matsutake (Ito et Imai) Sing. in pure culture. Transactions of the Mycological Society of Japan 16:406-415.
Ogawa W., N. Endo and A. Yamada (2014) Morphological and molecular phylogenetical analyses of Japanese Cantharellus. In: Abstracts of Papers Presented at the 58th Annual Meeting of the Mycological Society of Japan. B07. (In Japanese).
Ohta A. (1994a) Some cultural characteristics of mycelia of a mycorrhizal fungus, Lyophyllum shimeji. Mycoscience 35:83-87.
Ohta A. (1994b) Production of fruit-bodies of a mycorrhizal fungus, Lyophyllum shimeji, in pure culture. Mycoscience 35:147-151.
Ohta A. (1998a) Culture conditions for commercial production of Lyophyllum shimeji.Japanese Journal of Mycology 39:13-20. (In Japanese).
Ohta A. (1998b) Fruit-body production of two ectomycorrhizal fungi in the genus Hebeloma in pure culture. Mycoscience 39:15-19.
Phosri C., M. P. Martín and R. Watling (2013)Astraeus: hidden dimensions. IMA Fungus 4:347-356.
Shimomura N. (2013) Cultivation of gourmet mushroom Shouro (Rhizopogon roseolus). Business meeting of Tottori University in Tokyo. http://www.cjrd.tottori-u.ac.jp/.
Shimomura N., M. Matsuda, K. Ariyoshi and T. Aimi (2012) Development of mycelial slurries containing surfactant for cultivation of the edible ectomycorrhizal mushroom Rhizopogon roseolus (syn. Rhizopogon rubescens). Botany 90:839-844.
Suzuki K. (2005) Ectomycorrhizal ecophysiology and the puzzle of Tricholoma matsutake. Journal of the Japanese Forest Society 87:90-102. (In Japanese).
Tedersoo L. and K. Nara (2010) General latitudinal gradient of biodiversity is reversed in ectomycorrhizal fungi. New Phytologist 185:351-354.
Todo C. (2012) Fruit body occurrence after inoculation of Lyophyllum shimeji mycelial culture in Quercus serrata forest. Annual Reports of Hyogo Prefectural Research Institute for Agriculture, Forestry and Fishery: Forestry Section 57:12-15. (In Japanese).
Tsing A. L. (2015) The Mushroom at the End of the World: On the Possibility of Life in Capitalist Ruins. Princeton University Press. Oxford, U. K. 331 p.
Vaario L. M., A. Guerin-Laguette, W. M. Gill, F. Lapeyrie and K. Suzuki (2000) Only two weeks are required for Tricholoma matsutake to differentiate ectomycorrhizal Hartig net structures in roots of Pinus densiflora seedlings cultivated on artificial substrate. Journal of Forest Research 5:293-297.
Vaario L. M., A. Guerin-Laguette, N. Matsushita, K. Suzuki and F. Lapeyrie (2002) Saprobic potential of Tricholoma matsutake: growth over pine bark treated with surfactants. Mycorrhiza 12:1-5.
Visnovsky S. B., N. Cummings, A. Guerin-Laguette, Y. Wang, A. Yamada, H. Kobayashi, M. Kawai and A. R. Pitman (2014) Detection of the edible ectomycorrhizal fungus, Lyophyllum shimeji, colonizing seedlings of cultivated conifer species in New Zealand. Mycorrhiza 24:453-463.
Wang Y., I. R. Hall and L. A. Evans (1997) Ectomycorrhizal fungi with edible fruiting bodies 1. Tricholoma matsutake and related fungi. Economic Botany 51:311-327.
Watanabe K., M. Kawai and Y. Obatake (1994) Fruiting body formation of Lyophyllum shimeji in pure culture. Mokuzai Gakkiashi 40:879-882.
Yamada A. (2015) Ecology of Tricholoma matsutake as the ectomycorrhizal mushroom. JATAFF Journal 3:30-34. (In Japanese).
Yamada A. and N. Endo (2011) Ectomycorrhiza synthesis of Cantharellales mushrooms: morphological and physiological characteristics. In: The Japanese Forest Society Congress 122: L21. 22 March. http://doi.org/10.11519/jfsc.122.0.341.0 (In Japanese)
Yamada A. (2002) Utility of mycorrhizal mushrooms as food resources in Japan. Journal of the Faculty of Agriculture, Shinshu University 38:1-17. (In Japanese).
Yamada A., S. Kanekawa and M. Ohmasa (1999a) Ectomycorrhiza formation of Tricholoma matsutake on Pinus densiflora. Mycoscience 40:193-198.
Yamada A. and H. Kobayashi (2008) Future perspective in the cultivation of matsuake. Shinrin Kagaku 53:41-42. (In Japanese).
Yamada A., K. Maeda and M. Ohmasa (1999b) Ectomycorrhiza formation of Tricholoma matsutake isolates on seedlings of Pinus densiflora in vitro. Mycoscience 40:455-463.
Yamada A., K. Maeda, H. Kobayashi and H. Murata (2006) Ectomycorrhizal symbiosis in vitro between Tricholoma matsutake and Pinus densiflora seedlings that resembles naturally occurring ‘shiro’. Mycorrhiza 16:111-116.
Yamada A., K. Masuno and M. Fukuda (2012) Taxonomic consideration for Japanese Lophyllum shimeji and its allied species. Mushroom Science and Technology 20:9-15. (In Japanese).
Yamada A., T. Ogura and M. Ohmasa (2001) Cultivation of mushrooms of edible ectomycorrhizal fungi associated with Pinus densiflora by in vitro mycorrhizal synthesis I. Primordium and basidiocarp formation in open-pot culture. Mycorrhiza 11:59-66.
Yamanaka K., K. Namba and A. Tajiri (2000) Fruit body formation of Boletus reticulatus in pure culture. Mycoscience 41:189-191.
Yamanaka T. (2016) Researches on the cultivation of matsutake and truffles. Sanrin 1580:64-73. (In Japanese).
Yamanaka T., Y. Ota, M. Konno, M. Kawai, A. Ohta, H. Neda, Y. Terashima and A. Yamada (2014) The host ranges of conifer-associated Tricholoma matsutake, Fagaceae-associated T. bakamatsutake and T. fulvocastaneum are wider in vitro than in nature. Mycologia 106:397-406.
Author notes
akiyosh@shinshu-u.ac.jp