Articles
Mejoramiento de las Propiedades Físicas del Polibutilsuccinato Usando Estabilizantes de Origen Vegetal y Sintético
Improvement of the Physical Properties of Polybutylsuccinate Using Stabilizers of Vegetable and Synthetic Origin
Mejoramiento de las Propiedades Físicas del Polibutilsuccinato Usando Estabilizantes de Origen Vegetal y Sintético
Revista Politécnica, vol. 55, no. 1, pp. 71-82, 2025
Escuela Politécnica Nacional
Received: 28 March 2024
Accepted: 17 February 2024
Resumen: La producción de residuos plásticos no degradables ha generado una problemática ambiental bastante grave lo que provoca la búsqueda de polímeros biodegradables que puedan reemplazar a estos plásticos. Uno de estos es el polibutilsuccinato (PBS) que se caracteriza por su buena trabajabilidad, alta resistencia tanto térmica como química, así como alta biocompatibilidad comparable al polietileno. El presente estudio tiene como objetivo el mejoramiento de las propiedades físicas del PBS sintetizado por condensación adicionando estabilizantes vegetales y sintéticos. Se analizó la degradación térmica que sufre el material mediante análisis termogravimétrico y la reología del biopolímero mediante reología rotacional frente a condiciones de trabajo de extrusión para perfiles. Para esto se preparó probetas con polvo de cáscara de patata, ácido ferúlico y luperox. El estudio confirmó que la adición del 5 % en masa de polvo de cáscara de patata al PBS mejora su estabilidad térmica, logró alcanzar valores de velocidad de cizalla para su procesabilidad por extrusión y brinda una oportunidad para copolimerizarse, de tal manera que su uso sea viable.
Palabras clave: Celulosa, biopolímeros, ácido ferúlico, extrusión, patata.
Abstract: The production of non-degradable plastic waste has generated serious environmental problems, which has motivated the search for biodegradable polymers that can replace plastics. One of these is polybutylsuccinate (PBS), which is characterized by good workability, high thermal and chemical resistance, and high biocompatibility comparable to polyethylene. The objective of this study was to improve the physical properties of PBS synthesized by condensation by adding vegetables and synthetic stabilizers. The thermal degradation suffered by the material was analyzed by thermogravimetric analysis, and the rheology of the biopolymer by rotational rheology against extrusion working conditions. The test tubes were prepared using potato peel powder, ferulic acid, and luperox. The study confirmed that the addition of 5 % by mass of potato peel powder to PBS improves its thermal stability, achieves shear rate values for its processability by extrusion to profiles, and provides an opportunity to copolymerize, in such a way that its use is viable.
Keywords: Cellulose, biopolymers, ferulic acid, extrusion, potato.
(1. INTRODUCTION
The expansion of the global economy and advancement of the petrochemical industry in the 20th century boosted the production of plastics. Their low cost, versatility, and ease of production give them an advantage over traditional materials, such as wood, metal, and glass. Plastic is one of the most widely used materials in modern life, but it is also one of the slowest to decompose. Their applications range from the automotive industry to food packaging (Aliotta et al., 2022).
The widespread use of plastics and their incorrect waste management have generated large-scale environmental problems (Wiesinger et al., 2021). Currently, plastics represent between 12 and 25 % of solid urban waste (Cottom et al., 2024). This requires the implementation of eco-friendly alternatives, such as the development of bioplastics and efficient waste management, to mitigate the invasion of plastics on land and sea (Papadopoulou et al., 2023).
Nowadays, biodegradable materials have gained importance because of their inherent value in reducing the carbon footprint, helping the environment and ecosystems, saving fossil resources, and allowing the development of an innovative option based on renewable resources, which in turn degrades in a shorter time and generates energy savings in the production of plastics (Karthikumar et al., 2024; Pacheco et al., 2022).
Some biodegradable polymers are commercially available in the market, such as poly(butylene adipate-co-terephthalate) (PBAT), polyhydroxyalkanoates (PHAs), polycaprolactone (PCL), poly(propylene carbonate) (PPC), poly(butylene succinate) (PBS), poly(lactic acid) (PLA), and thermoplastic starch (Papadopoulou et al., 2023). However, further research and development of biodegradable polymers are needed to improve their mechanical properties, thermal stability, and lower production costs (Averous & Boquillon, 2004).
PBS has excellent physical properties and is easy to process. It is an aliphatic polyester synthesized by the polycondensation reaction of petroleum-derived aliphatic dicarboxylic acid (succinic acid) and 1,4-butanediol. The biodegradability of PBS is similar to that of cellulose (Huang et al., 2012; Marek et al., 2020). PBS is synthesized from succinic acid based on renewable resources, which reduces its carbon footprint (Muthuraj et al., 2014; Zhang et al., 2018). However, they are expensive commercial polymers that cannot compete with petrochemical polymers (Barrino et al., 2023; Martínez et al., 2020).
PBS is a white crystalline material with a melting point ranging from approximately 90 to 120°C. Its glass transition temperature (Tg), which falls between -45°C and -10°C, is comparable to the Tg values of polyethylene and polypropylene (Rafiqah et al., 2021). The physical properties of PBS are similar to those of polyethylene and polypropylene, including its density, tensile strength, and flexibility. The elongation at break is approximately 330%, and the tensile strength is 330 kg/cm². PBS also exhibits good impact resistance, a low coefficient of friction, and excellent chemical resistance, particularly to acids and oils. Additionally, it shows better fiber compatibility, heat resistance, and biodegradability under specific conditions when compared to poly lactic acid (PLA) and poly glycolic acid (PGA) (Muthuraj et al., 2014). Its low moisture absorption, high dimensional stability, and ability to be processed through conventional polymer processing techniques further enhance its suitability for various industrial applications (Rajgond et al., 2024).
Currently, polymers that are processed as films, fibers, and other objects have seen an increase in demand for polymers that have high-temperature stability. Stabilization (against both heat and light) is a major objective of polymer development (Miranzadeh et al., 2024). Stabilizers are increasingly needed in plastics to stabilize compounds during processing at higher temperatures and/or to provide stability for application during continuous exposure to elevated temperatures and/or outdoor conditions (Shekhar & Mondal, 2024). Over time, stabilizers have been derived to form fewer toxic forms, and have been recovered from biomass waste (Sohn et al., 2020).
Organic stabilizers can react with molecular oxygen in a process called autooxidation, which is initiated by heat, light, mechanical stress, catalyst residues, or reaction with impurities to form alkyl radicals (Marturano et al., 2023). In turn, the free radicals can react to degrade the polymer, causing brittleness, melt flow instability, loss of tensile properties, and discoloration (Fodil Cherif et al., 2020). Oxidation can be reduced with chain-breaking antioxidants to slow the rate of propagation or with preventive antioxidants to prevent the initial formation of free radicals. Antioxidants deactivate these sites by decomposing hydroperoxide or terminating the free radical reaction (Reano et al., 2016).
Ferulic acid (aromatic hydroxy acid) is of plant origin, and potato peel (phenolic and lignocellulosic compounds) recovered from biomass residues confers photoprotective and thermooxidative properties to the PBS chain (Dekker, 2005; Righetti et al., 2019). They are considered as primary antioxidants that stabilize polymers against oxidative degradation. These stabilizers usually have hindered phenolic groups in their chemical structure and act by scavenging free radicals (Reano et al., 2016).
The application of natural stabilizers improves thermal and mechanical properties during processing at high temperatures (Marturano et al., 2023). Thus, copolymerization can be carried out with other types of polymers to lower costs and maintain processability for the extrusion of PBS (Fujimaki, 1998; Muthuraj et al., 2014; Xu & Guo, 2010)
This study investigates the improvement in the physical properties of PBS when stabilizers of natural and synthetic origin are incorporated for extrusion processing in such a way that a useful and competitive biopolymer for industrial applications is achieved (Cecchi, 2021; Wang et al., 2007). The natural stabilizers used were ferulic acid, potato peel, and synthetic stabilizers, such as ditert-butyl peroxide (luperox). In addition, the Carreau Model was used to project the processability of the biopolymer by extrusion and evaluate its rheological behavior.
2. METHODOLOGY
2.1. Materials
The polybutylsuccinate biopolymer was synthesized from succinic acid CAS No 110-15-6, 1-4, butanediol, CAS No 110-63-4, methanol CAS No 67-56-1, chloroform CAS No 67-66- 3, and titanium tetrabutoxide (Ti(O(CH2)3CH3)4), CAS No. 5593-70-4, all with reactive grade, was used as the catalyst.
The following vegetable stabilizers were used: potato peel powder (Solanum tuberosum) produced by SüdStärke (Schrobenhausen, Germany) and ferulic acid (trans-4-hydroxy-3-methoxycinnamic acid), CAS No. 537-98-4; Luperox (ditert-butyl peroxide), CAS No. 110-05-4, was used as a synthetic stabilizer. All reagents were purchased from the Sigma-Aldrich brand of the Merck Company.
2.2. PBS synthesis
PBS was synthesized by a two-stage condensation method, as shown in Figure 1. For this process, a batch reactor with a volume of 5 L was used, which is typical for small-scale polymerization reactions. In the reactor, 150.0 g of succinic acid, 137.5 g of butanediol, 0.86 g of catalyst, and stabilizers were added according to the formulation. Table 1 shows the amount of stabilizer with respect to the biopolymer, where potato peel powder (A), ferulic acid (B), and Luperox (E) were added in mass percentage (Zhang et al., 2018).
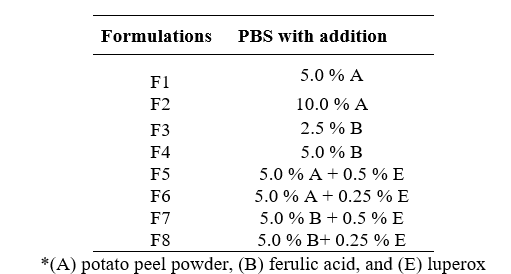
The molar ratio of succinic acid to butanediol was 1:1.2, and the catalyst to succinic acid ratio was 1:500.
Once the reactor was loaded, it was purged by adding nitrogen to generate an inert atmosphere. The nitrogen flow rate should be controlled at 20 to 50 mL/min to ensure proper purging without excessive gas consumption. The reactor was then immediately heated to 150 °C and stirred at 60 to 100 rpm for a period of three hours to facilitate esterification. The stirring speed should be low enough to ensure uniform mixing but not so fast as to cause unwanted turbulence or uneven heating. During this time, the water vapor generated in the first reaction was continuously removed via nitrogen purging and vacuum generation. The vacuum pressure within the reactor should be maintained at 10-20 mmHg to efficiently remove water without compromising the reaction conditions.
After the esterification, the system was heated to 210 °C for another three hours, where polycondensation occurred. It is at this stage that ferulic acid, potato peel powder, and luperox are added to enhance the physical properties of the PBS. Temperature control during this step is crucial, and the reaction temperature should be monitored using a calibrated thermocouple to ensure accuracy. Once the polycondensation was complete, the system was allowed to cool to room temperature, resulting in a solid white product.
The biopolymer obtained was then dissolved in chloroform and precipitated in excess methanol (three times the amount of chloroform). This purification step helps eliminate unreacted monomers and residual oligomers. Following precipitation, the polymer was dried under a vacuum to remove any residual solvents, ensuring the final product is pure and free from volatiles.
The final yield of PBS obtained from this method is approximately 150 g for each formulation, as shown in Table 1. This yield is consistent across the different formulations, with slight variations depending on the efficiency of the reaction and the purification steps employed. To verify the synthesis of PBS and check for any impurities, an FTIR (Fourier Transform Infrared Spectroscopy) analysis should be conducted on the final product.
2.3 Characterization of the biopolymer
To verify the composition of the samples, FTIR (Fourier Transform Infrared Spectroscopy) tests were performed, where the characteristic bands of the functional groups belonging to PBS were analyzed. A NICOLET 6700 S/N AHR071021 model with a resolution of 4 cm-1 was used. Smart Orbit reflection with a diamond crystal was used. The spectral width used is between 4000 and 400 cm-1.
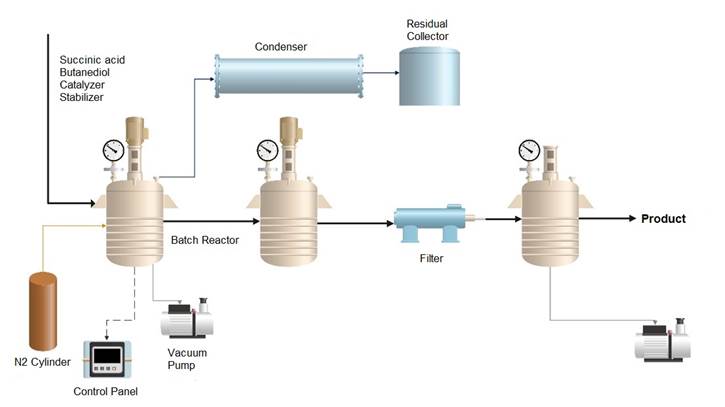
Figure 1
Synthesis of polybutylsuccinate by esterification and polycondensation in batch process
2.4 Determination of oxidation induction time (OIT)
For each analysis, experiments were carried out in duplicate using platinum crucibles. The OIT value is determined graphically by the oxidation induction time, with the onset or intersection of the tangents to the weight curve of a TGA before and immediately after weight loss, with respect to the time scale. Thermal analysis was carried out by thermogravimetry on a model Q500 thermobalance (TA Instruments), and thermogravimetric analysis was used to determine the oxidation induction time (OIT) in dynamic mode at a temperature ramp of 5 °C/min, from room temperature to 500 °C, in an air atmosphere at a flow rate of 30 mL/min, following the general test standard UNE-EN 11 35.
2.5 Rotational Rheology
Rheological tests were carried out in an AR-G2 rotational rheometer (TA Instruments) with a pressure of 0-5 bar a minimum torque of 10 μN.m. The test conditions to measure shear rate were a continuous ramp, test time of 6 min, measurement interval of 10-3 to 103.10 measurements per minute, logarithmic scale, test in the presence of nitrogen, and at 140 °C for extrusion.
3. RESULTS AND DISCUSSION
3.1 Characterization of PBS
The FTIR spectrums of polybutylsuccinate presented in Figure 2 and Figure 3 show an absorption band at 2945 cm-1, which is assigned to the stretching of the C-H bond. The intense band at 1712 cm-1 corresponds to the stretching vibration of the C=O carbonyl, characterizing the formation of the ester group. Furthermore, the peak at 1341 cm-1 is assigned to the stretching vibration of the -COO- bond. The signal at 1151.9 cm-1 is characteristic of the C-O-C stretching vibration in the repeat unit -OCH2CH2, and all bands indicate the presence of PBS (Abderrahim et al., 2015).
3.1.1 PBS with plant stabilizers
Figure 2a shows the infrared spectra of PBS with stabilizers, such as potato peel and ferulic acid, at different concentrations. The spectra did not show significant differences between the samples and the standard PBS, which can be attributed to the insertion of the stabilizer without alteration in the polymer structure (Barletta et al., 2022).
3.1.2 PBS with vegetable and synthetic stabilizers
Figure 2b shows the spectra of PBS with stabilizers of plant and synthetic origins at different concentrations. It can be verified that there was no chemical modification of the developed polymers.
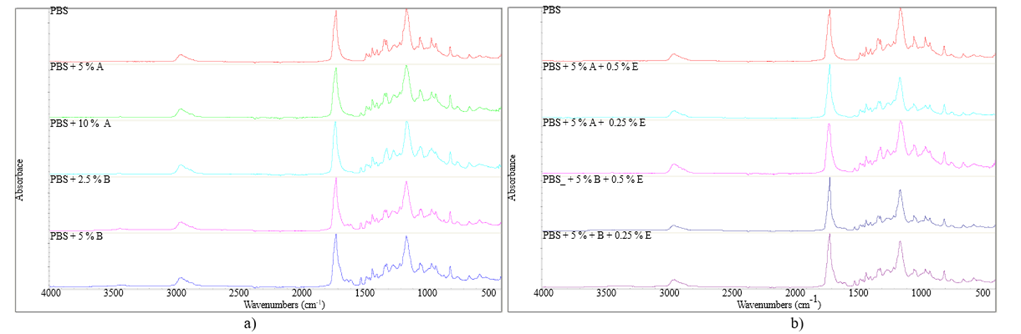
Figure 2
Spectra a) PBS with stabilizers of plant origin for extrusion b) PBS with stabilizers of plant and synthetic origin for extrusion (no chemical change upon stabilizer addition)
3.2 Oxidative thermal stability
3.2.1 PBS with plant stabilizers
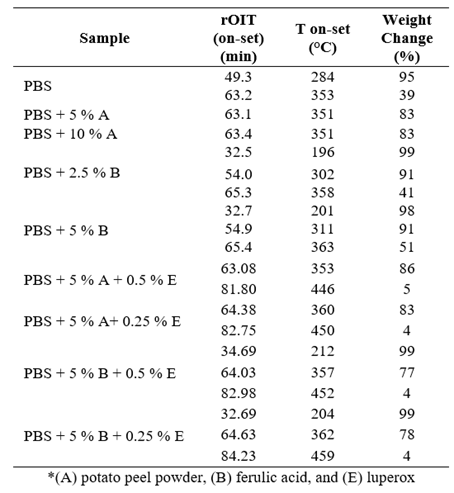
The thermogravimetric curves shown in Figure 3 provide insight into the mass loss profiles of the samples over time (TG curves), which were used to determine the oxidation induction time (OIT) and degradation temperatures. As indicated in Table 2, the addition of potato peel and ferulic acid to the PBS samples demonstrated a stabilizing effect on the polymer. The standard PBS exhibited a mass loss of 61 % at an onset temperature of 353 °C and an oxidation induction time (OIT) of 63.2 minutes. In contrast, the PBS + 5 % potato peel sample lost only 17 % of its weight at an onset temperature of 351 °C, with an OIT of 63.1 minutes. This suggests that the presence of potato peel stabilized the polymer, maintaining the oxidation temperature while slightly reducing the OIT. Increasing the proportion of potato peel further, while maintaining the same oxidation temperature (351 °C), led to a slight decrease in oxidation induction time. This trend of reduced mass loss and stabilized degradation temperatures in samples with stabilizers can be attributed to the effect of the additives, which mitigate oxidative degradation. As the volatile decomposition products are detected during the decomposition process, the stabilizing compounds delay the onset of significant mass loss, resulting in higher degradation temperatures compared to the unmodified PBS (Hiller et al., 2023; Georgousopoulou et al., 2016).
The PBS sample containing 2.5 % ferulic acid shows a weight loss of 59% at an onset temperature of 358 °C, with an oxidation induction time (OIT) of 65.3 minutes. When the ferulic acid content is increased to 5 %, the sample loses 49% of its weight at an onset temperature of 363 °C, with an OIT of 65.4 minutes. These results indicate that while ferulic acid contributes to improving the oxidation resistance of PBS, the potato peel stabilizer (added at both 5 % and 10 % concentrations) offers the most significant protective effect, allowing PBS to withstand oxidation for longer periods and reach higher degradation temperatures.
This suggests that potato peel provides similar stabilization in comparison to ferulic acid, due to its complex lignocellulosic structure and aromatic groups, capable of better reinforcing the polymer matrix and interacting with PBS at the molecular level, enhancing its thermal stability and resistance to oxidative degradation. These findings highlight the importance of the choice of stabilizers in tailoring the properties of biopolymers for various applications, particularly in environments where thermal and oxidative stability is critical (Olumurewa et al., 2022)
3.2.2 PBS with vegetable and synthetic stabilizers
Figure 4 illustrates that all additive combinations used in the study contributed to stabilizing the master polymer. Among the options, the most effective approach for stabilizing PBS against oxidative degradation is the combination of potato peel and luperox, irrespective of their proportions. This pairing offers enhanced stabilization due to the higher antioxidant content, which provides superior protection against thermal degradation of the polymer.
The F5 sample of PBS, which contains both potato peel fiber and luperox demonstrate significant stabilization, losing only 14 % of its weight at an onset temperature of 353 °C with an oxidation induction time (OIT) of 63.08 minutes before beginning irreversible thermal degradation. This behavior is similar to the effect observed when the percentage of luperox is reduced to 0.25 %, indicating the synergistic stabilizing effect of combining potato peel fiber and luperox in the PBS matrix. In comparison, the standard PBS sample lost more than 61 % of its weight at the same onset temperature (353 °C), with an OIT of 63.2 minutes, as shown in Table 2. The combination of ferulic acid and luperox in PBS samples showed slightly improved thermal stability, with a 23 % weight loss at an onset temperature of 357 °C and an OIT of 64.03 minutes. These results suggest that while ferulic acid and luperox improve oxidation resistance, the combination of potato peel fiber and luperox provides the most effective stabilization, as it significantly delays the onset of degradation and maintains a higher thermal stability for PBS.
Potato peel fibers, ferulic acid, and luperox can collectively enhance the physical properties of polybutylene succinate (PBS) through a combination of chemical and physical mechanisms. The addition of potato peel fibers, which are rich in cellulose and lignin, improves the PBS by reinforcing its structure (Olumurewa et al., 2022). The hydroxyl groups on the fibers form hydrogen bonds with the ester groups of PBS, strengthening the fiber-matrix interface and improving the tensile strength and rigidity (Bajwa et al., 2019). The fibrous structure also contributes to enhanced barrier properties by creating a tortuous path for gas or moisture diffusion, whereas Van der Waals forces provide additional adhesion at the interface (Joshi et al., 2020).
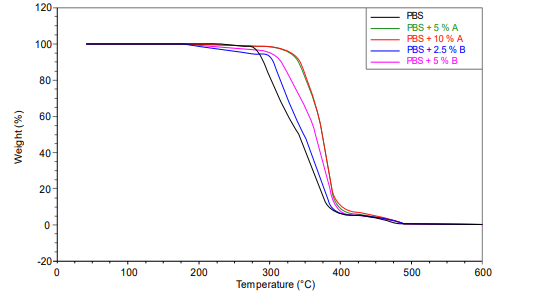
Figure 3
Thermogravimetric curves for the degradation temperature of the standard PBS (black) and PBS with stabilizers of plant origin (green, red, blue, fuchsia) for extrusion
Ferulic acid further contributed by introducing antioxidant properties and improving the thermal stability of PBS. Its phenolic hydroxyl groups interact with the polymer matrix through hydrogen bonding, whereas its aromatic rings participate in π-π stacking with the PBS chains. This dual interaction enhances the mechanical strength and resistance of the polymer to thermal degradation. Additionally, ferulic acid can act as a compatibilizer, facilitating better dispersion and phase integration when PBS is blended with other materials (Nanni et al., 2020).
Luperox, an organic peroxide, chemically modifies PBS by initiating crosslinking reactions during processing. Crosslinking improves the elasticity, tensile strength, and thermal resistance of the polymer. Peroxide action results in a denser and more interconnected polymer network, which is particularly beneficial for applications that require high durability and resistance to mechanical stress (Thirmizir et al., 2020).
Together, these additives improve PBS by leveraging mechanisms such as hydrogen bonding, Van der Waals forces, and phase compatibilization while simultaneously addressing mechanical, thermal, and oxidative stability. This multifunctional approach makes PBS composites with these additives suitable for a broader range of applications, including packaging and biodegradable materials.
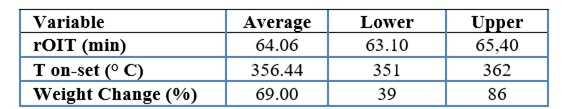
Three key variables were assessed: oxidative induction time (rOIT), thermal degradation onset temperature (T onset), and weight change (%), with the objective of identifying the most effective compositions to improve material stability and performance, as detailed in Table 4 (see Appendix 1).
The oxidative induction time (rOIT), with an average of 64 min, demonstrated good resistance to oxidative degradation, reflecting the stability of the results obtained. On the other hand, the thermal degradation onset temperature (T onset), with an average of 356 °C, indicates that the material can withstand high temperatures before degradation, confirming a significant improvement in its thermal stability. Regarding the weight loss during the degradation process, although considerable variability was observed, with an average of 69 %, this suggests that the stabilizers applied have managed to partially mitigate thermal degradation, although there are still opportunities to optimize the formulations. Together, plant-based (such as ferulic acid and potato fiber) and synthetic (such as luperox) stabilizers contribute significantly to improving the thermal and oxidative properties of PBS, thus broadening its potential for application in more demanding industrial environments.
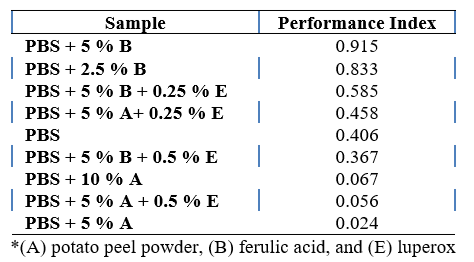
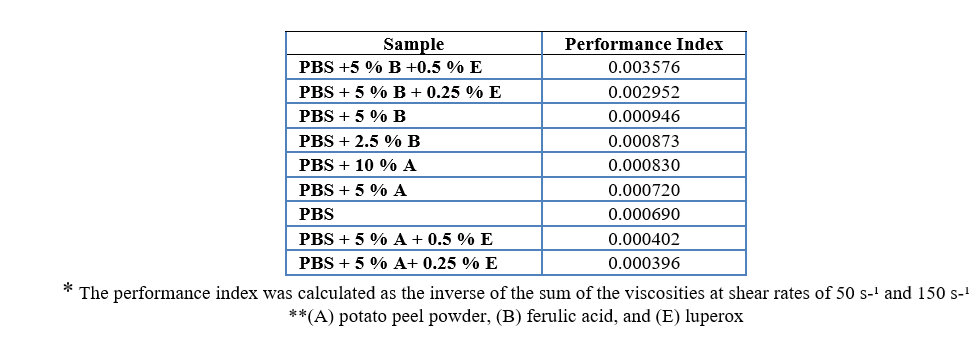
To assess overall performance, a performance index was calculated by normalizing the key variables and computing a weighted average. The PBS + 5 % ferulic acid and PBS + 2.5 % ferulic acid samples demonstrated the highest performance indices (Table 5, Appendix 2).
3.3 Rotational Rheology
3.3.1 PBS with plant stabilizers
In Figure 5 a, a Newtonian region is observed at a low shear rate between 0.001 and 1 s-1 where the molecules remain entangled and their viscosity remains high (Mezger, 2002). As the shear speed increased, it behaved like a pseudoplastic fluid, and its viscosity decreased because the molecules had less friction and were arranged linearly (Mathew et al., 2006; Syverud & Stenius, 2009; Kuo et al., 2018). This behavior has been verified with the data in Table 3 and the flow curves in Figure 5 a.
In the Newtonian region, it was observed that the viscosity was higher in the standard polymer than in the polymers with stabilizers. The addition of ferulic acid to potato peel interferes with the interactions and cohesion between the polymer molecules, resulting in a decrease in viscosity, an aspect that is related to the proportion of the stabilizer (Mattos et al., 2020). The viscosity decreased when the percentage of stabilizer in PBS was increased for both potato peels, from 5 % to 10 %, and ferulic acid, from 2.5 % to 5 %, as shown in Table 3.
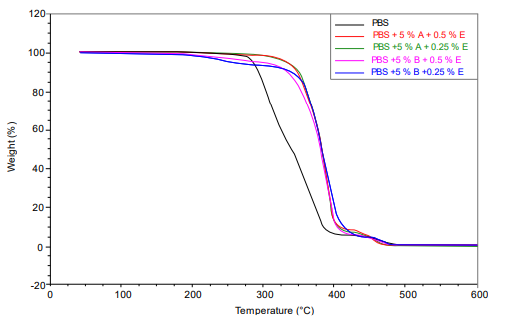
Figure 4
Thermogravimetric curves for the degradation temperature of the standard PBS (black) and PBS with stabilizers of plant and synthetic origin (red, green, fuchsia, blue) for extrusion
According to Figure 5 a, for all materials, flow curves are obtained in which shear rate values included in the extrusion range (10-1 000 s-1) are reached, which allows the evaluation of their behavior in the said (Dekker, 2005; Takeda & Takenaka, 2021).
All the materials experienced a typical drop in viscosity, and the differences in viscosity became shorter as we approached the shear ranges typical of the extrusion process. Therefore, it is not expected that there will be significant differences in the processability of the different formulations with stabilizers, with viscosities very similar or close enough to each other so that no important adjustment of the extrusion parameters is required (temperature profile, speed extrusion, etc.) to process one or another formulation interchangeably (García et al., 2020).
3.3.2 PBS with plant and synthetic stabilizers
Figure 5b shows flow curves as a function of viscosity and shear rate of PBS with a combination of stabilizers of vegetable and synthetic origin.
In Figure 5b, the flow curve of the standard PBS reaches values up to 159.6 s-1 shear rate, a different situation for samples with different combinations of different stabilizers where values lower than 100 s-1 shear rate are reached. A Newtonian region was observed at a low shear rate between 0,001 and 1 s-1, where the molecules remained entangled (higher friction) and their viscosity remained high. The addition of luperox (ditert-butyl peroxide) causes cross-linking between PBS chains, resulting in greater viscosity (Emami et al., 2002; Zapata et al., 2023). According to Fig. 5 b, the flow curve of PBS + 5 % potato fiber + 0.5 % luperox has a higher viscosity than that of standard PBS. By lowering the percentage of luperox to 0.25 %, the viscosity decreased, confirming the cross-linking of PBS. The combination of ferulic acid and luperox, as seen in the flow curve, had a lower viscosity than the standard and combined PBS because it reacted with luperox, decreasing the cross-linking of the polymer, as shown in Table 3 (Parisi et al., 2010).
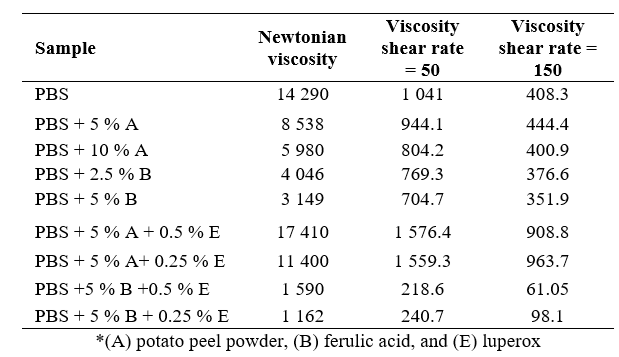
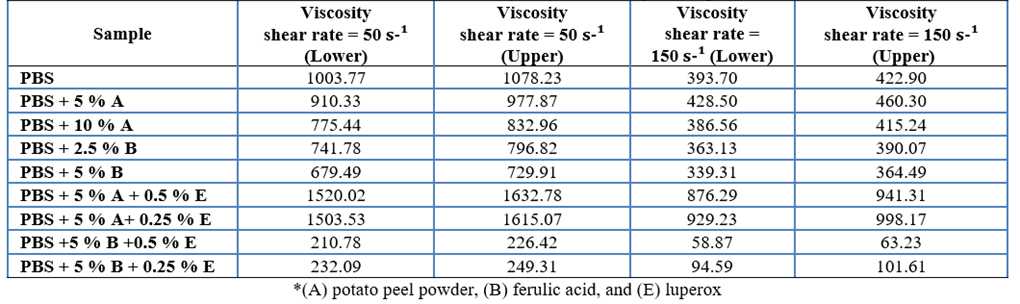
A statistical analysis of the rheological properties of Polybutylene Succinate (PBS) treated with vegetable and synthetic stabilizers was conducted. Viscosities were measured at shear rates of 50 s⁻¹ and 150 s⁻¹, with Newtonian viscosity as a reference. 95 % confidence intervals were calculated, considering a 5 % margin of error (Table 6, Appendix 3). The rheological performance index was derived from the inverse of the sum of the viscosities at both shear rates (Table 7, Appendix 4). The analysis showed that the combination of ferulic acid (B) and luperox (E) significantly reduced the PBS viscosity, enhancing its behavior under shear stress. PBS + 5 % ferulic acid + 0.5 % luperox and PBS + 5 % ferulic acid + 0.25 % luperox exhibited the best performances, indicating superior fluidity under the evaluated conditions. These findings suggest that the optimal combination of stabilizers improves both the processability and the potential for industrial applications of PBS.
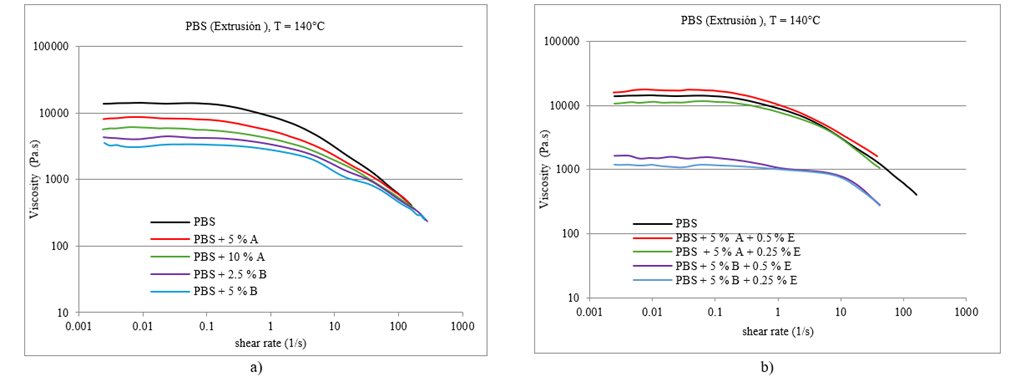
Figure 5
Flow curves of PBS formulations for extrusion a) PBS with plant-based stabilizers b) PBS with stabilizers of plant and synthetic origin
As the shear speed increased, it behaved like a pseudoplastic fluid. At high shear speeds, the chains untangle as much as possible and orient themselves parallel, reaching an optimal state of alignment. At this point, the viscosity reaches the lowest value and remains constant. It is feasible to model the behavior of the material in some extrusion processes using the Carreau model, which allows the curve to be projected at higher shear speed values that are not obtained in the rotational rheology test. In the pseudoplastic region, where information about the processability of the polymers is obtained according to Figure 6, all the samples will reach a shear speed value within the range used for extrusion processes. However, the PBS samples with ferulic acid and luperox have viscosities below 100 Pa.s, which makes their handling difficult in the extrusion process and will require temperature adjustment. The flow curve of the PBS + 5 % potato fiber + 0.25 % luperox sample, however, did not maintain the trend of the rotational rheology test when applying the projection by the Carreau model (Ladin et al., 2001).
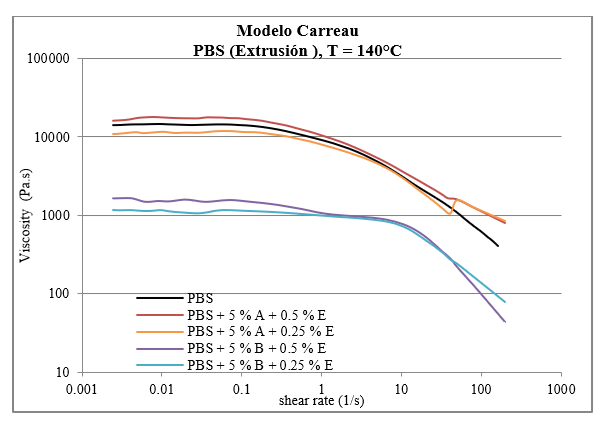
Figure 6
Flow curves projected by the Carreau model of the standard PBS, PBS with stabilizers of plant and synthetic origin for extrusion
4. CONCLUSIONS
The incorporation of natural stabilizers such as potato peel powder and ferulic acid into polybutylsuccinate (PBS) effectively enhances its thermal stability and rheological properties. The addition of ferulic acid improved the oxidation induction time and lowered the weight loss during thermal degradation compared with standard PBS. This stabilizing effect is attributed to the interaction of the phenolic hydroxyl groups with the polymer matrix through hydrogen bonding, which act as antioxidants and prevent oxidative degradation. Known for its photoprotective and thermooxidative properties, it demonstrates improved performance by increasing degradation onset temperature and maintaining the integrity of the polymer during processing. These enhancements make PBS more suitable for extrusion processes, enabling it to achieve the shear rates required for practical application in industrial settings. The combination of vegetable (potato peel and ferulic acid) and synthetic (luperox) stabilizers further enhanced thermal stability and processability of PBS. This synergy is evident in the superior oxidation induction times and reduced weight loss during degradation, particularly when ferulic acid is combined with luperox. The enhanced antioxidant activity from both natural and synthetic sources effectively protects the PBS matrix from oxidative and thermal degradation, thereby extending its usability in high-temperature environments. Additionally, rheological analysis confirmed that this combination reduced PBS viscosity within the shear rate range relevant to extrusion, improving its flow properties and making it a viable and competitive biopolymer for industrial applications.
The biopolymer polybutylsuccinate (PBS), particularly when improved with stabilizers of vegetable and synthetic origin, is versatile and can be utilized in various polymeric transformation processes. The improved thermal stability and rheological properties make it suitable for a range of applications, primarily in processes requiring high-temperature stability and good workability as extrusion and thermoforming.
REFERENCES
Abderrahim, B., Abderrahman, E., Mohamed, A., Fatima, T., Abdesselam, T., & Krim, O. (2015). Kinetic Thermal Degradation of Cellulose, Polybutylene Succinate and a Green Composite: Comparative Study. World Journal of Environmental Engineering, 3(4), 95-110. https://doi.org/10.12691/WJEE-3-4-1
Aliotta, L., Seggiani, M., Lazzeri, A., Gigante, V., & Cinelli, P. (2022). A brief review of Poly (Butylene Succinate) (PBS) and its main and applications. Polymer, 14, 844.
Averous, L., & Boquillon, N. (2004). Biocomposites based on plasticized starch: Thermal and mechanical behaviours. Carbohydrate Polymers, 56(2), 111-122. https://doi.org/10.1016/j.carbpol.2003.11.015
Bajwa, D. S., Pourhashem, G., Ullah, A. H., & Bajwa, S. G. (2019). A concise review of current lignin production, applications, products and their environmental impact. Industrial Crops and Products, 139, 111526. https://doi.org/https://doi.org/10.1016/j.indcrop.2019.111526
Barletta, M., Aversa, C., Ayyoob, M., Gisario, A., Hamad, K., Mehrpouya, M., & Vahabi, H. (2022). Poly(butylene succinate) (PBS): Materials, processing, and industrial applications. Progress in Polymer Science, 132, 101579. https://doi.org/https://doi.org/10.1016/j.progpolymsci.2022.101579
Barrino, F., De La Rosa-Ramírez, H., Schiraldi, C., López-Martínez, J., & Samper, M. D. (2023). Preparation and Characterization of New Bioplastics Based on Polybutylene Succinate (PBS). Polymers, 15(5), 1-15. https://doi.org/10.3390/polym15051212
Cecchi, T. (2021). Biocomposites from Food Waste BT - Biobased Products from Food Sector Waste: Bioplastics, Biocomposites, and Biocascading (T. Cecchi & C. De Carolis (eds.); pp. 287-310). Springer International Publishing. https://doi.org/10.1007/978-3-030-63436-0_7
Cottom, J. W., Cook, E., & Velis, C. A. (2024). A local-to-global emissions inventory of macroplastic pollution. Nature, 633(September). https://doi.org/10.1038/s41586-024-07758-6
Dekker, M. (2005). Handbook Of Polymer Synthesis.
Emami, S. H., Salovey, R., & Hogen-Esch, T. E. (2002). Peroxide-mediated crosslinking of poly(ethylene oxide). Journal of Polymer Science, Part A: Polymer Chemistry, 40(17), 3021-3026. https://doi.org/10.1002/pola.10367
Fodil Cherif, M., Trache, D., Benaliouche, F., Tarchoun, A. F., Chelouche, S., & Mezroua, A. (2020). Organosolv lignins as new stabilizers for cellulose nitrate: Thermal behavior and stability assessment. International Journal of Biological Macromolecules, 164, 794-807. https://doi.org/https://doi.org/10.1016/j.ijbiomac.2020.07.024
Fujimaki, T. (1998). Processability and properties of aliphatic polyesters, “BIONOLLE”, synthesized by polycondensation reaction. Polymer Degradation and Stability, 59(1-3), 209-214. https://doi.org/10.1016/s0141-3910(97)00220-6
García, H., Jaime-Fonseca, M., Borries-Medrano, E., & Vieyra, H. (2020). Extrusion parameters to produce a PLA-starch derived thermoplastic polymer. Revista Mexicana de Ingeniería Química, 19, 395-412.
Georgousopoulou, I.-N., Vouyiouka, S., Dole, P., & Papaspyrides, C. D. (2016). Thermo-mechanical degradation and stabilization of poly(butylene succinate). Polymer Degradation and Stability, 128, 182-192. https://doi.org/https://doi.org/10.1016/j.polymdegradstab.2016.03.012
Hiller, B. T., Azzi, J. L., & Rennert, M. (2023). Improvement of the Thermo-Oxidative Stability of Biobased Poly(butylene succinate) (PBS) Using Biogenic Wine By-Products as Sustainable Functional Fillers. Polymers, 15(11). https://doi.org/10.3390/polym15112533
Huang, C. L., Jiao, L., Zhang, J. J., Zeng, J. B., Yang, K. K., & Wang, Y. Z. (2012). Poly(butylene succinate)-poly(ethylene glycol) multiblock copolymer: Synthesis, structure, properties and shape memory performance. Polymer Chemistry, 3(3), 800-808. https://doi.org/10.1039/c2py00603k
Joshi, A., Sethi, S., Arora, B., Azizi, A. F., & Thippeswamy, B. (2020). Potato Peel Composition and Utilization BT - Potato: Nutrition and Food Security (P. Raigond, B. Singh, S. Dutt, & S. K. Chakrabarti (eds.); pp. 229-245). Springer Singapore. https://doi.org/10.1007/978-981-15-7662-1_13
Karthikumar, S., Kumar R, S., B, J., T, S., & Devi M, Y. (2024). Present trends and prospects of synthetic and bio-plasticizers. In I. Suyambulingam, D. Divakaran, S. M. Rangappa, & S. B. T.-S. F. /Plasticizers for P. C. Siengchin (Eds.), Woodhead Series in Composites Science and Engineering (pp. 211-236). Elsevier Science Ltd. https://doi.org/https://doi.org/10.1016/B978-0-443-15630-4.00009-9
Kuo, W. R., Huang, J. J., Fujimoto, N., & Lin, H. C. (2018). Physicochemical Properties of Biochar Derived from Agricultural and Forestry Processing Wastes as Cultural Medium Substance and Its Effect on Growth Quality of Vegetable Plug Seedlings. Journal of the Faculty of Agriculture, Kyushu University, 63(2), 361-370. https://doi.org/10.5109/1955655
Ladin, D., Park, C. B., Park, S. S., Naguib, H. E., & Cha, S. W. (2001). Study of Shear and Extensional Viscosities of Biodegradable PBS/CO2 Solutions. Journal of Cellular Plastics, 37(2), 109-148. https://doi.org/10.1106/72D3-9PX6-7C60-RD2X
Marek, A. A., Verney, V., Totaro, G., Sisti, L., Celli, A., Cionci, N. B., Di Gioia, D., Massacrier, L., & Leroux, F. (2020). Organo-modified LDH fillers endowing multi-functionality to bio-based poly(butylene succinate): An extended study from the laboratory to possible market. Applied Clay Science, 188, 105502. https://doi.org/https://doi.org/10.1016/j.clay.2020.105502
Martínez, L., Montes, I., Rivera, N., Díaz, I., Pérez, E., & Waldo, M. (2020). Thermal degradation of polypropylene reprocessed in a co-rotating twin-screw extruder: kinetic model and relationship between Melt Flow Index and Molecular weight. Revista Mexicana de Ingeniería Química, 395-412.
Marturano, V., Marotta, A., Agustin, S., Ambrogi, V., & Cerruti, P. (2023). Progress in Materials Science Recent advances in bio-based functional additives for polymers. Progress in Materials Science, 139(May 2022), 101186. https://doi.org/10.1016/j.pmatsci.2023.101186
Mathew, A. P., Chakraborty, A., Oksman, K., & Sain, M. (2006). The structure and mechanical properties of cellulose nanocomposites prepared by twin screw extrusion. ACS Symposium Series, 938, 114-131. https://doi.org/10.1021/bk-2006-0938.ch009
Mattos, A., Crocitti, A., & Carvalho, L. H. De. (2020). Properties of Biodegradable Films Based on.
Mezger, T. (2002). The Rheology Handbook_ For Users of Rotational and Oscillatory Rheometers (Issue October).
Miranzadeh, N., Najafi, M., & Ataeefard, M. (2024). Production of biodegradable packaging film based on PLA/starch: optimization via response surface methodology. Bulletin of Materials Science, 47(4), 273. https://doi.org/10.1007/s12034-024-03351-9
Muthuraj, R., Misra, M., & Mohanty, A. K. (2014). Biodegradable Poly(butylene succinate) and Poly(butylene adipate-co-terephthalate) Blends: Reactive Extrusion and Performance Evaluation. Journal of Polymers and the Environment, 22(3), 336-349. https://doi.org/10.1007/s10924-013-0636-5
Nanni, A., Ricci, A., Versari, A., & Messori, M. . (2020). Wine derived additives as poly(butylene succinate) (PBS) natural stabilizers for different degradative environments. Polymer Degradation and Stability, 182, 109381. https://doi.org/https://doi.org/10.1016/j.polymdegradstab.2020.109381
Olumurewa, J. A. V., Tobi, A., & Umuze, K. O. (2022). The use of agricultural wastes as biopolymers for food packaging. Regional Food Science and Technology, June.
Pacheco, M. P., Gómez, O. R. T., Escamilla, G. C., Aranda, S. D., & Velázquez, M. G. N. (2022). Obtaining and characterization of bioplastics based on potato starch, aloe, and graphene. Polimeros, 32(4). https://doi.org/10.1590/0104-1428.20220084
Papadopoulou, K., Klonos, P. A., Kyritsis, A., Mašek, O., Wurzer, C., Tsachouridis, K., Anastasiou, A. D., & Bikiaris, D. N. (2023). Synthesis and Study of Fully Biodegradable Composites Based on Poly(butylene succinate) and Biochar. Polymers, 15(4). https://doi.org/10.3390/polym15041049
Parisi, O. I., Puoci, F., Iemma, F., De Luca, G., Curcio, M., Cirillo, G., Spizzirri, U. G., & Picci, N. (2010). Antioxidant and spectroscopic studies of crosslinked polymers synthesized by grafting polymerization of ferulic acid. Polymers for Advanced Technologies, 21(11), 774-779. https://doi.org/https://doi.org/10.1002/pat.1499
Rafiqah, S. A., Khalina, A., Harmaen, A. S., Tawakkal, I. A., Zaman, K., Asim, M., Nurrazi, M. N., & Lee, C. H. (2021). A review on properties and application of bio‐based poly(Butylene succinate). Polymers, 13(9), 1-28. https://doi.org/10.3390/polym13091436
Rajgond, V., Mohite, A., More, N., & More, A. (2024). Biodegradable polyester-polybutylene succinate (PBS): a review. Polymer Bulletin, 81(7), 5703-5752. https://doi.org/10.1007/s00289-023-04998-w
Reano, A. F., Domenek, S., Pernes, M., Beaugrand, J., & Allais, F. (2016). Ferulic Acid-Based Bis/Trisphenols as Renewable Antioxidants for Polypropylene and Poly(butylene succinate). ACS Sustainable Chemistry and Engineering, 4(12), 6562-6571. https://doi.org/10.1021/acssuschemeng.6b01429
Righetti, M. C., Cinelli, P., Mallegni, N., Massa, C. A., Aliotta, L., & Lazzeri, A. (2019). Thermal, mechanical, viscoelastic and morphological properties of poly(lactic acid) based biocomposites with potato pulp powder treated with waxes. Materials, 12(6). https://doi.org/10.3390/ma12060990
Shekhar, N., & Mondal, A. (2024). Synthesis, properties, environmental degradation, processing, and applications of Polylactic Acid (PLA): an overview. Polymer Bulletin, 81(13), 11421-11457. https://doi.org/10.1007/s00289-024-05252-7
Sohn, Y. J., Kim, H. T., Baritugo, K. A., Jo, S. Y., Song, H. M., Park, S. Y., Park, S. K., Pyo, J., Cha, H. G., Kim, H., Na, J. G., Park, C., Choi, J. Il, Joo, J. C., & Park, S. J. (2020). Recent Advances in Sustainable Plastic Upcycling and Biopolymers. Biotechnology Journal, 15(6), 1-16. https://doi.org/10.1002/biot.201900489
Syverud, K., & Stenius, P. (2009). Strength and barrier properties of MFC films. Cellulose, 16(1), 75-85. https://doi.org/10.1007/s10570-008-9244-2
Takeda, E., & Takenaka, K. (2021). Polymer Extrusion. In Seikei-Kakou (Vol. 33, Issue 7). https://doi.org/10.4325/seikeikakou.33.222
Thirmizir, M. Z. A., Ishak, Z. A. M., & Salim, M. S. (2020). Compatibilization and Crosslinking in Biodegradable Thermoplastic Polyester Blends BT - Reactive and Functional Polymers Volume Two: Modification Reactions, Compatibility and Blends (T. J. Gutiérrez (ed.); pp. 23-89). Springer International Publishing. https://doi.org/10.1007/978-3-030-45135-6_3
Wang, X., Zhou, J., & Li, L. (2007). Multiple melting behavior of poly(butylene succinate). European Polymer Journal, 43(8), 3163-3170. https://doi.org/10.1016/j.eurpolymj.2007.05.013
Wiesinger, H., Wang, Z., & Hellweg, S. (2021). Deep Dive into Plastic Monomers , Additives , and Processing Aids. https://doi.org/10.1021/acs.est.1c00976
Xu, J., & Guo, B. H. (2010). Poly(butylene succinate) and its copolymers: Research, development and industrialization. Biotechnology Journal, 5(11), 1149-1163. https://doi.org/10.1002/biot.201000136
Zapata, R. C., Ramírez, D. P., Pérez, J. J. C., Rosales, H. D., Díaz, B. B., & Adame, J. A. A. (2023). Mechanical and thermal evaluation of PLA/PLA-g-MA/MCC composites via reactive extrusion. MRS Advances, 8(23), 1325-1329. https://doi.org/10.1557/s43580-023-00683-2
Zhang, Y., Yuan, W., & Liu, Y. (2018). Synthesis and characterization of bio-based poly(butylene succinate-co-10-hydroxydecanoate). Journal of Elastomers and Plastics, 50(4), 325-338. https://doi.org/10.1177/0095244317723181
APPENDIX 1
APPENDIX 2
APPENDIX 3
APPENDIX 4
Notes
Notes
Notes
Notes
Author notes
*luissantiago.carrera@espoch.edu.ec
