Abstract:
Introduction: Although prevention of the disease and its spread is the primary goal in the fight against the pandemic, studies on the correct management of those who have the disease and the predictability of the prognosis are also important. This study aimed to determine whether lymphocyte-C-reactive protein ratio, together with other inflammation markers, would be useful in predicting intensive care unit admission and mortality in Coronavirus disease 2019 cases.
Material and methods: 883 patients were followed in 758 wards and 125 intensive care units. Data of the patients included in the study were compared with those admitted to the service and intensive care unit, and with those who survived and developed mortality.
Results: According to the receiver operating characteristic analysis to distinguish the patients followed in the intensive care unit from the patients hospitalized in the ward that was determined that lymphocyte-C-reactive protein ratio, C-reactive protein ratio, CRP-albumin ratio, and neutrophil-lymphocyte ratio were moderate (70%–80%). D-dimer was good (80%–90%) predicting follow-up in intensive care unit. Increase in age, increase in lactate dehydrogenase and interleukin-6 levels, and uptake in tomography were determined as independent risk factors that increase intensive care unit admission. 243 (27.5%) of the patients were mortal. The mean age of the patients with a mortal course was 70±14 years, and mortality increased with increasing age. In the receiver operating characteristic analysis of patients with a mortal course that was determined that lymphocyte-C-reactive protein ratio, neutrophil-lymphocyte ratio, and D-dimer had a good (80–90%) ability to distinguish patients with a mortal course. Age, fever, and increases in lactate dehydrogenase and interleukin-6 levels were determined to be independent risk factors increasing mortality.
Conclusions: Low lymphocyte-C-reactive protein ratio and high D-Dimer, neutrophil-lymphocyte ratio, and CRP-albumin ratio can be used in clinical monitoring to reduce morbidity and mortality rates due to COVID-19.
Keywords: COVID-19, Biomarker, Lymphocite to CRP ratio, Neutrophil to lymphocite ratio, D-dimer.
Resumen:
Introducción: Si bien la prevención de la enfermedad y su propagación es el objetivo principal en la lucha contra la pandemia, también son importantes los estudios sobre el correcto manejo de los pacientes con la enfermedad y la previsibilidad del pronóstico. El objetivo de este estudio fue determinar si la proporción de linfocitos y proteína C reactiva, junto con otros marcadores inflamatorios, sería útil para predecir el ingreso a la unidad de cuidados intensivos y la mortalidad en casos de enfermedad por coronavirus en 2019.
Material y métodos: Se siguieron 883 pacientes en 758 salas y 125 unidades de cuidados intensivos. Los datos de los pacientes incluidos en el estudio se compararon con los ingresados en el servicio y unidad de cuidados intensivos, y con los que sobrevivieron y desarrollaron mortalidad.
Resultados: Según el análisis de las características operativas del receptor para distinguir los pacientes seguidos en la unidad de cuidados intensivos de los pacientes hospitalizados en el servicio, se encontró que la relación linfocitos a proteína C reactiva, proteína C reactiva a albúmina y neutrófilos a la proporción de linfocitos fue moderada y el dímero D fue bueno para predecir el seguimiento en la unidad de cuidados intensivos. Se encontró que el aumento de la edad, el aumento de los niveles de lactato deshidrogenasa e interleucina-6 y la captación en la tomografía eran factores de riesgo independientes para el ingreso a la unidad de cuidados intensivos. 243 (27,5%) de los pacientes fallecieron. En el análisis de las características operativas del receptor de pacientes con un desenlace fatal, se encontró que la proporción de linfocitos a proteína C reactiva, la proporción de neutrófilos a linfocitos y el dímero D tenían una buena capacidad para discriminar a los pacientes con un desenlace fatal. Se encontró que la edad, la fiebre y los niveles elevados de lactato deshidrogenasa e interleucina-6 eran factores de riesgo independientes para una mayor mortalidad.
Conclusiones: Una proporción baja de linfocitos a proteína C reactiva y una proporción alta de dímero D, neutrófilos a linfocitos y PCR a albúmina se pueden utilizar en el seguimiento clínico para reducir las tasas de morbilidad y mortalidad debido a COVID-19.
Palabras clave: COVID-19, Biomarcador, Ratio linfocito-PCR, Ratio neutrófilo-linfocito, Dímero D.
Original article
The Value of Lymphocyte-CRP Ratio Predicting the Prognosis in COVID-19 Patients
El valor de la relación linfocito-PCR para predecir el pronóstico en pacientes con COVID-19
Hospital San Pedro

Esta obra está bajo unaLicencia Creative Commons Atribución 4.0 Internacional.
Received: 30 June 2023
Corrected: 26 July 2023
Accepted: 23 August 2023
Coronavirus disease 2019 (COVID-19), which began in China in December 2019 and caused a pandemic, has caused a huge loss of life around the world. As of 16 February 2023, it is reported that approximately 756 million people have been infected and 6.8 million people have died worldwide. In Turkey, it is reported that 17 million people have been infected, and 101 thousand people have died [1]. The coronavirus has been a cause of mortality and morbidity, especially in the elderly and those with comorbidities, by causing multi-organ failure, manifested by laboratory and clinical dysfunction in various systems. However, it is difficult to predict the course and severity of the COVID-19 infection during hospital admission. Rapid assessment of the severity of the disease and the need for intensive care in the early stages of the disease is important for the course of the disease. Prognostic predictors are of great importance for the rapid response and optimal use of the healthcare system during the ongoing COVID-19 pandemic. The importance of having objective data to predict ICU admissions and mortality has increased even more during the pandemic period. Although the primary goal is to prevent the disease and stop its spread during the pandemic, studies on the correct management of patients and the predictability of the prognosis are also important [2].
The S1 subgroup of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) surface spike protein stimulates the production of angiotensin 1 by binding to the angiotensin-converting enzyme 2 (ACE2) receptor. Angiotensin converting enzyme (ACE) then converts angiotensin 1 to angiotensin 2, which binds to the angiotensin receptor and increases endothelin-1 levels. A diffuse inflammatory response occurs with the release of pro-inflammatory cytokines and interleukins. Large scale unregulated production of interleukin, particularly interleukin-6 (IL-6), stimulates multiple inflammatory pathways. It increases the production of acute phase reactants such as C-reactive protein (CRP) and the mobilization of neutrophils. This explains the relative lymphopenia seen in severe manifestations of SARS-CoV-2 infection, possibly with stress-induced neutrophilia [3, 4].
Typically, the lymphocyte to CRP ratio (LCR) is used as a prognostic marker for several types of cancer, including colorectal and gastric cancer [5, 6]. As hyperinflammation is known to be the main cause of poor prognosis in COVID-19 disease, combinations of biomarkers reflecting the inflammatory state may be a good alternative in this regard [7]. Since SARS-CoV-2 is highly correlated with high viral load, low lymphocyte count and high CRP value, we thought that low LCR might help predict disease severity. Therefore, the aim of this study was to determine whether LCR, together with other markers of inflammation, would be useful in predicting ICU admission and mortality in COVID 19 cases.
In our study, the information of 955 patients over 18 years of age who were hospitalised for more than three days in the pandemic service or intensive care units between 01-06-2020 and 28-02-2022 and who had a positive COVID-19 reverse transcription-polymerase chain reaction (COVID RT-PCR) test was scanned. 883 patients with complete information in the database were included in the study. The data of the patients included in the study were compared with those in the service and intensive care unit (ICU) group who survived and developed mortality.
After hospitalisation, blood tests including prognostic factors for COVID-19 disease (such as haemogram, alanine aminotransferase (ALT), aspartate aminotransferase (AST), urea, creatinine, D-dimer, ferritin, fibrinogen, CRP) are routinely checked. The routine tests studied and the data to be analysed were retrieved retrospectively from the hospital information system. Patient demographics, underlying diseases, ICU admission, and clinical characteristics such as need for mechanical ventilation, laboratory findings, treatments and outcomes were obtained from the hospital electronic database system.
The ethics committee of a tertiary education and research hospital approved the study (ethics committee decision no: 2/14, date: 20/01/2022).
Patient data collected in the study were analysed using the IBM Statistical Package for the Social Sciences (SPSS) for Windows 23.0 (IBM Corp., Armonk, NY). Frequency and percentage for categorical data and, mean and standard deviation for continuous data were used as descriptive values. "ANOVA Test" was used for comparisons between groups, and "Chi-square or Fisher's Exact Test" was used for comparison of categorical variables. The Post-Hoc Tukey test was used to determine which groups caused the significant difference in the variables that showed a significant difference because of the ANOVA test. ROC analysis was performed and the ROC curve was drawn for the parameters thought to have a distinctive effect on ICU admission and survival. Logistic regression analysis was also used to identify risk factors for ICU admission and survival. Results were considered statistically significant if the p-value was less than 0.05.
883 patients were evaluated in the study, of whom 758 were patients followed in the ward and 125 in the ICU. The mean age of the patients was 59±17 years, and 58.1% (n=513) were male. The mean age of the patients followed in the ICU was 69±15 years, and ICU admission was significantly higher in patients over 65 years of age. There was no difference in terms of ICU admission according to gender. The most common comorbidities in our patients were hypertension (HT), diabetes mellitus (DM), renal failure and malignancy. Patients with DM, chronic kidney disease, heart failure, peripheral arterial disease (PAH), cerebrovascular accident (CVO), chronic obstructive pulmonary disease (COPD) and solid tumours were more likely to be admitted to the ICU. Patients in the ICU had significantly lower saturations on first admission than those in the service. Patients who developed acute respiratory distress syndrome (ARDS), pulmonary thromboembolism (PTE), and stroke because of COVID-19 complications were significantly more likely to be followed in the ICU (Table 1). Creatinine, ALT, AST, lactate dehydrogenase (LDH), CRP, leukocyte count, lymphocyte count, D-dimer, ferritin, IL-6, neutrophil to lymphocyte ratio (NLR), C-reactive protein to albumin ratio (CAR), platelet to lymphocyte ratio (PLR) were significantly higher in patients followed in the ICU than in those followed in the service (p<0.05). LCR, haemoglobin and albumin levels were significantly lower in ICU patients (p<0.05) (Table 2).
Comparison of demographic data of the patients in the service or Intensive Care Unit (ICU)
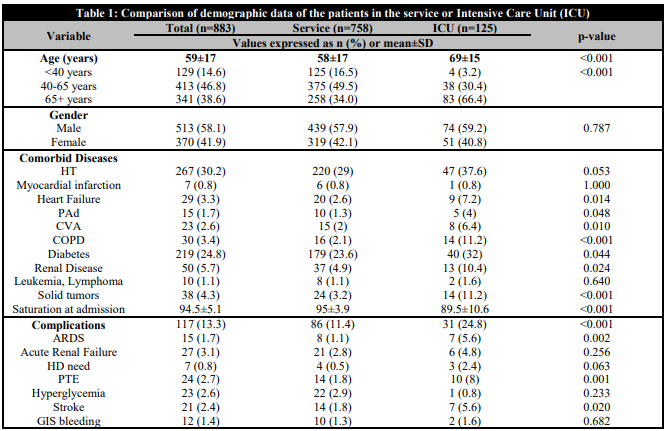
HT: hypertension; Pad: peripheral artery disease; CVA: cerebrovascular accident; COPD: chronic obstructive pulmonary disease; ARDS: acute respiratory distress syndrome; HD: hemodialysis; PTE: pulmonary thromboembolism; GIS: gastrointestinal system.
Comparison of the laboratory findings of the patients in the service or Intensive Care Unit (ICU)
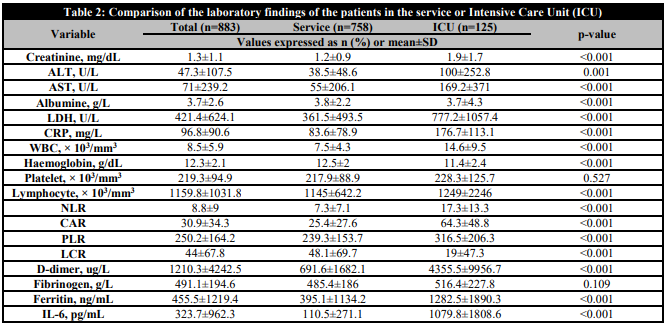
ALT: alanine aminotransferase; AST: aspartate aminotransferase; LDH: lactate dehydrogenase; CRP: C-reactive protein; WBC: white blood count; NLR: neutrophil lymphocyte ratio; CAR: CRP albumin ratio; PLR: platelet to lymphocyte ratio; LCR: lymphocyte to CRP ratio); IL-6: interleukin 6.
Table 3 shows the results of the ROC analysis performed to examine the differential effect of CRP, D-dimer, NLR, PLR, CAR, and LCR in discriminating patients followed in the ICU from those admitted to the ward. Using a cut-off value of <8.87 for LCR, the area under the curve was 74.3%. For other parameters, the area under the curve and cut-off values were respectively; 75.5% and 117 for CRP, 83% and 360 for D-dimer, 79.7% and 8.9 for NLR, 60.9% and 300.9 for PLR, 76.3% and 46.3% for CAR. The area under the curve shows the statistical significance of the discriminatory ability of the diagnostic test. In our study, LCR, CRP, CAR, NLR were found to be moderate (70-80%), PLR weak (60-70%) and D-dimer good (80-90%) tests for predicting ICU follow-up.
ROC Analysis Result of Laboratory Parameters of Intensive Care Unit admission
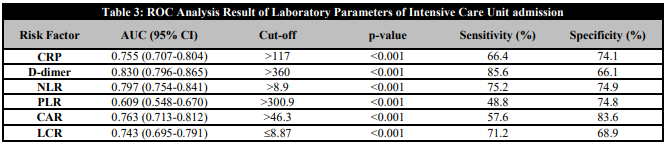
CRP: C-reactive protein; NLR: neutrophil lymphocyte ratio; CAR: CRP albumin ratio; PLR: platelet to lymphocyte ratio; LCR: lymphocyte to CRP ratio).
The distribution of risk factors influencing ICU admission of the patients in the study is shown in Table 4. Age, presence of comorbidities, cough, dyspnoea, ALT, LDH, CRP, leukocyte count, D-dimer, IL-6, NLR, PLR and CAR increase and LCR decrease and CT scan uptake were found to increase ICU admission in univariate analysis. The presence of comorbidity in the patients increased ICU admission 2.18 fold, shortness of breath 1.65 fold, and increased creatinine levels 1.45 fold. When the parameters that showed a significant difference in the univariate analysis were re-evaluated in the multivariate analysis, increasing age, increasing LDH, increasing IL-6 levels and uptake on tomography were identified as independent risk factors for increasing ICU admission.
Evaluation of Risk Factors Affecting Intensive Care Unit Admission
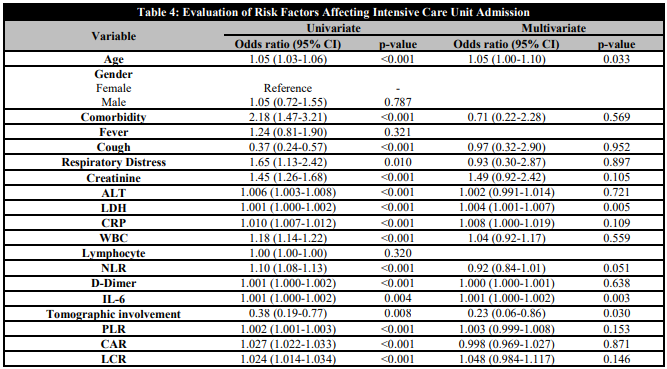
ALT: alanine aminotransferase; LDH: lactate dehydrogenase; CRP: C-reactive protein; WBC: white blood count; NLR: neutrophil lymphocyte ratio; CAR: CRP albumin ratio; PLR: platelet to lymphocyte ratio; LCR: lymphocyte to CRP ratio); IL-6: interleukin 6.
Of the 883 patients included in the study, 640 survived and 243 (27.5%) died. The mean age of patients with a mortal outcome was 70±14 years, and it was observed that mortality increased with increasing age, especially in patients over 65 years of age, mortality was significantly higher than in other age groups (p<0.05). There was no difference in mortality according to sex. Patients with DM, chronic kidney disease (CKD), heart failure, PAH, CVO, COPD and solid tumours had higher mortality. Mortality was significantly higher in patients with low oxygen saturation on hospital admission. Mortality was significantly higher in patients with complications, and mortality was significantly higher in patients who developed acute renal failure, ARDS, PTE, and stroke (Table 5). The levels of creatinine, ALT, AST, LDH, CRP, leukocyte count, D-dimer, fibrinogen, ferritin, IL-6, NLR, CAR, PLR were significantly higher in the patients who survived (p<0.05). LCR, haemoglobin and albumin levels were significantly lower (p<0.05) (Table 6).
Comparison of demographic data of patients who survived and exitus
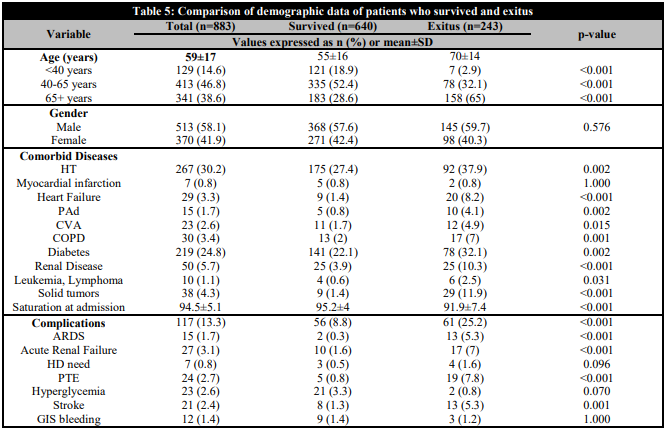
HT: hypertension; Pad: peripheral artery disease; CVA: cerebrovascular accident; COPD: chronic obstructive pulmonary disease; ARDS: acute respiratory distress syndrome; HD: hemodialysis; PTE: pulmonary thromboembolism; GIS: gastrointestinal system.
Comparison of Laboratory Data According to Survival and Exitus of Patients
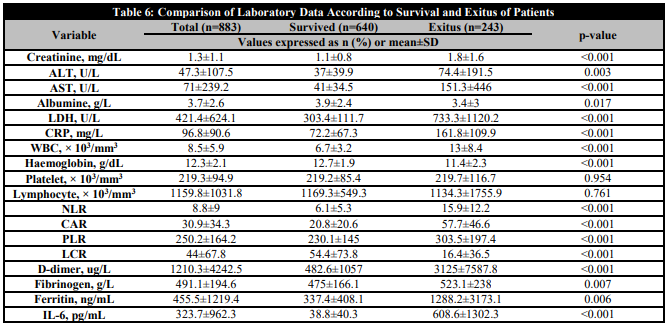
ALT: alanine aminotransferase; AST: aspartate aminotransferase; LDH: lactate dehydrogenase; CRP: C-reactive protein; WBC: white blood count; NLR: neutrophil lymphocyte ratio; CAR: CRP albumin ratio; PLR: platelet to lymphocyte ratio; LCR: lymphocyte to CRP ratio); IL-6: interleukin 6.
Table 7 shows the results of the ROC analysis to examine the differential effect of CRP, D-dimer, NLR, PLR, CAR and LCR in separating the patients who died from those who survived. The area under the curve for LCR was 80.6%, while the cut-off value was <8.83. For other parameters, the area under the curve and cut-off values were 76.4% and 116.5% for CRP, 82.9% and 458 for D-dimer, 81.6% and 8.9% for NLR, 61.3% and 210.9 for PLR, 78% and 37.1% for CAR. It was found that it was able to discriminate patients with a mortal course, with weak PLR (60-70%), CRP and CAR moderate (70-80%), LCR, NLR and D-dimer at a good level (80-90%).
ROC Analysis Result of Laboratory Data Affecting Mortality
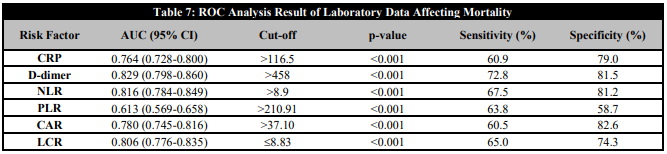
CRP: C-reactive protein; NLR: neutrophil lymphocyte ratio; CAR: CRP albumin ratio; PLR: platelet to lymphocyte ratio; LCR: lymphocyte to CRP ratio).
An examination of the risk factors affecting mortality in the patients included in the study is shown in Table 8. In univariate analysis, age, presence of additional diseases, presence of fever, cough and shortness of breath, creatinine, ALT, LDH, CRP, leukocyte count, D-dimer and IL-6 elevation, uptake on tomography, increase in NLR, PLR and CAR, and decrease in LCR were identified as factors increasing mortality. The presence of comorbidity in patients increased mortality 2.53 times, fever 2.27 times, shortness of breath 2.23 times, and the increase in creatinine values 1.45 times. When the parameters that showed a significant difference in univariate analysis were re-evaluated in multivariate analysis, age, fever, increase in LDH and IL-6 levels were identified as independent risk factors increasing mortality.
Evaluation of Risk Factors Affecting Mortality
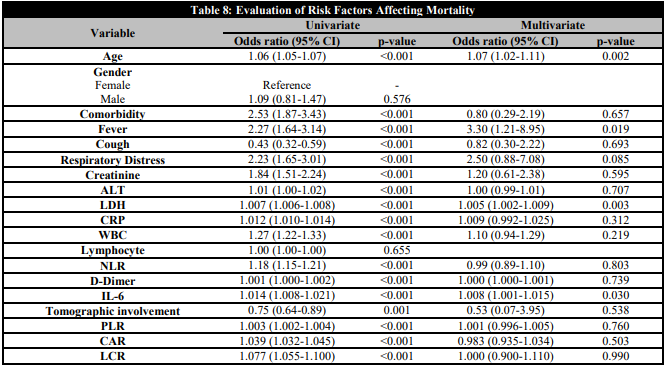
ALT: alanine aminotransferase; LDH: lactate dehydrogenase; CRP: C-reactive protein; WBC: white blood count; NLR: neutrophil lymphocyte ratio; CAR: CRP albumin ratio; PLR: platelet to lymphocyte ratio; LCR: lymphocyte to CRP ratio); IL-6: interleukin 6.
In the COVID-19 pandemic, a large number of patients required intensive care and a significant proportion of them died [8]. Optimal care of patients during the pandemic period requires early diagnosis of patients at risk of severe illness and identification of patients who may require ICU admission and mechanical ventilation. In order to prioritise these patients and use resources efficiently, the clinical determinants of disease progression need to be well understood. This is particularly important when considering which patients should receive early and effective treatment. This will help improve patient prognosis and limit the devastating impact of the epidemic.
Although the lung is the main target organ in COVID-19 disease, COVID-19 is now recognised as a multi-system infection involving the haematological and immunological systems. As the prognosis of COVID-19 disease depends on the response of the haematological system, it is not surprising to see changes in routine blood tests. Some parameters can be used to differentiate disease severity [9, 10]. SARS-CoV-2 has been shown to cause lymphopenia by directly causing cytotoxic-mediated destruction of circulating lymphocytes [7, 11]. As CRP levels have been shown to be elevated prior to the onset of lymphopenia, the LCR may be sensitive to detect the early part of the inflammatory cascade. Therefore, a low LCR representing systemic inflammation may be a marker for in-hospital complications and mortality [12]. In our study, we tested the comparison of LCR with other COVID markers in a large data pool of patients in our hospital cohort, its value in predicting ICU admission and mortality, and its compatibility with the literature.
A meta-analysis of 16 studies reported that 251 of 1832 patients died and the crude mortality rate was 13.7% [13]. The mortality rate in hospitalized patients reported by Zhou et al. was 28.3% [14]. There are studies in which the mortality rate can reach 61.5% in critically ill patients with COVID-19 [15]. Data from China and Italy showed that COVID-19 mortality occurs mostly in adults aged 60 years and older [16]. In our study, in accordance with the literature, the mortality rate increased with increasing age, and it was significantly higher than in other age groups, especially over 65 years of age, with a mortality rate of 46.3% in the group of patients over 65 years of age. In our study of hospitalised patients, the mortality rate was 27.5%, which is in line with the literature. Age is an independent predictor of both ICU stay and mortality. The reason for these different mortality rates may be due to the different sample sizes and case inclusion criteria used in the studies. Previous studies have shown that decreased immunity and underlying comorbidities in elderly patients may have an impact on poor prognosis [17, 18].
Many researchers have pointed out that COVID-19 infections are more severe and have a high mortality rate in people with comorbidities such as hypertension, cardiovascular disease, obesity, diabetes, and cancer. Other predisposing conditions include autoimmune disease, chronic kidney disease, and chronic lung disease such as asthma, neurological conditions such as dementia, liver disease, Down syndrome and alcohol use [19, 20, 21]. The most common comorbidities among the patients in our study were HT, DM, renal failure and malignancy. It was observed that patients with DM, CKD, heart failure, PAH, SVO, COPD and solid tumours were more likely to be admitted to the ICU and these patients had significantly higher mortality. Consistent with the literature, the presence of comoborditis alone was associated with a 2.18-fold increase in ICU admission and a 2.53-fold increase in mortality.
When Xie et al. examined the effect of hypoxaemia on mortality in COVID-19 patients; they reported a strong association between hypoxaemia and worse clinical outcomes. As patients' hypoxaemia worsened, the length of stay in intensive care and mortality increased. They found that dyspnoea was also independently associated with mortality in multivariate analyses [22]. In our study, we observed that patients with high hypoxaemia on the first day of hospitalisation had significantly longer ICU follow-up and were more likely to die. Similarly, we found that the presence of dyspnea increased ICU admission 1.65 times and mortality 2.23 times.
In a meta-analysis of 32 studies with 10,491 confirmed COVID-19 patients, lymphopenia, thrombocytopenia, and elevated D-dimer, CRP, PCT, creatinine kinase, AST, ALT, creatinine, and LDH were associated with worse clinical risk [23]. In another study conducted in our country, AST, ALT, LDH, ferritin, INR and d-dimer levels were found to help predict disease severity in COVID-19 [24]. Yang et al. reported that age, WBC, NLR, LMR (lymphocyte-monocyte ratio), PLR, CRP and d-NLR (derived NLR ratio) ratios were significantly higher in severe patients than in other patients, while the lymphocyte count was significantly lower [24]. Wang et al. reported that CAR was significantly higher in severe COVID-19 patients than in the non-severe group. Multivariate regression analyses also showed that CAR is an independent risk factor for COVID-19 severity [25]. Consistent with the literature, in our study, creatinine, ALT, AST, LDH, CRP, leukocyte count, D-dimer, ferritin, IL-6, NLR, CAR, PLR levels were significantly higher in patients followed up in the ICU and in patients with a fatal outcome (p< 0.05). LCR, haemoglobin and albumin levels were significantly lower in ICU patients (p<0.05). LCR, CRP, CAR and NLR were found to be moderate (70-80%) and D-dimer good (80-90%) predictors of ICU stay. Patients with a fatal outcome were found to have poor PLR (60-70%), moderate CRP and CAR (70-80%) and good (80-90%) LCR, NLR and D-dimer. In univariate analysis, age, presence of comorbidity, cough, shortness of breath, ALT, LDH, CRP, WBC, D-dimer, IL-6 elevation, NLR, PLR and CAR increase, CT uptake and decrease in LCR were found to be significant in predicting ICU admission and mortality.
Jemaa et al. found a higher CLR in ward patients compared with healthy controls. They also observed a significant increase in CLR in patients who were followed up in the ICU, when comparing ward and ICU patients. Importantly, this study observed that the CLR remained high in the group that died, whereas these values returned to normal more quickly in the surviving patients [26]. Similar results have been documented in other studies showing that the LCR can discriminate between COVID-19 infected patients of different severity (mild/moderate, severe and critically ill) [27, 28]. In a cohort of 84 patients, Turan et al. investigated the relationship between the rates of various biomarkers and COVID-19 severity, mortality and need for intensive care. The LCR measures disease severity (AUC = 0.766, p < 0.001, sensitivity 89.29%, specificity 53.57%), mortality (AUC = 0.696, p = 0.029, sensitivity 45.45%, specificity 90.41%), and ICU need (AUC = 0.746, p < 0.001, sensitivity 92.31%, specificity 49.30%) [29]. Miao Yang et al. found similar results in a cohort of 108 patients [30]. In our study, in agreement with the literature, LCR predicted ICU follow-up (AUC = 0.743, p < 0.001, sensitivity 71.2%, specificity 68.9%) and mortality (AUC = 0.806, p < 0.001, sensitivity 65%, specificity 74.3%). The LCR was significantly lower in the intensive care unit group and in patients with a fatal outcome.
Both NLR and LCR are used to highlight a relative lymphopenia compared with elevated neutrophil counts and CRP levels. The LCR is more sensitive in detecting the early part of the inflammatory cascade, as CRP levels have been shown to rise before neutrophilia or lymphopenia occurs. Based on this information, we thought that LCR would be a better marker than both NLR and CRP. However, in our study, NLR and CRP were better at predicting both ICU follow-up and mortality. In the study by Turan et al, similar to our study, NLR and CRP were better at predicting poor clinical outcome and mortality [29]. We attribute this to the fact that the data of our patient population were obtained from the examination of patients on the day of admission with clinical deterioration, and not at the early stage of the disease when the diagnosis of COVID-19 was made.
It is also important to note that although a high NLR is known to be associated with the severity of COVID-19-related complications, its use may be limited in certain situations. For example, patients on high-dose steroids may have a falsely elevated NLR due to neutrophilia. Similarly, the NLR may be falsely suppressed in immunocompromised patients with neutropenia due to bone marrow infiltration from chemotherapy or primary malignancy. In these cases, the LCR may be a reliable marker for estimating disease severity, as it is not affected by the above confounding factors. However, both LCR and NLR values should not be used as absolute indicators and require interpretation in the clinical context. Early prediction of in-hospital complications can help in the timely and effective allocation of available resources. Physicians can take therapeutic measures in patients with increased NLR and decreased LCR. As all of the data in our study were obtained from the blood tests taken on the first day of hospitalisation, before the patient received steroid treatment, there was no steroid-induced false elevation of NLR [30].
Some limitations of our study should be mentioned. This study was a single-centre, retrospective study. Thus, the possibility of missing asymptomatic or mildly affected patients, the inability to monitor parameter changes in patients with rapid clinical exacerbation based on a single blood result, and the uncertainty of the effect of patient comorbidity and treatment on blood results were the limitations of the study.
In conclusion, low LCR and high D-dimer, NLR and CAR can be used in clinical monitoring to reduce morbidity and mortality rates due to COVID-19. In patients with low LCR or high NLR, early supportive treatment can be applied to improve a better prognosis and reduce mortality and costs. The ease of measurement and low cost of these markers may increase their utility. Despite the large number of patients here, prospective studies with larger patient series are needed to more clearly reveal the relationship between LCR, NLR, CAR and D-dimer levels, and the presence of COVID-19 and disease severity.
The authors have no conflict of interest to declare. The authors declared that this study has received no financial support.
alpertahmaz@hotmail.com
Comparison of demographic data of the patients in the service or Intensive Care Unit (ICU)

HT: hypertension; Pad: peripheral artery disease; CVA: cerebrovascular accident; COPD: chronic obstructive pulmonary disease; ARDS: acute respiratory distress syndrome; HD: hemodialysis; PTE: pulmonary thromboembolism; GIS: gastrointestinal system.
Comparison of the laboratory findings of the patients in the service or Intensive Care Unit (ICU)

ALT: alanine aminotransferase; AST: aspartate aminotransferase; LDH: lactate dehydrogenase; CRP: C-reactive protein; WBC: white blood count; NLR: neutrophil lymphocyte ratio; CAR: CRP albumin ratio; PLR: platelet to lymphocyte ratio; LCR: lymphocyte to CRP ratio); IL-6: interleukin 6.
ROC Analysis Result of Laboratory Parameters of Intensive Care Unit admission

CRP: C-reactive protein; NLR: neutrophil lymphocyte ratio; CAR: CRP albumin ratio; PLR: platelet to lymphocyte ratio; LCR: lymphocyte to CRP ratio).
Evaluation of Risk Factors Affecting Intensive Care Unit Admission

ALT: alanine aminotransferase; LDH: lactate dehydrogenase; CRP: C-reactive protein; WBC: white blood count; NLR: neutrophil lymphocyte ratio; CAR: CRP albumin ratio; PLR: platelet to lymphocyte ratio; LCR: lymphocyte to CRP ratio); IL-6: interleukin 6.
Comparison of demographic data of patients who survived and exitus

HT: hypertension; Pad: peripheral artery disease; CVA: cerebrovascular accident; COPD: chronic obstructive pulmonary disease; ARDS: acute respiratory distress syndrome; HD: hemodialysis; PTE: pulmonary thromboembolism; GIS: gastrointestinal system.
Comparison of Laboratory Data According to Survival and Exitus of Patients

ALT: alanine aminotransferase; AST: aspartate aminotransferase; LDH: lactate dehydrogenase; CRP: C-reactive protein; WBC: white blood count; NLR: neutrophil lymphocyte ratio; CAR: CRP albumin ratio; PLR: platelet to lymphocyte ratio; LCR: lymphocyte to CRP ratio); IL-6: interleukin 6.
ROC Analysis Result of Laboratory Data Affecting Mortality

CRP: C-reactive protein; NLR: neutrophil lymphocyte ratio; CAR: CRP albumin ratio; PLR: platelet to lymphocyte ratio; LCR: lymphocyte to CRP ratio).
Evaluation of Risk Factors Affecting Mortality

ALT: alanine aminotransferase; LDH: lactate dehydrogenase; CRP: C-reactive protein; WBC: white blood count; NLR: neutrophil lymphocyte ratio; CAR: CRP albumin ratio; PLR: platelet to lymphocyte ratio; LCR: lymphocyte to CRP ratio); IL-6: interleukin 6.
